Pathological Subtrochanteric Femoral Fracture Secondary to Multiple Myeloma: Diagnostic Nuances & Bone Scintigraphy
Key Takeaway
Multiple myeloma presenting as a pathological femoral fracture often involves elderly patients with pre-existing vague symptoms. Diagnostic imaging shows comminuted lytic lesions and a widespread lytic pattern. Crucially, Tc-99m bone scans typically show 'cold' or equivocal lesions due to osteoclast predominance, a key differentiator from other metastatic processes.
Patient Presentation & History
We present the case of Mr. J.A., an 82-year-old male, who presented to the emergency department following a mechanical fall from standing height. The patient reported immediate, severe pain in his left hip and thigh, rendering him unable to bear weight.
His past medical history is significant for well-controlled hypertension and type 2 diabetes mellitus. He reports a 6-month history of vague, intermittent lower back pain, which he attributed to "getting old," and had not sought medical attention for it. He denied any preceding trauma to the left hip or thigh. Prior to the fall, he noted increasing fatigue and mild unintentional weight loss of approximately 5 kg over the past year. He denied fever, chills, night sweats, or recent infections. There was no history of previous fractures or known osteoporosis. He is independent with all activities of daily living (ADLs) and resides at home with his wife. Social history includes a remote history of smoking, quit 20 years prior, and occasional alcohol consumption.
His current medications include lisinopril and metformin. No known drug allergies.
Clinical Examination
Upon arrival, Mr. J.A. was alert, oriented, and in obvious distress due to pain. Vital signs were stable.
Inspection:
The left lower extremity was externally rotated and shortened, characteristic of a proximal femoral fracture. There was no obvious gross deformity, skin tenting, or open wound. No ecchymosis was immediately visible.
Palpation:
Direct palpation over the left greater trochanter and anterior hip joint elicited severe pain. No crepitus was appreciated due to patient guarding and pain. Distal pulses (dorsalis pedis and posterior tibial) were 2+ bilaterally, strong and symmetric. Skin temperature was normal.
Range of Motion (ROM):
Active and passive range of motion of the left hip was severely limited and painful, particularly with any attempt at rotation or flexion. The patient guarded significantly. Examination of the contralateral hip and knee showed full, pain-free ROM.
Neurological/Vascular Assessment:
Neurological examination of the left lower extremity revealed intact sensation to light touch in the L2-S1 dermatomes. Motor strength was limited by pain (approximately 3/5 with hip flexion/extension, 4/5 with knee flexion/extension, 5/5 with ankle dorsiflexion/plantarflexion), but no focal neurological deficits were identified. Capillary refill in the left toes was brisk, less than 2 seconds. The overall vascular status of the limb was reassuring.
Given the mechanism of injury (low-energy fall in an elderly patient) and the patient's reported preceding symptoms (back pain, fatigue, weight loss), a high index of suspicion for underlying pathological fracture was immediately raised, rather than a purely traumatic event.
Imaging & Diagnostics
Initial plain radiographs of the left hip were obtained upon presentation.
X-ray Findings:
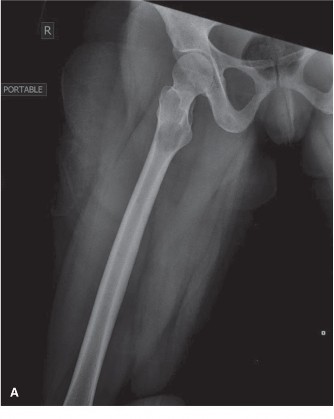
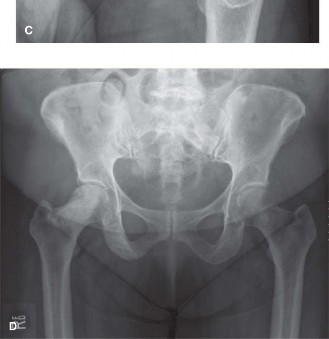
The anteroposterior (AP) and lateral views of the left hip demonstrated a comminuted, subtrochanteric fracture of the left femur. The fracture pattern involved significant cortical destruction and a large lytic lesion extending proximally into the intertrochanteric region and distally into the subtrochanteric area. The margins of the lytic lesion appeared ill-defined in several areas, raising strong suspicion for a neoplastic process. There was no evidence of periosteal reaction or overt sclerotic changes commonly associated with benign lesions or healing fractures.

(Figure 1: Initial AP radiograph of the left hip demonstrating a comminuted subtrochanteric fracture with an underlying large lytic lesion of the proximal femur.)
Further Imaging Indications:
Given the radiographic evidence of a pathological fracture, an expedited workup was initiated to identify the primary pathology and stage the disease.
- Computed Tomography (CT) Scan of the Left Femur: A CT scan was immediately performed to better delineate the extent of bone destruction, cortical involvement, and soft tissue component. The CT confirmed an extensive lytic lesion involving a significant portion of the proximal femur, with substantial cortical thinning and impending pathological fracture, which had now fully propagated. The medullary canal was extensively involved. No overt soft tissue mass was identified, but the fracture hematoma obscured precise differentiation. CT provided crucial information for surgical planning regarding the integrity of the remaining bone stock and potential need for adjuvant fixation strategies.
- Skeletal Survey: A full skeletal survey (AP and lateral views of skull, spine, pelvis, bilateral humeri, and femora) was performed to assess for multifocal involvement. This revealed numerous other lytic lesions in the vertebral bodies (T8, L2, L4), skull, and bilateral humeri, without evidence of osteoblastic response. This widespread lytic pattern is highly suggestive of a disseminated neoplastic process.
-
Bone Scintigraphy (Technetium-99m Bone Scan):
A whole-body technetium-99m methylene diphosphonate (Tc-99m MDP) bone scan was performed. Classically, bone scans detect increased osteoblastic activity, which is characteristic of most metastatic lesions (e.g., prostate, breast). However, in multiple myeloma, lesions are predominantly lytic due to increased osteoclast activity and decreased osteoblast activity, often resulting in
"cold" lesions or areas of normal uptake
rather than increased uptake. In Mr. J.A.'s case, the bone scan demonstrated a patchy, equivocal uptake pattern. Specifically, the major lytic lesion in the left proximal femur showed only mildly increased peripheral uptake, with a relative photopenic center. Several vertebral lesions identified on the skeletal survey showed no or minimal uptake. This "cold" or equivocal scan is a critical nuance in diagnosing multiple myeloma.

(Figure 2: Whole-body Technetium-99m MDP bone scan demonstrating equivocal uptake in the left proximal femur and a "cold" spot in a vertebral body, highlighting the atypical presentation of multiple myeloma on bone scintigraphy.) - Magnetic Resonance Imaging (MRI) of the Spine: Given the equivocal bone scan and the patient's reported back pain, an MRI of the entire spine was performed to evaluate for cord compression and better characterize the extent of marrow involvement. The MRI demonstrated diffuse marrow infiltration consistent with plasma cell dyscrasia, with multiple focal lesions showing low signal on T1-weighted images and high signal on T2-weighted and STIR sequences, confirming involvement of several vertebral bodies, sacrum, and pelvic bones. Importantly, there was no immediate spinal cord compromise. MRI is considered superior to bone scans for detecting early myeloma lesions and assessing marrow involvement.
- Positron Emission Tomography-Computed Tomography (PET/CT) with FDG: An FDG-PET/CT was subsequently performed. This imaging modality assesses metabolic activity and is highly sensitive for active myeloma lesions, including purely lytic ones. In Mr. J.A., the FDG-PET/CT revealed hypermetabolic foci corresponding to all known lesions on the skeletal survey and MRI, including the primary lesion in the left proximal femur and numerous other widespread lesions, confirming widespread active disease. This modality has emerged as a crucial tool for staging and assessing response to therapy in multiple myeloma.
-
Laboratory Investigations:
- Complete Blood Count (CBC): Mild normocytic anemia (Hemoglobin 9.8 g/dL).
- Renal Function Tests: Elevated creatinine (1.8 mg/dL) with an estimated GFR of 38 mL/min/1.73m², consistent with myeloma kidney.
- Calcium: Hypercalcemia (corrected calcium 11.8 mg/dL), a common paraneoplastic manifestation.
- Serum Protein Electrophoresis (SPEP) and Immunofixation Electrophoresis (IFIX): Detected a monoclonal M-spike in the gamma region, identified as IgG kappa light chain.
- Serum Free Light Chain Assay: Markedly elevated kappa free light chains with an abnormal kappa/lambda ratio.
- Urinalysis and Urine Protein Electrophoresis (UPEP) with IFIX: Bence Jones proteinuria (kappa light chains) detected.
- Bone Marrow Biopsy: Performed from the posterior iliac crest, revealed >60% plasma cells, unequivocally diagnostic of multiple myeloma. Cytogenetics showed specific high-risk features.
The combination of imaging and laboratory findings confirmed a diagnosis of Stage III Multiple Myeloma with widespread lytic bone disease and pathological subtrochanteric fracture of the left femur, associated with myeloma-related renal dysfunction and hypercalcemia.
Differential Diagnosis
The presentation of a pathological fracture in an elderly patient mandates a thorough differential diagnosis, particularly when systemic symptoms are present and imaging reveals widespread lytic lesions.
| Feature | Multiple Myeloma | Metastatic Carcinoma (e.g., Lung, Kidney, Thyroid, Breast) | Primary Bone Sarcoma (e.g., Chondrosarcoma, Fibrosarcoma) |
|---|---|---|---|
| Clinical Presentation | Elderly patient, fatigue, weight loss, back pain, recurrent infections, hypercalcemia, renal dysfunction. Pathological fracture. | Elderly patient, constitutional symptoms, history of known primary malignancy (often), organ-specific symptoms, pathological fracture. | Variable age (often younger for osteosarcoma, older for chondrosarcoma), localized pain, swelling, palpable mass. Pathological fracture less common. |
| Radiographic Features | "Punched-out" lytic lesions without sclerotic borders or periosteal reaction. Diffuse osteopenia. Skull lesions common. | Predominantly lytic or blastic lesions (depending on primary). Often "moth-eaten" or permeative pattern. May have associated soft tissue mass. | Highly variable: Lytic, sclerotic, mixed. Often large, destructive mass with cortical breakthrough and soft tissue component. Periosteal reaction (Codman's triangle, sunburst) common in high-grade sarcomas. |
| Bone Scan (Tc-99m) | Classically "cold" or equivocal lesions due to lack of osteoblastic activity. May show peripheral uptake. | Typically "hot" lesions due to increased osteoblastic activity surrounding the tumor, particularly for prostate/breast. | Variable uptake, often "hot" but can be equivocal depending on tumor type and activity. |
| MRI Features | Diffuse marrow infiltration, focal lesions (low T1, high T2/STIR). Crucial for marrow involvement & spinal cord assessment. | Focal lesions with variable signal characteristics. May show significant soft tissue component. | Large, heterogeneous mass with extensive bone destruction, often with aggressive features and marrow invasion. |
| PET/CT (FDG) | Highly sensitive for active lesions, showing increased metabolic activity (hypermetabolic foci). Excellent for staging. | High sensitivity and specificity for metabolically active metastases. Excellent for staging and response. | Highly avid lesions; useful for determining tumor extent and identifying distant metastases. |
| Laboratory Markers | M-spike (SPEP/IFIX), Bence Jones proteinuria (UPEP), elevated free light chains, hypercalcemia, renal dysfunction. | Elevated tumor markers (e.g., PSA, CEA, CA 19-9) if primary known. Elevated calcium/ALP (less specific). | Elevated ALP and LDH can be seen. Specific markers generally absent. Definitive diagnosis by biopsy. |
| Biopsy Findings | Monoclonal plasma cell proliferation in bone marrow. | Histological features of primary tumor (e.g., adenocarcinoma, renal cell carcinoma). | Malignant mesenchymal cells with specific features based on tumor type (e.g., chondroblastic, osteoblastic differentiation). |
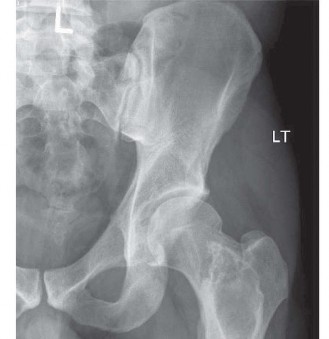
|
|||
| (Figure 3: Coronal T1-weighted MRI demonstrating diffuse marrow signal abnormalities and focal lytic lesions in the pelvis and proximal femora, consistent with widespread marrow infiltration by multiple myeloma.) |
Surgical Decision Making & Classification
Given the diagnosis of a pathological subtrochanteric femur fracture secondary to multiple myeloma, surgical intervention was deemed necessary for pain control, restoration of mobility, and prevention of further devastating collapse. Non-operative management was considered inappropriate due to the complete fracture, the patient's age and desire for mobility, and the inherent instability of the lesion.
Classification Systems & Considerations:
- Mirels' Scoring System for Impending Pathological Fractures: While the fracture was already complete, Mirels' score is a valuable tool to assess the risk of fracture in existing lesions. This system evaluates lesion site, size, character (lytic/blastic/mixed), and pain. A score of 9 or higher generally indicates a high risk and often prompts prophylactic fixation. In Mr. J.A.'s case, with a large lytic lesion at a weight-bearing site and significant pain, the Mirels' score would have been high (likely >10), supporting fixation if it had been identified prior to fracture.
- AO/OTA Classification: The fracture was classified as an AO/OTA 32-A3 fracture, indicating a subtrochanteric simple or wedge fracture. However, this system primarily describes morphology and does not account for the underlying oncological pathology, which dictates critical aspects of surgical strategy and prognosis.
- Enneking Classification: This system is used for staging musculoskeletal sarcomas but provides useful principles for considering the extent of tumor and surgical margins for all bone tumors. For multiple myeloma, which is a systemic disease, local control is palliative rather than curative. The lesion was intra-compartmental (T1) based on imaging, but the widespread systemic nature means it is a disseminated disease.
Decision for Operative Management:
- Pain Relief: Immediate, substantial pain relief from fracture stabilization.
- Mobility & Function: Restoration of weight-bearing ability and ability to ambulate, crucial for an elderly patient's independence and quality of life. Without fixation, the patient would be bed-bound with ongoing pain.
- Local Control: Stabilization of the bone to prevent further progression of the fracture and potential for neurovascular compromise.
- Facilitate Adjuvant Therapy: Stable fixation allows the patient to undergo systemic chemotherapy and radiation therapy more comfortably and efficiently, without exacerbating fracture pain or instability.
- Bone Healing Potential: While pathological fractures often have impaired healing, stabilization prevents displacement and optimizes the chances of some callus formation or osseointegration of the implant, especially with cement augmentation.
Pre-operative Optimization:
Prior to surgery, the patient's hypercalcemia was managed with intravenous fluids and bisphosphonates (Zoledronic acid). Renal function was monitored closely. Hematology/Oncology consultation was obtained to discuss systemic treatment plans post-operatively. Blood products were cross-matched due to potential intraoperative blood loss and anemia.
Surgical Technique / Intervention
The primary goal of surgical intervention for this pathological subtrochanteric fracture was to achieve stable fixation, provide immediate weight-bearing capability, and address the compromised bone quality due to myeloma involvement.
Patient Positioning:
The patient was positioned supine on a fracture table with the left leg in traction, allowing for controlled reduction and fluoroscopic imaging in both AP and lateral planes. The contralateral leg was abducted and flexed to allow for free fluoroscopy arc. Care was taken to pad all pressure points given the patient's age and comorbidities.
Approach:
A standard lateral approach to the proximal femur was utilized. A longitudinal incision, approximately 10-12 cm in length, was made centered over the greater trochanter, extending distally. The fascia lata was incised, and the vastus lateralis was identified and split longitudinally to expose the lateral cortex of the proximal femur.
Reduction Techniques:
Reduction was achieved predominantly through indirect means using the fracture table traction and internal rotation to correct the external rotation deformity. A large-fragment reduction clamp was used to reduce the fracture fragments. The significant bone loss and comminution made direct reduction challenging and increased the risk of further fragmenting the already compromised bone. Gentle manipulation and external rotation were employed to achieve an acceptable alignment, approximately 5-10 degrees of valgus to offset potential varus collapse. Fluoroscopy was used extensively to confirm reduction in both planes.
Fixation Construct:
Given the patient's age, the comminuted nature of the fracture, the extensive lytic destruction, and the systemic disease, an
intramedullary (IM) nail
was selected for fixation. Specifically, a long cephalomedullary nail (e.g., a trochanteric entry femoral nail) was chosen to bypass the entire lesion and provide durable fixation throughout the diaphysis, extending distal to the compromised bone. This also offers rotational stability and load sharing.
- Entry Point: A standard piriformis fossa or greater trochanteric entry point was made using an awl.
- Reaming: Reaming was performed carefully to avoid iatrogenic perforation of the thinned cortex. The reamer size was chosen to allow for cement augmentation later.
- Nail Insertion: A long cephalomedullary nail of appropriate length and diameter was inserted. Proximal interlocking screws were placed through the nail into the femoral head/neck fragments under fluoroscopic guidance, aiming for bicortical purchase where bone quality allowed. Due to the lytic nature, careful screw selection and purchase were crucial.
-
Polymethylmethacrylate (PMMA) Cement Augmentation:
This was a critical step for enhancing immediate stability and load-bearing capacity, particularly important in oncologic bone where healing is delayed or absent.
- Once the IM nail was provisionally secured, the medullary canal, particularly within the defect created by the lytic lesion, was irrigated and dried.
- A bone cement gun was used to inject low-viscosity PMMA cement into the fracture gap and surrounding bone defects through a small cortical window or directly around the nail (if a fenestrated nail was used, though standard nails can also be augmented). This was done carefully to avoid extravasation into surrounding soft tissues or the joint.
- The cement was allowed to polymerize, providing immediate structural support and filling the lytic defects. This created a hybrid osteo-cement-implant construct.
- Distal interlocking screws were then placed to secure the nail and complete the construct.
-
Wound Closure:
The wound was copiously irrigated. The vastus lateralis was repaired, fascia lata closed, and subcutaneous tissue and skin approximated in layers.

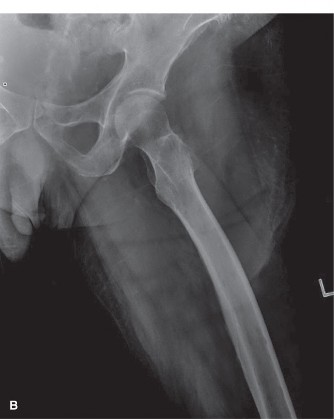
(Figure 4: Post-operative AP radiograph of the left femur demonstrating stable fixation of the subtrochanteric fracture with a long cephalomedullary nail and polymethylmethacrylate cement augmentation around the fracture site.)
Post-Operative Protocol & Rehabilitation
The post-operative course for patients with pathological fractures secondary to multiple myeloma requires careful coordination between orthopedic trauma surgery, oncology, physical therapy, and pain management.
Immediate Post-Operative Period (Days 0-7):
- Pain Management: Aggressive, multimodal pain management protocol including patient-controlled analgesia (PCA) or continuous epidural infusion, transitioning to oral opioids and non-opioid adjuncts. Early mobilization helps reduce reliance on narcotics.
- Weight-Bearing Status: Immediate weight-bearing as tolerated (WBAT) on the operative limb was encouraged due to the stable construct and cement augmentation. This is crucial for early mobilization and prevention of complications associated with prolonged bed rest in the elderly.
- Deep Vein Thrombosis (DVT) Prophylaxis: Pharmacological DVT prophylaxis (e.g., low molecular weight heparin) initiated post-operatively and continued for at least 4-6 weeks, along with mechanical prophylaxis (sequential compression devices).
- Physical Therapy (PT): Initiation of out-of-bed activity on post-operative day 1. Focus on seated exercises, transfers, and gait training with assistive devices (walker). Range of motion exercises for hip and knee were started to prevent stiffness.
- Oncology Consultation: Daily liaison with oncology team to finalize systemic treatment plan (chemotherapy, bisphosphonates) which typically commences once the patient is medically stable and pain is controlled. Bisphosphonate therapy (e.g., zoledronic acid) is crucial for managing bone pain, reducing skeletal-related events (SREs), and inhibiting osteoclast activity in myeloma.
- Renal Function & Calcium Monitoring: Close monitoring of renal function, serum calcium, and electrolytes, given the patient's pre-existing myeloma-related nephropathy and hypercalcemia.
Subacute Phase (Weeks 2-6):
- Continued PT: Progression of ambulation with less assistance, strengthening exercises for hip abductors, quadriceps, and core musculature. Emphasis on functional independence.
- Occupational Therapy (OT): Assessment and training for ADLs, home safety, and adaptive equipment needs.
- Systemic Therapy: Systemic chemotherapy regimen (e.g., proteasome inhibitor-based or immunomodulatory drug-based regimen) was initiated as per oncology recommendations.
- Radiation Therapy: While not routinely indicated for disseminated disease, localized external beam radiation therapy might be considered for isolated painful lesions, or if there was concern for local progression despite systemic therapy, or if residual disease threatened the construct. In this case, stable fixation allowed systemic therapy to be the primary oncologic treatment.
Long-Term Follow-up (Months and Years):
- Orthopedic Follow-up: Regular clinical and radiographic assessment to monitor implant integrity, bone healing (if any), and rule out local progression or periprosthetic fracture. Radiographs typically obtained at 6 weeks, 3 months, 6 months, and annually.
- Oncology Follow-up: Lifelong follow-up with the oncology team for disease monitoring, management of systemic therapy, and assessment for relapse or new skeletal events.
- Fracture Healing: Expectation for true "fracture healing" in the traditional sense is often diminished in pathological fractures due to poor bone quality and the effects of chemotherapy/radiation. The goal is implant longevity and functional stability rather than complete radiographic union. The PMMA augmentation provides immediate structural support independent of biological healing.
Pearls & Pitfalls (Crucial for FRCS/Board Exams)
Pearls:
- High Index of Suspicion: Any low-energy fracture in an elderly patient, especially with vague constitutional symptoms (weight loss, fatigue, back pain) or abnormal labs (anemia, hypercalcemia, renal dysfunction), mandates immediate workup for underlying pathological etiology. "Don't just fix the fracture, find the cause."
- Bone Scan Nuances in Myeloma: Multiple myeloma classically presents with purely lytic lesions due to osteoclast activation and suppressed osteoblast activity. This means technetium-99m bone scans can be "cold" or equivocal , leading to false negatives. Relying solely on a bone scan for screening for myeloma is a significant pitfall.
-
Preferred Imaging for Myeloma:
- Skeletal Survey: Remains the gold standard for initial assessment of lytic lesions in myeloma due to its ability to visualize pure osteolysis.
- MRI: Best for evaluating bone marrow involvement, detecting early lesions, and assessing spinal cord compression.
- FDG-PET/CT: Highly sensitive for metabolically active lesions, staging, and assessing response to treatment, including those "cold" on bone scan. It's often superior to bone scan for active myeloma.
-
Surgical Principles for Pathological Fractures:
- Long Bypass: The fixation construct must bypass the entire lesion and extend at least two cortical diameters (or preferably one full bone diameter) past the tumor burden proximally and distally to prevent propagation or periprosthetic fracture. For long bones, an IM nail spanning joint to joint is often ideal.
- Cement Augmentation: Polymethylmethacrylate (PMMA) augmentation is critical. It provides immediate mechanical stability, fills lytic defects, and acts as a stress riser buffer. It does not allow for bone ingrowth but ensures stable load sharing.
- Stable Fixation: The goal is immediate load-bearing and functional stability. A rigid construct is paramount, often achieved with IM nails or plates with supplemental cement or locking screws.
- Biological Augmentation: While not in this case, consider allograft/autograft/stem cells for benign lesions or isolated metastases with long prognosis. For myeloma, mechanical augmentation with cement is preferred.
- Multidisciplinary Approach: Management of pathological fractures due to systemic malignancies requires close collaboration with oncology, radiation oncology, pain management, and physical therapy. Early consultation facilitates optimal systemic treatment.
- Mirels' Score: Understand and apply Mirels' score for assessment of impending pathological fractures to guide prophylactic fixation.
Pitfalls:
- Misinterpreting Bone Scans: Assuming a negative or equivocal bone scan rules out widespread metastatic disease, especially in the context of myeloma. Always correlate with plain radiographs, CT, MRI, and patient symptoms.
- Inadequate Fixation: Choosing a short implant or an unstable construct that does not adequately bypass the entire compromised bone segment, leading to early mechanical failure or periprosthetic fracture.
- Ignoring Systemic Disease: Focusing solely on the fracture fixation without addressing the underlying oncological diagnosis. This compromises patient prognosis and overall quality of life.
- Delayed Diagnosis: Attributing constitutional symptoms or vague pain to "old age" without appropriate diagnostic workup, delaying diagnosis of malignancy and potentially leading to a pathological fracture as the first presentation.
- Failure to Augment: Not using cement augmentation in highly lytic lesions or compromised bone, risking early implant loosening, cutout, or inadequate stability for weight-bearing.
- Underestimating Post-Operative Complications: Elderly patients with malignancy are at higher risk for DVT, PE, infection, delayed wound healing, and medical complications. Aggressive prophylaxis and vigilance are essential.
- Inappropriate Weight-Bearing Restrictions: Placing unnecessary weight-bearing restrictions on a stable, cemented construct. This can lead to deconditioning, DVT, and pneumonia in an elderly population already at risk.
Clinical & Radiographic Imaging
You Might Also Like