Chondroblastoma: Comprehensive Pathology, Diagnosis, and Surgical Management
Key Takeaway
Chondroblastoma is a rare, benign cartilaginous neoplasm characteristically arising in the epiphyses of long bones in adolescents and young adults. Presenting with joint pain mimicking synovitis, it requires meticulous diagnostic imaging to differentiate from giant cell tumors. Standard treatment involves extended intralesional curettage with mechanical and chemical adjuvants, followed by defect reconstruction using bone graft or polymethylmethacrylate (PMMA) cement to minimize recurrence while preserving joint congruity.
Introduction and Epidemiology
Chondroblastoma, historically referred to as Codman’s tumor, is a rare, benign, but locally aggressive cartilaginous neoplasm. According to extensive institutional series, including data from the Mayo Clinic, this tumor represents approximately 1% of all primary bone tumors. It exhibits a striking predilection for the epiphyses or apophyses of long bones, making its anatomical location a critical component of its diagnostic profile.
The demographic distribution heavily favors adolescents and young adults, typically presenting in patients between 10 and 25 years of age. There is a documented 2:1 male predominance. The most frequent sites of occurrence include the distal femur, proximal tibia, and proximal humerus. While long bone epiphyses are the classic locations, chondroblastoma can also arise in apophyseal regions such as the greater trochanter of the femur or the greater tuberosity of the humerus. In older patient populations, the tumor demonstrates an atypical tendency to manifest in flat bones, including the talus, calcaneus, and pelvis. Multicentric or polyostotic disease is exceedingly rare and should prompt a broader differential diagnosis or genetic workup.
Clinical Presentation
The clinical presentation of chondroblastoma is often insidious, leading to delayed diagnosis. Most patients complain of progressive, deep-seated pain that is exacerbated by activity and weight-bearing. Because the tumor is almost exclusively epiphyseal, the pain is frequently referred to the adjacent joint.
Clinical Pearl: A young patient presenting with chronic, unexplained joint pain, a persistent effusion, and symptoms mimicking chronic synovitis, meniscal pathology, or other intra-articular derangements must be evaluated for an underlying epiphyseal bone lesion.
Physical examination may reveal localized tenderness, a palpable mass (rarely, unless there is significant cortical breach), muscle atrophy secondary to disuse, and restricted range of motion in the affected joint. Soft-tissue extension is extremely rare in primary chondroblastoma, distinguishing it from more aggressive malignancies.
Diagnostic Imaging
Radiographic evaluation is paramount in the diagnosis and surgical planning of epiphyseal lesions. The imaging profile of chondroblastoma is highly characteristic, though it shares features with other aggressive benign tumors.
Plain Radiography
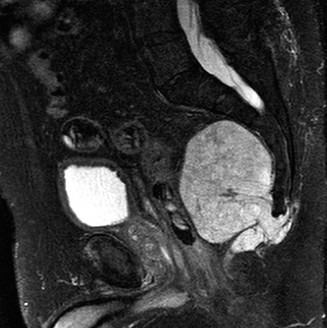
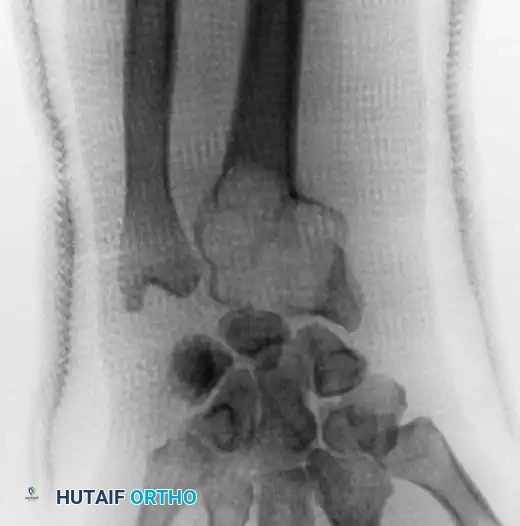
On plain radiographs, chondroblastoma typically appears as a well-circumscribed, eccentrically or centrally located lytic lesion confined to the epiphysis or apophysis. A defining feature is the presence of a surrounding rim of reactive sclerotic bone. Intralesional calcification—often described as a stippled or "fluffy" chondroid matrix—is visible on plain films in 30% to 50% of cases.

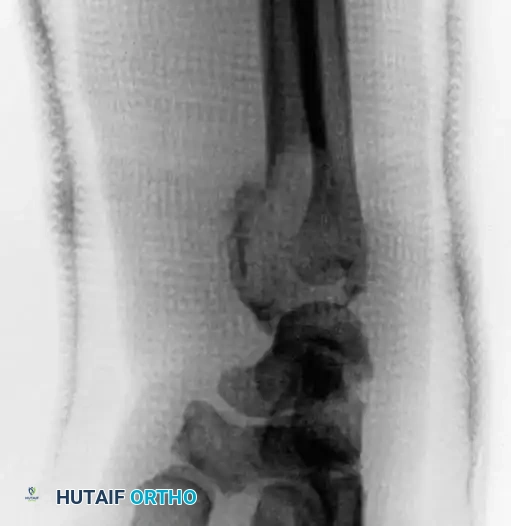
In skeletally immature patients, a well-circumscribed epiphyseal lesion that crosses an open physis (growth plate) into the metaphysis is virtually diagnostic of chondroblastoma.
Advanced Imaging (MRI and CT)
Magnetic Resonance Imaging (MRI) is essential for evaluating the extent of the lesion, assessing articular cartilage integrity, and identifying secondary aneurysmal bone cysts (ABCs), which are present in up to 20% of patients. MRI typically reveals extensive bone marrow edema surrounding the lesion, which can be disproportionate to the tumor's size, further explaining the severe joint pain.
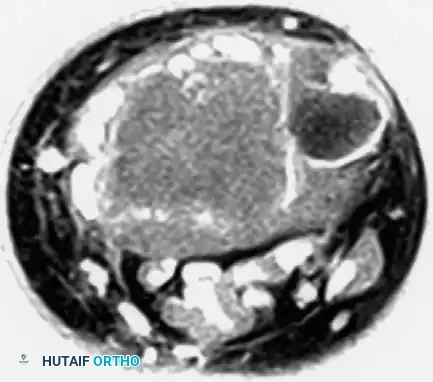
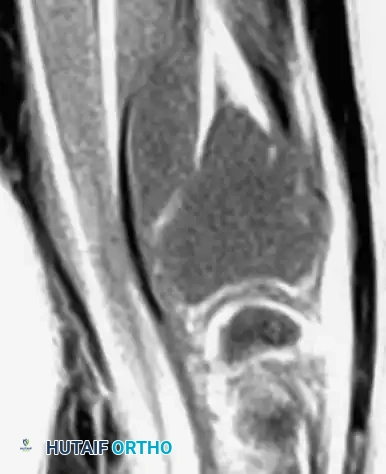
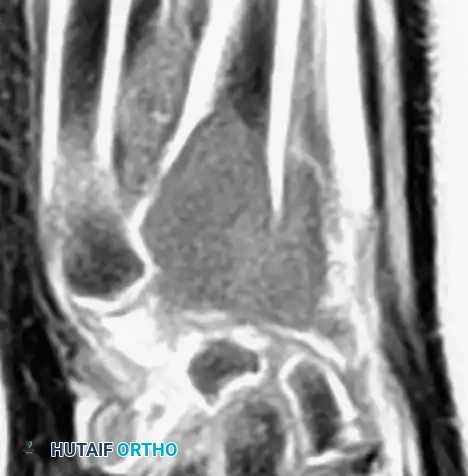
Computed Tomography (CT) is highly sensitive for detecting subtle intralesional calcifications and delineating the exact three-dimensional anatomy of the cortical margins, which is critical for planning the surgical approach and cortical window.
Histopathology and Differential Diagnosis
Microscopic Features
The definitive diagnosis of chondroblastoma relies on histopathological examination. Macroscopically, the tissue is gritty and grayish-pink. Microscopically, the tumor is highly cellular, composed of sheets of mononuclear cells (chondroblasts) set within a background of eosinophilic chondroid matrix.
The chondroblasts are polygonal with distinct cytoplasmic borders, eosinophilic cytoplasm, and characteristic longitudinally clefted ("coffee bean") nuclei. A pathognomonic feature is the presence of fine, pericellular network calcifications that surround individual chondroblasts, creating the classic "chicken wire" appearance. Multinucleated osteoclast-like giant cells are abundant and often scattered throughout the lesion.
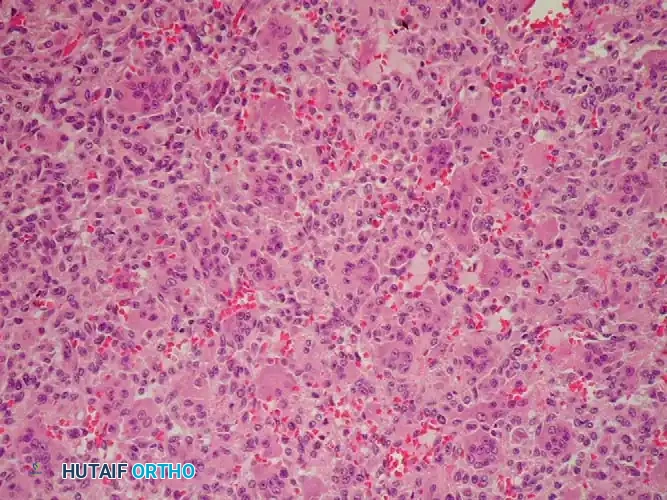
Differential Diagnosis
In adult patients, the differential diagnosis for an epiphyseal lytic lesion includes:
1. Giant Cell Tumor (GCT): Unlike chondroblastoma, GCTs typically lack a sclerotic reactive rim, do not exhibit intralesional calcification, and frequently have a soft-tissue component due to cortical destruction. However, because chondroblastomas contain abundant giant cells, distinguishing the two requires careful identification of the mononuclear cell morphology and chondroid matrix.
2. Clear Cell Chondrosarcoma: A low-grade malignancy that also occurs in the epiphysis but typically affects older adults (30-50 years) and features cells with abundant clear cytoplasm.
3. Infection (Brodie's Abscess): Can present with a sclerotic rim and extensive edema, but lacks neoplastic cellularity.
Surgical Indications and Preoperative Planning
Chondroblastomas are typically classified as Enneking Stage 2 (active, benign) and more rarely as Stage 3 (aggressive, benign). They are generally less locally destructive than giant cell tumors.
The primary goal of treatment is complete eradication of the tumor to prevent local recurrence, preservation of joint congruity, and restoration of bone stock.
Surgical Warning: While sparing the open growth plate in a pediatric patient is highly desirable to prevent angular deformity or limb length discrepancy, adequate and aggressive intralesional curettage must always take precedence over physeal preservation. Incomplete curettage guarantees recurrence, which will ultimately cause greater physeal and articular destruction.
Operative Technique: Extended Intralesional Curettage
The gold standard treatment for Enneking Stage 2 chondroblastoma is extended intralesional curettage supplemented with local adjuvants and defect reconstruction.
1. Positioning and Approach
The patient is positioned based on the anatomical location of the tumor. A tourniquet is utilized to provide a bloodless field, which is critical for visualizing the tumor margins and ensuring complete curettage.
* Proximal Humerus: Deltopectoral approach.
* Distal Femur: Lateral or medial subvastus approach, depending on the lesion's eccentricity.
* Proximal Tibia: Anterolateral or anteromedial approach.
2. Cortical Windowing
Under fluoroscopic guidance, a cortical window is created. The window must be large enough to allow visualization of the entire cavity. A high-speed burr or osteotome is used to remove the overlying cortex. If the cortex is uninvolved, it can be elevated as a trapdoor and preserved for later closure.
3. Intralesional Excision and Adjuvant Therapy
The gross tumor is removed using a series of angled and straight curettes. Because chondroblastoma can have a firm, gritty texture, aggressive scraping is required.
Once gross tumor is removed, the cavity undergoes extended curettage. A high-speed mechanical burr is used to break down the reactive sclerotic rim, extending the margin by 1 to 2 millimeters into normal cancellous bone. This step is vital to eradicate microscopic disease extending into the surrounding trabeculae.
Following mechanical burring, chemical or thermal adjuvants are applied:
* Phenol (followed by alcohol neutralization): Causes protein coagulation and cell death.
* Hydrogen Peroxide: Provides mechanical bubbling to clear debris and mild chemical cytotoxicity.
* Argon Beam Coagulation: Delivers uniform thermal necrosis to the cavity walls.
4. Defect Reconstruction
The resulting cavitary defect must be filled to support the articular surface and prevent pathological fracture.
* Bone Grafting: Autograft (e.g., iliac crest) or allograft chips are packed tightly into the defect. This is preferred in pediatric patients to allow biological incorporation and remodeling, especially near an open physis.
* Polymethylmethacrylate (PMMA): Bone cement provides immediate structural stability, allows for early weight-bearing, and generates an exothermic reaction that acts as an additional thermal adjuvant. Furthermore, the radiopaque nature of PMMA makes postoperative surveillance for local recurrence (which appears as a radiolucent halo around the cement) much easier.
Operative Technique: En Bloc Resection and Reconstruction
While extended curettage is standard, Enneking Stage 3 lesions, massive recurrences, or aggressive epiphyseal tumors (such as Giant Cell Tumors that heavily destroy the articular surface) may necessitate en bloc resection. When the subchondral bone is entirely destroyed, joint salvage becomes impossible.
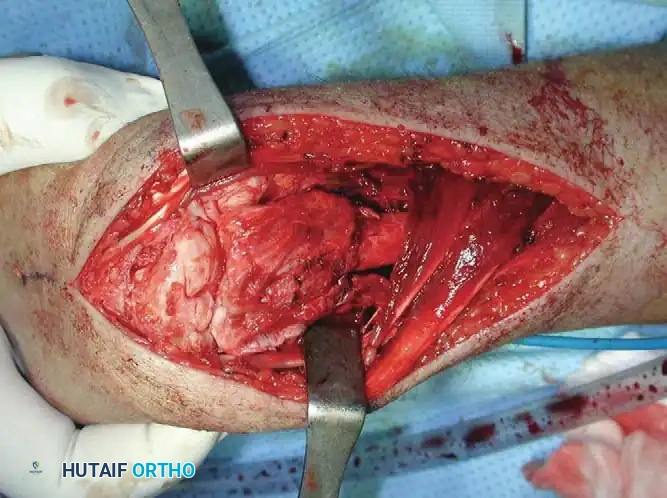
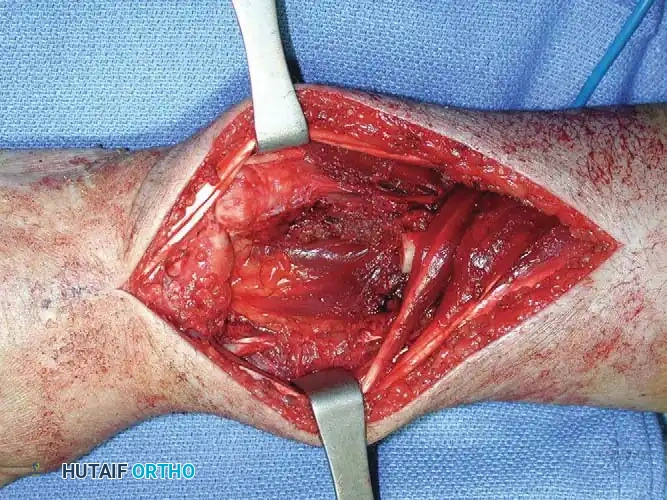
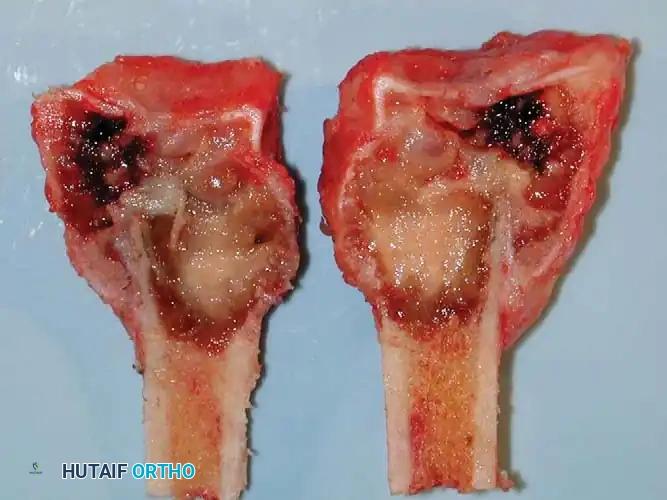
Resection and Structural Grafting
In cases involving the distal radius (a common site for aggressive epiphyseal tumors like GCT), en bloc resection of the distal radius is performed. The tumor is excised with wide margins, sacrificing the radiocarpal joint.
To reconstruct the defect and restore forearm length, a structural autograft is harvested. The contralateral proximal fibula is an excellent anatomical match for the distal radius.
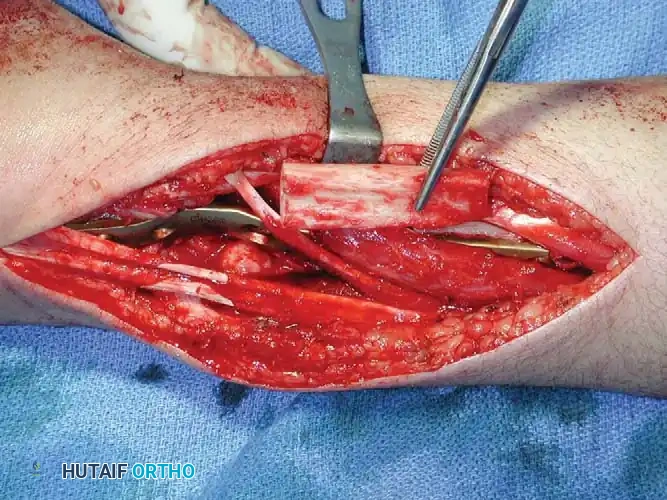
The fibular autograft is contoured and impacted into the medullary canal of the remaining radial diaphysis. A rigid wrist fusion plate is then applied dorsally, spanning from the radial shaft, across the fibular graft, and securing to the carpal bones (typically the capitate and third metacarpal) to achieve a stable radiocarpal arthrodesis.
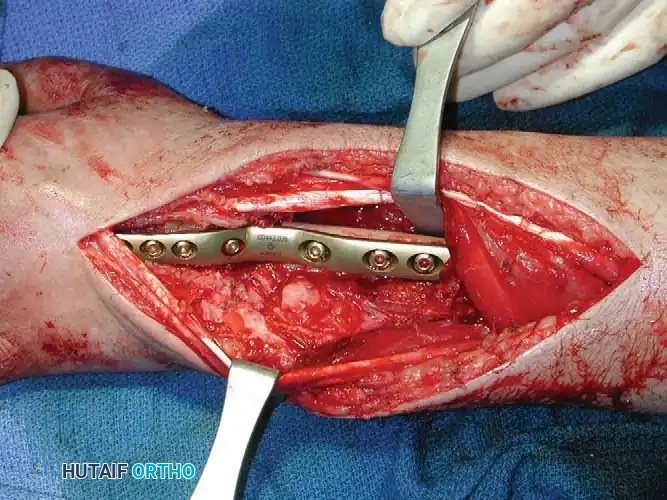
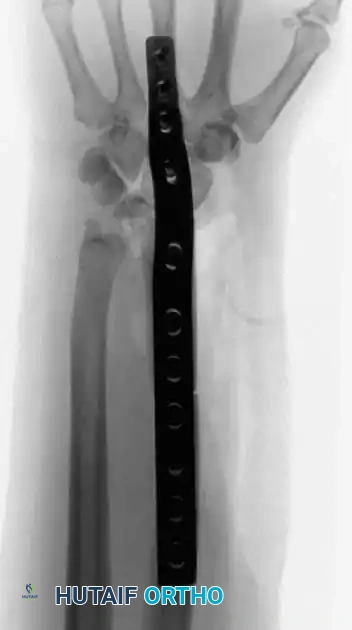
Management of Axial and Inoperable Lesions
When epiphyseal-type tumors arise in the axial skeleton, such as the sacrum, surgical morbidity increases exponentially. Resection of sacral lesions can result in massive hemorrhage, loss of bowel/bladder control, and severe spinopelvic instability.
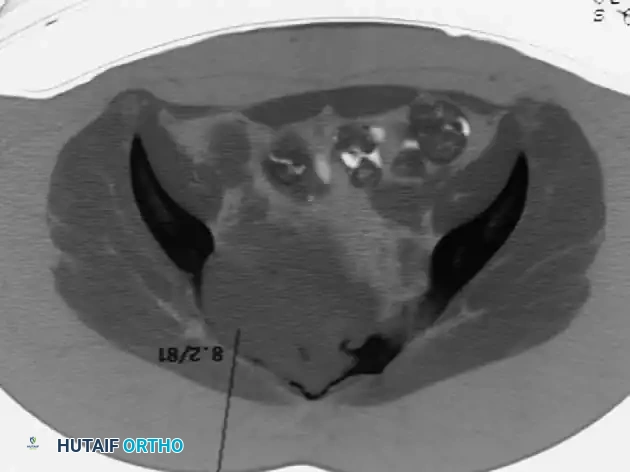
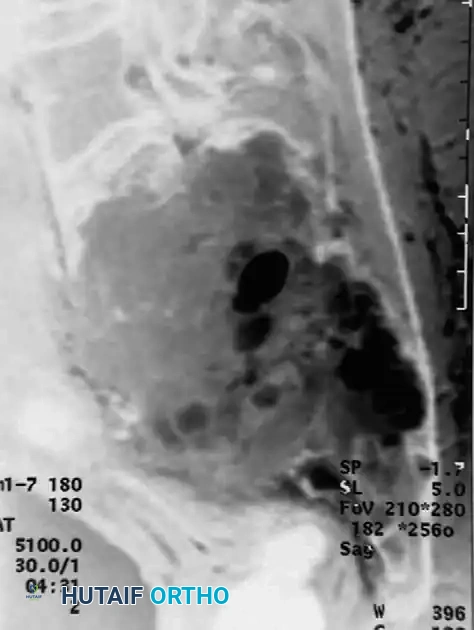
For large, destructive lesions in the sacrum (as seen in aggressive GCTs or rare axial chondroblastomas), CT-guided core needle biopsy is mandatory to confirm the diagnosis. If the morbidity of surgical extirpation is deemed unacceptably high, patients are referred for definitive radiation therapy. Serial arterial embolization may also be employed to reduce tumor volume and alleviate pain.
Postoperative Protocol and Rehabilitation
Postoperative management is dictated by the anatomical site and the method of reconstruction.
* Curettage and PMMA: Patients can often be allowed immediate weight-bearing as tolerated, given the high compressive strength of the cement. Early range of motion (ROM) is encouraged to prevent joint stiffness.
* Curettage and Bone Graft: Patients are typically restricted to protected, non-weight-bearing or partial weight-bearing status for 6 to 12 weeks until radiographic evidence of graft incorporation is observed.
* En Bloc Resection and Arthrodesis: The limb is immobilized in a splint or cast for 2 to 4 weeks to allow soft tissue healing, followed by progressive rehabilitation of adjacent non-fused joints. Heavy lifting is restricted until complete bony union of the structural graft is achieved.
Complications and Oncologic Surveillance
Local Recurrence
The local recurrence rate for chondroblastoma ranges from 10% to 20%, largely dependent on the adequacy of the initial curettage. Recurrences typically present within the first 2 to 3 years postoperatively.
Pitfall: Failure to adequately burr the reactive sclerotic rim or address a secondary aneurysmal bone cyst component significantly increases the risk of local recurrence.
Recurrent lesions are generally treated with a repeat extended curettage and bone grafting or cementation, similar to the primary lesion.
Pulmonary Metastases
Despite its benign histological classification, chondroblastoma has the capacity to metastasize to the lungs in approximately 1% of patients. These "benign pulmonary metastases" are histologically identical to the primary tumor and do not imply malignant transformation. They are typically slow-growing and should be treated with surgical wedge resection (metastasectomy), which is often curative. True malignant transformation of a chondroblastoma into a chondrosarcoma is exceedingly rare and is usually associated with prior radiation therapy.
Surveillance Protocol
Rigorous oncologic surveillance is mandatory. Radiographs of the primary surgical site and cross-sectional imaging (CT) or radiographs of the chest should be obtained every 6 months for at least the first 3 years postoperatively. Following this high-risk period, surveillance should continue on an annual basis for up to 10 years to monitor for late recurrences or delayed pulmonary involvement.
📚 Medical References
- arising from a nonepiphyseal site, Skeletal Radiol 24:220, 1995.
- Brien EW, Mirra JM, Kerr R: Benign and malignant cartilage tumors of bone and joint: their anatomic and theoretical basis with an emphasis on radiology, pathology and clinical biology: 1. the intramedullary cartilage tumors, Skeletal Radiol 26:325, 1997.
- Brien EW, Mirra JM, Kessler S, et al: Benign giant cell tumor of bone with osteosarcomatous transformation (“dedifferentiated” primary malignant GCT): report of two cases, Skeletal Radiol 26:246, 1997.
- Brigman BE, Kumagai SG, McGuire MH: Rotationplasty after failed limb-sparing tumor surgery: a report of two cases, Clin Orthop Relat Res 415:254, 2003.
- Brostrom LA, Strander H, Nilsonne U: Survival in osteosarcoma in relation to tumor size and location, Clin Orthop Relat Res 167:250, 1982.
- Bullough PG: Bullough and Vigorita’s orthopaedic pathology, 3rd ed, London, 1997, Mosby-Wolfe. Burk CD, Belasco JB, O’Neill JA, et al: Pulmonary metastases and bone sarcomas: surgical removal of lesions appearing after adjuvant chemotherapy, Clin Orthop Relat Res 262:88, 1991.
- Butler MS, Roberson WW, Rate W, et al: Skeletal sequelae of radiation therapy for malignant childhood tumors, Clin Orthop Relat Res 251:235, 1990.
- Callaghan JJ, Salvati EA, Pellicci PM, et al: Evaluation of benign acetabular lesions with excision through the Ludloff approach, Clin Orthop Relat Res 237:170, 1988.
- Campanacci M, Baldini N, Boriani S, et al: Giant-cell tumor of bone, J Bone Joint Surg 69A:106, 1987.
- Campanacci M, Capanna R, Picci P: Unicameral and aneurysmal bone cysts, Clin Orthop Relat Res 204:25, 1986.
- Capanna R, Biagini R, Ruggieri P, et al: Temporary resectionarthrodesis of the knee using an intramedullary rod and bone cement, Int Orthop 13:253, 1989.
- Cara JA, Canadell J: Limb salvage for malignant bone tumors in young children, J Pediatr Orthop 14:112, 1994.
- Carli M, Colombatti R, Oberlin O, et al: High-dose melphalan with autologous stem-cell rescue in metastatic rhabdomyosarcoma, J Clin Oncol 17:2796, 1999.
- Carli M, Colombatti R, Oberlin O, et al: European intergroup studies (mmt4-89 and mmt4-91) on childhood metastatic rhabdomyosarcoma: fi nal results and analysis of prognostic factors, J Clin Oncol 22:4735, 2004.
- Carrell WB: Transplantation of the fi bula in the same leg, J Bone Joint Surg 20:627, 1938.
- Casadei R, Ruggieri P, Moscato M, et al: Aneurysmal bone cyst and giant cell tumor of the foot, Foot Ankle Int 17:487, 1996.
- Caterini R, Manili M, Spinelli M, et al: Epiphyseal chondroblastoma of bone: long-term effects on skeletal growth and articular function in 15 cases treated surgically, Arch Orthop Trauma Surg 111:327, 1992.
- Cha S, Schultz E, McHeffey-Atkinson B, et al: Malignant lymphoma involving the pelvis, Skeletal Radiol 25:783, 1996.
- Chakravarti A, Spiro IJ, Hug EB, et al: Megavoltage radiation therapy for axial and inoperable giant-cell tumor of bone, J Bone Joint Surg 81A:1566, 1999.
- Chandrasekhar B, Sloan GM, Beatty JD: The external oblique myocutaneous fl ap for extended
You Might Also Like