Lateral Condyle Fractures: Optimize ORIF to Prevent Tardy Ulnar Nerve
Key Takeaway
For anyone wondering about Lateral Condyle Fractures: Optimize ORIF to Prevent Tardy Ulnar Nerve, Lateral condyle fractures are breaks in the distal humerus, frequently seen in children, involving the metaphysis, physis, or articular surface. Classified by Milch types and displacement, these fractures require careful management. Long-term complications, such as progressive valgus deformity, can potentially result in a **tardy ulnar nerve** palsy, impacting elbow function years after the initial injury.
Introduction and Epidemiology of Lateral Condyle Fractures
Lateral condyle fractures of the distal humerus represent a critical diagnostic and therapeutic challenge in pediatric orthopedic traumatology. These injuries involve the lateral aspect of the distal humerus, encompassing varying degrees of the metaphysis, physis, epiphysis, and articular surface. Constituting approximately 10% to 15% of all pediatric elbow fractures, they are second in frequency only to supracondylar fractures of the distal humerus. The peak incidence occurs in children between the ages of four and ten years, corresponding to a period of rapid physeal growth and evolving ossification centers.

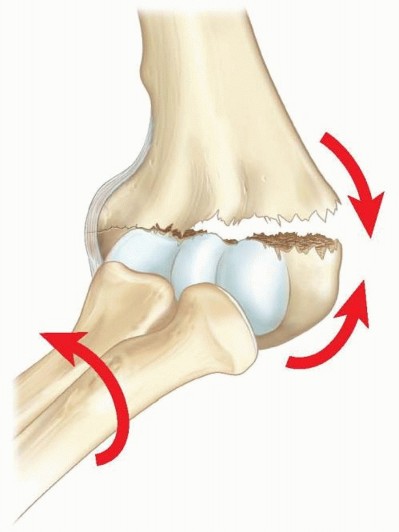
The clinical significance of lateral condyle fractures lies in their inherent instability and their propensity for profound long-term complications if mismanaged. Unlike supracondylar fractures, which are predominantly extra-articular, lateral condyle fractures are intra-articular injuries driven by both shear forces and the continuous traction of the common extensor origin. Nondisplaced fractures may hinge entirely on the intact articular cartilage, rendering them deceptively stable on initial radiographic evaluation. However, failure to recognize occult displacement or adequately stabilize these fractures leads directly to nonunion, progressive cubitus valgus, and the classic delayed presentation of tardy ulnar nerve palsy.
Pathogenesis and Mechanism of Injury
The typical mechanism of injury involves a fall on an outstretched hand, generating complex forces across the pediatric elbow. Two primary biomechanical theories explain the propagation of the fracture line. The first describes an avulsion mechanism resulting from sudden adduction of a supinated forearm with the elbow in extension. This places immense tension on the lateral collateral ligament complex and the extensor musculature, avulsing the lateral condyle.

The second theory posits a push-off mechanism, wherein an axial load transmitted through the forearm is combined with a valgus force. The radial head impacts the capitellum, driving a shear fracture through the lateral condyle. While lateral condyle fractures typically present as isolated injuries, high-energy variants may be associated with elbow joint subluxation, radial head fractures, or olecranon fractures. Understanding these deforming forces is critical for the orthopedic surgeon, as the same muscular and ligamentous forces that cause the fracture will continuously act to displace it prior to definitive union.
Surgical Anatomy and Biomechanics of the Pediatric Elbow
A profound understanding of the developmental anatomy of the distal humerus is requisite for optimizing open reduction and internal fixation. Proximally, lateral condyle fractures almost universally include a portion of the posterolateral metaphysis. The fracture line then propagates distally along the physis before exiting either through the ossification center of the capitellum or medial to it, through the unossified trochlear groove.
Osteology and Physis Development
The articular cartilage of the distal humerus may or may not be violated depending on the fracture pattern. The capitellum is the first secondary ossification center to appear in the distal humerus, typically visible radiographically between six months and two years of age. The trochlea, however, remains largely cartilaginous until eight to ten years of age. This discrepancy in ossification complicates radiographic interpretation, as a significant portion of the fracture line traverses radiolucent cartilage.

The vascular supply to the lateral condyle is tenuous and highly specific. The primary arterial inflow enters posteriorly via branches of the radial collateral artery and the posterior descending branch of the profunda brachii. These vessels penetrate the non-articular posterior surface of the lateral condyle. Consequently, posterior soft tissue dissection during surgical approaches must be strictly avoided to prevent iatrogenic avascular necrosis of the capitellum.

Ligamentous and Muscular Attachments
The distal fracture fragment serves as the origin for the extensor carpi radialis longus, extensor carpi radialis brevis, and the supinator muscles. Furthermore, the lateral collateral ligament complex remains firmly attached to this fragment. In the setting of a displaced fracture, the anterior and posterior portions of the elbow joint capsule are typically torn. The continuous resting tone of the extensor musculature exerts a distal, posterior, and rotatory force on the lateral condyle fragment, explaining the high rate of secondary displacement in fractures treated non-operatively.
Classification Systems for Lateral Condyle Fractures
Classification systems guide both treatment algorithms and prognostic expectations. The Milch classification categorizes fractures based on the anatomic location where the fracture line exits distally.

Milch type I fractures traverse the metaphysis and physis, extending directly across the ossification center of the capitellum. This pattern is less common and is biomechanically analogous to a Salter-Harris type IV fracture. Because the fracture line exits lateral to the trochlear groove, the radioulnar articulation remains structurally sound, conferring relative lateral stability to the elbow.
Milch type II fractures are significantly more common and inherently more unstable. The fracture extends from the metaphysis, tracks along the physis, and exits through the unossified trochlea, medial to the capitellar ossification center. Radiographically, this may mimic a Salter-Harris type II injury due to the unossified nature of the trochlea, but functionally and anatomically, it involves the articular cartilage and behaves as a Salter-Harris type IV injury. Crucially, the fracture line disrupts the trochlear crista. Loss of this stabilizing ridge allows lateral translation and subluxation of the radius and ulna, drastically increasing the instability of the joint.

A secondary numeric classification system focuses on the degree of displacement, which directly dictates the necessity for operative intervention:
* Stage I: Fractures involve the metaphysis and physis without violating the articular cartilage hinge. Displacement is minimal.
* Stage II: Fractures cross the articular surface completely but exhibit minimal displacement (typically defined as less than 2 mm).
* Stage III: Completely displaced fractures crossing the metaphysis, physis, and articular surface. The continuous pull of the extensor mass frequently results in severe rotation of the distal fragment, necessitating open reduction.
Indications and Contraindications for Surgical Intervention
The overarching goal of managing lateral condyle fractures is the restoration of articular congruity and physeal alignment to prevent the cascade of nonunion, deformity, and neurologic compromise. The natural history of these injuries is highly dependent on initial displacement and the biological viability of the physis.

While completely nondisplaced fractures with an intact articular hinge may heal with cast immobilization, the risk of delayed union or secondary displacement remains significant. This is largely due to the fracture being bathed in synovial fluid, which contains fibrinolytic enzymes that inhibit hematoma consolidation, combined with the relatively poor metaphyseal circulation and the constant deforming forces of the extensor musculature.
Operative vs Non Operative Management Criteria
| Clinical Scenario | Treatment Modality | Rationale and Biomechanical Considerations |
|---|---|---|
| Nondisplaced (<2 mm) on all views | Non-Operative (Long Arm Cast) | Articular hinge is intact. Requires strict weekly radiographic surveillance to detect delayed displacement. |
| Displaced (2 mm or greater) | Operative (CRPP or ORIF) | Disruption of articular cartilage. High risk of nonunion and cubitus valgus if left unreduced. |
| Rotated Fragment (Stage III) | Operative (ORIF) | Closed reduction is nearly impossible due to extensor muscle pull. Direct visualization required for articular reduction. |
| Delayed Presentation (>3 weeks) | Operative (In Situ Fixation vs ORIF) | High risk of AVN with aggressive late mobilization. Management depends on functional deficit and degree of union. |
| Associated Elbow Dislocation | Operative (ORIF) | Severe soft tissue disruption. Requires rigid internal fixation to restore joint stability and permit early motion. |
Contraindications to immediate open reduction are exceedingly rare but may include severe, life-threatening polytrauma taking precedence, or active local soft tissue infection. In cases of delayed presentation (greater than three to four weeks), aggressive open reduction and extensive soft tissue stripping carry a prohibitively high risk of avascular necrosis. In such late presentations, in situ fixation or acceptance of the nonunion with planned late reconstructive osteotomy may be indicated.
Pre Operative Planning and Patient Positioning
Meticulous preoperative planning is essential to ensure a successful surgical outcome and mitigate the risk of late complications. The diagnostic workup relies heavily on high-quality orthogonal radiography.
Imaging Modalities and Templating
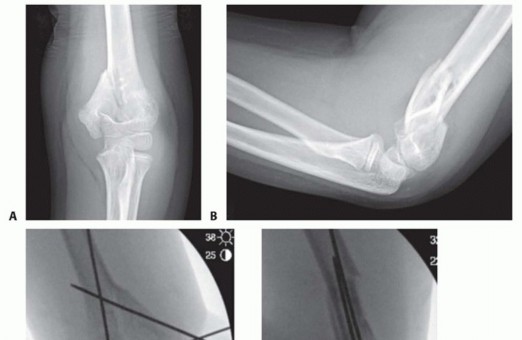
Standard anteroposterior and lateral radiographs of the elbow are mandatory. However, the true degree of displacement is frequently underestimated on standard views due to the posterolateral trajectory of the fracture fragment. An internal oblique radiograph is critical, as it profiles the lateral condyle and often reveals the maximum extent of the metaphyseal gap.

In cases where the articular congruity is questionable or the fracture pattern is obscured by unossified cartilage, advanced imaging may be utilized. Magnetic Resonance Imaging provides excellent visualization of the cartilaginous anlage and the exact exit point of the fracture line. Alternatively, intraoperative arthrography can be an invaluable, cost-effective tool to dynamically assess the integrity of the articular surface under fluoroscopy before committing to an open versus closed procedure.

Operating Room Setup and Anesthesia
The procedure is performed under general anesthesia. The patient is positioned supine on the operating table with the affected extremity extended onto a radiolucent hand table. A non-sterile tourniquet is applied high on the brachium to ensure a bloodless surgical field, which is vital for identifying the cartilaginous articular surface during open reduction.

The C-arm fluoroscopy unit should be positioned parallel to the operating table, entering from the head or the foot, allowing for unobstructed orthogonal imaging without compromising the sterile field. The surgeon typically sits in the axilla, providing direct access to the lateral aspect of the elbow.
Detailed Surgical Approach and Technique
The transition from a displaced lateral condyle fracture to a tardy ulnar nerve palsy is paved by inadequate surgical technique. The primary objective of the operation is absolute anatomic reduction of the articular surface, followed by stable internal fixation that resists the deforming forces of the extensor musculature.
Surgical Approach to the Lateral Elbow
A direct lateral approach is utilized. The incision is centered over the lateral epicondyle, extending proximally along the lateral supracondylar ridge and distally toward the radial head.

Following skin incision and subcutaneous dissection, the deep fascia is incised. The internervous plane between the anconeus (innervated by the radial nerve) and the extensor carpi ulnaris (innervated by the posterior interosseous nerve) can be utilized, though frequently the trauma itself has created a traumatic window directly to the fracture site.

The critical principle during dissection is the absolute preservation of the posterior soft tissue attachments. The posterior aspect of the lateral condyle harbors the tenuous vascular supply to the capitellum. Stripping this tissue will precipitate avascular necrosis. Dissection should be strictly limited to the anterior aspect of the fracture, exposing the joint capsule anteriorly to visualize the articular surface. The hematoma is meticulously evacuated using suction and gentle curettage to allow clear visualization of the fracture margins.
Fracture Reduction Strategies
Reduction of a displaced, rotated lateral condyle fracture requires a combination of precise manual manipulation and direct visualization. The forearm is typically pronated; this maneuver relaxes the extensor musculature that originates on the lateral epicondyle, thereby reducing the displacing tension on the fragment.

A small dental pick or a single-pronged skin hook can be carefully inserted into the metaphyseal portion of the fragment to derotate and manipulate it into position. The surgeon must directly visualize the articular cartilage anteriorly to confirm an anatomic reduction. Relying solely on the metaphyseal read is a recognized pitfall, as metaphyseal comminution can mask articular malreduction. Once anatomic reduction is achieved visually, it is provisionally held with a dental pick or a small provisional K-wire and confirmed via intraoperative fluoroscopy.
Internal Fixation Techniques
Rigid internal fixation is paramount to counteract the synovial fluid environment and muscular forces. Fixation is most commonly achieved using two or three smooth Kirschner wires (typically 0.062-inch or 1.6 mm in diameter, depending on the child's size).

The trajectory of the wires is critical for biomechanical stability. The wires should be divergent to maximize pull-out strength and rotational control. The first wire is typically directed from the lateral epicondyle, aiming proximally and medially to engage the intact medial cortex of the distal humerus. The second wire is driven more anteriorly or posteriorly, maintaining a divergent path while avoiding the olecranon fossa.

In older children or adolescents with substantial metaphyseal fragments, a partially threaded cancellous screw may be utilized for interfragmentary compression. However, screws must never cross the open physis. For the vast majority of pediatric patients, smooth K-wires provide excellent stability while minimizing iatrogenic physeal injury. The pins are typically bent and left protruding through the skin to facilitate removal in the clinic, though some surgeons prefer to bury them beneath the skin to reduce pin tract infection rates.
Complications and Management Strategies
The complication profile of lateral condyle fractures is severe and unforgiving. The primary focus of the orthopedic surgeon must be the prevention of nonunion, which is the direct precursor to progressive deformity and neurologic compromise.
Nonunion and Cubitus Valgus
Nonunion of the lateral condyle is a catastrophic complication stemming from inadequate initial immobilization, failure to recognize displacement, or technically deficient internal fixation. The continuous pull of the extensor mass, combined with the fibrinolytic synovial environment, prevents bridging callus formation.

As the nonunion persists, the lateral column of the distal humerus fails to grow synchronously with the medial column. Furthermore, the ununited fragment migrates proximally. This structural collapse leads to a progressive cubitus valgus deformity. The carrying angle of the elbow increases significantly, altering the biomechanics of the entire upper extremity.

The most insidious consequence of this progressive cubitus valgus is tardy ulnar nerve palsy. As the valgus angle increases, the ulnar nerve is subjected to chronic traction and friction as it traverses the cubital tunnel behind the medial epicondyle. This traction neuropathy can present years or even decades after the initial injury. Patients typically report insidious onset of paresthesias in the ring and small fingers, intrinsic muscle weakness, and eventual clawing of the hand.

Management of tardy ulnar nerve palsy requires surgical intervention. The primary procedure is an anterior transposition of the ulnar nerve (either subcutaneous or submuscular) to relieve the tension across the medial epicondyle. However, addressing the nerve alone is often insufficient. A corrective medial closing wedge or lateral opening wedge supracondylar osteotomy of the humerus is frequently required to correct the mechanical axis and prevent recurrent neuropathy.
Complication Rates and Salvage Procedures
| Complication | Pathophysiology | Prevention and Management |
|---|---|---|
| Lateral Spurring | Periosteal elevation and new bone formation laterally. Very common. | Usually asymptomatic and remodels over time. Excision rarely indicated unless mechanically symptomatic. |
| Nonunion | Inadequate fixation, synovial fluid bathing, poor vascularity. | Optimize initial ORIF. Late management: In situ fixation with bone grafting vs corrective osteotomy. |
| Cubitus Valgus | Lateral column collapse secondary to nonunion or premature physeal closure. | Anatomic reduction. Late management requires corrective supracondylar osteotomy. |
| Tardy Ulnar Nerve Palsy | Chronic traction neuropathy due to progressive cubitus valgus. | Prevent nonunion. Late management: Anterior ulnar nerve transposition and corrective osteotomy. |
| Avascular Necrosis | Iatrogenic disruption of posterior blood supply during surgical approach. | Strictly avoid posterior dissection. Maintain anterior exposure only. |
Post Operative Rehabilitation Protocols
Following definitive fixation, the extremity is immobilized to protect the repair and facilitate osseous union. The elbow is placed in a well-padded long arm cast or posterior splint with the elbow at 90 degrees of flexion and the forearm in neutral rotation.

Immobilization and Mobilization Phases
Clinical and radiographic follow-up is typically performed at one week to ensure maintenance of reduction and assess for pin tract erythema. The total duration of immobilization is generally three to four weeks. At this juncture, radiographs are obtained out of cast to evaluate for bridging callus across the metaphyseal fracture line.

Once early union is confirmed, the Kirschner wires are removed in the outpatient clinic. The patient is then transitioned to a removable splint and initiated on an active range of motion protocol. Passive stretching and forceful manipulation by physical therapists must be strictly avoided in the pediatric elbow, as this significantly increases the risk of heterotopic ossification and myositis ossificans, which can severely limit terminal motion. Most children will regain functional range of motion through normal daily activities and play within six to twelve weeks following pin removal.
Summary of Key Literature and Guidelines
The management of lateral condyle fractures has evolved, but the core tenet of achieving absolute articular congruity remains unchallenged in the orthopedic literature.

Evidence Based Best Practices
Contemporary literature emphasizes the utility of the internal oblique radiograph for accurate classification of displacement. Studies by Song et al. have demonstrated that fractures appearing minimally displaced on standard AP views frequently show significant gapping on internal oblique projections, necessitating a shift from non-operative to operative management.
Furthermore, long-term outcome studies, such as those utilizing Flynn's criteria, consistently demonstrate that functional and cosmetic outcomes are directly correlated with the quality of the initial articular reduction. The literature unequivocally supports aggressive open reduction and internal fixation for fractures displaced greater than 2 mm, as the morbidity of surgery is vastly outweighed by the catastrophic sequelae of nonunion, cubitus valgus, and the subsequent development of tardy ulnar nerve palsy. The orthopedic surgeon must approach these fractures with a high index of suspicion, meticulous surgical technique, and a profound respect for the delicate vascular anatomy of the pediatric elbow.
Clinical & Radiographic Imaging





















You Might Also Like