Cartilage Lesions: Comprehensive Surgical Management of Chondromas and Osteochondromas
Key Takeaway
Benign cartilage lesions, including chondromas and osteochondromas, frequently present as incidental radiographic findings but can cause pathologic fractures or neurovascular compromise. Surgical management requires a meticulous approach to differentiate benign pathology from chondrosarcoma. This guide details the indications, advanced imaging criteria, and step-by-step operative techniques—such as extended curettage for enchondromas and en bloc resection for osteochondromas—ensuring optimal outcomes and minimizing recurrence in orthopedic oncology.
CARTILAGE LESIONS: AN ACADEMIC OVERVIEW
Benign cartilaginous tumors represent a significant proportion of osseous neoplasms encountered in orthopedic practice. While frequently asymptomatic and discovered incidentally, these lesions demand rigorous clinical and radiographic evaluation to exclude malignant transformation, particularly to secondary chondrosarcoma. The two most prevalent entities in this category are chondromas (including enchondromas and periosteal chondromas) and osteochondromas.
This masterclass provides an exhaustive, evidence-based framework for the diagnosis, biomechanical considerations, and surgical management of these cartilage lesions, tailored for the practicing orthopedic oncologist, consultant surgeon, and senior resident.

CHONDROMA
Chondromas are benign, hamartomatous lesions composed of mature hyaline cartilage. They can affect individuals across all age groups and may arise in any bone preformed in cartilage. Epidemiologically, they are the most common primary bone tumor of the small tubular bones of the hands and feet, particularly the phalanges.
Depending on their anatomical location relative to the cortex, they are classified into:
* Enchondromas: Arising centrally within the medullary canal.
* Periosteal (Juxtacortical) Chondromas: Arising on the surface of the bone, deep to the periosteum.
Pathophysiology and Syndromic Associations
Enchondromas are believed to originate from displaced fragments of physeal cartilage that escape normal endochondral ossification and proliferate within the metaphysis or diaphysis.
While most enchondromas are solitary, multiple enchondromatosis is a recognized clinical entity:
* Ollier Disease: A rare, non-hereditary condition characterized by multiple enchondromas distributed asymmetrically within the large and small tubular bones, as well as flat bones. The profound failure of normal endochondral ossification leads to severe skeletal deformities, including limb shortening (due to lack of epiphyseal growth), metaphyseal broadening, and bowing of long bones.
* Maffucci Syndrome: A variant of multiple enchondromatosis associated with multiple soft-tissue hemangiomas (spindle cell hemangioendotheliomas).
Clinical Pearl: The risk of malignant transformation to chondrosarcoma is significantly elevated in syndromic presentations. Approximately 25% of patients with Ollier disease will develop a sarcoma by age 40. Patients with Maffucci syndrome carry an even higher lifetime risk of malignant transformation, approaching 100% when considering both osseous and extra-osseous malignancies.
Clinical Presentation and Diagnostics
Solitary enchondromas are overwhelmingly asymptomatic. They are typically discovered incidentally during radiographic evaluation for unrelated trauma or following a pathological fracture through the structurally weakened bone.
Radiographic Evaluation
Plain radiographs are the cornerstone of diagnosis. Enchondromas present as well-circumscribed, benign-appearing radiolucent lesions with characteristic intralesional calcifications. These calcifications are classically described as "stippled," "punctate," "flocculent," or "popcorn-like" rings and arcs, representing the enchondral ossification of the hyaline cartilage lobules.
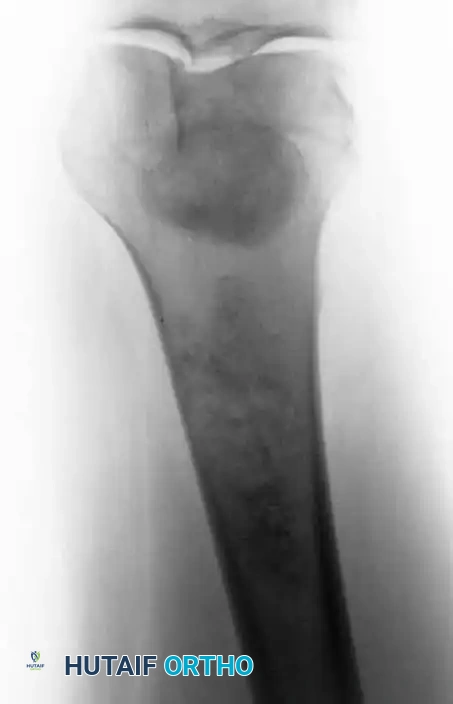
Anteroposterior radiograph of the distal femur showing a calcified lesion without cortical destruction, consistent with an enchondroma.
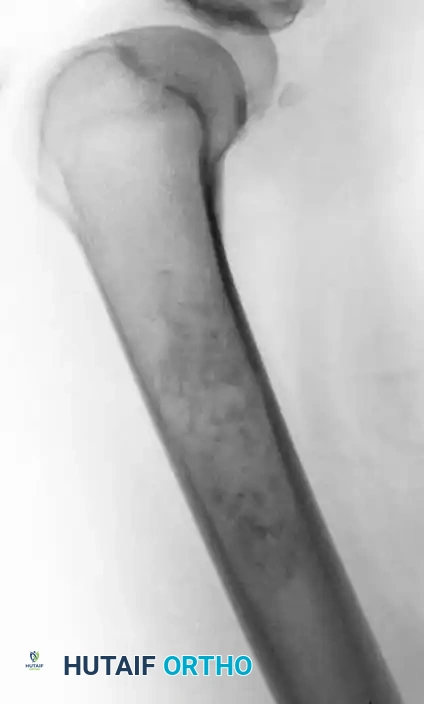
Lateral radiograph of the same distal femur lesion.
In the small bones of the hands and feet, enchondromas frequently cause considerable endosteal erosion and expansion of the overlying cortex, leading to a thinned, "eggshell" appearance. However, in more proximal locations (e.g., proximal humerus, distal femur), deep endosteal erosion—defined as scalloping exceeding two-thirds of the cortical thickness—is a highly suspicious radiographic hallmark of chondrosarcoma.
Advanced Imaging
When plain radiographs are equivocal, or when assessing for malignant transformation, advanced imaging is mandatory.
-
Computed Tomography (CT): CT is the optimal modality for evaluating the integrity of the cortex and quantifying the depth of endosteal scalloping.
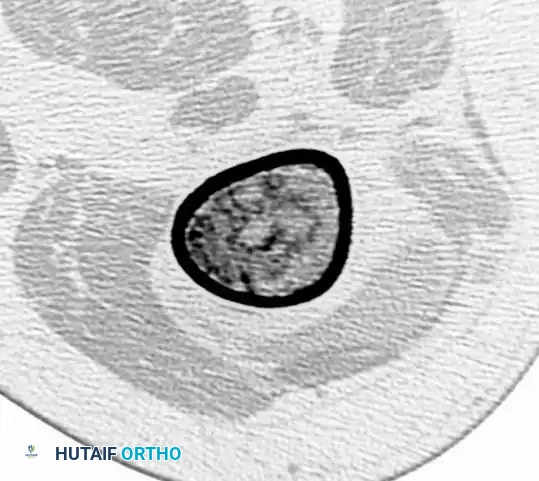
CT scan confirming the absence of cortical destruction, supporting the diagnosis of a benign enchondroma rather than a chondrosarcoma. -
Magnetic Resonance Imaging (MRI): MRI is invaluable for assessing the marrow extent and identifying any associated soft-tissue mass. An associated soft-tissue mass is never present with an uncomplicated enchondroma; its presence is pathognomonic for a malignant process (chondrosarcoma). On MRI, cartilage lesions typically demonstrate low-to-intermediate signal intensity on T1-weighted images and characteristically high signal intensity on T2-weighted images due to the high water content of hyaline cartilage.
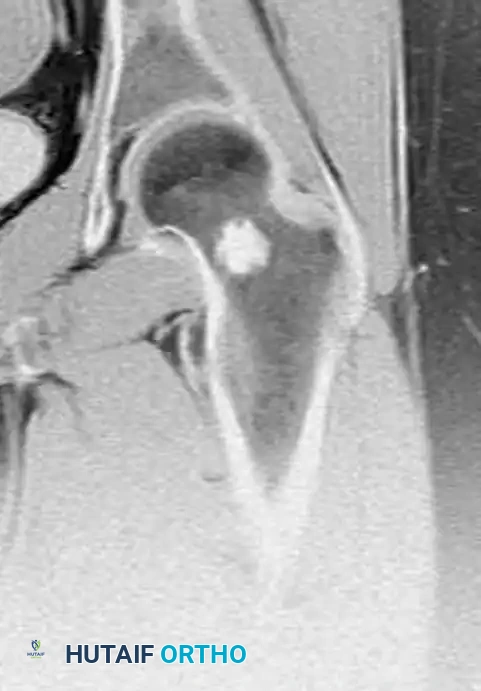
MRI (T1-weighted) demonstrating a dark, well-circumscribed medullary lesion.
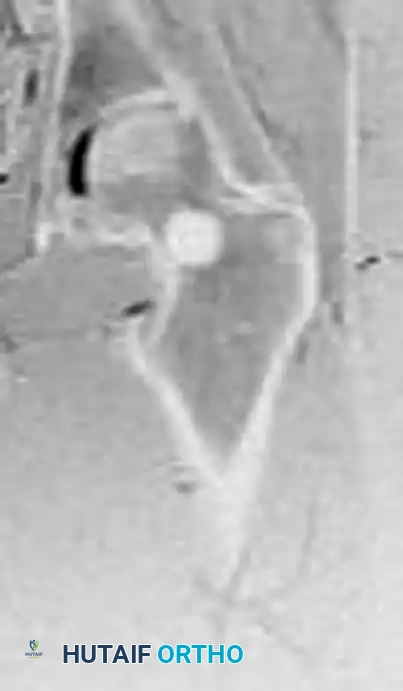
MRI (T2-weighted) of the same lesion.
Histopathology
The microscopic appearance of a benign enchondroma consists of lobules of mature, bland, hypocellular hyaline cartilage. The differentiation between a benign enchondroma and a low-grade (Grade 1) chondrosarcoma is notoriously one of the most challenging dilemmas in musculoskeletal pathology.
Surgical Warning: Proximally located cartilage tumors (e.g., pelvis, proximal femur, proximal humerus) must appear strictly bland and hypocellular. Any degree of hypercellularity, nuclear atypia, or myxoid change in a proximal lesion should raise immediate suspicion for chondrosarcoma. Conversely, enchondromas of the hand, juxtacortical chondromas, and lesions in Ollier disease may exhibit relative hypercellularity and mild atypia while remaining entirely benign.
Surgical Management of Chondromas
Indications for Surgery
- Symptomatic Lesions: Pain directly attributable to the lesion (after rigorously excluding other causes, such as rotator cuff pathology in proximal humerus lesions).
- Pathological Fracture: Often requires allowing the fracture to heal first, followed by definitive treatment of the lesion, though simultaneous management may be indicated depending on the bone involved.
- Radiographic Progression: Enlargement of the lesion or progressive endosteal scalloping on serial imaging.
- Diagnostic Uncertainty: When malignancy cannot be excluded clinically or radiographically.
Operative Technique: Extended Curettage
For symptomatic or growing enchondromas, extended curettage is the gold standard, offering excellent local control with low recurrence rates.
1. Positioning and Approach:
* The patient is positioned based on the anatomical location of the lesion. A tourniquet is utilized where applicable to ensure a bloodless field, aiding in the visual differentiation of tumor tissue from normal marrow.
* A direct surgical approach is utilized, ensuring that the biopsy tract (if previously performed) is excised in line with the definitive incision.
2. Cortical Windowing:
* A longitudinal cortical window is created over the lesion using a high-speed burr or osteotomes. The window must be large enough to allow complete visualization and access to the entire extent of the intramedullary tumor. Oval windows are preferred to minimize stress risers and subsequent fracture risk.
3. Intralesional Curettage:
* Meticulous curettage is performed using a series of angled and straight curettes. The gross appearance of the tumor is typically lobulated, avascular, and pearly blue-white. All visible tumor must be evacuated until normal-appearing cancellous bone is reached.
4. Extended Adjuvant Treatment:
* To eradicate microscopic disease residing in the cancellous bone interstices, the cavity is treated with a high-speed burr to extend the margin by 1-2 mm.
* Chemical or thermal adjuvants are then applied. Options include:
* Phenol (89%): Applied carefully with cotton-tipped applicators, followed by thorough neutralization with absolute alcohol and copious saline lavage.
* Hydrogen Peroxide or Argon Beam Coagulation: Used for both tumoricidal effect and hemostasis.
* Liquid Nitrogen (Cryotherapy): Highly effective but carries a risk of adjacent bone necrosis and postoperative fracture.
5. Reconstruction:
* The resulting cavitary defect can be managed in several ways depending on its size and location:
* Observation: Small defects in non-weight-bearing bones may be left to fill with hematoma and eventually ossify.
* Bone Grafting: Autograft (iliac crest) or allograft (cancellous chips) can be packed into the defect to restore structural integrity.
* Polymethylmethacrylate (PMMA) Bone Cement: Cement provides immediate structural stability and generates an exothermic reaction that acts as an additional thermal adjuvant. It also allows for easy radiographic monitoring for recurrence (seen as a radiolucent line developing between the cement mantle and host bone).
6. Postoperative Protocol:
* Weight-bearing restrictions are dictated by the size of the cortical window and the method of reconstruction. Prophylactic internal fixation may be required if the mechanical integrity of the bone is severely compromised (e.g., >50% cortical diameter involvement).
OSTEOCHONDROMA
Osteochondromas (exostoses) are the most common benign bone tumors, accounting for approximately 35% of all benign bone lesions. Rather than true neoplasms, they are widely considered developmental malformations originating from the periosteum as small, aberrant cartilaginous nodules.
Pathophysiology and Biomechanics
The lesion consists of a bony mass—often forming a distinct stalk—produced by the progressive endochondral ossification of a growing cartilaginous cap. Because their growth is driven by this cartilage cap, the growth of an osteochondroma parallels the skeletal growth of the patient. Crucially, growth typically ceases when skeletal maturity is reached and the adjacent physis closes.
Osteochondromas can occur on any bone preformed in cartilage but have a strong predilection for the metaphyses of long bones, particularly near rapidly growing physes. The most common locations are the distal femur, proximal tibia, and proximal humerus. They are directed away from the adjacent joint due to the mechanical forces of overlying muscle and tendon traction during bone growth.
Clinical Presentation
Approximately 90% of patients present with a solitary lesion. Many are entirely asymptomatic and discovered incidentally. When symptoms do occur, they are usually mechanical in nature:
* Cosmetic Deformity: A palpable, painless mass.
* Mechanical Irritation: Pain caused by the exostosis rubbing against overlying muscles or tendons, frequently leading to the formation of an overlying reactive bursa (exostosis bursata). This bursa may occasionally contain osteocartilaginous loose bodies.
* Neurovascular Compromise: Pressure on adjacent structures can cause significant morbidity. Examples include false aneurysms (pseudoaneurysms) of the popliteal artery from a distal femoral osteochondroma, or common peroneal nerve palsy from a proximal fibular lesion.
* Fracture: Trauma may cause a fracture through the stalk of a pedunculated osteochondroma.
Multiple Hereditary Exostoses (MHE)
Multiple hereditary exostoses (also known as diaphyseal aclasis) is an autosomal dominant condition with variable penetrance, linked to mutations in the EXT1 and EXT2 tumor suppressor genes.
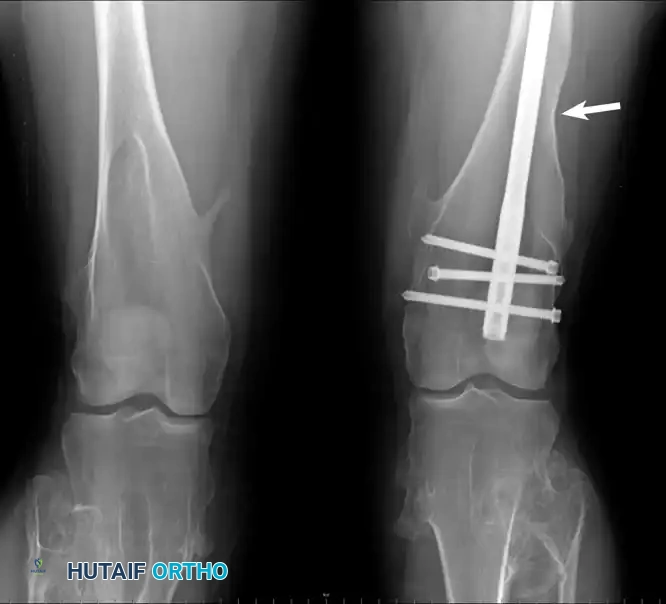
Clinical photograph and radiograph of the knees in a 22-year-old woman with multiple hereditary exostoses. Note the profound metaphyseal broadening, tibial angulation, and a healed femoral fracture sustained post-resection.
Patients with MHE present with numerous osteochondromas and associated profound disturbances in skeletal growth. These include abnormal tubulation of bones (producing broad, blunt, "Erlenmeyer flask" metaphyses), bowing of the radius, shortening of the ulna (leading to ulnar deviation of the hand), and valgus deformities of the knee and ankle. MHE occurs in approximately 1 in 50,000 individuals and is more frequently symptomatic than solitary lesions.
Imaging and Malignant Transformation
Osteochondromas are classified morphologically into two types:
1. Pedunculated: Having a narrow stalk. The stalk is characteristically directed away from the adjacent physis.
2. Sessile: Having a broad base without a distinct stalk.
The radiographic hallmark of an osteochondroma is that the cortical and cancellous bone of the lesion are in direct, uninterrupted continuity with the corresponding components of the parent host bone.
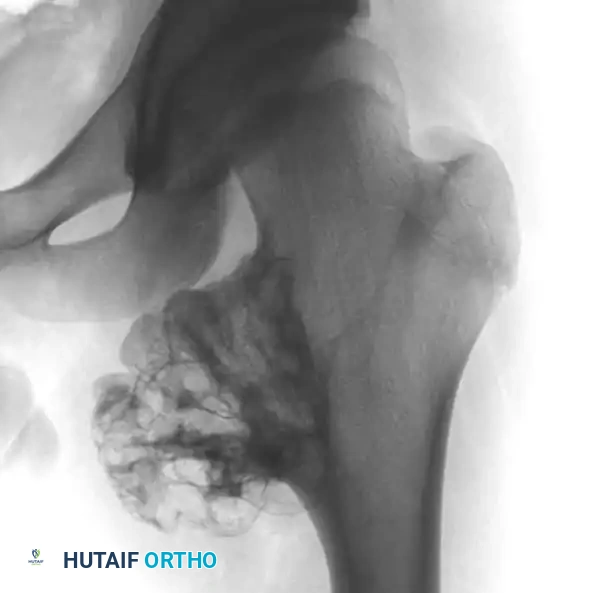
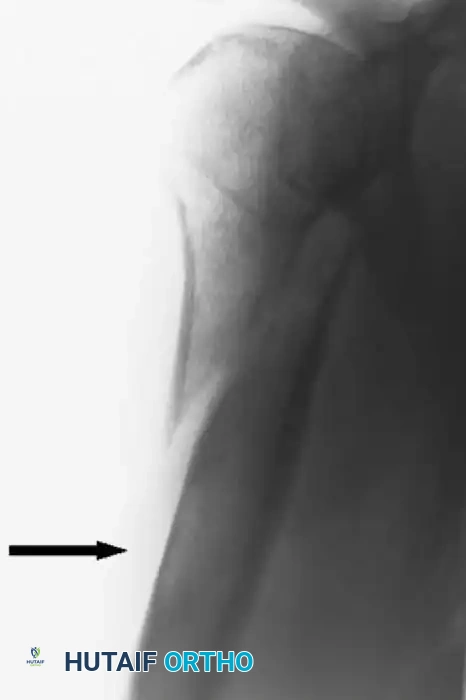
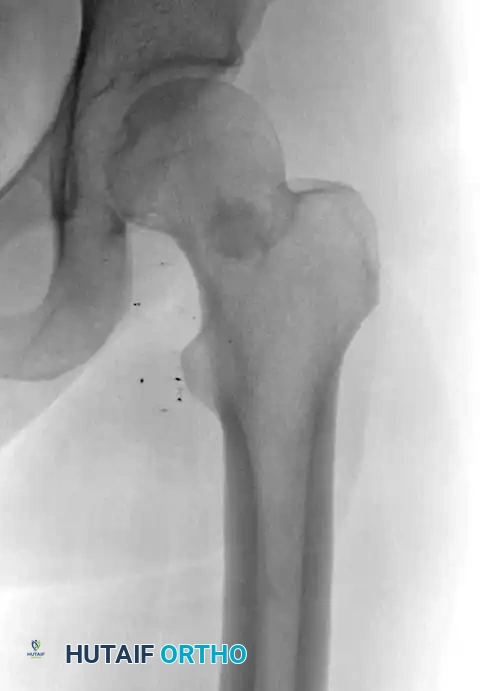
Radiograph demonstrating a pedunculated osteochondroma of the proximal femur.
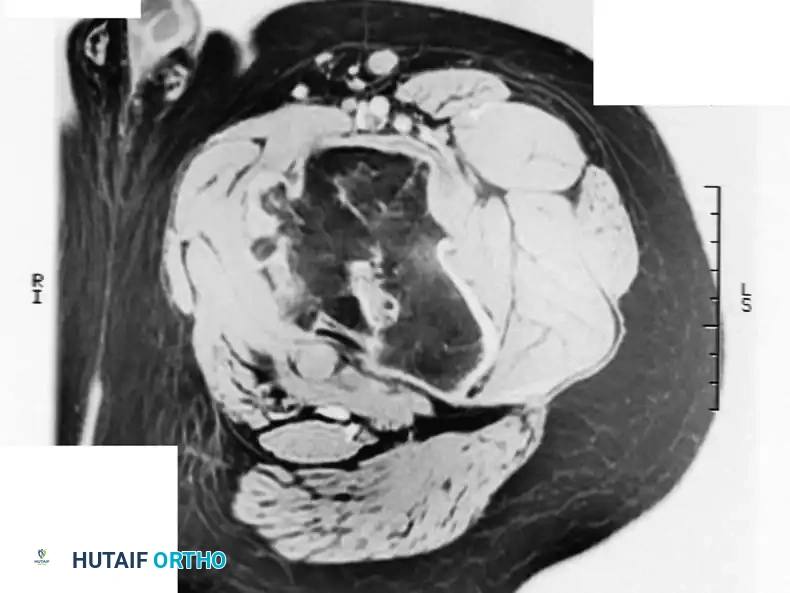
MRI confirming that the cortex and medullary canal of the lesion are in direct continuity with the host bone.
The cartilaginous cap is radiolucent and usually invisible on plain radiographs unless it contains irregular calcifications. In adults, the cap is typically only a few millimeters thick. In actively growing children, it may be up to 2 cm thick.
Malignant Degeneration
Malignant transformation to a secondary peripheral chondrosarcoma is the most feared complication. The incidence is estimated at <1% for solitary osteochondromas and up to 5% for patients with MHE.
Surgical Warning: Malignant transformation must be highly suspected if a previously quiescent lesion in a skeletally mature adult begins to grow rapidly or becomes painful.
Advanced imaging (MRI or CT) is mandatory to evaluate the thickness of the cartilage cap. A cartilage cap thicker than 2 cm in a skeletally mature adult is highly suggestive of malignant transformation.
Surgical Management of Osteochondromas
Indications for Surgery
- Pain or mechanical symptoms (e.g., snapping tendons, restricted range of motion).
- Neurovascular compression.
- Significant cosmetic deformity.
- Clinical or radiographic suspicion of malignant transformation (rapid growth, cap >2 cm, new onset pain in an adult).
Operative Technique: En Bloc Resection
The goal of surgery is the complete, en bloc resection of the osteochondroma, including its entire cartilaginous cap and overlying perichondrium, to prevent recurrence.
1. Approach and Exposure:
* A longitudinal incision is made over the most prominent aspect of the mass.
* Careful dissection is required to protect adjacent neurovascular structures, which may be displaced or draped over the lesion.
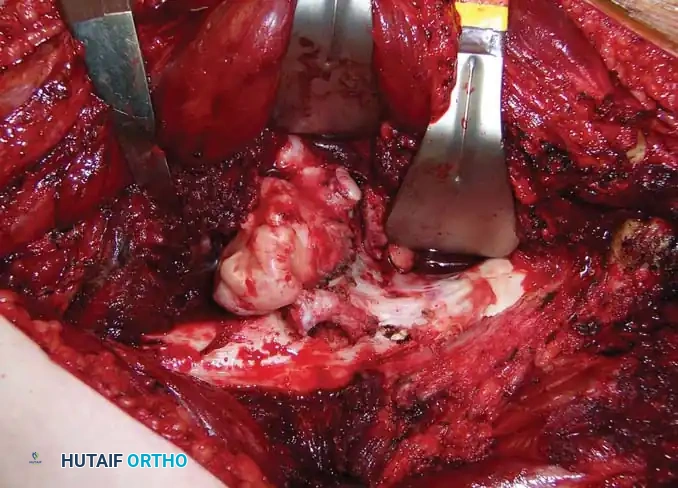
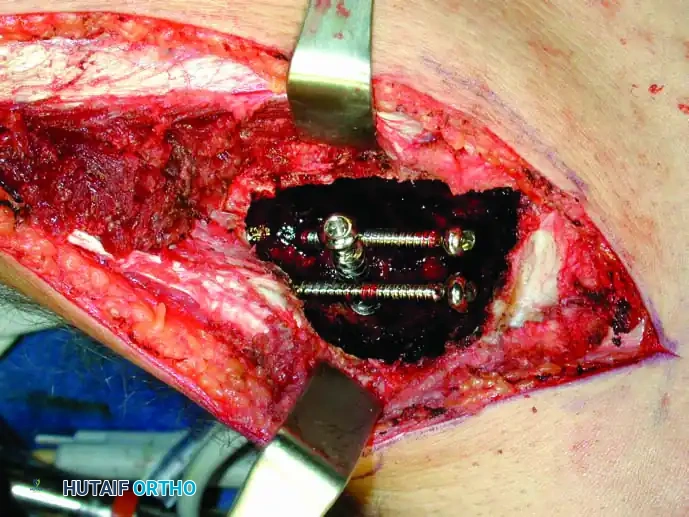
Intraoperative photograph exposing the cartilaginous cap of a proximal femur osteochondroma.
2. Extraperiosteal Dissection:
* The dissection must remain extraperiosteal over the cap of the tumor. Violating the perichondrium or leaving fragments of the cartilage cap behind is the primary cause of local recurrence.
* Any overlying bursa should be excised in continuity with the lesion.
3. Osteotomy:
* For pedunculated lesions, the stalk is exposed down to its base at the host bone cortex.
* An osteotome or oscillating saw is used to perform an osteotomy flush with the normal cortical contour of the host bone. Care must be taken not to notch the host bone excessively, which could create a stress riser.
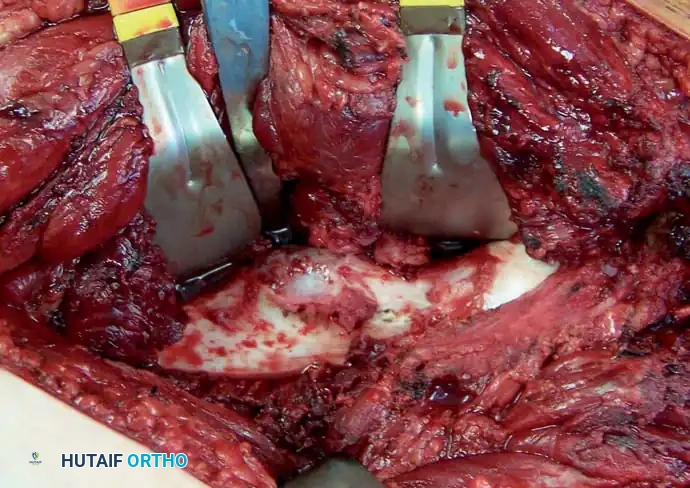
Intraoperative photograph following flush resection of the lesion, restoring the normal cortical contour.
4. Sessile Lesions:
* Sessile lesions require a more extensive, broad-based osteotomy. The margins must be carefully planned to ensure complete removal of the cap while preserving the structural integrity of the parent bone. Prophylactic fixation may be necessary if a massive sessile lesion resection compromises the bone's load-bearing capacity.
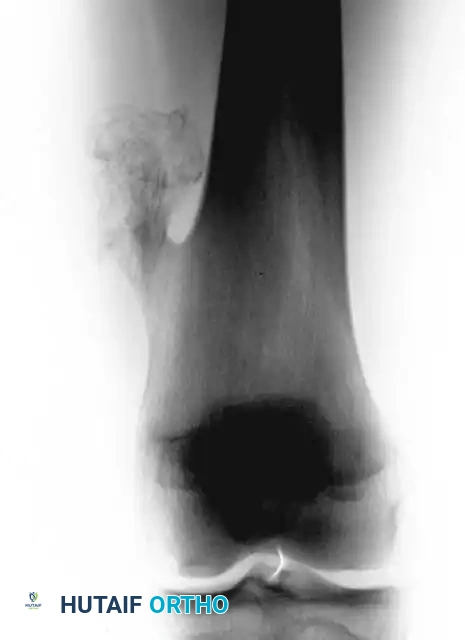
Radiograph of a broad-based, sessile osteochondroma on the distal femur.
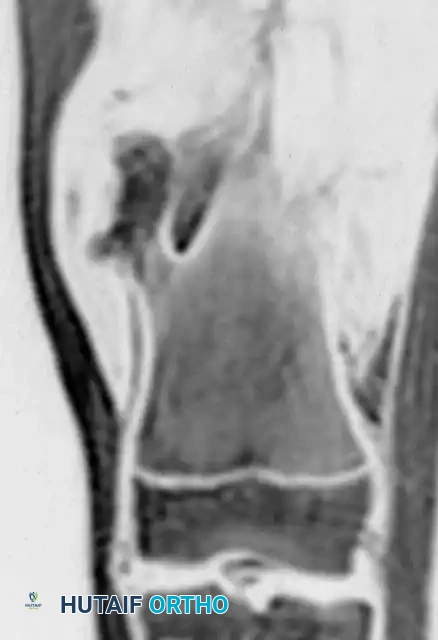
MRI of the distal femur sessile lesion.
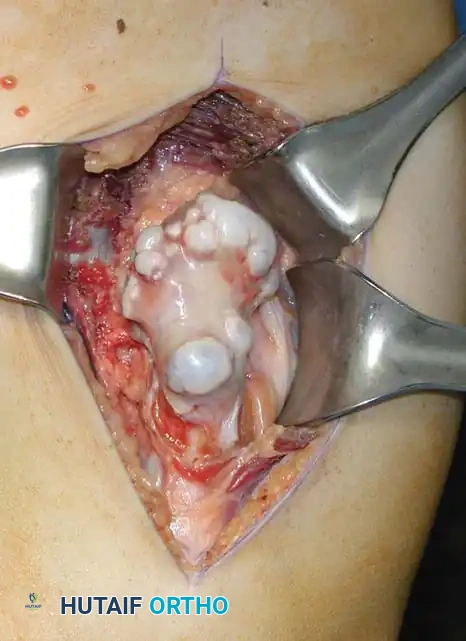
Intraoperative exposure of the distal femur osteochondroma.
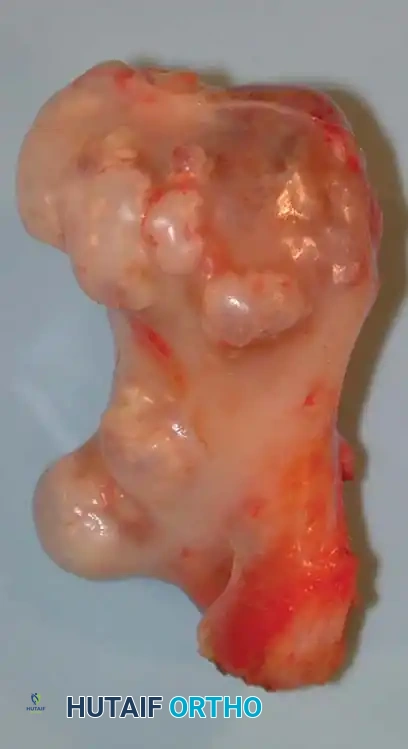
Gross photograph of the resected specimen.
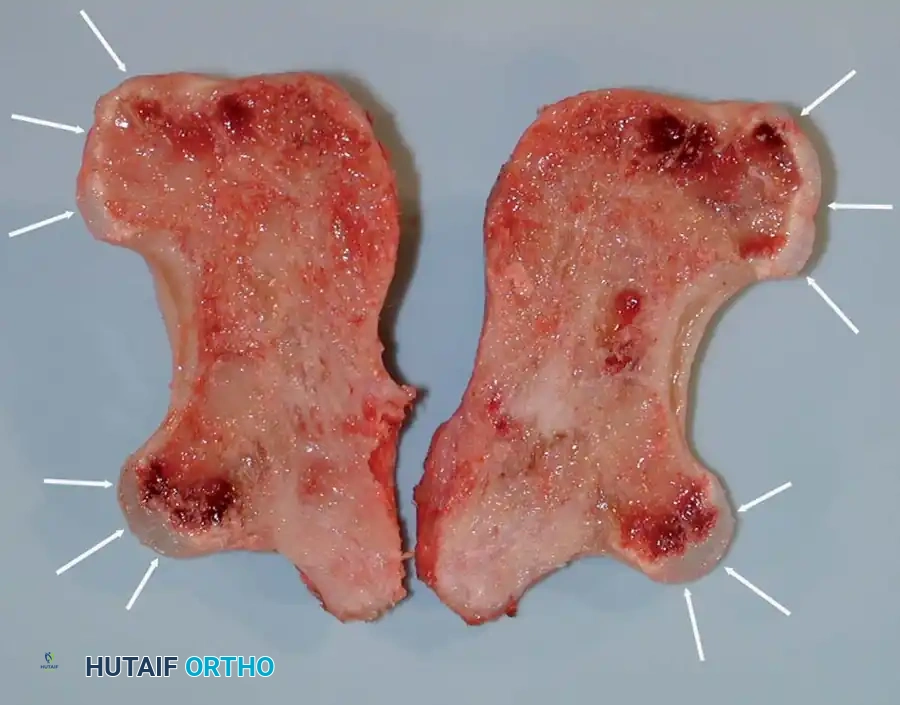
Photograph of the bisected specimen. Note the thin cartilage caps (arrows), measuring only 3 to 4 mm thick, confirming benignity.
5. Pediatric Considerations:
* In growing children, extreme care must be taken to avoid injuring the adjacent open physis during the osteotomy. Resection is often delayed until skeletal maturity unless symptoms are severe, to minimize the risk of iatrogenic growth arrest.
6. Postoperative Protocol:
* Routine hemostasis is achieved, and the wound is closed in layers.
* Early range of motion is encouraged to prevent adhesions of overlying muscles and tendons.
* Weight-bearing is typically permitted as tolerated, unless a massive sessile resection dictates a period of protected weight-bearing.
Subungual Exostosis
A distinct but related entity is the subungual exostosis, which typically develops on the dorsal aspect of the distal phalanx, most commonly the great toe. Unlike true osteochondromas, these lesions frequently have a definitive history of antecedent trauma.
Clinically, they present as a painful, firm mass elevating the nail bed, often leading to nail dystrophy or ulceration. The diagnosis is confirmed radiographically, distinguishing it from a true osteochondroma by its location and lack of medullary continuity. Surgical excision, including the cartilaginous cap and a
📚 Medical References
- cartilage lesions in ankles with lateral ligament injury: an arthroscopic study, Am J Sports Med 21:120, 1993.
- Taillard W, Meyer JM, Garcia A, et al: The sinus tarsi syndrome, Int Orthop 5:117, 1981.
- Taranow WS, Bisignani GA, Towers JD, et al: Retrograde drilling of osteochondral lesions of the medial talar dome, Foot Ankle Int 20:474, 1999.
- Taylor DC, Englehardt DL, Bassett FH III: Syndesmosis sprains of the ankle: the infl uence of heterotopic ossifi cation, Am J Sports Med 20:146, 1992.
- Teitz CC, Harrington RM: A biochemical analysis of the squeeze test for sprains of the syndesmotic ligaments of the ankle, Foot Ankle Int 19:489, 1998.
- Thein R, Eichenblat M: Arthroscopic treatment of sports-related synovitis of the ankle, Am J Sports Med 20:496, 1992.
- Thermann H, Zwipp H, Tscherne H: Treatment algorithm of chronic ankle and subtalar instability, Foot Ankle Int 18:163, 1997.
- Thompson JP, Loomer RL: Osteochondral lesions of the talus in a sports medicine clinic: a new radiographic technique and surgical approach, Am J Sports Med 12:460, 1984.
- Tochigi Y, Takahashi K, Yamagata M, et al: Infl uence of the interosseous talocalcaneal ligament injury on stability of the ankle-subtalar joint complex—a cadaver experimental study, Foot Ankle Int 21:486, 2000.
- Tochigi Y, Yoshinaga K, Wada Y, et al: Acute inversion injury of the ankle: magnetic resonance imaging and clinical outcomes, Foot Ankle Int 19:730, 1998.
- Tohyama H, Yasuda K, Ohkoshi Y, et al: Anterior drawer test for acute anterior talofi bular ligament injuries of the ankle. How much load should be applied during the test? Am J Sports Med 31:226, 2003.
- Vaes P, De Boeck H, Handleberg F, et al: Comparative radiologic study of the infl uence of ankle joint bandages on ankle stability, Am J Sports Med 13:46, 1985.
- Vammen S, Petersen LG, Kaalund S, et al: The effect of musculus extensor digitorum brevis transfer for chronic lateral ankle instability, Foot Ankle Int 19:563, 1998.
- Van Dijk CN, Bossuyt PM, Marti RK: Medial ankle pain after lateral ligament rupture, J Bone Joint Surg 78B:562, 1996.
- Van Dijk CN, Lim LS, Bossuyt PM, et al: Physical examination is suffi cient for the diagnosis of sprained ankles, J Bone Joint Surg 78B:958, 1996.
- van Hellemondt FJ, Louwerens JWK, Sijbrandij ES, et al: Stress radiograph and stress examination of the talocrural and subtalar joint on helical computed tomography, Foot Ankle Int 18:482, 1997.
- Veltri DM, Pagnani MJ, O’Brien SJ, et al: Symptomatic ossifi cation of the tibiofi bular syndesmosis in professional football players: a sequela of the syndesmotic ankle sprain, Foot Ankle Int 16:285, 1995.
- Verhaven EFC, Shahabpour M, Handelberg FWJ, et al: The accuracy of three-dimensional magnetic resonance imaging in the diagnosis of ruptures of the lateral ligaments of the ankle, Am J Sports Med 19:583, 1991.
- Watson-Jones R: Fractures and joint injuries, 3rd ed, Baltimore, 1946, Williams & Wilkins. Wilson JD: Sprains and ruptures of ligaments of the ankle joint, Clin Orthop 3:109, 1954.
- Wood JJ, Malek MA, Frassica FJ, et al: Autologous cultured chondrocytes: adverse events reported to the United States Food and Drug Administration, J Bone Joint Surg 88A:503, 2006.
- Woods K, Harris I: Osteochondritis dissecans of the talus in identical twins, J Bone Joint Surg 77B:331, 1995.
- Younes C, Fowles JV, Antoun R: Long-term results of surgical reconstruction for chronic lateral instability of the ankle: comparison of Watson-Jones and Evans techniques, J Trauma 28:1330, 1988.
- Zinman BC, Wolfson N, Reis ND: Osteochondritis dissecans of the dome of the talus: computed tomography scanning in diagnosis and follow-up, J Bone Joint Surg 70A:1017, 1988.
You Might Also Like