Challenging Oncology Cases: Aneurysmal Bone Cyst Diagnosis
Key Takeaway
We review everything you need to understand about Challenging Oncology Cases: Aneurysmal Bone Cyst Diagnosis. Aneurysmal Bone Cysts (ABCs) are destructive, reactive bone lesions characterized by multiple blood-filled cavities. Radiographically, they present as eccentric, expanded radiolucent lesions, retaining a thin periosteal rim. Histologically, they feature cavernous blood spaces with multinucleated giant cells and no cellular atypia. MRI often confirms diagnosis with characteristic fluid-fluid levels, important for distinguishing these **oncology cases aneurysmal** manifestations.
Patient Presentation & History
A 16-year-old male presented to the Orthopedic Trauma Clinic with a 6-month history of insidious onset left shoulder pain, exacerbated acutely after a minor fall while playing basketball. He reported localized aching pain, which progressively worsened over time, limiting his ability to participate in sports. The acute exacerbation followed an axial load mechanism onto an outstretched arm, leading to immediate severe pain and inability to move the left arm. There was no history of fever, weight loss, night sweats, or other constitutional symptoms.
His past medical history was unremarkable. He denied any regular medications, allergies, or prior surgeries. Family history was non-contributory for bone tumors or genetic conditions. He was a non-smoker and did not consume alcohol. The patient was a student, engaged in a physically active lifestyle prior to the onset of symptoms.
Clinical Examination
On arrival, the patient was alert, oriented, and in moderate distress due to pain.
General Inspection:
Visible swelling and ecchymosis were noted over the left shoulder region, with a subtle anterior prominence. The patient held his left arm adducted and internally rotated, resisting any attempts at passive movement.
Palpation:
Significant tenderness was elicited over the proximal humerus. A firm, expansile mass was palpable, particularly anteriorly, which felt warm to touch. No obvious crepitus was appreciated with minimal passive motion, but the patient guarded heavily. No pulsatile mass was noted.
Range of Motion (ROM):
Active ROM was severely restricted in all planes due to pain. Passive ROM was also significantly limited by pain and guarding, demonstrating only approximately 10 degrees of abduction and external rotation before resistance.
Neurological Assessment:
Distal neurological examination revealed intact motor function (deltoid, biceps, triceps, wrist/hand musculature strength 5/5) and sensation (light touch, pinprick) in the radial, ulnar, and median nerve distributions. No specific neurological deficits were identified, indicating no overt brachial plexus involvement or nerve compression.
Vascular Assessment:
Radial and ulnar pulses were bilaterally palpable, strong, and equal. Capillary refill in the digits was brisk (<2 seconds). No signs of acute vascular compromise were present.
Systemic Examination:
No lymphadenopathy was identified. Cardiopulmonary examination was unremarkable.
Imaging & Diagnostics
Plain Radiographs
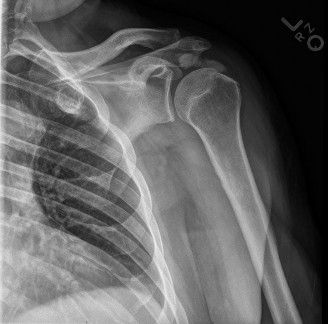
Initial anteropoposterior (AP) and lateral radiographs of the left shoulder and humerus demonstrated a large, expansile, purely lytic lesion involving the metadiaphyseal region of the proximal humerus, measuring approximately 7 cm in its greatest dimension. The lesion showed characteristic "blow-out" or "ballooning" of the cortex, with a very thin, attenuated cortical shell. Internal septations were visible, giving a soap-bubble or multicystic appearance. A pathological fracture was evident through the thinnest cortical margin, specifically involving the posteromedial cortex of the proximal humeral shaft, with minimal displacement. No aggressive periosteal reaction or overt evidence of matrix mineralization was observed, which would suggest an osteoid-producing tumor.
(Figure 1: Initial AP radiograph of the left humerus demonstrating an expansile lytic lesion with characteristic "blow-out" appearance and pathological fracture.)
Computed Tomography (CT) Scan
A computed tomography (CT) scan of the left humerus was performed to better delineate the bony architecture, cortical integrity, and precise extent of the lesion, particularly in relation to the articular surface and surrounding soft tissues. The CT confirmed the expansile, multiloculated nature of the lesion, showing multiple fluid-fluid levels within various compartments. The thin, remodeled cortex was clearly visualized, with focal areas of complete cortical breach corresponding to the pathological fracture site. No significant solid components or calcified matrix were identified within the lesion, which would be highly suspicious for osteosarcoma or chondrosarcoma. The CT provided critical information for surgical planning regarding the stability of the remaining bone and the size of the defect after potential curettage.
Magnetic Resonance Imaging (MRI)
Magnetic resonance imaging (MRI) of the left shoulder and humerus was obtained for comprehensive characterization of the lesion's internal architecture and its relationship to neurovascular structures and the rotator cuff. On T1-weighted images, the lesion appeared largely hypointense, while T2-weighted images showed heterogeneous signal intensity with multiple high-signal intensity locules separated by low-signal intensity septa. Crucially, multiple prominent fluid-fluid levels were observed within these locules on T2-weighted sequences, representing layering of serosanguinous fluid and cellular debris, which is highly suggestive, though not pathognomonic, of an Aneurysmal Bone Cyst (ABC). Post-contrast T1-weighted images demonstrated enhancement of the septa and peripheral rim of the lesion but no significant solid enhancing component. The articular cartilage and glenoid labrum appeared intact. The deltoid and rotator cuff muscles were edematous adjacent to the fracture but otherwise unremarkable. The brachial plexus and axillary vessels were displaced but not directly invaded.

(Figure 2: Coronal T2-weighted MRI image demonstrating multiple fluid-fluid levels (arrows) within the lesion, a characteristic feature of an Aneurysmal Bone Cyst.)
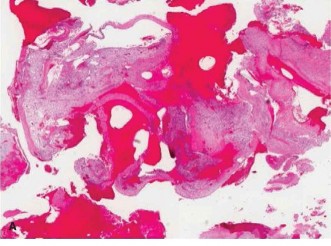
Biopsy
Given the diagnostic challenge, particularly differentiating ABC from telangiectatic osteosarcoma (TOS) due to their radiographic and MRI similarities, a CT-guided core needle biopsy was performed. Multiple samples were taken from different areas of the lesion, specifically targeting both solid-appearing septa and the walls of the cystic spaces.
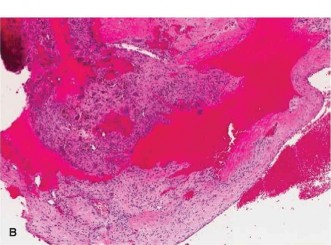
Histopathological examination revealed blood-filled cystic spaces separated by fibrous septa containing spindle cells, osteoclast-like giant cells, and reactive woven bone formation. Importantly, there was no evidence of cellular atypia, pleomorphism, or malignant osteoid production, thus ruling out telangiectatic osteosarcoma. These findings were entirely consistent with an Aneurysmal Bone Cyst.
Laboratory Investigations
Routine laboratory tests including complete blood count (CBC), erythrocyte sedimentation rate (ESR), C-reactive protein (CRP), serum calcium, phosphate, and alkaline phosphatase were all within normal limits. These results helped exclude inflammatory or metabolic bone diseases and did not suggest overt malignancy.
Differential Diagnosis
The differential diagnosis for an expansile lytic lesion of bone, especially in a young patient presenting with a pathological fracture, is broad and includes both benign and malignant entities. The presence of fluid-fluid levels on MRI is a significant clue but not entirely specific, necessitating a careful approach. The primary differentials considered in this case were:
| Feature | Aneurysmal Bone Cyst (ABC) | Telangiectatic Osteosarcoma (TOS) | Giant Cell Tumor (GCT) | Unicameral Bone Cyst (UBC) / Simple Bone Cyst (SBC) |
|---|---|---|---|---|
| Age Group | Typically <20 years (peak 10-20) | Often <30 years (peak 10-20) | Typically 20-40 years | Typically <20 years (peak 3-14) |
| Location | Metaphysis of long bones, vertebrae, flat bones | Metaphysis of long bones | Epiphysis/Metaphysis of long bones (distal femur, proximal tibia, distal radius) | Metaphysis of long bones (proximal humerus, proximal femur) |
| Radiographs | Expansile, lytic, "blow-out" or "ballooning," thin cortex, internal septations, eccentric | Expansile, lytic, aggressive periosteal reaction (Codman's triangle), permeative margins, soft tissue mass, osteoid matrix (subtle/absent) | Epiphyseal/Metaphyseal, purely lytic, expansile, sharp but non-sclerotic margins, no matrix, cortical breach common | Centrally located, lytic, oblong, "fallen leaf" sign, sclerotic margin, no periosteal reaction |
| CT Findings | Multiloculated cystic lesion, thin septations, fluid-fluid levels, subtle cortical breach | Destructive, aggressive cortical destruction, soft tissue component, subtle osteoid matrix, hemorrhage/necrosis | Lytic, expansile, well-defined margins, often cortical breach, no internal matrix | Thin cortical shell, homogeneous fluid density, may have septations, minimal expansion |
| MRI Findings | Multiple fluid-fluid levels (key), heterogeneous T1/T2 signal, enhancing septa, no solid components | Fluid-fluid levels (less common, usually fewer and less prominent), solid enhancing components, soft tissue mass, marrow edema, aggressive features | Heterogeneous T1/T2, no fluid-fluid levels, strong peripheral and septal enhancement, solid components, subchondral involvement | Homogeneous T1/T2 fluid signal, no fluid-fluid levels, non-enhancing thin wall |
| Histopathology | Blood-filled spaces, fibrous septa, bland spindle cells, reactive woven bone, scattered osteoclast-like giant cells. NO cellular atypia. | Atypical osteoblasts, malignant osteoid production, severe pleomorphism, necrotic areas, blood-filled spaces (may mimic ABC) | Uniform mononuclear stromal cells, numerous uniformly distributed osteoclast-like giant cells, no osteoid production | Simple serous fluid-filled cavity, fibrous lining, few giant cells, no solid components |
| Prognosis | Benign, high local recurrence rate if incompletely treated | Malignant, high metastatic potential | Benign but locally aggressive, high recurrence if incompletely treated, small metastatic potential | Benign, often self-limiting, low recurrence |
Surgical Decision Making & Classification
Given the patient's age, the symptomatic nature of the lesion, the progressive pain, radiographic evidence of an active and expansile process, and especially the presence of a pathological fracture, operative intervention was deemed necessary. Conservative management with casting or bracing alone would not adequately address the underlying structural instability or the progressive nature of the lesion, and carried a high risk of non-union or refracture due to the extensive lytic destruction.
Classification
According to the Enneking Staging System for benign bone tumors, this lesion was classified as an Enneking Stage 2 (Active) or potentially Stage 3 (Aggressive) due to its expansile nature, cortical breach, and pathological fracture. Active lesions are typically contained by a reactive rim but show evidence of growth. Aggressive lesions extend beyond the reactive bone into soft tissue or violate the cortex significantly, often leading to fracture. While Stage 3 benign lesions share some features with low-grade malignancies, histopathology confirmed the benign nature of this ABC.
Pre-operative Planning
Pre-operative planning involved:
1.
Confirmation of Diagnosis
: The biopsy was crucial to definitively rule out telangiectatic osteosarcoma, which would necessitate a radically different treatment paradigm (neoadjuvant chemotherapy and wide en bloc resection).
2.
Assessment of Extent
: CT and MRI scans provided detailed anatomical information regarding the lesion's boundaries, relationship to the glenohumeral joint, and the neurovascular bundle. This guided the surgical approach and the extent of required curettage.
3.
Blood Loss Management
: ABCs can be highly vascular. Pre-operative selective arterial embolization was considered but ultimately deemed unnecessary as the lesion was not excessively large or overtly hypervascular on imaging, and the anticipated blood loss could be managed intraoperatively.
4.
Reconstruction Strategy
: Given the pathological fracture and the significant bone defect anticipated post-curettage, the reconstruction plan involved extensive curettage, adjuvant therapy, followed by structural augmentation. Options included autogenous cancellous bone grafting (from the ipsilateral iliac crest) combined with polymethylmethacrylate (PMMA) cement or allograft, and internal fixation to restore structural stability and facilitate early rehabilitation. An intramedullary (IM) nail was favored for its load-sharing capabilities in the diaphysis, extending into the metaphysis, while a plate would allow for better contouring in the proximal metaphysis and head region. Given the proximal humerus location, a locked plate seemed more appropriate for securing the humeral head fragments and bridging the defect.
Surgical Technique / Intervention
The patient was positioned in the supine position on a radiolucent operating table with a beanbag, allowing for adequate access to the left shoulder and ipsilateral iliac crest. General anesthesia was administered, and prophylactic antibiotics were given.
A standard deltopectoral approach was utilized to access the proximal humerus. The cephalic vein was identified and retracted laterally. The deltoid muscle was carefully elevated off the humerus. The pathological fracture site was identified, along with the thinned, expanded cortex of the lesion.
- Exposure and Debridement : The fracture hematoma was evacuated. The attenuated cortical shell overlying the ABC was carefully incised, and the cystic lesion was exposed. As expected, the cyst was filled with blood, serous fluid, and fibrous septations. Gross inspection during surgery confirmed the characteristic appearance of an ABC, without any obvious solid or overtly malignant-appearing tissue.
- Intralesional Curettage : Meticulous intralesional curettage was performed using various-sized curettes. The entire inner lining of the cyst wall was aggressively removed, taking care to scrape all fibrous septa and abnormal tissue. A high-speed burr was then used to meticulously debride the remaining bone margins, ensuring all visible pathological tissue was removed and creating a smooth, sclerotic bone cavity. This process is crucial to reduce the high recurrence rate associated with ABCs.
- Adjuvant Therapy : Following thorough curettage, adjuvant therapy with cryosurgery (liquid nitrogen application) was utilized. This involved creating freeze-thaw cycles by applying liquid nitrogen directly to the cavity walls for two cycles of 60 seconds each. This thermal necrosis helps destroy any residual microscopic tumor cells, further reducing the risk of local recurrence. Phenol or argon beam coagulation are alternative adjuvants.
-
Defect Reconstruction and Fixation
:
- The significant bone defect created by the pathological fracture and curettage necessitated structural support. An autogenous cancellous bone graft was harvested from the ipsilateral posterior iliac crest to provide osteoconductive and osteoinductive properties. This graft was packed into the largest areas of the defect.
- To provide immediate structural stability and to serve as an additional adjuvant (due to exothermic reaction), polymethylmethacrylate (PMMA) bone cement was mixed and carefully injected into the remaining cavity, encompassing the bone graft. The cement was allowed to polymerize, creating a stable, load-bearing construct within the humerus.
- Due to the pathological fracture and the large defect, internal fixation was essential. A pre-contoured locking proximal humeral plate was selected. The plate was carefully applied to the lateral aspect of the humerus, bridging the defect and providing multi-planar stability to the humeral head and shaft. Locking screws were inserted proximally into the humeral head fragments and distally into the humeral shaft, ensuring secure fixation. The aim was to achieve stable osteosynthesis that would allow early rehabilitation.

(Figure 3: Post-operative AP radiograph showing the locking proximal humeral plate and screws providing stable fixation, with bone cement filling the defect after curettage.)
- Closure : The wound was thoroughly irrigated. The deltopectoral interval was closed, and subcutaneous tissues and skin were closed in layers. A drain was placed to manage post-operative bleeding.
Post-Operative Protocol & Rehabilitation
Immediate Post-operative Phase (0-2 weeks)
- Pain Management : Multi-modal analgesia including oral opioids, NSAIDs (if no contraindications), and paracetamol.
- Wound Care : Daily dressing changes, drain removal typically on post-op day 1-2 when output is minimal.
- Immobilization : The arm was placed in a sling for comfort and protection, but passive motion was initiated early.
-
Rehabilitation
:
- Day 1 Post-op : Gentle passive range of motion (PROM) exercises for the elbow, wrist, and hand to prevent stiffness. Pendulum exercises for the shoulder were initiated, avoiding active shoulder abduction/flexion.
- Weight Bearing : Non-weight-bearing restrictions for the affected upper extremity.
Early Rehabilitation Phase (2-6 weeks)
- Sling Use : Continued sling use for protection and comfort, but removed for exercises.
-
Rehabilitation
:
- Shoulder PROM : Gradually increased passive range of motion with therapist assistance, aiming for full PROM by 6 weeks.
- Gentle Active-Assisted ROM (AAROM) : Started around week 4, as pain allows, focusing on forward flexion and external rotation.
- Scapular Stabilization Exercises : Isometric contractions to activate peri-scapular musculature.
- Avoidance : No active abduction or lifting against gravity until radiographic signs of healing and stability are evident. No pushing, pulling, or lifting more than 1 kg.
Intermediate Rehabilitation Phase (6-12 weeks)
- Radiographic Assessment : Follow-up radiographs at 6 weeks to assess implant position, cement consolidation, and any signs of recurrence or instability.
-
Rehabilitation
:
- Active Range of Motion (AROM) : Progressed to active motion exercises as tolerated, focusing on restoring functional range of motion.
- Light Strengthening : Isometric strengthening exercises for rotator cuff and deltoid.
- Progression : Gradually introduced light resistance exercises with elastic bands.
- Weight Bearing : Partial weight-bearing restrictions may be eased depending on radiographic and clinical assessment of stability.
Advanced Rehabilitation Phase (>12 weeks)
-
Rehabilitation
:
- Progressive Strengthening : Gradual increase in resistance and complexity of strengthening exercises.
- Functional Training : Sport-specific or occupation-specific training.
- Return to Activity : Return to light activities/sports usually by 4-6 months, with full unrestricted activity often allowed by 6-9 months, contingent on complete pain resolution, full range of motion, and radiographic evidence of bone remodeling and no recurrence.
- Follow-up : Long-term clinical and radiographic follow-up is crucial for ABCs, typically at 3, 6, 12 months, and then annually for several years to monitor for local recurrence, which can occur up to 5 years post-operatively.
Pearls & Pitfalls (Crucial for FRCS/Board Exams)
Pearls
- Diagnostic Challenge : Always consider Telangiectatic Osteosarcoma (TOS) in the differential for ABC, especially with pathological fracture or aggressive imaging features. Histological distinction is paramount.
- Fluid-Fluid Levels : While highly characteristic of ABC on MRI, fluid-fluid levels are not pathognomonic and can be seen in other lesions like TOS, simple bone cysts, chondroblastoma, fibrous dysplasia, and even metastatic disease.
- Meticulous Curettage : The cornerstone of ABC treatment. Inadequate curettage is the primary reason for local recurrence. A high-speed burr should be used to achieve clear margins.
- Adjuvant Therapy : Local adjuvants (e.g., cryotherapy, phenol, argon beam, radiofrequency ablation, hydrogen peroxide) are essential after curettage to destroy residual cells and significantly reduce recurrence rates, particularly for aggressive or recurrent lesions. PMMA cement also provides thermal necrosis.
- Structural Support : For large defects or pathological fractures, robust internal fixation combined with bone graft (autograft, allograft) and/or PMMA cement is crucial to prevent re-fracture and facilitate early mobilization.
- Biopsy Technique : Obtain multiple, representative samples from both solid and cystic areas of the lesion to avoid sampling error and ensure an accurate diagnosis.
- Recurrence Monitoring : ABCs have a significant recurrence rate, particularly in skeletally immature patients and after incomplete treatment. Long-term clinical and radiological surveillance is mandatory.
- Molecular Markers : Detection of USP6 gene rearrangement (a fusion gene characteristic of primary ABC) via FISH or RT-PCR can be a valuable diagnostic tool, especially in challenging or recurrent cases, or to differentiate from secondary ABCs.
Pitfalls
- Misdiagnosis : Confusing ABC with TOS is a critical error, leading to insufficient treatment for TOS (which requires neoadjuvant chemotherapy and wide resection) or overtreatment for ABC. Careful pathological review is non-negotiable.
- Incomplete Resection/Curettage : The most common pitfall leading to recurrence. Ensure all septa and cyst lining are thoroughly debrided, particularly in irregular cavities.
- Growth Plate Injury : In skeletally immature patients, aggressive curettage or adjuvant use (e.g., cryotherapy) near the physis can lead to growth arrest or deformity. Careful surgical planning and technique are required to minimize iatrogenic damage.
- Neurovascular Injury : Large or aggressive ABCs, particularly in locations like the proximal humerus, can abut or encase neurovascular structures. Careful dissection and clear visualization are paramount to avoid iatrogenic injury.
- Secondary ABC : Not all ABCs are primary; some develop secondary to other underlying bone lesions (e.g., fibrous dysplasia, chondroblastoma, GCT). If recurrence or an atypical presentation occurs, reconsider the possibility of a secondary ABC.
- Mechanical Failure : Insufficient internal fixation or poor choice of graft/cement can lead to hardware failure, non-union, or refracture, especially in weight-bearing or highly mobile bones.
- Ignoring Malignant Transformation : Although rare, malignant transformation of ABC has been reported, often following radiation therapy. A persistent or rapidly progressive recurrence warrants re-biopsy.
Clinical & Radiographic Imaging
You Might Also Like