Advanced Perioperative Pain Management for Shoulder Surgery: Regional Anesthesia & Neuroanatomy
Key Takeaway
Advanced perioperative pain management for shoulder surgery emphasizes regional anesthesia, especially peripheral nerve blocks (PNBs), in a multimodal approach. This significantly reduces opioid use, minimizes side effects, and facilitates faster recovery. Understanding the shoulder's neuroanatomy is crucial for effective, targeted pain control and optimal patient outcomes.
Introduction & Epidemiology
Adequate management of perioperative pain following shoulder surgery is a critical determinant of post-operative recovery, patient satisfaction, and functional outcomes. The incidence of shoulder pathologies requiring surgical intervention, ranging from arthroscopic debridement and rotator cuff repair to total shoulder arthroplasty, continues to rise. Effective pain control is paramount not only for humane patient care but also for mitigating adverse physiological responses to stress, facilitating early mobilization, and optimizing participation in rehabilitation protocols.
Historically, post-operative pain management heavily relied on systemic opioid analgesia. While effective, this approach is associated with a spectrum of undesirable side effects including nausea, vomiting, constipation, respiratory depression, sedation, pruritus, and the potential for opioid-induced hyperalgesia and prolonged opioid dependence. Given the ongoing global opioid crisis, strategies that minimize opioid consumption while maintaining superior analgesia are highly valued. Regional anesthesia, particularly peripheral nerve blocks (PNBs), has emerged as a cornerstone of modern multimodal perioperative pain management for shoulder surgery.
The evolution of PNBs, from William Halsted’s pioneering use of cocaine in 1885 for local anesthesia, has seen significant advancements, particularly in the last three decades with the advent of ultrasound guidance. The increasing trend in the utilization of PNBs reflects a growing body of evidence supporting their efficacy in:
* Decreasing hospital length of stay, facilitating the transition from inpatient to outpatient surgical models.
* Significantly reducing opioid requirements and their associated side effects.
* Enhancing patient engagement in physical therapy, thereby improving functional recovery.
* Elevating overall patient satisfaction.
* Demonstrating improved cost-effectiveness by reducing resource utilization and complications.
A comprehensive understanding of the relevant anatomy, indications, contraindications, and potential complications associated with regional anesthetic techniques is indispensable for orthopedic surgeons and anesthesiologists. This guide aims to provide a high-yield overview of contemporary perioperative pain management strategies for shoulder surgery, emphasizing the role of regional anesthesia within a multimodal framework.
Surgical Anatomy & Biomechanics
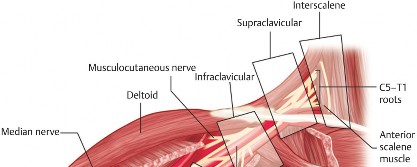
Effective perioperative pain management for shoulder surgery necessitates a profound understanding of the neuroanatomy supplying the shoulder girdle. The primary sensory innervation to the glenohumeral joint, acromioclavicular (AC) joint, and surrounding musculature is derived from the brachial plexus, with contributions from cervical spinal nerves C3-C4.
The
brachial plexus
is formed by the ventral rami of spinal nerves C5, C6, C7, C8, and T1. These roots emerge between the anterior and middle scalene muscles, forming three trunks, six divisions, three cords, and numerous terminal branches.
*
Roots (C5-T1):
Emerge from the intervertebral foramina. The C5-C6 roots coalesce to form the upper trunk; C7 continues as the middle trunk; C8-T1 unite to form the lower trunk. The roots are the target for interscalene blocks.

*
Trunks (Upper, Middle, Lower):
Located in the supraclavicular fossa, superior to the first rib. These are the targets for supraclavicular blocks.
*
Divisions (Anterior, Posterior):
Each trunk divides into anterior and posterior divisions as they pass posterior to the clavicle. These are rarely targeted directly.
*
Cords (Lateral, Posterior, Medial):
Formed in the axilla, named for their relationship to the axillary artery. The cords give rise to the major terminal nerves of the upper extremity.
Key Nerves of the Brachial Plexus Relevant to Shoulder Pain:
*
Suprascapular Nerve (C5, C6):
Arises from the upper trunk. It supplies motor innervation to the supraspinatus and infraspinatus muscles and sensory innervation to the superior and posterior aspects of the glenohumeral joint, as well as the AC joint and subacromial bursa. It is a critical target for pain control, especially in rotator cuff pathology and shoulder arthroplasty.
*
Axillary Nerve (C5, C6):
Arises from the posterior cord. Provides motor innervation to the deltoid and teres minor, and sensory innervation to the lateral shoulder (regimental badge area). While primarily motor, its sensory contribution is relevant.
*
Lateral Pectoral Nerve (C5, C6, C7):
Arises from the lateral cord. Motor to pectoralis major.
*
Medial Pectoral Nerve (C8, T1):
Arises from the medial cord. Motor to pectoralis major and minor.
*
Long Thoracic Nerve (C5, C6, C7):
Arises directly from roots, motor to serratus anterior.
*
Dorsal Scapular Nerve (C5):
Arises directly from C5 root, motor to rhomboids and levator scapulae.
*
Musculocutaneous Nerve (C5, C6, C7):
Arises from the lateral cord. Motor to biceps and brachialis; sensory to lateral forearm (lateral antebrachial cutaneous nerve). While typically not a primary target for shoulder analgesia, it can be involved in block spread.
Cervical Plexus Contributions:
The shoulder also receives sensory innervation from branches of the
cervical plexus
(C3-C4), specifically the supraclavicular nerves. These nerves innervate the skin over the superior aspect of the shoulder and clavicle. For surgical procedures involving extensive skin incisions or periosteal stripping (e.g., total shoulder arthroplasty with an deltopectoral approach), a superficial cervical plexus block can complement brachial plexus blocks.
Pathophysiology of Postoperative Shoulder Pain:
Postoperative pain arises from a complex interplay of nociceptive stimuli from incisional wounds, deep tissue manipulation, joint distension, muscle spasm, and inflammatory responses.
*
Incisional Pain:
Directly related to skin and subcutaneous tissue trauma.
*
Deep Somatic Pain:
Originates from manipulated muscles, tendons, ligaments, and the joint capsule. The glenohumeral joint capsule is richly innervated, particularly its anterior and posterior aspects.
*
Bone Pain:
From osteotomies (e.g., glenoid and humeral reaming in arthroplasty) or fixation sites.
*
Inflammation:
Surgical trauma induces a localized inflammatory cascade, releasing prostaglandins, bradykinin, and other mediators that sensitize peripheral nociceptors.
Biomechanics of Recovery and Pain:
Effective pain management directly impacts the biomechanics of recovery. Uncontrolled pain leads to muscle guarding, limited range of motion (ROM), and patient reluctance to participate in rehabilitation. This can result in:
*
Shoulder Stiffness:
Delayed or incomplete ROM recovery.
*
Muscle Atrophy:
Disuse further compromises shoulder stability and strength.
*
Delayed Functional Return:
Impaired ability to perform activities of daily living.
*
Chronic Pain Syndromes:
Persistent neuropathic or centralized pain, often requiring further intervention.
Regional anesthesia, by blocking nerve conduction in the perioperative period, interrupts the afferent pain signals, reduces central sensitization, and allows for earlier, more comfortable engagement in the necessary biomechanical restoration processes.
Indications & Contraindications
Indications for Regional Anesthesia and Multimodal Analgesia in Shoulder Surgery
The use of regional anesthesia and a multimodal analgesia approach is indicated for nearly all types of shoulder surgery, particularly those associated with moderate to severe postoperative pain. The primary goal is to optimize pain control, minimize opioid consumption, and enhance functional recovery.
General Indications:
*
All major shoulder surgeries:
Including total shoulder arthroplasty (TSA), reverse total shoulder arthroplasty (rTSA), rotator cuff repair (arthroscopic or open), capsular shift/labral repair, and fracture fixation.
*
Outpatient surgical procedures:
Facilitates early discharge and improved patient experience.
*
Patients with comorbidities:
Especially those at high risk for opioid-related adverse effects (e.g., sleep apnea, obesity, elderly, chronic pulmonary disease).
*
History of opioid abuse or dependence:
To minimize new opioid prescriptions.
*
Expected significant postoperative pain:
As assessed by the surgeon, anesthesiologist, and patient expectations.
*
Enhanced recovery after surgery (ERAS) protocols:
A core component of modern ERAS pathways.
Specific Block Indications:
*
Interscalene Brachial Plexus Block (ISB):
Provides excellent analgesia for procedures involving the glenohumeral joint and proximal humerus. Most effective for rotator cuff repair, instability surgery, and shoulder arthroplasty.
*
Suprascapular Nerve Block (SSNB):
Offers targeted analgesia for superior and posterior shoulder pain, particularly useful for rotator cuff repair, AC joint pathology, and as an adjunct to ISB or for patients where ISB is contraindicated.
*
Pectoral Nerve Blocks (PECS I/II):
Useful for procedures involving the anterior chest wall and pectoral region, such as subpectoral biceps tenodesis, clavicle ORIF, or procedures with extensive anterior dissection.
*
Superficial Cervical Plexus Block:
Augments analgesia for procedures with significant skin incisions over the clavicle or superior shoulder, often combined with an ISB for TSA/rTSA.
Contraindications for Regional Anesthesia
Absolute and relative contraindications must be carefully considered during pre-operative planning to ensure patient safety.
Absolute Contraindications:
*
Patient refusal:
The patient's autonomy must be respected.
*
Allergy to local anesthetics:
A rare but critical contraindication.
*
Local infection at the injection site:
Increases the risk of spreading infection to deeper tissues or the neural structures.
*
Severe coagulopathy or therapeutic anticoagulation (not reversible):
Significant risk of hematoma formation around neural structures, potentially leading to nerve compression and permanent injury. This requires careful discussion with hematology and the surgical team regarding timing and reversal strategies.
Relative Contraindications:
*
Pre-existing neurological deficit in the affected limb:
Can obscure the diagnosis of block-related nerve injury or worsen an existing deficit. Careful pre-block neurological examination and documentation are crucial.
*
Peripheral neuropathy:
May increase sensitivity to local anesthetic neurotoxicity or complicate assessment of block efficacy and resolution.
*
Previous surgery or radiation in the neck/supraclavicular region:
May alter anatomy, making block placement more challenging and increasing the risk of complications.
*
Severe pulmonary disease (for ISB):
Due to the risk of phrenic nerve palsy and subsequent hemidiaphragmatic paresis, which can compromise respiratory function.
*
Unstable vital signs:
Requires stabilization before block placement.
*
Lack of patient cooperation:
Essential for safe and effective block placement.
Operative vs. Non-Operative Indications for Pain Management Strategies
| Indication Type | Surgical Procedure Examples | Non-Surgical Pain Management Examples | Role of Regional Anesthesia / Multimodal Analgesia |
|---|---|---|---|
| Operative | Rotator Cuff Repair | - | Primary strategy for perioperative pain control, reducing opioid use, facilitating early rehabilitation. ISB, SSNB, PECS blocks, multimodal oral regimen. |
| Total Shoulder Arthroplasty (TSA) / Reverse TSA | - | Essential for managing severe postoperative pain. Continuous ISB catheters, ISB, SSNB, Superficial Cervical Plexus Block, multimodal oral/IV regimen. | |
| Shoulder Instability Repair (Labral Repair, Capsular Shift) | - | Significant pain reduction, particularly during early post-operative period. ISB, multimodal oral regimen. | |
| Proximal Humerus Fracture Fixation | - | Critical for acute pain management and allowing early physical therapy. ISB, continuous ISB. | |
| AC Joint Reconstruction / Distal Clavicle Excision | - | Effective analgesia for local incisional and deep pain. ISB, SSNB, local infiltration. | |
| Subacromial Decompression / Biceps Tenodesis | - | Good for moderate pain relief and early mobilization. ISB, SSNB, PECS II. | |
| Non-Operative | Acute Rotator Cuff Tendinopathy | Oral NSAIDs, Acetaminophen, Physical Therapy, Corticosteroid Injections | Limited role for acute PNBs, but SSNB can provide diagnostic/therapeutic relief for chronic suprascapular nerve entrapment or persistent posterior shoulder pain. |
| Glenohumeral Osteoarthritis | Oral NSAIDs, Acetaminophen, Physical Therapy, Intra-articular Corticosteroid or Viscosupplementation Injections | SSNB or suprascapular nerve pulsed radiofrequency ablation may be considered for refractory pain in non-surgical candidates. | |
| Adhesive Capsulitis (Frozen Shoulder) | Oral NSAIDs, Physical Therapy, Intra-articular Corticosteroid Injections | Suprascapular nerve blocks can be used as a therapeutic adjunct to facilitate physical therapy and improve ROM in refractory cases. | |
| Chronic Regional Pain Syndrome (CRPS) Type I | Multimodal pharmacotherapy (gabapentinoids, TCAs), Physical Therapy, Sympathetic Blocks | Interscalene blocks are not primary, but stellate ganglion blocks (a sympathetic block) may be indicated for CRPS of the upper extremity, though less specific for shoulder pain itself. |
Pre-Operative Planning & Patient Positioning
Thorough pre-operative planning is essential to optimize perioperative pain management for shoulder surgery. This involves a collaborative discussion between the orthopedic surgeon, anesthesiologist, and the patient.
Pre-Operative Evaluation and Discussion:
-
Patient Education and Expectation Management:
- Discuss anticipated pain levels, the benefits of regional anesthesia (faster recovery, less opioid use), and potential side effects or complications.
- Explain the multimodal approach, including the roles of regional blocks, non-opioid analgesics, and opioids.
- Set realistic expectations regarding pain relief; complete pain eradication is often not achievable.
- Provide information on post-operative pain scales and reporting methods.
-
Medical History Review:
- Identify pre-existing neurological deficits in the operative limb. Document baseline sensory and motor function meticulously.
- Assess for significant cardiac or pulmonary comorbidities that might influence the choice of regional block (e.g., severe COPD might contraindicate ISB due to phrenic nerve palsy risk).
- Evaluate for bleeding diatheses, anticoagulant use, or antiplatelet medications. Discuss with the hematology service or prescribing physician regarding holding or reversing agents according to ASRA guidelines.
- Assess for previous adverse reactions to local anesthetics or opioids.
- Identify current opioid use or history of opioid use disorder. This dictates a more aggressive opioid-sparing strategy.
-
Anesthesia Consult:
- The anesthesiologist will determine the most appropriate regional block technique(s) based on surgical invasiveness, patient factors, and potential contraindications.
- Discuss the option of a continuous peripheral nerve block (CPNB) catheter for prolonged analgesia versus a single-shot block. CPNB is often preferred for more invasive procedures like shoulder arthroplasty.
-
Prescription of Oral Multimodal Analgesics:
- Initiate non-opioid medications pre-operatively if appropriate (e.g., celecoxib, gabapentinoids). This "pre-emptive analgesia" can reduce post-operative pain and opioid requirements.
- Consider a low-dose oral opioid prescription for breakthrough pain, with clear instructions on responsible use.
Patient Positioning for Regional Block Placement:
Optimal positioning is crucial for safe and effective block placement, typically performed pre-operatively in a dedicated block room or the operating room prior to sterile surgical draping. Ultrasound guidance is the standard of care for peripheral nerve blocks.
-
Interscalene Block (ISB):
- Position: Supine with the head turned approximately 45 degrees to the contralateral side. The ipsilateral arm is adducted, and the shoulder is slightly depressed.
- Anatomical Landmarks: Sternocleidomastoid muscle, anterior and middle scalene muscles. The brachial plexus roots (C5-C7) are typically visualized between the anterior and middle scalene muscles, lateral to the carotid artery and internal jugular vein.
- Ultrasound Probe Placement: High-frequency linear array transducer placed transversely across the neck, superior to the clavicle, at the level of the cricoid cartilage.
-
Needle Trajectory:
In-plane or out-of-plane approach, directed towards the "traffic light" sign or the stacked "bunch of grapes" appearance of the brachial plexus.

-
Supraclavicular Block:
- Position: Supine with the head turned to the contralateral side. Arm adducted.
- Anatomical Landmarks: Clavicle, subclavian artery, first rib. The brachial plexus trunks/divisions are superficial to the subclavian artery, superior to the first rib.
- Ultrasound Probe Placement: High-frequency linear array transducer placed transversely in the supraclavicular fossa, superior to the clavicle, aimed to visualize the "cluster of grapes" lateral to the subclavian artery.
- Needle Trajectory: In-plane approach, often directed from lateral to medial, avoiding the pleura and subclavian artery.
-
Suprascapular Nerve Block (SSNB):
- Position: Sitting or lateral decubitus. For ultrasound-guided posterior approach, the patient can be prone or sitting with the arm across the chest to protract the scapula.
- Anatomical Landmarks: Scapular spine, suprascapular notch (or spinoglenoid notch).
- Ultrasound Probe Placement: High-frequency linear array transducer placed over the suprascapular notch or spinoglenoid notch, identifying the nerve alongside the suprascapular artery and vein.
- Needle Trajectory: In-plane approach towards the nerve.
-
Pectoral Nerve Blocks (PECS I/II):
- Position: Supine with the arm abducted to 90 degrees.
- Anatomical Landmarks: Pectoralis major and minor muscles, axillary artery and vein.
- Ultrasound Probe Placement: High-frequency linear array transducer placed at the level of the third or fourth rib in the midclavicular line, scanning laterally towards the axilla to identify the fascial planes between the pectoralis major/minor (PECS I) and between pectoralis minor and serratus anterior (PECS II).
- Needle Trajectory: In-plane approach to deposit local anesthetic in the respective fascial planes.
Patient Positioning for Surgical Procedure:
After block placement, the patient is positioned for surgery. Proper positioning is paramount for surgical access, patient safety, and optimal visualization.
-
Beach Chair Position:
- Indications: Most common for arthroscopic shoulder procedures, rotator cuff repair, instability repair, and open procedures like TSA/rTSA.
- Technique: Patient is supine on the operating table. Torso is elevated to approximately 45-70 degrees. The head is secured in a headrest (e.g., Mayfield or doughnut pillow) to prevent cervical hyperextension or lateral flexion. Legs are often slightly flexed at the hips and knees to prevent posterior subluxation of the knees. The operative arm is free-draped, allowing full mobility.
- Key Considerations: Ensure adequate padding at pressure points (heels, sacrum, occiput). Monitor cerebral perfusion, especially in hypotensive patients, as cerebral autoregulation may be compromised. Adequate blood pressure monitoring at the level of the brain (e.g., using an arterial line) is advisable.
-
Lateral Decubitus Position:
- Indications: Less common now for arthroscopy due to beach chair popularity, but still used for some complex fracture cases or specific approaches.
- Technique: Patient lies on the non-operative side. Axillary roll placed under the dependent axilla to protect the brachial plexus. Dependent arm is extended forward, padded. Dependent leg flexed, non-dependent leg straight with pillow between. Torso secured with beanbag and straps. Operative arm suspended in traction for arthroscopy.
- Key Considerations: Protect neural structures (brachial plexus, ulnar nerve, peroneal nerve). Ensure stable positioning to prevent patient movement during traction.
Following positioning, the surgical site is prepped and draped. The regional block should be assessed for efficacy before the start of the surgical incision to allow for supplementation if necessary.
Detailed Surgical Approach / Technique (Pain Management Focused)
This section focuses on the detailed techniques of regional anesthesia pertinent to shoulder surgery, as these are the "surgical approaches" from a pain management perspective. Each block requires precise anatomical identification, typically guided by ultrasound, and careful needle manipulation to ensure optimal local anesthetic delivery and minimize complications.
1. Interscalene Brachial Plexus Block (ISB)
The ISB is the most commonly employed regional anesthetic technique for shoulder surgery, providing robust analgesia for procedures involving the glenohumeral joint, proximal humerus, and clavicle.
Anatomy (Ultrasound-Guided):
*
Patient Position:
Supine, head turned 45 degrees to contralateral side.
*
Transducer Placement:
High-frequency linear array transducer (10-15 MHz) placed transversely at the level of the cricoid cartilage.
*
Identification:
Identify the sternocleidomastoid (SCM) muscle anteriorly. Scan laterally to identify the anterior scalene muscle (ASM) and then the middle scalene muscle (MSM). The brachial plexus roots (typically C5, C6, C7) appear as hypoechoic, round or oval structures ("traffic light" or "bunch of grapes" appearance) between the ASM and MSM. The phrenic nerve lies superficial to the ASM. The deep cervical fascia surrounds the scalene muscles and brachial plexus.

Technique (Single-Shot):
1.
Skin Preparation:
Sterile prep and drape.
2.
Local Anesthetic:
Inject a small wheal of local anesthetic (e.g., lidocaine 1%) at the needle insertion site.
3.
Needle Insertion:
A 22-gauge, 5-cm block needle (e.g., echogenic bevel) is inserted in-plane or out-of-plane. For an in-plane approach, the needle is typically inserted from lateral to medial, aiming towards the space between the anterior and middle scalene muscles, adjacent to the brachial plexus.
4.
Target Confirmation:
Advance the needle under direct ultrasound visualization until the tip is positioned within the fascial plane surrounding the brachial plexus, typically posterior to C5/C6 roots.
5.
Hydrodissection/Injection:
After negative aspiration for blood, slowly inject a small test dose (e.g., 1-2 mL) of local anesthetic. Observe for "hydrodissection" – the spread of local anesthetic circumferentially around the nerve roots. This confirms proper needle tip placement.
6.
Full Injection:
Administer the full dose of local anesthetic (e.g., 15-20 mL of ropivacaine 0.5% or bupivacaine 0.5% for a single shot). Observe for continued spread around the nerves.

Continuous Interscalene Block (C-ISB):
*
Technique:
Similar initial setup. A larger gauge needle (e.g., 17-18 gauge Tuohy) is used to facilitate catheter placement. Once the needle tip is optimally positioned, a multi-orifice catheter is threaded 3-5 cm beyond the needle tip into the interscalene groove. The catheter is then secured with skin adhesive and a transparent dressing.
*
Infusion:
A continuous infusion of dilute local anesthetic (e.g., ropivacaine 0.2% at 5-10 mL/hr with patient-controlled boluses) is initiated post-operatively.
2. Supraclavicular Brachial Plexus Block
This block targets the brachial plexus at the trunk/division level, just superior to the clavicle. It provides excellent anesthesia for the entire arm and hand but may spare innervation to the superior shoulder via C3/C4 supraclavicular nerves. Less risk of phrenic nerve palsy compared to ISB.
Anatomy (Ultrasound-Guided):
*
Patient Position:
Supine, head turned to contralateral side, arm adducted.
*
Transducer Placement:
High-frequency linear array transducer placed transversely in the supraclavicular fossa, just superior to the clavicle, aimed to visualize the "cluster of grapes" (brachial plexus trunks/divisions) lateral to the pulsatile subclavian artery and superior to the first rib. The pleura should also be visualized deep to the first rib.
Technique:
1.
Skin Prep/Local Anesthetic:
Sterile prep, skin wheal.
2.
Needle Insertion:
A 22-gauge, 5-cm needle is typically inserted in-plane from a lateral-to-medial direction, aiming towards the "corner pocket" formed by the subclavian artery and the first rib, where the lower trunk is often located.
3.
Target Confirmation/Injection:
Once the needle tip is adjacent to the brachial plexus, after negative aspiration, inject 20-25 mL of local anesthetic (e.g., ropivacaine 0.5%). Observe for spread around the plexus. Careful visualization of the pleura is critical to avoid pneumothorax.
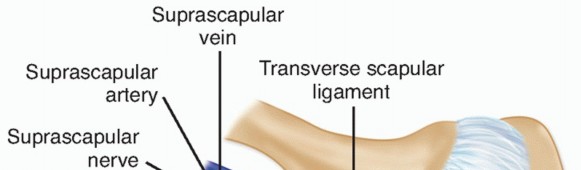
3. Suprascapular Nerve Block (SSNB)
The suprascapular nerve provides significant sensory innervation to the glenohumeral and AC joints, making its block highly effective for posterior and superior shoulder pain.
Anatomy (Ultrasound-Guided Posterior Approach):
*
Patient Position:
Sitting, prone, or lateral decubitus with arm across chest.
*
Transducer Placement:
High-frequency linear array transducer placed over the scapular spine, scanning medially towards the suprascapular notch or laterally towards the spinoglenoid notch.
*
Identification:
At the suprascapular notch (inferior to the superior transverse scapular ligament), identify the suprascapular nerve as a hyperechoic structure often with the suprascapular artery and vein. At the spinoglenoid notch, the nerve can be identified exiting the notch, deep to the infraspinatus.
Technique:
1.
Skin Prep/Local Anesthetic:
Sterile prep, skin wheal.
2.
Needle Insertion:
A 22-gauge, 5-8 cm needle is advanced in-plane towards the identified nerve at either the suprascapular or spinoglenoid notch.
3.
Target Confirmation/Injection:
After negative aspiration, inject 5-10 mL of local anesthetic (e.g., bupivacaine 0.25% or ropivacaine 0.5%) around the nerve.
4. Pectoral Nerve Blocks (PECS I and PECS II)
These fascial plane blocks target the pectoral nerves and medial/lateral cutaneous branches of the intercostal nerves, providing analgesia to the anterior shoulder, chest wall, and sometimes the axilla. Useful for subpectoral biceps tenodesis, anterior capsule pain, or trauma.
Anatomy (Ultrasound-Guided):
*
Patient Position:
Supine, arm abducted to 90 degrees.
*
Transducer Placement:
High-frequency linear array transducer placed in the mid-clavicular line at the level of the 3rd or 4th rib, and scanned laterally towards the axilla.
*
Identification:
Identify the pectoralis major (PM) and pectoralis minor (PMin) muscles. For PECS II, visualize the serratus anterior (SA) muscle deep to PMin. Identify the thoracoacromial artery and sometimes the lateral pectoral nerve in the plane between PM and PMin.
Technique:
1.
Skin Prep/Local Anesthetic:
Sterile prep, skin wheal.
2.
PECS I Block:
*
Needle Insertion:
A 22-gauge, 5-8 cm needle is inserted in-plane, aiming for the fascial plane between PM and PMin.
*
Injection:
After negative aspiration, inject 10-15 mL of local anesthetic (e.g., ropivacaine 0.25%).
3.
PECS II Block:
*
Needle Insertion:
After performing PECS I, the needle is advanced further through PMin into the fascial plane between PMin and SA.
*
Injection:
After negative aspiration, inject an additional 10-15 mL of local anesthetic. This targets the medial/lateral pectoral nerves, as well as intercostal nerves (T2-T4) and the long thoracic nerve.
5. Multimodal Systemic Analgesia
Regional anesthesia forms the foundation but is complemented by systemic medications to provide comprehensive pain control and reduce opioid reliance.
- NSAIDs/COX-2 Inhibitors: (e.g., ibuprofen, celecoxib) Administered pre-operatively, intra-operatively (if allowed), and post-operatively. Contraindicated in patients with renal impairment, peptic ulcer disease, or significant cardiovascular risk. Caution in fracture healing.
- Acetaminophen: (e.g., Tylenol) Regular dosing provides opioid-sparing effects. Administered pre-operatively and post-operatively (oral or IV).
- Gabapentinoids: (gabapentin, pregabalin) Administered pre-operatively. Reduce post-operative pain intensity, opioid consumption, and opioid side effects. Start at lowest effective dose due to sedation risk.
- Corticosteroids: (e.g., dexamethasone) Single intra-operative dose can reduce post-operative nausea, vomiting, and pain, likely by reducing inflammation.
- Opioids: Reserved for breakthrough pain. Utilize short-acting opioids (e.g., oxycodone, hydromorphone) titrated to effect. Avoid long-acting opioids in the acute post-operative phase. Patient-controlled analgesia (PCA) can be useful for IV opioids.
- Ketamine: Low-dose ketamine infusion (subanesthetic doses) can be used intra-operatively and post-operatively for its NMDA receptor antagonism, reducing central sensitization and opioid requirements, especially in chronic pain patients or those on chronic opioids.
- Lidocaine Infusion: IV lidocaine can provide systemic analgesia, particularly for neuropathic pain components, and may reduce opioid consumption.
Combining these techniques provides a synergistic effect, optimizing analgesia while minimizing the individual side effects of each agent.
Complications & Management
Complications related to perioperative pain management, particularly regional anesthesia, can range from minor and self-limiting to severe and life-threatening. A thorough understanding of these risks and their management is crucial.
Complications of Regional Anesthesia
-
Phrenic Nerve Palsy:
- Incidence: Nearly 100% with interscalene block (ISB) due to the close proximity of the phrenic nerve to the brachial plexus in the interscalene groove.
- Clinical Presentation: Ipsilateral hemidiaphragmatic paresis, leading to a mild decrease in pulmonary function. Symptoms typically minimal in healthy patients but can be significant in those with severe pre-existing pulmonary disease (e.g., COPD, emphysema, restrictive lung disease).
- Management: Reassure patient. Monitor respiratory status. Avoid ISB in patients with severe respiratory compromise. Suprascapular or supraclavicular blocks are alternatives with lower phrenic nerve involvement.
-
Horner's Syndrome:
- Incidence: ~75% with ISB.
- Clinical Presentation: Unilateral ptosis, miosis, anhidrosis, and enophthalmos. Caused by local anesthetic spread to the stellate ganglion (part of the sympathetic chain).
- Management: Reassure patient. Self-resolves with block regression. No specific treatment required.
-
Recurrent Laryngeal Nerve Palsy:
- Incidence: 1-10% with ISB.
- Clinical Presentation: Hoarseness, dysphagia (rare). Caused by local anesthetic spread to the recurrent laryngeal nerve.
- Management: Reassure patient. Self-resolves with block regression.
-
Local Anesthetic Systemic Toxicity (LAST):
- Incidence: Rare (~0.01-0.03%), but potentially life-threatening.
- Clinical Presentation: Early signs (CNS): perioral numbness, metallic taste, tinnitus, lightheadedness, confusion, muscle twitching, seizures. Late signs (Cardiovascular): bradycardia, hypotension, ventricular arrhythmias, asystole.
- Management: Immediate cessation of local anesthetic injection. Call for help, initiate ACLS protocol. Administer 20% lipid emulsion (Intralipid) : bolus 1.5 mL/kg IV over 1 minute, then infusion 0.25 mL/kg/min (increase to 0.5 mL/kg/min if cardiovascular instability persists). Continue for at least 10 minutes after hemodynamic stability. Avoid vasopressin, calcium channel blockers, beta-blockers.
-
Nerve Injury:
- Incidence: Transient deficits up to 5-10%; permanent injury <0.01-0.02%.
- Clinical Presentation: Persistent numbness, weakness, pain beyond the expected duration of the block. Can be due to direct needle trauma, intraneural injection, hematoma compression, or local anesthetic neurotoxicity.
- Management: Meticulous technique, ultrasound guidance, avoid paresthesia on injection, negative aspiration. If injury suspected, neurological consultation, EMG/NCS, and symptomatic treatment.
-
Pneumothorax:
- Incidence: Rare with ISB, higher with supraclavicular (~0.5-1%).
- Clinical Presentation: Dyspnea, pleuritic chest pain, cough. May be delayed.
- Management: Careful ultrasound visualization of the pleura. If suspected, chest X-ray. Small pneumothorax may be observed. Larger or symptomatic pneumothorax requires chest tube insertion.
-
Infection:
- Incidence: Very rare (<0.01%), but serious.
- Clinical Presentation: Localized erythema, warmth, swelling, pain at injection site; fever, sepsis (if severe).
- Management: Strict aseptic technique. Antibiotics, possibly drainage.
-
Hematoma:
- Incidence: Increased risk in anticoagulated patients.
- Clinical Presentation: Swelling, ecchymosis, pain. Can compress nerves.
- Management: Hold anticoagulants per ASRA guidelines. Apply direct pressure. Surgical decompression if neurological deficit develops.
-
Vascular Puncture:
- Incidence: Can occur with any block.
- Clinical Presentation: Blood in syringe on aspiration (arterial or venous), rapid onset of LAST symptoms (if intravascular injection).
- Management: Withdraw needle, apply pressure. Re-evaluate needle position under ultrasound.
Complications of Poorly Managed Postoperative Pain
- Increased Opioid Consumption and Opioid-Related Side Effects: Nausea, vomiting, sedation, constipation, respiratory depression, pruritus.
- Prolonged Hospital Stay: Due to inadequate pain control and side effects.
- Impaired Rehabilitation: Patient reluctance to participate in physical therapy, leading to stiffness, muscle atrophy, and delayed functional recovery.
- Development of Chronic Postoperative Pain: Persistent pain beyond 3-6 months, potentially evolving into neuropathic pain or centralized pain syndromes.
- Patient Dissatisfaction: Affects overall patient experience and perception of care quality.
- Increased Risk of Postoperative Delirium/Cognitive Dysfunction: Especially in elderly patients.
Complications and Salvage Strategies Table
| Complication | Incidence (Approx.) | Etiology / Risk Factors | Clinical Presentation | Salvage Strategies / Management |
|---|---|---|---|---|
| Phrenic Nerve Palsy (ISB) | ~100% | Spread to phrenic nerve (C3-C5) in interscalene groove | Ipsilateral hemidiaphragmatic paresis, mild dyspnea. | Reassurance, monitor respiratory status. Avoid ISB in severe pulmonary disease. |
| Horner's Syndrome (ISB) | ~75% | Spread to stellate ganglion. | Ptosis, miosis, anhidrosis (ipsilateral). | Reassurance, self-resolves. |
| Recurrent Laryngeal Nerve Palsy (ISB) | 1-10% | Spread to recurrent laryngeal nerve. | Hoarseness, dysphagia. | Reassurance, self-resolves. |
| Local Anesthetic Systemic Toxicity (LAST) | 0.01-0.03% (severe) | Intravascular injection or rapid systemic absorption. | CNS (tinnitus, seizures), Cardiovascular (bradycardia, asystole). | Stop injection, ABCs, 20% Lipid Emulsion (Intralipid) , ACLS. |
| Nerve Injury | <0.01-0.02% (permanent) | Direct trauma, intraneural injection, compression, neurotoxicity. | Persistent numbness, weakness, pain beyond block duration. | Meticulous technique, ultrasound. Neurological consult, EMG/NCS, symptomatic treatment. |
| Pneumothorax (Supraclavicular) | ~0.5-1% | Needle puncture of pleura. | Dyspnea, pleuritic chest pain. | Ultrasound, Chest X-ray. Observation vs. Chest tube. |
| Infection | <0.01% | Breach of aseptic technique. | Local erythema, warmth, pain; fever, sepsis. | Strict aseptic technique. Antibiotics, drainage if abscess. |
| Hematoma | Variable | Vascular puncture, coagulopathy. | Swelling, ecchymosis. Can cause nerve compression. | Hold anticoagulants. Direct pressure. Surgical decompression if neurological deficit. |
| Failed Block / Inadequate Analgesia | 5-10% | Improper needle placement, insufficient dose, anatomical variation. | Persistent severe pain. | Re-evaluate, supplemental local infiltration, rescue opioid analgesia, consider alternative block or PNB. |
| Post-operative Nausea & Vomiting (PONV) | 20-30% | Opioids, anesthetic agents, patient factors. | Nausea, emesis. | Prophylactic antiemetics (dexamethasone, ondansetron). Reduce opioid use. |
| Opioid-Induced Pruritus | 10-20% | Opioid receptor activation in CNS. | Generalized itching. | Nalbuphine, diphenhydramine. Reduce opioid dose. |
| Urinary Retention | 5-10% | Opioids, surgical stress. | Inability to void. | Alpha-blockers (e.g., tamsulosin), bladder scan, catheterization. |
| Chronic Postoperative Pain | 5-10% | Inadequate acute pain control, central sensitization. | Pain persisting >3-6 months. | Multidisciplinary pain management, pain clinic referral, psychological support. |
Post-Operative Rehabilitation Protocols
Effective perioperative pain management is not merely about comfort but is a fundamental enabler of successful post-operative rehabilitation. The primary goal of pain control in the context of rehabilitation is to create a physiological environment where the patient can actively and comfortably participate in their prescribed physical therapy, thereby accelerating recovery and optimizing functional outcomes.
Early Mobilization and Pain Control Synergy:
- Pain as a Barrier: Uncontrolled pain leads to protective muscle guarding, apprehension, and an inability or unwillingness to move the affected limb. This directly impedes early range of motion (ROM) exercises and therapeutic strengthening.
-
Regional Anesthesia's Role:
Peripheral nerve blocks, particularly continuous catheters, provide targeted, prolonged analgesia that allows patients to overcome this barrier. The ability to perform exercises with reduced pain translates to:
- Improved ROM: Patients can tolerate passive, assisted, and active ROM within safe surgical limits, preventing stiffness and adhesive capsulitis.
- Enhanced Muscle Activation: Reduced pain facilitates neuromuscular control and early activation of rotator cuff and periscapular muscles, critical for stability and function.
- Reduced Inhibition: Lower pain levels decrease central sensitization and fear-avoidance behaviors, promoting a more positive and proactive engagement in therapy.
- Earlier Discharge: Patients who achieve functional milestones faster and manage pain effectively are candidates for earlier discharge from inpatient facilities or expedited progression in outpatient settings.
Phased Rehabilitation and Pain Management Considerations:
Rehabilitation protocols for shoulder surgery typically involve distinct phases, each with specific pain management implications.
Phase I: Immediate Post-Operative (0-6 weeks) – Protection & Early Motion
- Goals: Pain control, protect surgical repair, reduce swelling, initiate passive/assisted ROM.
-
Pain Management Focus:
- Continuous Peripheral Nerve Block (CPNB): If placed, the continuous infusion provides consistent baseline analgesia. Boluses can be administered before physical therapy sessions to maximize comfort during exercises.
- Transition to Oral Multimodal Analgesia: As the CPNB infusion rate is tapered or a single-shot block wears off, patients are transitioned to scheduled oral non-opioids (NSAIDs, acetaminophen, gabapentinoids) and short-acting opioids for breakthrough pain. This transition must be carefully managed to avoid abrupt pain escalation.
- Ice/Cryotherapy: Regular application to reduce swelling and pain, particularly effective for superficial incisional pain.
- Sling Management: Proper sling use is essential for protection but can sometimes contribute to discomfort or stiffness if not fitted correctly. Pain management should facilitate comfort even when in the sling.
- Rehabilitation Activities: Pendulum exercises, passive ROM (PROM) within protected arcs (e.g., flexion, external rotation for rotator cuff repair), scapular stabilization exercises. Pain control during these activities is paramount to achieve desired ROM and prevent guarding.
Phase II: Moderate Protection & Early Strengthening (6-12 weeks)
- Goals: Progress ROM, initiate active ROM (AROM), light isometric strengthening.
-
Pain Management Focus:
- Oral Multimodal Analgesia: Continue scheduled non-opioids. Opioid use should be significantly reduced or eliminated by this phase. Focus on managing activity-related pain.
- Topical Analgesics: Consider topical NSAIDs or capsaicin cream for localized pain.
- Education: Patients are educated on the difference between "good pain" (muscle soreness from exercise) and "bad pain" (sharp, joint pain) and how to manage activity levels.
- Rehabilitation Activities: AROM, light isometric exercises for rotator cuff and deltoid, progressive scapular strengthening. Good pain control allows for greater effort and consistency in these exercises.
Phase III: Minimum Protection & Advanced Strengthening (12-24 weeks)
- Goals: Full AROM, progressive strengthening, proprioception, return to functional activities.
-
Pain Management Focus:
- Opioid-Free: Patients should ideally be opioid-free. Pain should be managed with non-pharmacological methods and PRN non-opioids.
- Focus on Function: Pain management at this stage often revolves around mitigating exercise-induced soreness or minor discomfort as strength and endurance improve.
- Rehabilitation Activities: Resistance training, eccentric exercises, plyometrics (if appropriate), sport-specific training.
Measuring Rehabilitation Progress and Pain:
- Visual Analog Scale (VAS) or Numeric Rating Scale (NRS): Regularly used to quantify pain levels at rest and with activity.
- Functional Outcome Scores: Objective measures like the American Shoulder and Elbow Surgeons (ASES) score, Constant-Murley score, or Western Ontario Rotator Cuff Index (WORC) correlate improved function with effective pain management.
- Range of Motion (ROM) Assessment: Objective measurement of joint mobility.
- Patient Diaries: Encouraging patients to track pain levels, medication use, and rehabilitation participation can provide valuable insights for adjustment of pain management strategies.
In summary, perioperative pain management is an integral component of the rehabilitation process for shoulder surgery. By optimizing pain control through regional anesthesia and a multimodal approach, clinicians enable patients to engage fully in their recovery, thereby enhancing their functional return and overall satisfaction.
Summary of Key Literature / Guidelines
The landscape of perioperative pain management for shoulder surgery has been significantly shaped by extensive research, leading to robust evidence-based guidelines. The shift towards opioid-sparing strategies and enhanced recovery after surgery (ERAS) protocols underscores the critical role of regional anesthesia and multimodal analgesia.
Key Evidence and Literature Highlights:
-
Superiority of Regional Anesthesia: Numerous randomized controlled trials (RCTs) and meta-analyses consistently demonstrate the superiority of brachial plexus blocks (particularly interscalene blocks) compared to systemic analgesia alone for shoulder surgery.
- Reduced Pain Scores: Patients receiving ISB report significantly lower pain scores in the immediate post-operative period (0-24 hours).
- Decreased Opioid Consumption: PNBs drastically reduce opioid requirements, minimizing associated side effects. Studies consistently show reductions of 50-70% in opioid consumption.
- Improved Patient Satisfaction: Better pain control leads to higher patient satisfaction with their surgical experience.
- Shorter Length of Stay: Facilitation of early discharge, particularly for outpatient procedures.
- Faster Functional Recovery: By allowing earlier and more comfortable participation in rehabilitation.
-
Continuous Peripheral Nerve Blocks (CPNBs): For major shoulder surgeries such as total shoulder arthroplasty, continuous interscalene catheters provide prolonged analgesia (up to 2-3 days), which is particularly beneficial.
- Benefits: Sustain opioid-sparing effects, improve sleep quality, and facilitate early physical therapy engagement in the immediate post-operative phase.
- Challenges: Catheter dislodgement, infection risk (low), and management of home infusions.
- Evidence: Meta-analyses support CPNBs over single-shot blocks for prolonged analgesia and reduced opioid consumption following major orthopedic surgery.
-
Multimodal Analgesia: The synergistic use of various analgesic agents with different mechanisms of action is the cornerstone of modern perioperative pain management.
- NSAIDs/COX-2 Inhibitors: Effective for inflammatory pain. Concerns exist regarding fracture healing (especially non-selective NSAIDs) and cardiovascular/renal risks, but short-term use is generally safe and beneficial.
- Acetaminophen: Provides baseline analgesia and opioid-sparing effects.
- Gabapentinoids (Gabapentin/Pregabalin): Reduce acute and chronic post-operative pain, decrease opioid consumption, and mitigate opioid-related side effects. Evidence supports their use as part of a multimodal regimen, particularly for shoulder arthroplasty.
- Corticosteroids (Dexamethasone): Single intra-operative dose has anti-inflammatory and antiemetic properties, contributing to overall pain and PONV reduction.
- Local Infiltration Analgesia: Direct infiltration of local anesthetic into the surgical site, capsule, and surrounding tissues can complement regional blocks, particularly for the superficial cervical plexus sensory distribution often missed by ISB.
-
Role of Ultrasound Guidance: The widespread adoption of ultrasound guidance has revolutionized regional anesthesia.
- Improved Safety: Direct visualization of nerves, vessels, and surrounding structures significantly reduces the risk of nerve injury, vascular puncture, and pneumothorax.
- Increased Efficacy: Precise needle placement leads to more reliable and effective blocks.
- Faster Onset: Optimized local anesthetic spread.
-
Complication Management: Literature emphasizes prompt recognition and management of complications like Local Anesthetic Systemic Toxicity (LAST) with lipid emulsion, and careful patient selection for blocks to minimize risks like phrenic nerve palsy in susceptible individuals.
Key Guidelines and Recommendations:
-
American Society of Regional Anesthesia and Pain Medicine (ASRA) Guidelines:
- Provide comprehensive recommendations on the safe practice of regional anesthesia, including anticoagulant management for neuraxial and peripheral nerve blocks.
- Emphasize the use of ultrasound guidance for PNBs.
- Detail the recognition and management of LAST.
-
Enhanced Recovery After Surgery (ERAS) Society Guidelines:
- ERAS protocols for shoulder surgery strongly advocate for multimodal analgesia with a central role for regional anesthesia.
- Recommendations include pre-emptive analgesia, opioid-sparing regimens, and early mobilization facilitated by effective pain control.
-
American Academy of Orthopaedic Surgeons (AAOS) Clinical Practice Guidelines:
- While not always exclusively focused on pain, these guidelines often implicitly support optimal pain management to achieve the functional outcomes they recommend for various shoulder procedures.
-
Consensus Statements on Interscalene Block Alternatives:
- Recent literature explores alternatives or adjuncts to ISB to mitigate phrenic nerve palsy (e.g., lower volume ISB, supraclavicular blocks, suprascapular nerve blocks, combined SSNB with superficial cervical plexus block), particularly for patients with compromised pulmonary function or for more distal shoulder procedures where full brachial plexus blockade may not be necessary.
In conclusion, the current paradigm for perioperative pain management in shoulder surgery integrates a multimodal approach, with ultrasound-guided regional anesthesia as a cornerstone. This strategy aims to provide superior analgesia, minimize opioid-related morbidity, accelerate rehabilitation, and ultimately enhance patient satisfaction and functional recovery. Continuous adherence to established guidelines and a commitment to evidence-based practice are essential for optimizing patient care.
Clinical & Radiographic Imaging

You Might Also Like