Managing Complex Open Tibia Fractures with Neurovascular Injury: A Case Study
Key Takeaway
A Gustilo Type IIIC open tibia fracture involves severe soft tissue damage, significant bone comminution, and arterial injury requiring repair. Assessment includes detailed clinical examination for neurovascular status, Gustilo-Anderson classification, and calculating the Mangled Extremity Severity Score (MESS) to guide urgent limb salvage versus amputation decisions, supported by immediate imaging.
Patient Presentation & History
Mr. J.S. is a 35-year-old male, previously fit and healthy with no significant medical comorbidities or regular medications, who presented to the emergency department following a high-speed motorcycle vs. stationary object collision. He was ejected from the motorcycle, sustaining significant blunt and penetrating trauma. Paramedics reported a conscious patient with a Glasgow Coma Scale (GCS) of 15, complaining of severe left lower extremity pain. A pressure dressing was applied to a profusely bleeding wound on the left leg at the scene, and a rigid splint was used for immobilization. On arrival, vital signs were stable following 1L of crystalloid infusion: blood pressure 110/70 mmHg, heart rate 98 bpm, respiratory rate 16/min, oxygen saturation 99% on room air.
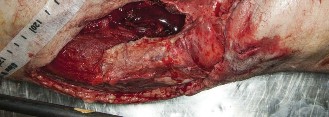
His left lower extremity was noted to have a grotesque deformity with obvious angulation and rotation, associated with a large, irregular laceration on the anteromedial aspect of the leg, approximately 15 x 8 cm in size. The wound was heavily contaminated with road debris, motorcycle parts, and grease. Exposed bone fragments, identified as the distal tibia, were visible within the wound. There was active pulsatile hemorrhage, which was temporarily controlled with direct pressure and a tourniquet applied proximally in the emergency department, pending immediate surgical assessment. A detailed trauma survey (ATLS protocol) revealed no other life-threatening injuries. Given the high-energy mechanism and profound neurovascular compromise, an urgent orthopedic trauma consultation was initiated.
Clinical Examination
Upon focused examination of the left lower extremity after initial resuscitation:
Inspection
- Gross open deformity of the mid-distal leg with significant soft tissue avulsion and degloving, primarily on the anteromedial aspect.
- An extensive, irregular open wound, estimated at 15 x 8 cm, with exposed, comminuted distal tibial shaft and metaphyseal fragments.
- Muscle damage, specifically of the tibialis anterior and extensor hallucis longus, was evident.
- Heavy contamination within the wound, including gravel, soil, and metallic fragments.
- Distal to the injury, the foot was pallid and cool to touch.
- Significant swelling was observed proximally and distally, indicating profound tissue trauma.
Palpation
- Gross instability and crepitus palpable at the fracture site.
- Tense compartments, particularly the anterior and lateral compartments, suggesting potential compartment syndrome, although the open nature of the wound provides some decompression.
- Distal pulses (dorsalis pedis and posterior tibial) were absent by palpation. Handheld Doppler interrogation confirmed absent flow in both vessels. Capillary refill in the toes was significantly prolonged (>4 seconds) despite the tourniquet being temporarily released.
- Profound tenderness across the entire lower leg.
Range of Motion
- Grossly unassessable due to severe pain, instability, and the nature of the open injury. Any attempts at passive motion elicited severe pain and further instability.
- Adjacent joint mobility (knee and ankle) could not be fully assessed but appeared mechanically sound from a skeletal perspective, though the ankle was rendered non-functional due to the distal tibial injury.
Neurological/Vascular Assessment
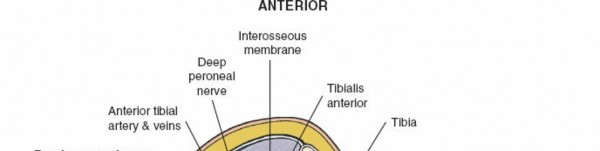
- Vascular: As noted, dorsalis pedis and posterior tibial pulses were absent via palpation and Doppler. The foot was cool and mottled. This strongly indicated an arterial injury compromising distal perfusion.
- Neurological: Sensory examination revealed diminished sensation in the superficial and deep peroneal nerve distributions and absence of sensation in the tibial nerve distribution distal to the injury. Motor function of the ankle and toes was absent. This suggested a combined neurovascular insult, likely involving the posterior tibial artery, peroneal artery, and tibial nerve, given the posteromedial location of the major neurovascular bundle and the extent of soft tissue disruption.
The initial clinical assessment pointed towards a Gustilo-Anderson Type IIIC open fracture of the left tibia and fibula, with associated complete transection or severe injury to the posterior tibial artery and likely the tibial nerve. The MESS (Mangled Extremity Severity Score) score was immediately calculated as:
* Skeletal/Soft-tissue injury: High (Grade 3 – severe crush, degloving)
* Limb Ischemia: High (Grade 2 – pulseless, paresthesias but viable)
* Shock: Low (Grade 0 – systolic BP > 90 mmHg)
* Age: Low (Grade 0 – <30 years, adjusted for age 35 to 1, or kept 0 for simplicity if using strict <30)
This would yield a high MESS score, indicating a significant risk for amputation.
Imaging & Diagnostics
Following the initial clinical assessment and stabilization, a series of urgent imaging studies were obtained.
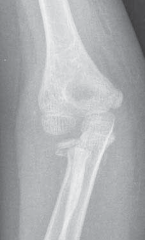
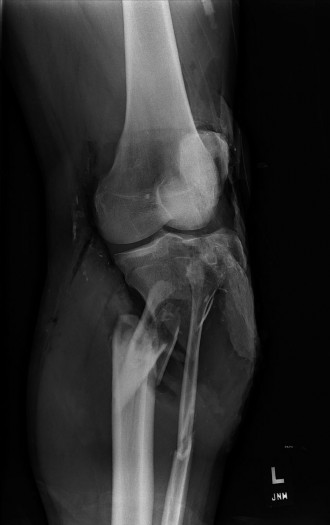
X-ray Findings
Anteroposterior and lateral radiographs of the left tibia and fibula were obtained (initial images are often acquired on a gurney in the ED, thus may be sub-optimal but critical for rapid assessment).
*
AP View:
Revealed a highly comminuted, segmental fracture of the mid-distal tibial shaft, extending into the metaphysis. There was significant shortening and displacement. A large butterfly fragment was noted medially. Several small radiopaque foreign bodies consistent with road debris were visible within the soft tissues surrounding the fracture. The fibula also demonstrated a comminuted fracture at a similar level.
*
Lateral View:
Confirmed the severe comminution and displacement. The open wound was directly communicated with the fracture site, as evidenced by air within the soft tissues and gross bone protrusion. Significant cortical defects and areas of bone loss were apparent.

*
Ankle and Knee Views:
Standard AP/Lateral views of the ankle and knee were performed to rule out concomitant intra-articular fractures, which were negative in this case.
CT Angiogram (CTA)
Due to the profound vascular compromise, an urgent CTA of the left lower extremity was performed. This was crucial for precisely delineating the extent of arterial injury and guiding vascular surgical planning.
*
Findings:
The CTA confirmed complete transection of the posterior tibial artery approximately 5 cm distal to the popliteal trifurcation, with significant spasm and thrombosis of the proximal and distal ends. The anterior tibial artery and peroneal artery demonstrated flow, but with signs of intimal injury and reduced caliber distally. This finding was critical: while the posterior tibial artery was completely out, the remaining two vessels, though compromised, offered potential for collateral flow if the posterior tibial artery could be reconstructed. Significant soft tissue swelling and hematoma were also noted.

Other Diagnostics
- Laboratory Studies: Complete blood count (CBC) showed a normocytic anemia consistent with acute blood loss. Coagulation profile (PT/INR, PTT) was within normal limits. Electrolytes and renal function were monitored due to potential for rhabdomyolysis from crush injury, though initial levels were within normal limits.
- Microbiology: Wound cultures were taken after initial debridement to guide targeted antibiotic therapy.
Templating
Pre-operative planning involved assessing the required length for revascularization and considering options for skeletal stabilization. Given the extensive comminution and bone loss, external fixation was immediately templated as the initial damage control orthopedic (DCO) measure. The plan was to use a delta frame or a hybrid ring fixator for initial stability, allowing for soft tissue management and vascular repair. Definitive fixation options, such as an intramedullary nail or plate fixation, would be re-evaluated after soft tissue healing and vascular patency were secured.
Differential Diagnosis
When confronted with a severe open fracture with neurovascular compromise, the primary "differential diagnosis" shifts from identifying the specific fracture type (which is usually obvious) to determining the optimal treatment strategy, particularly distinguishing between aggressive limb salvage and primary amputation. This decision hinges on a multitude of factors, each contributing to the overall prognosis for functional recovery.
| Feature / Criteria | Limb Salvage (Extensive Reconstruction) | Primary Amputation (Below-Knee or Above-Knee) | Staged Salvage (Damage Control Orthopaedics) |
|---|---|---|---|
| Injury Severity (Gustilo) | Typically Type IIIC; potentially IIIB with severe vascular injury. | Type IIIC with irreparable vascular injury, extensive segmental tissue loss, or severe nerve injury. | Type IIIC with high contamination, significant soft tissue edema, or patient instability. |
| MESS Score | High (≥7) but with potentially modifiable factors (e.g., reconstructible vascular injury, short ischemia). | Very High (≥7), often with additional factors like prolonged warm ischemia, extensive nerve loss. | High (≥7), where initial definitive management is contraindicated due to local or systemic factors. |
| Vascular Status | Reconstructible vascular injury (e.g., segmental transection with suitable vessel ends/donor graft). | Irreparable vascular injury; multiple vessel transections; extensive vessel damage beyond repair. | Reconstructible vascular injury, but requiring staged approach due to other injury burden/contamination. |
| Soft Tissue Injury | Extensive soft tissue loss, degloving, contamination, but reconstructible with local/free flaps. | Massive soft tissue loss, non-viable muscle, extensive crush injury, high infection risk without viable tissue. | Extensive, contaminated soft tissue injury requiring serial debridements before definitive closure. |
| Bone Loss/Comminution | Segmental bone loss, severe comminution, reconstructible with bone grafting/transport techniques. | Massive segmental bone loss, irrecoverable bone stock, or severe osteomyelitis risk. | Significant bone loss/comminution, amenable to planned reconstruction after initial debridement/stabilization. |
| Nerve Injury | Complete transection of single major nerve, potentially repairable (grafting). | Irreparable complete transection of multiple major nerves, especially tibial nerve with high motor loss. | Nerve injury assessed during staged procedures, potentially deferred for secondary repair. |
| Ischemia Time | Short warm ischemia time (<6 hours ideally) for successful revascularization. | Prolonged warm ischemia time (>8-12 hours), leading to irreversible tissue damage and reperfusion injury. | Ischemia time managed acutely with temporary shunts if definitive repair is delayed. |
| Patient Factors | Younger, healthy, high functional demands, psychologically robust, good social support. | Elderly, significant comorbidities, low functional demands, poor psychosocial support, prolonged ICU stay predicted. | Polytrauma patient requiring stabilization of other injuries; hemodynamic instability. |
| Resource Availability | Access to specialized microsurgical, plastic, and trauma orthopedic teams. | Standard trauma surgical team for emergent amputation. | Access to experienced trauma teams for serial procedures and reconstructive options. |
| Expected Outcome | Prolonged rehabilitation, potential for good functional limb, albeit with potential long-term issues (pain, stiffness, residual deficit). | Shorter initial hospital stay, potentially faster return to function with prosthetic, but psychological impact. | Bridging strategy to achieve salvage, delaying definitive reconstruction until optimal conditions. |
For Mr. J.S., the critical decision-making centered around whether his vascular injury was repairable within a reasonable warm ischemia window and if the overall tissue damage was compatible with a functional outcome. The severe neurovascular deficit, high Gustilo classification (IIIC), and high MESS score pushed the initial assessment towards a very guarded prognosis for salvage. However, the patient's young age, otherwise healthy status, and the immediate availability of a multidisciplinary trauma team (orthopedic trauma, vascular, and plastic surgery) argued strongly for an attempt at limb salvage, provided the vascular injury was amenable to repair and significant irreversible tissue damage had not yet occurred. The CTA findings, showing a reconstructible posterior tibial artery, further supported this approach.
Surgical Decision Making & Classification
The decision for limb salvage versus primary amputation in Mr. J.S.'s case was made within minutes of his arrival, based on the rapid assessment and imaging.
Why Operative Intervention?
Given the open nature of the fracture, gross contamination, active bleeding, and profound vascular compromise, immediate operative intervention was unequivocally indicated. Non-operative management was not an option. The goals were clear:
1. Control hemorrhage.
2. Restore vascularity to the limb.
3. Debride contaminated and non-viable tissue.
4. Stabilize the skeletal injury.
5. Obtain soft tissue coverage.
6. Prevent infection and achieve a functional limb.
Specific Classifications Utilized:
-
Gustilo-Anderson Classification:
Mr. J.S.'s injury was classified as a
Gustilo-Anderson Type IIIC Open Tibia/Fibula Fracture
.
- Type III: High-energy injury, extensive soft tissue damage, high contamination.
- Type C: Arterial injury requiring repair, regardless of the extent of soft tissue injury. This automatically elevates the injury to Type IIIC. The extent of the soft tissue envelope loss and contamination also qualified it as at least Type IIIB, but the vascular injury was the definitive factor.
-
Mangled Extremity Severity Score (MESS Score):
- Skeletal/Soft-tissue injury: High-energy (IIIC) with extensive degloving, crush, and comminution = 3 points.
- Limb Ischemia: Pulselessness with paresthesias and prolonged capillary refill, but potentially viable if revascularized within 6 hours = 2 points.
- Shock: Normotensive upon arrival = 0 points.
- Age: 35 years = 1 point (scores 0 for <30, 1 for 30-50, 2 for >50).
- Total MESS Score = 3 + 2 + 0 + 1 = 6.
- A MESS score of ≥ 7 is often cited as a strong predictor for amputation. While Mr. J.S.'s score was 6, it was very close to the threshold and highlighted the severe nature of the injury and the high risk of failure if salvage was attempted. This score served as a powerful prognostic indicator but was not the sole determinant for the final decision.
- Tscherne and Oestern Classification (for Closed Soft Tissue Injury): While primarily for closed injuries, the principles apply to assessing the severity of soft tissue damage. In this open injury context, the extensive degloving and muscle contusion would equate to a grade IV (severe crushing, compartment syndrome, irreversible damage), further emphasizing the complexity.
- AO/OTA Fracture Classification: The specific AO/OTA classification for the tibial fracture was 42-C3 (complex, multi-fragmentary distal third diaphyseal fracture) with an associated fibular fracture. This detailed classification aided in planning the skeletal stabilization strategy.
Salvage vs. Amputation Decision Factors:
The decision to attempt limb salvage was based on a comprehensive assessment weighing multiple critical factors:
*
Reconstructible Vascular Injury:
The CTA confirmed a complete transection of the posterior tibial artery but suggested reconstructible ends, and importantly, the anterior tibial and peroneal arteries showed some flow, potentially allowing for collateral circulation. The warm ischemia time was estimated to be within the acceptable window (patient arrived within 2-3 hours of injury).
*
Patient Age and Health:
Mr. J.S. was young and otherwise healthy, suggesting good healing potential and resilience for a prolonged rehabilitation.
*
Absence of Other Life-Threatening Injuries:
His stable systemic condition allowed the focus to be entirely on limb reconstruction.
*
Multidisciplinary Team Availability:
Immediate access to experienced orthopedic trauma, vascular, and plastic surgeons at a Level I trauma center was paramount.
*
Potential for Functional Outcome:
Despite severe nerve injury, the potential for a functional, albeit compromised, limb was deemed superior to an amputation in this young, active patient, especially if nerve grafting could be performed.
*
Patient Wishes:
Although not directly involved in the immediate emergency decision, the patient's pre-injury activity level and implied desire for limb retention were factors considered in the overall approach.
The decision was made to proceed with an aggressive attempt at limb salvage, acknowledging the high risks of infection, non-union, and potential eventual amputation. This was communicated to the patient’s next-of-kin.
Surgical Technique / Intervention
Mr. J.S. was immediately transferred to the operating theatre for emergent surgery. The procedure involved a coordinated multi-specialty approach with Orthopedic Trauma, Vascular Surgery, and Plastic Surgery teams.
1. Patient Positioning
- The patient was positioned supine on a radiolucent operating table to allow for easy access to the entire lower extremity, C-arm fluoroscopy, and potential harvest sites (e.g., contralateral saphenous vein, free flap donor sites). A tourniquet was placed high on the thigh but not inflated initially, reserving it for specific stages.
2. Initial Debridement and Exploration (Orthopedic Trauma & Vascular)
- The wound was thoroughly irrigated with copious amounts of normal saline (minimum 9 liters).
- Aggressive, but judicious, excisional debridement of all non-viable skin, subcutaneous tissue, muscle, and contaminated bone fragments was performed. All road debris and foreign bodies were meticulously removed. The deep fascia was opened to fully explore all compartments and relieve any potential occult compartment syndrome.
- The fracture hematoma was evacuated. Exposed bone ends were debrided of periosteum and non-viable fragments.
- Following debridement, the wound was re-examined. The complete transection of the posterior tibial artery and an associated partial transection of the tibial nerve were confirmed. The anterior tibial artery and peroneal artery, while present, showed significant contusion.
3. Vascular Reconstruction (Vascular Surgery)
- Harvest of Autologous Graft: The contralateral greater saphenous vein was harvested from the non-injured leg, using a skip incision technique to obtain a sufficient length (approximately 15 cm) for an interposition graft.
- Arterial Repair: The transected ends of the posterior tibial artery were prepared. A reverse saphenous vein interposition graft was anastomosed end-to-end to the proximal and distal segments of the posterior tibial artery using microsurgical techniques (8-0 Prolene sutures).
- Confirmation of Patency: Post-anastomosis, blood flow was confirmed distally by Doppler signal, pulsatile flow, and improved color and warmth of the foot. An intraoperative angiogram was performed to confirm graft patency and rule out distal emboli or spasm.
4. Skeletal Stabilization (Orthopedic Trauma)
-
Damage Control External Fixation:
Given the extensive soft tissue injury, contamination, and the need for serial debridements and soft tissue coverage, immediate definitive internal fixation was contraindicated. A monolateral external fixator (e.g., LRS or a hybrid ring fixator construct) was applied to the tibia.
- Proximal pins were inserted into the tibia in safe zones, away from major neurovascular structures.
- Distal pins were inserted into the calcaneus and potentially the talus, ensuring stability of the ankle joint and providing a stable platform for length restoration and alignment.
-
The frame was constructed to achieve appropriate length, alignment, and rotation, providing provisional stability to the highly comminuted tibial and fibular fractures. This allowed for subsequent soft tissue procedures without disturbing the vascular repair or skeletal alignment.
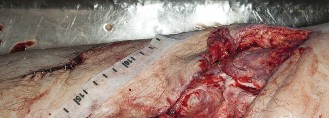
5. Initial Soft Tissue Management (Orthopedic Trauma & Plastic Surgery)
- Following skeletal stabilization, the wound was copiously irrigated again.
- A vacuum-assisted closure (VAC) device was applied over the extensive wound and exposed bone. This serves to manage exudate, promote granulation tissue formation, and prepare the wound bed for definitive soft tissue coverage.
- The tourniquet was released. The patient was monitored closely for reperfusion injury and compartment syndrome post-revascularization.
6. Tibial Nerve Repair (Optional/Delayed)
- Due to the degree of contamination and soft tissue loss, immediate primary repair of the transected tibial nerve was not performed during the initial surgery. The plan was for a delayed nerve grafting procedure by the plastic surgery team once the wound bed was clean and stable, typically after definitive soft tissue coverage.
7. Post-Operative Management in OR
- Broad-spectrum intravenous antibiotics (e.g., a cephalosporin and an aminoglycoside, or meropenem if highly contaminated) were continued, pending culture results.
- The patient was transferred to the intensive care unit (ICU) for close monitoring of the revascularized limb, fluid balance, and systemic parameters.
Subsequent Staged Procedures:
- Serial Debridements: The patient underwent multiple planned surgical debridements (every 24-48 hours) in the operating room to remove any further necrotic or non-viable tissue and to re-irrigate the wound. The VAC dressing was changed with each debridement.
- Definitive Soft Tissue Coverage (Plastic Surgery): After 5 days and 3 debridements, a clean, granulating wound bed was achieved. Given the large defect and exposed bone, a free tissue transfer was planned. A latissimus dorsi free flap was harvested and microvascularly anastomosed to recipient vessels in the thigh or proximal leg to cover the extensive soft tissue defect over the tibia. The flap was monitored closely for viability.
- Delayed Primary or Definitive Skeletal Fixation (Orthopedic Trauma): Approximately 2-3 weeks post-injury, after successful flap take and stability of the soft tissue envelope, the external fixator was revised for better stability, or converted to definitive internal fixation (e.g., intramedullary nailing or plate fixation) if conditions allowed. In this case, given the segmental bone loss and extensive soft tissue reconstruction, the external fixator was maintained and modified into a limb reconstruction frame (e.g., Ilizarov or Taylor Spatial Frame) to address the bone defect with bone transport or gradual compression/distraction osteogenesis. This approach allowed for concomitant treatment of bone loss and non-union potential.
- Bone Grafting: Autologous cancellous bone grafting from the iliac crest was planned once the soft tissue envelope was stable and the risk of infection minimized, likely several weeks to months after the initial injury.
- Tibial Nerve Grafting: After initial wound healing and flap integration, and once the overall condition permitted, a sural nerve graft was used to reconstruct the tibial nerve defect.
Post-Operative Protocol & Rehabilitation
The post-operative course for a complex open fracture managed with limb salvage is protracted and requires a highly individualized, multidisciplinary approach.
Immediate Post-Operative Phase (Weeks 0-2)
- ICU Monitoring: Close observation for flap viability (Doppler, clinical assessment), signs of compartment syndrome, infection, and systemic complications.
- Antibiotics: Continued broad-spectrum IV antibiotics, tailored to wound cultures. Duration guided by infectious disease consultation, often 6 weeks or more for Gustilo III fractures, especially if osteomyelitis is suspected.
- Wound Care: Meticulous care of the free flap (if used), donor site, and external fixator pin sites. Regular VAC changes until flap coverage is achieved.
- Vascular Watch: Strict neurovascular checks of the revascularized limb. Anticoagulation (e.g., aspirin, heparin drip) as per vascular surgeon protocol to maintain graft patency.
- Pain Management: Multimodal analgesia including regional blocks, NSAIDs, and opioid titration.
- DVT Prophylaxis: Pharmacological (low molecular weight heparin) and mechanical (sequential compression devices) prophylaxis.
- Mobilization: Strict non-weight bearing on the affected limb. Gentle passive range of motion (PROM) for adjacent joints (hip, knee, and foot/ankle where possible without stressing the fracture site or flap) to prevent stiffness, initiated by physical therapy.
Intermediate Phase (Weeks 2-12)
- Wound and Flap Management: Continued monitoring for infection or flap complications. Progression of wound healing.
- Skeletal Management: External fixator adjustments (e.g., for bone transport, compression). Serial radiographs to assess fracture healing and bone defect management.
-
Early Rehabilitation:
- Physical Therapy (PT): Focus on maintaining strength in uninjured muscles, balance, core stability. Non-weight bearing gait training with crutches or walker. Gradual passive and active-assisted range of motion for knee and ankle, avoiding stress on the fracture site and soft tissue reconstruction.
- Occupational Therapy (OT): Assessment for activities of daily living (ADLs) and adaptive equipment.
- Nerve Healing: Regular neurological assessment. Referral to nerve specialist or physiatrist.
Late Phase (Months 3-12+)
- Fracture Consolidation: Progression to weight-bearing as fracture healing allows (determined by radiographic evidence and clinical stability). Transition from external fixator to definitive internal fixation or continued management in the frame.
- Bone Grafting/Reconstruction: If staged, further bone grafting procedures may be performed once soft tissues are healed and infection risk is low.
- Nerve Recovery: Continued monitoring of nerve function. EMG studies at 3-6 months. Consideration for further nerve reconstruction if no recovery.
-
Intensified Rehabilitation:
- PT: Progressive weight-bearing exercises, strengthening of the entire lower extremity, gait retraining, proprioceptive exercises, balance training. Focus on restoring functional ambulation.
- OT: Addressing specific functional deficits related to nerve injury, range of motion, and strength. Return to work/hobbies assessment.
-
Complication Surveillance:
- Infection: Persistent vigilance for osteomyelitis, flap infection.
- Non-union/Malunion: Careful follow-up with regular radiographs. Intervention if necessary (revision surgery, further bone grafting).
- Hardware Complications: Pin tract infections, loosening, breakage.
- Chronic Pain: Neuropathic pain (CRPS), soft tissue pain. Aggressive pain management, potentially involving pain specialists.
- Psychological Impact: Significant emotional and psychological distress is common. Early referral for psychological support or counseling is crucial.
The overall rehabilitation trajectory for Mr. J.S. would extend for 18-24 months, potentially longer, with a realistic expectation of achieving a functional, albeit possibly compromised, limb for activities of daily living and light work. Full pre-injury function is often elusive in such severe injuries.
Pearls & Pitfalls (Crucial for FRCS/Board Exams)
Pearls for Complex Open Fractures and Salvage Decisions:
- Time is Tissue (The Golden Hour): For Type IIIC injuries with vascular compromise, early revascularization (<6-8 hours warm ischemia time) is paramount to improve salvage rates and minimize reperfusion injury.
- Damage Control Orthopaedics (DCO): Initial, rapid debridement and external fixation is the standard. Do not attempt definitive internal fixation in a highly contaminated, unstable wound. "Fix the patient, then fix the fracture."
- Multidisciplinary Approach: Successful salvage requires a coordinated team: trauma surgeons, orthopedic trauma surgeons, vascular surgeons, plastic surgeons, infectious disease specialists, and rehabilitation specialists.
- Meticulous Debridement: The most crucial step. All non-viable tissue (skin, subcutaneous fat, muscle, bone fragments) must be excised. "The solution to pollution is dilution" – copious irrigation. Serial debridements are often necessary.
- Soft Tissue First: Achieving a healthy, vascularized soft tissue envelope is more critical than immediate definitive bone fixation. The "fix the envelope, then the skeleton" principle.
- Vascular Repair First (after debridement): Prioritize restoration of circulation after initial debridement. Skeletal stabilization should then be performed without compromising the vascular repair.
- Prognostic Tools (e.g., MESS Score): Use scores like MESS as a guide, not a rigid rule. They quantify severity but do not dictate the decision. Patient factors, surgeon experience, and institutional resources are equally important.
- Realistic Expectations: Counsel the patient and family transparently about the long, arduous recovery, potential for multiple surgeries, and the high complication rates (infection, non-union, chronic pain, residual deficits), even if salvage is successful.
- Consider Nerve Management: While often delayed, assess major nerve integrity. Early nerve coaptation or grafting can significantly improve long-term functional outcomes if conditions allow.
- Nutritional Support: These patients are catabolic; aggressive nutritional support is vital for healing and preventing infection.
Pitfalls to Avoid:
- Inadequate Debridement: The most common cause of persistent infection (osteomyelitis) and salvage failure. Leaving devitalized tissue is a setup for disaster.
- Delayed Revascularization: Prolonged warm ischemia time (>8 hours) significantly increases the risk of limb loss, non-functional limb, and systemic complications from reperfusion injury.
- Premature Definitive Internal Fixation: Placing plates or intramedullary nails into a contaminated or uncertain wound environment dramatically increases the risk of deep infection and osteomyelitis, often necessitating hardware removal and re-do surgery.
- Underestimating Soft Tissue Needs: Failing to secure adequate, healthy soft tissue coverage (local flap, free flap) over exposed bone or hardware will inevitably lead to complications.
- Ignoring Compartment Syndrome: Especially post-revascularization, the reperfusion phenomenon can lead to acute compartment syndrome, requiring emergent fasciotomies. Monitor closely.
- Focusing Solely on Bone: "Don't operate on an X-ray." The soft tissue envelope, vascularity, and nerve function are often more critical for limb function than perfect bone alignment in these complex cases.
- Lack of Psychological Support: The journey for a salvage patient is physically and psychologically taxing. Failing to address their mental health needs can hinder rehabilitation.
- Poor Pin Site Care: Neglecting external fixator pin sites can lead to pin tract infections, which can track deep and complicate subsequent surgeries or lead to osteomyelitis.
- Early Weight Bearing: Premature weight-bearing before adequate bone healing or soft tissue maturation can lead to hardware failure, non-union, or flap breakdown.
- Failure to Monitor for Systemic Complications: Sepsis, ARDS, rhabdomyolysis, and acute kidney injury can occur, especially in high-energy trauma and crush injuries. Vigilant monitoring is essential.
Clinical & Radiographic Imaging
You Might Also Like