of Musculoskeletal Tumors: Diagnostic and Surgical Strategies
Key Takeaway
The management of musculoskeletal tumors requires a rigorous, multidisciplinary approach. Diagnostic evaluation hinges on patient age, precise imaging, and systematic staging before any biopsy is performed. Surgical intervention ranges from intralesional curettage with local adjuvants to wide en bloc resection and complex limb salvage reconstruction. Strict adherence to oncologic principles ensures optimal local control and maximizes patient survival and functional outcomes.
Introduction to Musculoskeletal Oncology
The management of musculoskeletal tumors represents one of the most complex and high-stakes domains within orthopedic surgery. Optimal patient outcomes demand a highly coordinated, multidisciplinary team comprising an orthopedic oncologist, musculoskeletal radiologist, specialized pathologist, radiation oncologist, and medical oncologist. Depending on the anatomical location and extent of the lesion, the surgical team may also require the expertise of vascular, thoracic, or plastic surgeons to facilitate en bloc resections and complex soft-tissue reconstructions.
The orthopedic surgeon must be intimately versed in the principles of oncological surgery—ranging from the nuances of diagnostic imaging and biopsy tract placement to the biomechanics of limb salvage reconstruction.
Diagnostic Evaluation
The Clinical Assessment
An exhaustive history and physical examination remain the foundational steps in evaluating a patient with a suspected musculoskeletal neoplasm.
History:
Patients typically present with pain, a palpable mass, or an incidental radiographic finding.
* Pain Characteristics: While benign lesions may cause activity-related pain (often due to mechanical weakening of the bone), progressive pain that occurs at rest and awakens the patient at night is a hallmark red flag for bone malignancy. Notably, osteoid osteoma classically presents with severe night pain that is exquisitely responsive to NSAIDs.
* Age: Patient age is arguably the single most critical demographic data point. Most benign and malignant musculoskeletal neoplasms exhibit strict age predilections, which drastically narrows the differential diagnosis.
* Genetics and Demographics: Family history is vital in conditions like multiple hereditary exostoses (MHE) or neurofibromatosis. Race is rarely diagnostic, though Ewing sarcoma is exceptionally rare in patients of African descent.
Physical Examination:
The examination must document the mass's size, location, shape, consistency, mobility, and local temperature. The surgeon must assess for regional muscle atrophy, neurological deficits, and vascular compromise.
* Lymphadenopathy: While lymph node metastases are generally rare in soft-tissue sarcomas, they are notably present in specific subtypes: rhabdomyosarcoma, epithelioid sarcoma, clear cell sarcoma, and synovial sarcoma.
* Cutaneous Markers: Café-au-lait spots may indicate neurofibromatosis, while cutaneous hemangiomas can suggest underlying vascular malformations or syndromes (e.g., Maffucci syndrome).
Radiographic and Imaging Principles
Conventional radiography remains the most critical initial diagnostic test for evaluating bone lesions. The biological aggressiveness of a tumor can often be deduced from its radiographic margins. Inactive benign lesions typically exhibit well-marginated borders with a sclerotic rim of reactive bone. Conversely, aggressive lesions demonstrate a wide, ill-defined zone of transition, cortical destruction, and aggressive periosteal reactions (e.g., Codman’s triangle, "onion-skinning," or "sunburst" patterns).
Clinical Pearl: Never order advanced imaging (MRI/CT) without first obtaining high-quality, orthogonal plain radiographs of the entire involved bone. Plain films provide unparalleled information regarding the lesion's matrix (osteoid vs. chondroid) and its effect on host bone.
Site-Specific Differential Diagnoses
The anatomical location of the lesion within the bone is highly predictive of its histology:
- Epiphyseal Lesions:
- Chondroblastoma (Age 10–25)
- Giant Cell Tumor (Age 20–40, skeletally mature)
- Clear Cell Chondrosarcoma (Rare)
- Diaphyseal Lesions:
- Ewing Sarcoma (Age 5–25)
- Lymphoma (Adults)
- Fibrous Dysplasia (Age 5–30)
- Adamantinoma (Predominantly in the tibia)
- Langerhans Cell Histiocytosis (Age 5–30)
- Spinal Lesions (Older than 40 Years):
- Metastatic Carcinoma
- Multiple Myeloma
- Hemangioma
- Chordoma (Sacrum)
- Spinal Lesions (Younger than 30 Years):
- Vertebral Body: Histiocytosis, Hemangioma
- Posterior Elements: Osteoid Osteoma, Osteoblastoma, Aneurysmal Bone Cyst (ABC)
- Multiple Lesions:
- Histiocytosis, Enchondromatosis, Osteochondromatosis, Fibrous Dysplasia, Multiple Myeloma, Metastases, Hyperparathyroidism (Brown tumors).
Advanced Imaging Modalities
- Magnetic Resonance Imaging (MRI): The gold standard for local staging. MRI accurately delineates the intraosseous and extraosseous extent of the tumor, skip metastases, and the relationship of the tumor mass to critical neurovascular bundles. Any soft-tissue mass deep to the fascia or larger than 5 cm must be treated as a sarcoma until proven otherwise.
- Computed Tomography (CT): Superior for assessing subtle cortical erosion, matrix ossification/calcification, and localizing the nidus of an osteoid osteoma. A non-contrast CT of the chest is mandatory for detecting pulmonary metastases.
- Technetium-99m Bone Scans: Utilized to detect polyostotic disease or skeletal metastases. Note that bone scans may be falsely negative in multiple myeloma and certain aggressive renal cell carcinomas due to a lack of osteoblastic response.
- Positron Emission Tomography (PET): Increasingly utilized for systemic staging, guiding biopsy to the most metabolically active region, and monitoring neoadjuvant chemotherapy response.
Laboratory Investigations and Nutritional Optimization
While rarely diagnostic in isolation, laboratory tests guide the systemic evaluation.
* Nutritional Status: Serum albumin < 3.5 g/dL or total lymphocyte count < 1500/mL significantly increases the risk of postoperative wound complications and surgical site infections.
* Tumor Markers & Systemic Labs: Elevated ESR is seen in infection, metastases, and small round blue cell tumors (Ewing, lymphoma). Serum protein electrophoresis (SPEP) and urine protein electrophoresis (UPEP) are mandatory if myeloma is suspected. Prostate-specific antigen (PSA) is required for older males. Hypercalcemia suggests metastatic disease, myeloma, or hyperparathyroidism.
Metastases of Unknown Origin
In patients over the age of 40 presenting with a new, painful, destructive bone lesion, metastatic carcinoma and multiple myeloma are the most likely diagnoses, even in the absence of a known primary malignancy. The most common primary sources for skeletal metastases are breast, prostate, lung, kidney, and thyroid carcinomas. However, when the primary is unknown at presentation, lung and renal cell carcinomas are the most frequent culprits.
The Rougraff Protocol
Rougraff et al. established a highly effective, systematic algorithm for evaluating suspected metastases of unknown origin. Crucially, a biopsy must not be performed until this evaluation is complete. Premature biopsy can compromise limb salvage if the lesion is actually a primary bone sarcoma, or lead to catastrophic hemorrhage if the lesion is a hypervascular renal or thyroid metastasis.
The evaluation includes:
1. Comprehensive history and physical exam (including thyroid, breasts, prostate, and lymph nodes).
2. Laboratory analysis: CBC, ESR, comprehensive metabolic panel, LFTs, alkaline phosphatase, SPEP, and PSA.
3. Plain radiographs of the involved bone and chest.
4. Whole-body Technetium-99m bone scan.
5. CT scan of the chest, abdomen, and pelvis.
Surgical Warning: Routine mammography is not indicated as an initial screening tool in this specific scenario, as breast cancer rarely presents with skeletal metastases without a previously known primary breast mass.
Clinical Case Example: Metastatic Renal Cell Carcinoma
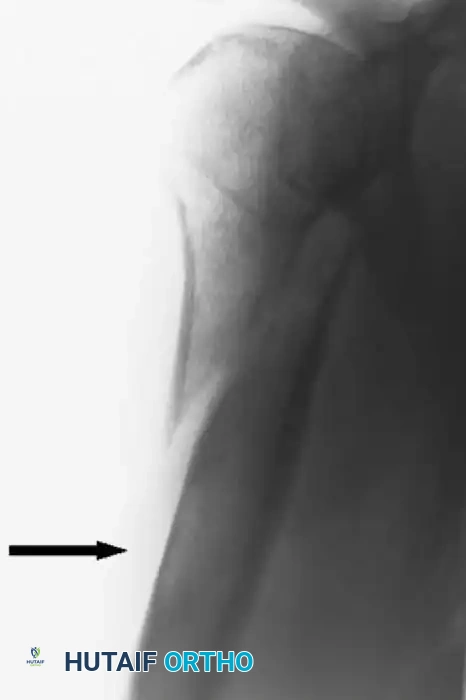
Consider the case of an 81-year-old male presenting with a humeral fracture after minimal trauma, with no known history of malignancy.

Figure 1A: Initial radiograph demonstrating a subtle, ill-defined radiolucency in the proximal humerus. The lesion was initially missed, and the patient was treated conservatively.

Figure 1B: Radiograph 10 weeks post-injury showing rapid, aggressive progression of the malignant process with extensive cortical destruction and pathological fracture.
Following referral to an orthopedic oncology center, a full staging workup was initiated. A bone scan revealed multiple sites of disease, and a CT of the abdomen was performed.
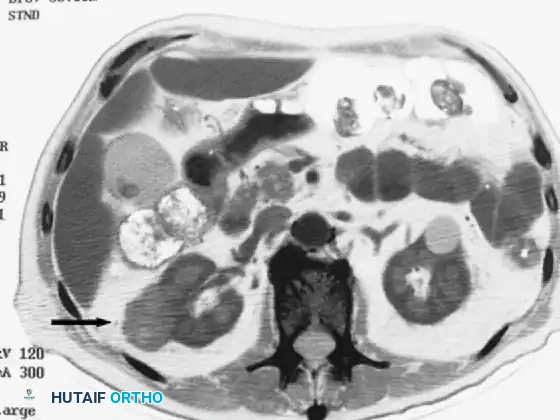
Figure 1C: Axial CT of the abdomen revealing a large, heterogeneous mass in the right kidney (arrow), confirming the primary source as renal cell carcinoma.
This case underscores the necessity of the Rougraff protocol. Had a biopsy been performed immediately on the humerus without prior imaging, the surgeon would have encountered massive, potentially life-threatening hemorrhage typical of renal cell metastases.
Principles of Biopsy
The biopsy is the final, definitive step in the diagnostic workup. A poorly planned biopsy can alter the surgical staging, necessitate a larger resection, or even convert a limb-salvage candidate into an amputee.
Core Biopsy Principles:
1. Placement: The biopsy tract must be placed perfectly in line with the planned definitive surgical incision. The entire tract (skin to bone) is considered contaminated and must be excised en bloc with the tumor during definitive resection.
2. Approach: Always use a longitudinal incision. Transverse incisions contaminate multiple muscle compartments and complicate future flap coverage.
3. Compartmentalization: Do not cross fascial planes unnecessarily. Go directly through the muscle belly rather than between muscles to avoid contaminating intermuscular planes.
4. Hemostasis: Meticulous hemostasis is mandatory. A postoperative hematoma disseminates tumor cells throughout the compartment. If a drain is used, it must exit directly in line with the surgical incision, close to the wound, so the drain tract can also be excised.
5. Cortical Windows: If the bone must be fenestrated to access a medullary lesion, the window should be round or oval to minimize stress risers, and the defect should be plugged with bone wax or PMMA to prevent tumor spillage into the soft tissues.
Surgical Techniques: Curettage and Local Adjuvants
Intralesional curettage is the workhorse procedure for benign and locally aggressive bone tumors (e.g., Giant Cell Tumor, Aneurysmal Bone Cyst, Chondroblastoma). The goal is macroscopic removal of the tumor while preserving the structural integrity of the host bone.
Step-by-Step Surgical Approach for Extended Curettage
- Positioning and Exposure: The patient is positioned to allow full access to the lesion. A tourniquet is utilized to minimize blood loss and provide a clear visual field, but the limb is not exsanguinated with an Esmarch bandage to prevent systemic embolization of tumor cells; instead, the limb is elevated for 3 minutes prior to inflation.
- Cortical Window Creation: A high-speed burr or osteotome is used to create a cortical window large enough to visualize the entire cavity.
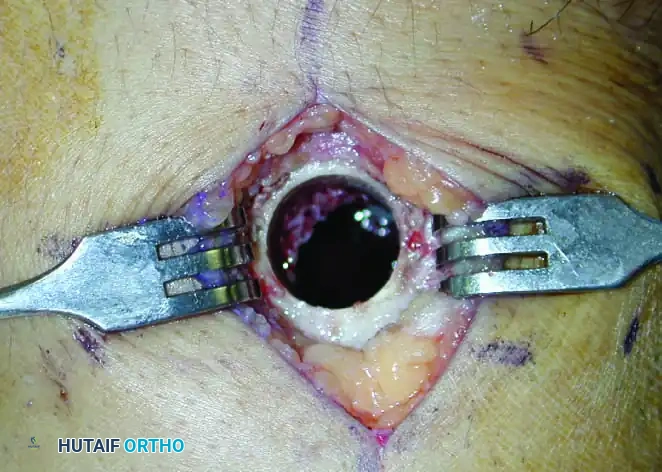
Figure 2: Intraoperative photograph demonstrating a meticulously prepared circular cortical window. Note the smooth edges designed to minimize stress risers and prevent iatrogenic fracture.
- Intralesional Excision: Using a series of progressively sized curettes, the tumor is systematically evacuated. The surgeon must meticulously scrape the walls of the cavity until normal, healthy cancellous bone is encountered.
- High-Speed Burring: A high-speed burr is then used to extend the curettage by 2–3 mm into the reactive bone rim. This breaks down the microscopic ridges where tumor cells hide.
- Chemical/Thermal Adjuvants: To further reduce local recurrence rates, adjuvants are applied to the cavity walls:
- Phenol (89%): Causes chemical necrosis. Must be neutralized with alcohol.
- Liquid Nitrogen: Causes thermal necrosis via freeze-thaw cycles. Carries a risk of postoperative fracture.
- Argon Beam Coagulation: Provides superficial thermal necrosis and excellent hemostasis.
- Reconstruction: The defect is typically packed with Polymethylmethacrylate (PMMA) bone cement.
- Biomechanics of PMMA: PMMA provides immediate structural stability and high compressive strength, allowing for early weight-bearing. Furthermore, the exothermic reaction of curing PMMA generates local temperatures up to 70°C, providing an additional layer of thermal necrosis to any remaining microscopic tumor cells.
Principles of Resection and Reconstruction
For malignant bone and soft-tissue sarcomas, the primary goal is complete eradication of the tumor with negative margins, prioritizing patient survival over limb preservation.
Surgical Margins (Enneking System)
- Intralesional: The dissection passes directly through the tumor. Leaves macroscopic disease. (Acceptable only for benign lesions).
- Marginal: The dissection passes through the reactive zone/pseudocapsule. Leaves microscopic satellite lesions.
- Wide: The dissection passes entirely through normal, healthy tissue outside the reactive zone. The tumor is removed en bloc with a cuff of normal tissue. This is the standard of care for sarcomas.
- Radical: The entire anatomical compartment containing the tumor is removed from origin to insertion.
Amputation versus Limb Salvage
Advancements in neoadjuvant chemotherapy, advanced imaging, and modular endoprosthetics have made limb salvage possible in 80-90% of extremity sarcomas.
Absolute Indications for Amputation:
1. Inability to achieve wide surgical margins without sacrificing critical neurovascular structures that cannot be reconstructed.
2. A non-functional limb post-resection (e.g., loss of the sciatic nerve in a distal femur resection often results in a limb functionally inferior to a modern prosthesis).
3. Severe infection or fungating tumor mass precluding safe soft-tissue coverage.
Reconstruction Options in Limb Salvage:
When wide resection is achieved, the resulting skeletal defect must be reconstructed. Options include:
* Modular Megaprostheses: Provide immediate stability and early mobilization. Subject to long-term aseptic loosening and mechanical failure, especially in young, active patients.
* Osteoarticular Allografts: Biological reconstruction. High rates of non-union, infection, and late fracture.
* Allograft-Prosthesis Composites (APC): Combines the biological soft-tissue attachment of an allograft with the articular durability of a prosthesis.
* Vascularized Fibular Grafts: Often used in conjunction with massive allografts (the "hot dog" technique) to promote biological union and hypertrophy over time.
Postoperative Protocols and Rehabilitation
Postoperative management in orthopedic oncology is highly individualized based on the extent of resection and the type of reconstruction.
* Wound Care: Meticulous attention to the surgical incision is paramount, especially if the patient is scheduled to receive postoperative radiation or chemotherapy, which severely impairs wound healing. Flap viability must be monitored closely.
* DVT Prophylaxis: Cancer patients are inherently hypercoagulable. Extended chemical prophylaxis (e.g., LMWH) is mandatory unless contraindicated by bleeding risks.
* Weight-Bearing: Patients with cemented megaprostheses are typically allowed immediate weight-bearing as tolerated. Patients with biological reconstructions (allografts/fibular grafts) require strict non-weight-bearing or protected weight-bearing for 3 to 6 months until radiographic union is confirmed.
* Surveillance: Routine oncologic surveillance (local MRI, chest CT) is instituted based on the tumor's grade and histology to monitor for local recurrence and distant metastasis.
By adhering strictly to these diagnostic and surgical principles, the orthopedic oncologist can navigate the complexities of musculoskeletal tumors, ensuring the highest probability of oncologic clearance while maximizing the patient's functional independence.
📚 Medical References
- musculoskeletal tumors for cytodiagnosis and DNA analysis, Acta Orthop Scand 58:523, 1987.
- Alexander AH: Thoracolumbar needle biopsy, Orthopedics 11:1473, 1988.
- Alho A, Karaharju EO, Korkala O, et al: Allogeneic grafts for bone tumor: 21 cases of osteoarticular and segmental grafts, Acta Orthop Scand 60:143, 1989.
- Alman BA, De Bari A, Krajbich JI: Massive allografts in the treatment of osteosarcoma and
You Might Also Like