Operative Management of Benign and Aggressive Bone Tumors: The Giant Cell Tumor Masterclass
Key Takeaway
Giant cell tumors (GCTs) of bone are locally aggressive neoplasms typically arising in the epiphyses of long bones in young adults. Standard treatment involves extended intralesional curettage, high-speed burring, and adjuvant therapies such as argon beam coagulation. Defect reconstruction with polymethylmethacrylate (PMMA) bone cement provides immediate biomechanical stability and facilitates early radiographic detection of local recurrence.
Introduction to Benign and Aggressive Tumors of Bone
The biological behavior of the osseous lesions described in this spectrum bridges the gap between purely benign entities and frankly malignant sarcomas. While these tumors are frequently managed with joint-salvaging intralesional procedures—most notably extended curettage—their capacity for aggressive local destruction necessitates meticulous surgical technique. In select cases, marginal or wide en bloc resection is mandated to achieve local control.
Furthermore, systemic involvement, though statistically rare, represents a critical consideration in the comprehensive management of these patients. Giant cell tumors (GCTs) and chondroblastomas possess the potential to metastasize to the lungs, which can occasionally be fatal. Similarly, Langerhans cell histiocytosis may manifest with multisystemic organ involvement beyond the skeletal system. This guide provides an exhaustive, postgraduate-level analysis of the Giant Cell Tumor of bone, detailing its pathophysiology, clinical presentation, and step-by-step surgical management.
Epidemiology and Pathophysiology of Giant Cell Tumors
Giant cell tumors represent approximately 5% of all primary bone neoplasms. They exhibit a distinct demographic profile, predominantly affecting young adults between the ages of 20 and 40, with a slight female predominance.
Anatomic Distribution
The anatomic predilection of GCTs is highly characteristic:
* Distal Femur: The most common site of occurrence.
* Proximal Tibia: The second most common location, making the knee joint the epicenter for the majority of these lesions.
* Distal Radius: The third most common site; notably, tumors in this location frequently exhibit a more aggressive biological and clinical behavior.
* Axial Skeleton: Spinal involvement is exceedingly rare, with the exception of the sacrum.
GCTs are typically solitary lesions. However, synchronous or metachronous multicentricity occurs in 1% to 2% of cases. The academic consensus remains divided on whether multicentric disease represents multiple primary de novo lesions or osseous metastases originating from a single primary source.
Metastatic Potential and Malignant Transformation
Although histologically classified as benign, GCTs possess a well-documented metastatic potential. Pulmonary metastases occur in approximately 3% of patients. The clinical course of these metastases is highly variable:
* Some patients experience spontaneous regression.
* Others remain entirely asymptomatic for decades.
* A subset develops progressive, fatal pulmonary compromise, despite the metastatic deposits remaining histologically "benign." The overall disease-specific mortality rate for patients with pulmonary metastases is approximately 15%.
Clinical Pearl: Patients presenting with locally recurrent lesions or primary tumors that exhibit aggressive radiographic features (Campanacci Stage 3) are at a statistically significantly higher risk for developing pulmonary metastases. Routine thoracic staging is mandatory.
Malignant giant cell tumors are exceptionally rare, representing less than 5% of all cases, and are subclassified into:
1. Primary Malignant GCT: Defined as a high-grade sarcoma arising de novo within a lesion that otherwise contains areas typical of a benign GCT.
2. Secondary Malignant GCT: A sarcoma arising at the site of a previously treated GCT, most commonly associated with a history of therapeutic radiation.
Clinical Presentation and Evaluation
The hallmark symptom of a giant cell tumor is progressive, insidious pain. Initially, the pain is mechanically related to weight-bearing or activity, but as the cortex expands and microfractures occur, the pain frequently becomes evident at rest.
Unless a pathological fracture has occurred, the pain is rarely described as severe. However, due to the lytic nature of the tumor and its propensity to destroy subchondral bone, pathological fractures are present at the time of initial examination in 10% to 30% of patients. Joint effusion, decreased range of motion, and a palpable mass may be present in advanced cases with cortical breakthrough.
Imaging Characteristics
Accurate radiographic staging is the cornerstone of preoperative planning.
Plain Radiography
Radiographic findings are often pathognomonic. GCTs are purely lytic lesions eccentrically located within the epiphyses of long bones, almost universally abutting the subchondral bone plate.
* Skeletal Maturity: While rare in skeletally immature patients, when they do occur in this population, they typically arise in the metaphysis. It is theorized that all GCTs originate in the metaphysis and migrate into the epiphysis following physeal closure.
* Margins: The zone of transition is often poorly defined. In less aggressive (Stage 1) tumors, a partial rim of reactive sclerosis may be visible.
* Cortical Involvement: The lesion frequently expands, thins, or frankly breaches the cortex. However, direct intra-articular extension is rare because the subchondral bone plate, though thinned, usually remains intact.
* Matrix: Matrix calcification or ossification is absent within the intraosseous compartment but may be observed if there is soft-tissue extension, local recurrence, or pulmonary metastasis.
Magnetic Resonance Imaging (MRI)
MRI is indispensable for determining the exact intraosseous extent of the tumor, evaluating the integrity of the subchondral bone, and identifying soft-tissue extension.

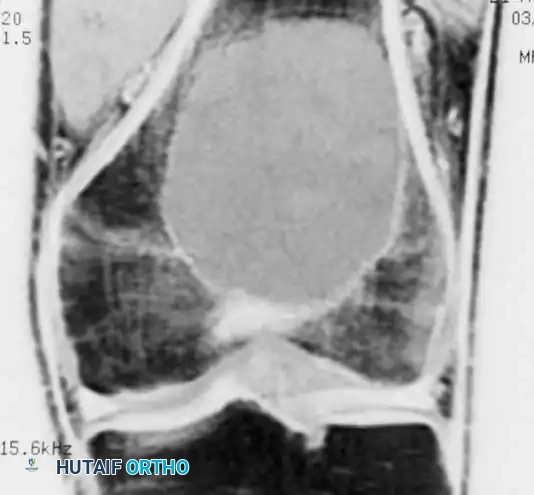
Coronal MRI demonstrating a large, eccentrically located giant cell tumor in the distal femur extending to the subchondral plate.
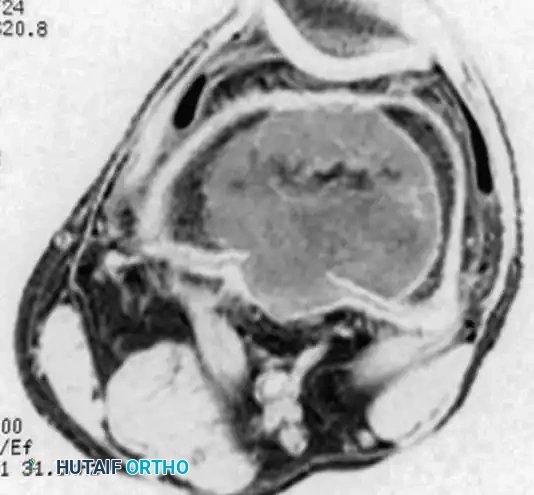
Axial MRI of the same distal femoral lesion, highlighting cortical thinning and the precise intramedullary boundaries of the tumor.
- Signal Characteristics: The lesion typically demonstrates low-to-intermediate signal intensity on T1-weighted images and heterogeneous high signal intensity on T2-weighted images.
- Secondary Aneurysmal Bone Cyst (ABC): MRI is highly sensitive for detecting fluid-fluid levels, which are indicative of a secondary ABC—a phenomenon occurring in up to 20% of GCT patients.
Histopathology
Microscopically, giant cell tumors are characterized by a dual-cell population:
1. Multinucleated Giant Cells: These osteoclast-like cells are abundant, typically containing 40 to 60 nuclei per cell.
2. Mononuclear Stromal Cells: This is the true neoplastic component of the tumor.
Pathology Pitfall: The defining histological feature that distinguishes a true GCT from other giant-cell-rich lesions (e.g., chondroblastoma, brown tumor of hyperparathyroidism) is that the nuclei of the mononuclear stromal cells are morphologically identical to the nuclei within the multinucleated giant cells.
Additional histological features may include areas of storiform spindle cell formation, reactive woven bone, foamy macrophages, and cystic spaces consistent with secondary ABC formation. Despite numerous historical attempts, no histological grading system has proven to possess reliable prognostic significance regarding local recurrence or metastatic potential.
Surgical Management: Principles and Decision Making
Giant cell tumors are locally aggressive, with the vast majority presenting as Campanacci Stage 2 (active, intact cortex) or Stage 3 (aggressive, cortical breach). Historically, simple curettage yielded unacceptable local recurrence rates exceeding 50%.
Modern surgical management relies on extended intralesional curettage, which has successfully reduced recurrence rates to between 5% and 15%. This dramatic improvement is attributed to advanced MRI preoperative planning, aggressive mechanical burring, and the routine use of chemical or thermal adjuvants.
Defect Reconstruction: Bone Graft vs. PMMA Cement
Following extended curettage, the surgeon must reconstruct the cavitary defect. Options include autograft, allograft, synthetic bone substitutes, or polymethylmethacrylate (PMMA) bone cement.
While bone grafting theoretically restores normal joint biomechanics and replenishes bone stock, it carries two significant disadvantages:
1. Prolonged protected weight-bearing is required to prevent subchondral collapse and pathological fracture during graft incorporation.
2. Local tumor recurrence is notoriously difficult to distinguish radiographically from normal graft resorption and remodeling.
The Case for PMMA Bone Cement:
We routinely advocate for the use of PMMA bone cement to fill the post-curettage cavity. The advantages are multifold:
* Immediate Biomechanical Stability: Allows for rapid postoperative rehabilitation and early weight-bearing.
* Thermal Necrosis: The exothermic reaction of polymerizing PMMA (reaching temperatures up to 70°C - 100°C) provides an additional adjuvant effect, causing thermal necrosis of microscopic residual tumor cells.
* Surveillance Efficacy: PMMA creates a dense, radiopaque cast of the cavity. Any subsequent local recurrence is easily identifiable on plain radiographs as a progressive, expanding radiolucent halo at the cement-bone interface.
Step-by-Step Surgical Technique: Extended Curettage and Cementation
The following technique describes the approach for a typical distal femoral giant cell tumor.
1. Positioning and Approach
- The patient is positioned supine on a radiolucent operating table.
- A sterile tourniquet is applied to the proximal thigh to ensure a bloodless surgical field, which is critical for identifying residual tumor tissue.
- A standard lateral or medial approach is utilized, depending on the eccentric location of the tumor. The vastus lateralis or medialis is elevated to expose the metaphyseal cortex.
2. Cortical Window Creation
- A large cortical window must be created. Crucially, the window must be at least as large as the maximum diameter of the underlying lesion.
- A window that is too small forces the surgeon to work "around the corner," virtually guaranteeing that residual tumor cells will be left adjacent to the near-side cortex.
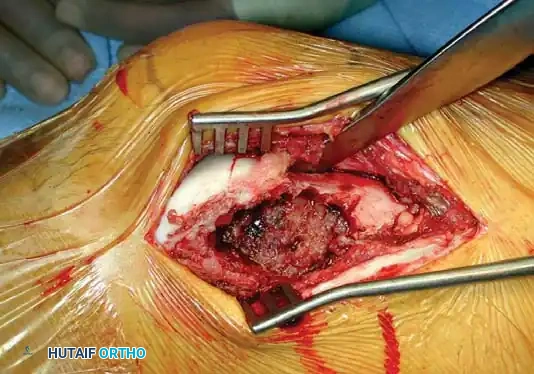
Intraoperative photograph demonstrating the creation of a wide cortical window, exposing the hemorrhagic, friable tissue characteristic of a giant cell tumor.
3. Intralesional Curettage
- Gross tumor removal is performed using a series of progressively larger curettes. The tumor typically has a soft, fleshy, and highly vascular consistency.
- Curettage is carried meticulously down to the subchondral bone plate. Extreme care must be taken not to breach the articular cartilage.
4. Extended Curettage (High-Speed Burr)
- Once gross tumor is removed, a high-speed power burr is utilized to aggressively enlarge the cavity by 1 to 2 cm in all directions.
- This step breaks down the reactive bony ridges and exposes the normal cancellous bone, eliminating microscopic tumor extensions within the marrow spaces.
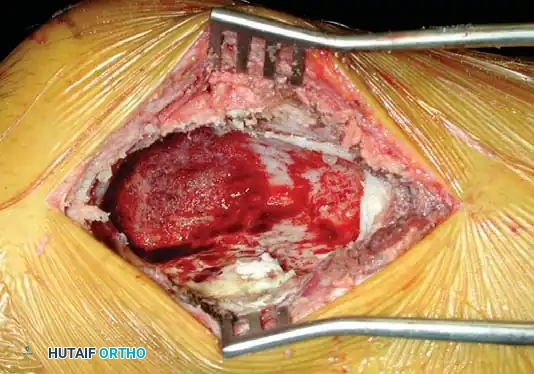
Intraoperative view of the osseous cavity following aggressive extended curettage with a high-speed burr. The cavity is now visually free of gross tumor.
5. Adjuvant Therapy
- To eradicate any remaining microscopic disease, an adjuvant is applied. Options include liquid nitrogen (cryotherapy), phenol, electrocautery, or an argon beam coagulator.
- Author's Preference: We prefer the argon beam coagulator. It is highly effective, easy to control, and avoids the severe complications associated with liquid nitrogen or phenol (e.g., adjacent nerve injury, severe soft-tissue necrosis, delayed wound healing, and secondary pathological fractures).
6. Structural Augmentation (Rebar Technique)
- Because the subchondral bone plate is often paper-thin following aggressive curettage, packing PMMA directly against it can lead to thermal injury to the articular cartilage or mechanical point-loading.
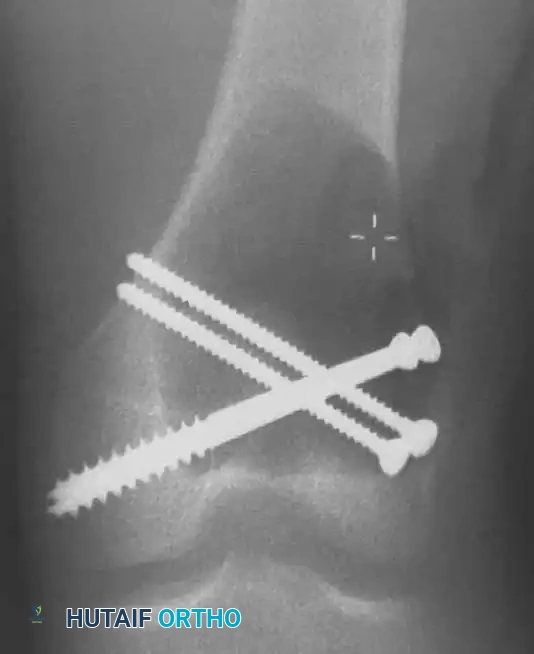
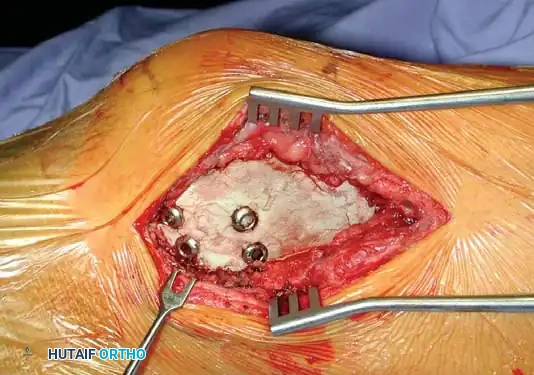
- To prevent this, structural cortical screws are placed across the cavity. These act as "rebar," distributing the mechanical load from the joint surface into the diaphyseal cortical bone.
Intraoperative photograph showing the placement of multiple structural screws within the cavity to support the subchondral plate and anchor the cement mantle.
Intraoperative fluoroscopy confirming the optimal trajectory and depth of the structural screws, ensuring they do not breach the articular surface.
7. Cementation (PMMA Application)
- The tourniquet is temporarily deflated to ensure hemostasis, then re-inflated. The cavity is thoroughly irrigated and dried.
- Standard viscosity PMMA bone cement is introduced into the cavity in a doughy state. It is digitally packed firmly around the screws and into all crevices of the burred cavity to ensure a perfect interdigitation with the host bone.
Intraoperative photograph demonstrating the PMMA bone cement packed tightly into the cavity, enveloping the structural screws.
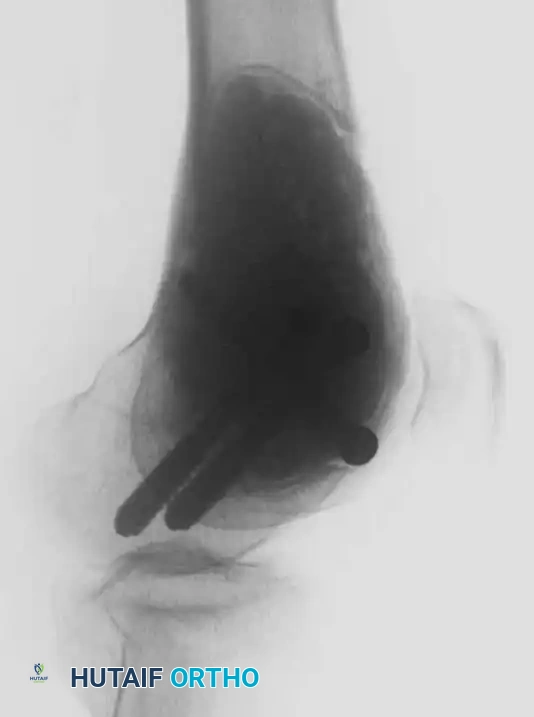
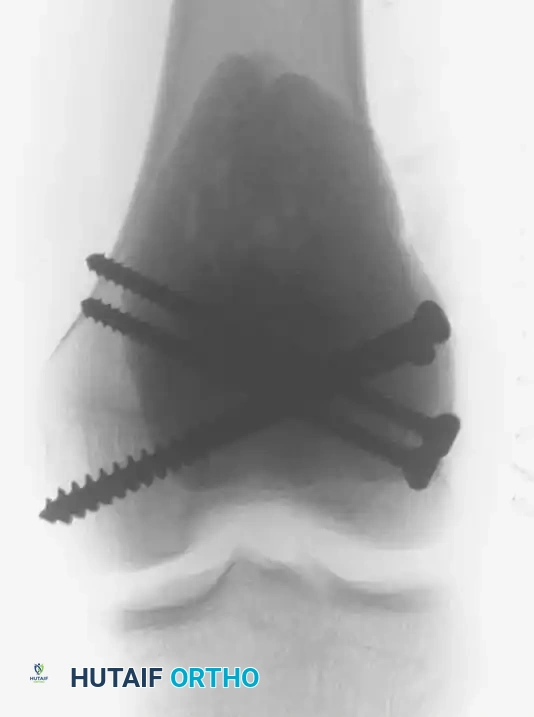
Postoperative Anteroposterior (AP) and Lateral radiographs demonstrating the final reconstruction. The radiopaque cement mantle and structural screws provide immediate stability.
Management of Advanced and Unresectable Lesions
While extended curettage is the gold standard, it may be oncologically or biomechanically inadequate for massive Stage 3 tumors with extensive cortical destruction or joint involvement.
Primary Resection and Reconstruction
- Knee Joint: Massive lesions of the distal femur or proximal tibia may require en bloc resection followed by reconstruction with a hemicondylar osteoarticular allograft or a modular rotating-hinge endoprosthesis.
- Distal Radius: Aggressive lesions here frequently destroy the carpal articular surface. Primary wide resection is often indicated, followed by reconstruction using a proximal fibular autograft (performed either as a wrist arthroplasty or a total wrist arthrodesis).
- Expendable Bones: For lesions located in the proximal fibula, distal ulna, or ribs, primary en bloc resection without structural reconstruction is the treatment of choice.
Inoperable Lesions
For surgically inaccessible tumors, particularly within the sacrum, pelvis, or complex spinal segments, alternative modalities must be employed:
* Serial Embolization: Can reduce tumor volume and alleviate pain.
* Radiation Therapy: May be utilized for local control; however, extreme caution is advised. Radiation of a benign GCT carries a well-documented risk of inducing secondary sarcomatous transformation.
* Medical Management: The advent of Denosumab (a monoclonal antibody against RANKL) has revolutionized the treatment of unresectable GCTs, inducing profound tumor necrosis and ossification, though its role as a neoadjuvant prior to curettage remains debated due to higher post-surgical recurrence rates.
Postoperative Protocol and Oncologic Surveillance
Patients diagnosed with giant cell tumors require rigorous, long-term oncologic follow-up. The vast majority of local recurrences and pulmonary metastases manifest within the first 3 years postoperatively, though late recurrences up to 20 years later have been documented.
Surveillance Schedule
- Baseline Staging: High-resolution Chest CT and plain radiographs of the primary site immediately post-op.
- Years 1 and 2: Clinical exam, local radiographs, and chest radiographs every 3 to 4 months.
- Year 3: Clinical exam and imaging every 6 months.
- Year 4 and Beyond: Annual evaluation indefinitely.
Note: Any suspicious nodule on a routine chest radiograph mandates an immediate dedicated Chest CT.
Managing Recurrence
- Local Recurrence: Typically presents as a progressive, expanding radiolucent zone adjacent to the PMMA cement mantle. Soft-tissue recurrences may present as a palpable mass or peripheral ossification, best evaluated with MRI. Treatment consists of repeat biopsy (to rule out malignant transformation) followed by repeat extended curettage or en bloc resection.
- Pulmonary Metastases: If identified, surgical metastasectomy (wedge resection) is the treatment of choice and can be curative. Chemotherapy has limited efficacy, and radiation is strictly reserved for symptomatic, surgically inoperable pulmonary lesions.
Surgical Warning: Never assume a recurrent giant cell tumor is benign without histological confirmation. Secondary malignant transformation must always be excluded prior to attempting a joint-salvaging re-curettage.
You Might Also Like

