Proximal Femoral Atypical Cartilaginous Tumor/Low-Grade Chondrosarcoma: A Diagnostic Imaging Case Study
Key Takeaway
Proximal femoral chondrosarcoma diagnosis integrates patient history, physical exam, and key imaging. Radiographs, CT, and MRI reveal characteristic expansile intramedullary lytic lesions with "rings and arcs" calcifications and significant endosteal scalloping, crucial for identifying this low-grade cartilaginous tumor.
Patient Presentation & History
A 62-year-old male presented to the orthopedic oncology clinic with an 8-month history of a dull, aching pain localized to his left hip and proximal thigh. The pain was insidious in onset, progressively worsened over time, and was particularly noticeable at night, disrupting his sleep. He reported no specific traumatic event, though he occasionally experienced a "catching" sensation with certain movements. The pain had become debilitating, significantly impacting his ambulation and activities of daily living. He denied any fever, chills, night sweats, or unexplained weight loss.
His past medical history was significant for well-controlled hypertension and hyperlipidemia. He was a non-smoker and consumed alcohol socially. He had no personal or family history of malignancies or genetic syndromes predisposing to cartilage tumors such as Ollier's disease or Maffucci syndrome. His primary occupation was a retired accountant, and he lived independently.
Clinical Examination
On inspection of the left hip and thigh, there was subtle fullness over the greater trochanteric region, with no obvious skin changes, erythema, or edema. No limb length discrepancy was appreciated. Gait analysis revealed an antalgic gait with a Trendelenburg sign on the left side, indicative of abductor insufficiency or pain.
Palpation revealed deep tenderness over the lateral aspect of the proximal femur and greater trochanter. A firm, non-mobile, mildly tender mass was palpable in the proximal vastus lateralis region, approximately 5 x 4 cm in size, fixed to the underlying bone. There was no palpable warmth.
Range of motion of the left hip was restricted and painful, particularly with internal rotation and abduction. Flexion was limited to 90 degrees, extension to 0 degrees, abduction to 20 degrees (compared to 45 degrees on the contralateral side), adduction to 15 degrees, internal rotation to 10 degrees, and external rotation to 25 degrees. Pain was reproduced at the end ranges of motion.
Neurological examination of the left lower extremity revealed intact motor strength (5/5 in all major muscle groups), normal sensation to light touch and pinprick in all dermatomes, and symmetrical deep tendon reflexes (2+ patellar and Achilles reflexes bilaterally). Distal pulses (femoral, popliteal, dorsalis pedis, posterior tibial) were all palpable and symmetrical bilaterally. There were no signs of vascular compromise.
Imaging & Diagnostics
Initial diagnostic workup commenced with plain radiographs of the left hip and femur.
Radiographs
Anteroposterior and lateral radiographs of the left proximal femur were obtained. These revealed an expansile, intramedullary lytic lesion originating in the subtrochanteric region and extending proximally into the intertrochanteric area. Characteristic findings included a lobulated appearance with "rings and arcs" type calcifications within the matrix, suggestive of cartilage formation. Significant endosteal scalloping, exceeding two-thirds of the cortical thickness, was evident, along with focal cortical thickening and remodeling. There was no obvious periosteal reaction or overt cortical destruction at this stage.

Figure 1: Anteroposterior radiograph of the left proximal femur demonstrating a lobulated, expansile intramedullary lesion with internal "rings and arcs" calcifications and significant endosteal scalloping.
Computed Tomography (CT)
A CT scan of the left hip and pelvis was subsequently performed to further characterize the lesion's morphology, cortical integrity, and medullary extent. The CT provided superior detail regarding the calcified matrix and the extent of endosteal erosion, confirming significant scalloping of the inner cortex (approximately 75-80% of cortical thickness) over an extended segment. It precisely delineated the intramedullary extent, demonstrating involvement from the lesser trochanter proximally to approximately 8 cm distal to the lesser trochanter. The calcified chondroid matrix was clearly visible, and there was no definitive evidence of cortical breach or significant extraosseous soft tissue mass, which suggested a lower-grade lesion, although high-grade components can sometimes be focal. The CT also assisted in pre-operative templating for potential reconstructive options.

Figure 2: Coronal and axial CT images of the proximal femur showing the intramedullary lesion with extensive endosteal scalloping, characteristic internal calcifications, and precise delineation of cortical integrity.
Magnetic Resonance Imaging (MRI)
An MRI of the left femur was indispensable for assessing the true extent of marrow involvement, identifying potential skip lesions, evaluating the relationship to neurovascular structures, and characterizing the soft tissue component more thoroughly. The MRI demonstrated a lobulated mass that was T1 hypointense and markedly T2 hyperintense, typical of hyaline cartilage. Internal septations showed moderate enhancement post-contrast. There was no definitive evidence of neurovascular encasement. Importantly, the MRI confirmed the absence of skip lesions within the femur and delineated the proximal and distal extent of marrow involvement, crucial for surgical margin planning. The high T2 signal suggested a cartilaginous lesion, and the aggressive endosteal scalloping and focal areas of cortical remodeling were concerning for an atypical cartilaginous tumor (ACT) or low-grade chondrosarcoma.

Figure 3: Axial T2-weighted MRI image demonstrating a lobulated, hyperintense lesion within the proximal femur, characteristic of a cartilaginous tumor, with clear delineation of marrow involvement.
Systemic Staging
A CT scan of the chest, abdomen, and pelvis was performed for systemic staging, revealing no evidence of metastatic disease. A bone scintigraphy (Tc-99m MDP bone scan) was also performed to rule out other osseous lesions or metastases, which was negative for additional uptake beyond the primary lesion.
Biopsy
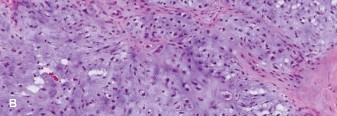
Given the strong suspicion for a cartilaginous tumor, an image-guided core needle biopsy was performed. The biopsy was planned carefully to ensure the biopsy tract could be incorporated into the definitive surgical resection to prevent tumor cell seeding. Histopathological analysis revealed a cartilaginous neoplasm characterized by increased cellularity, nuclear atypia, and occasional binucleated chondrocytes within a hyaline matrix. Mitotic figures were rare. Based on these features, the pathology was consistent with a Grade II Chondrosarcoma .
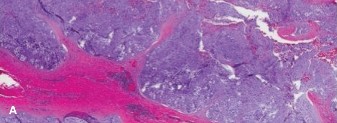
Figure 4: Histopathology slide demonstrating features of Grade II Chondrosarcoma, including increased cellularity, nuclear atypia, and binucleation within a cartilaginous matrix (Hematoxylin and Eosin stain).
Differential Diagnosis
The differential diagnosis for a lytic, calcified lesion in the proximal femur, particularly in an older adult, is broad and includes both benign and malignant entities. Key differentials considered in this case include:
| Feature | Chondrosarcoma (Primary) | Enchondroma (Atypical Cartilaginous Tumor) | Metastatic Carcinoma | Giant Cell Tumor |
|---|---|---|---|---|
| Demographics | 40-70 years, M>F | Any age, often 20-50, no sex predilection | >50 years, M=F, depends on primary origin | 20-40 years, F>M |
| Location | Pelvis, proximal femur, humerus, ribs (central/appendicular skeleton common) | Small bones of hands/feet, long bone metaphysis/diaphysis (often incidental) | Any bone, often axial skeleton, long bone metaphysis | Epiphyseal/Metaphyseal (often subarticular) of long bones (e.g., distal femur, proximal tibia, distal radius) |
| Pain | Persistent, dull ache, progressive, often nocturnal, unrelated to activity | Usually asymptomatic, incidental finding; pain if pathological fracture or malignant transformation (ACT/G1 CS) | Constant, deep ache, progressive, often nocturnal, weight-bearing exacerbates | Localized, progressive pain, joint effusion, restricted ROM |
| Radiographs | Intramedullary lytic lesion, endosteal scalloping >2/3 cortical thickness, "rings and arcs" calcification, cortical destruction/thickening, soft tissue mass (high grade) | Well-circumscribed lytic lesion, often central, punctate calcifications, no endosteal scalloping or minimal <2/3, quiescent | Lytic or blastic, "moth-eaten" or permeative pattern, cortical destruction, pathological fracture common | Purely lytic, eccentric, expansile, non-sclerotic margin, no calcifications, "soap bubble" appearance |
| CT Findings | Cortical erosion, medullary extension, calcified matrix, precise scalloping | Intra-lesional calcifications, no cortical destruction, stable | Cortical breach, soft tissue component, often multifocal, destructive | Thin, remodeled cortex, often extends to subarticular bone, no calcification |
| MRI Findings | Lobulated T2 hyperintensity, internal septations, bone marrow edema, extra-osseous extension, signal heterogeneity for higher grades | T2 hyperintensity, often quiescent, lack of significant edema or cortical involvement, stable over time | Heterogeneous signal, T1 hypointense, T2 variable, soft tissue mass, marrow replacement, often widespread | T1 hypointense, T2 heterogeneous (fluid levels common), hemosiderin deposits, often large soft tissue component |
| Biopsy/Histology | Cartilage matrix, increased cellularity, nuclear atypia, binucleation, mitotic figures (grade dependent). Myxoid change. | Mature hyaline cartilage, low cellularity, no atypia, no mitoses, quiescent nuclei. Stable over time. | Atypical epithelial cells, specific to primary (e.g., adenocarcinoma, squamous cell carcinoma) | Uniform ovoid mononuclear stromal cells, numerous multinucleated osteoclast-like giant cells |
| Treatment | En bloc wide surgical resection | Observation for asymptomatic; curettage for symptomatic or ACTs | Depends on primary, often palliative radiotherapy, chemotherapy, or stabilization | Extended curettage with adjuvant (e.g., phenol, cryotherapy, argon beam), or en bloc resection for recurrence/aggressive |
Surgical Decision Making & Classification
The definitive diagnosis of Grade II Chondrosarcoma necessitated aggressive surgical intervention. Chondrosarcomas are notoriously resistant to both chemotherapy and radiation therapy, making wide en bloc surgical resection the cornerstone of treatment for local control and prevention of recurrence.
Enneking Staging System
Based on the comprehensive imaging and biopsy results, this case was classified according to the Enneking Staging System for Musculoskeletal Sarcoma:
*
Grade (G)
: G2 (High-grade, as Grade II is considered intermediate to high grade in many contexts, requiring similar aggressive surgical management as high-grade tumors due to recurrence risk).
*
Site (T)
: T1 (Intraosseous, as the tumor was contained within the cortex without significant extraosseous extension, though with extensive endosteal scalloping).
*
Metastases (M)
: M0 (No regional or distant metastases).
Therefore, the tumor was staged as Stage IIA , indicating a high-grade tumor confined within the cortical bone. This staging mandates a wide surgical margin.
Operative vs. Non-operative Decision
For symptomatic, biopsy-proven chondrosarcoma of Grade II or higher, operative management is universally indicated. Non-operative management is generally reserved for asymptomatic, incidental atypical cartilaginous tumors (ACTs) or Grade I enchondromas, which are managed with close radiological surveillance. The patient's progressive pain, functional limitation, and the Grade II diagnosis firmly dictated the need for surgical resection.
Pre-operative Planning
A multidisciplinary tumor board convened, comprising orthopedic oncologists, musculoskeletal radiologists, pathologists, and medical oncologists. The consensus was for wide en bloc resection of the proximal femur, followed by reconstruction.
Key aspects of pre-operative planning included:
1.
Oncologic Margins
: Based on MRI and CT findings, osteotomy levels were planned proximally through the femoral neck and distally approximately 10 cm beyond the visualized tumor on MRI, ensuring a minimum 2 cm clear margin in bone.
2.
Reconstruction Options
: Given the substantial bone defect (approximately 18-20 cm segment of the proximal femur, including the femoral head/neck, greater and lesser trochanters, and a significant portion of the subtrochanteric region), a proximal femoral replacement endoprosthesis was deemed the most appropriate reconstructive choice. Alternatives like allograft-prosthesis composites or custom prostheses were considered but the modular endoprosthesis offered advantages in terms of availability, durability, and restoration of function.
3.
Soft Tissue Considerations
: The planned resection would necessitate meticulous dissection to preserve the femoral neurovascular bundle and reattachment of critical abductor and adductor musculature post-resection.
4.
Blood Loss Management
: Anticipating significant blood loss, pre-operative autologous blood donation and availability of banked blood products were arranged.
Surgical Technique / Intervention
Patient Positioning and Approach
The patient was positioned in a lateral decubitus position on a radiolucent operating table, allowing for optimal access to the lateral aspect of the hip and thigh. The limb was prepped and draped in a sterile fashion from the costal margin to the foot.
A standard modified Hardinge approach was utilized. An incision was made originating just distal to the anterior superior iliac spine, extending distally along the anterior border of the greater trochanter and then curving posteriorly along the lateral aspect of the thigh for approximately 25 cm. The tensor fascia lata was incised in line with its fibers. The vastus lateralis was incised longitudinally and reflected anteriorly to expose the lateral femoral cortex.
Tumor Resection
Careful subperiosteal dissection was performed. The gluteus medius and minimus insertions on the greater trochanter were carefully elevated, and the piriformis and obturator internus tendons were identified and released from their insertions to allow for femoral neck osteotomy. The biopsy tract was meticulously excised en bloc with the tumor to prevent local recurrence from seeding.
The femoral artery, vein, and nerve were identified anteriorly and protected throughout the procedure.
Osteotomy sites were marked based on pre-operative templating, confirmed with intraoperative fluoroscopy. The proximal osteotomy was performed obliquely through the femoral neck, just distal to the acetabular rim, preserving the acetabulum. The distal osteotomy was performed approximately 10 cm distal to the lesser trochanter, ensuring a wide margin. A reciprocating saw was used to perform the osteotomies.
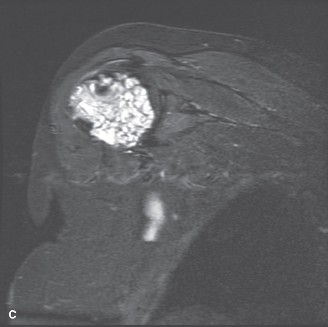
Figure 5: Intraoperative view during tumor resection. The proximal femur with the chondrosarcoma (tumor outlined) is being carefully dissected from surrounding soft tissues, with neurovascular structures protected.
The resected specimen included the femoral head, neck, greater and lesser trochanters, and a significant portion of the proximal femoral diaphysis. The specimen was sent for immediate pathological assessment of the margins, which confirmed wide, tumor-free margins both proximally and distally.
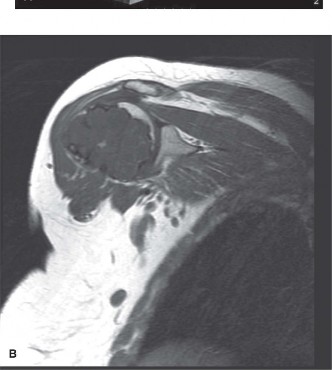
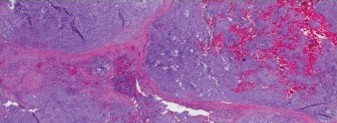
Figure 6: Gross pathology specimen of the resected proximal femur. The intramedullary chondrosarcoma is clearly visible, demonstrating its extent and characteristic lobulated appearance with calcified areas.
Reconstruction
A modular proximal femoral replacement endoprosthesis was selected. The femoral canal was reamed sequentially to fit the distal stem component. The appropriate length of the femoral stem and proximal body segment, including the femoral head component, were assembled. The stem was cemented into the femoral canal for robust fixation. The acetabulum was assessed and found to be pristine, so no acetabular component was required. The prosthetic femoral head was reduced into the native acetabulum. The stability and range of motion of the hip were assessed, demonstrating good stability through a full arc of motion.
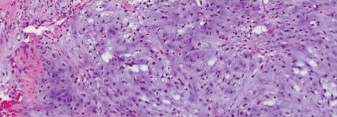
Figure 7: Intraoperative image showing the implantation of the modular proximal femoral replacement endoprosthesis. The distal stem is seated, and the proximal body with the femoral head component is being assembled and aligned for reduction into the acetabulum.
The greater trochanteric fragment, which had been osteotomized and preserved, was reattached to the prosthetic body using multiple non-absorbable sutures through drill holes, restoring the abductor lever arm. The vastus lateralis and other muscular layers were meticulously reapproximated to cover the prosthesis and restore soft tissue integrity.
Wound Closure
After thorough irrigation and hemostasis, two suction drains were placed. The incision was closed in layers: deep fascia, subcutaneous tissue, and skin with staples. A sterile dressing was applied.
Post-Operative Protocol & Rehabilitation
Immediate Post-Operative Care
- Pain Management : Multi-modal analgesia, including epidural catheter, PCA pump, and oral medications, was instituted.
- DVT Prophylaxis : Subcutaneous low molecular weight heparin was administered, along with intermittent pneumatic compression devices.
- Wound Care : Drains were monitored and removed when output was minimal. Wound staples were removed at 2 weeks.
- Early Mobilization : Out of bed to chair with assistance on Post-operative Day 1.
Rehabilitation Phases
Phase I: Early Protected Mobilization (Weeks 0-6)
*
Weight Bearing
: Toe-touch weight bearing (TTWB) or partial weight bearing (PWB) (20-25 lbs) with crutches or walker to protect soft tissue healing and trochanteric reattachment. Full weight bearing was deferred until radiographic evidence of trochanteric healing and robust muscle recovery.
*
Range of Motion
: Gentle active and passive range of motion exercises for the hip, focusing on flexion up to 90 degrees and limiting adduction and internal rotation to prevent dislocation (although less of a concern with this specific reconstruction and approach, general hip precautions were observed).
*
Muscle Activation
: Isometric gluteal sets, quadriceps sets, ankle pumps. Gentle hip abduction exercises within pain limits.
*
Gait Training
: With maximum assistance, focusing on proper technique and minimizing compensatory patterns.
Phase II: Progressive Strengthening and Balance (Weeks 6-12)
*
Weight Bearing
: Gradually increased to weight bearing as tolerated (WBAT) with crutches, progressing to a single cane as tolerated, based on surgeon approval and radiographic assessment of trochanteric healing.
*
Strengthening
: Progressive resistance exercises for hip abductors, extensors, and flexors (e.g., side-lying leg raises, prone hip extension, standing mini-squats). Core stability exercises.
*
Balance Training
: Single-leg stance exercises, tandem walking.
*
Cardiovascular
: Stationary cycling with low resistance.
Phase III: Advanced Functional Training (Week 12 onwards)
*
Strength and Endurance
: Continue progressive strengthening, incorporating higher resistance and functional movements.
*
Proprioception
: Advanced balance and agility drills.
*
Return to Activity
: Gradual return to light recreational activities, avoiding high-impact sports. Patient education regarding activity modification and limitations of the prosthesis.
Long-term Follow-up and Surveillance
- Clinical Follow-up : Every 3 months for the first 2 years, then every 6 months for the next 3 years, and annually thereafter.
-
Imaging Surveillance
:
- Plain radiographs of the hip: Performed at each follow-up visit to monitor for component loosening, periprosthetic osteolysis, or heterotopic ossification.
- Chest CT scan: Annually for the first 5 years to screen for pulmonary metastases, which, while less common in chondrosarcoma than in osteosarcoma, can occur.
- Oncologic Surveillance : Vigilance for local recurrence, which typically presents as increasing pain, palpable mass, or new radiographic changes.

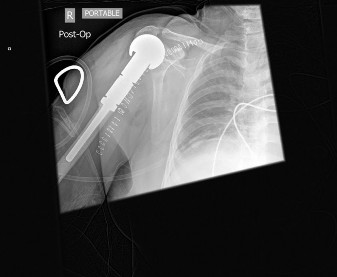
Figure 8: Post-operative anteroposterior radiograph of the left hip demonstrating the well-positioned proximal femoral replacement endoprosthesis, with excellent alignment and filling of the femoral canal.
Pearls & Pitfalls (Crucial for FRCS/Board Exams)
Pearls
- Chondrosarcoma is a Surgical Disease : Unlike many other sarcomas, chondrosarcoma is largely resistant to conventional chemotherapy and radiation. Wide en bloc surgical resection with clear margins is the only curative treatment.
- Biopsy Planning is Paramount : The biopsy tract must be meticulously planned to allow for its complete excision en bloc with the tumor during definitive surgery. An improperly placed biopsy can compromise limb salvage or increase local recurrence risk.
- Image Interpretation : Recognize the classic radiological signs: lobulated appearance, "rings and arcs" calcifications, and especially endosteal scalloping greater than 2/3 of the cortical thickness as a key indicator differentiating low-grade chondrosarcoma/ACT from quiescent enchondroma.
- Multidisciplinary Approach : Effective management requires a tumor board discussion involving orthopedic oncologists, radiologists, pathologists, and often medical oncologists, even when chemotherapy/radiation are not primary treatments.
- Enneking Staging : Crucial for guiding surgical aggressiveness. A Stage IIA lesion (high-grade, intraosseous) requires wide margins.
- Proximal Femoral Reconstruction : Modular endoprostheses offer excellent functional outcomes for large defects after proximal femoral resection, allowing early weight-bearing and rehabilitation. Soft tissue reattachment (especially trochanteric) is critical for abductor function and stability.
- Long-Term Follow-up : Lifelong surveillance is important due to the potential for late local recurrence or distant metastasis, particularly for higher-grade lesions.
Pitfalls
- Misdiagnosis of Low-Grade Chondrosarcoma as Enchondroma : This is a common and dangerous pitfall. Relying solely on radiographs for differentiating these lesions can lead to inadequate treatment. Persistent pain, rapid growth, or aggressive imaging features (e.g., marked endosteal scalloping, soft tissue extension) mandate biopsy.
- Inadequate Surgical Margins : Performing an intralesional or marginal resection for chondrosarcoma almost guarantees local recurrence, often with dedifferentiation to a higher-grade tumor. This can lead to re-resection, amputation, or poor prognosis.
- Ignoring Pain in Cartilaginous Lesions : While enchondromas are typically asymptomatic, any new or worsening pain in a known cartilaginous lesion, especially in a long bone or central skeleton, should raise suspicion for malignant transformation (to secondary chondrosarcoma) or primary chondrosarcoma, and warrant thorough investigation.
- Fragmenting the Tumor : During resection, care must be taken to avoid tumor spillage or fragmentation, which can lead to local recurrence or seeding. A wide soft tissue cuff around the tumor is vital.
- Failure to Address Soft Tissue Defects : Extensive resections, especially in the pelvis or proximal femur, can lead to significant soft tissue defects. Inadequate coverage or muscle reattachment can compromise function, increase infection risk, or contribute to dislocation of prosthetic implants.
- Over-reliance on Ancillary Treatments : Assuming chemotherapy or radiation will effectively manage chondrosarcoma is a critical error. These modalities have limited roles, primarily for specific subtypes (e.g., mesenchymal chondrosarcoma) or palliative settings.
- Biopsy Complications : Inappropriate biopsy technique can lead to pathological fracture, infection, or tumor seeding outside the planned resection field. This compromises limb salvage and can necessitate more extensive surgery or amputation.
Clinical & Radiographic Imaging
You Might Also Like