Kienböck Disease: Vascularized Bone Graft & Capitate Shortening Osteotomy Masterclass
Key Takeaway
This masterclass guides fellows through the intricate surgical management of Kienböck disease, combining vascularized bone grafting with capitate shortening osteotomy. We will meticulously detail preoperative planning, patient positioning, comprehensive dorsal wrist anatomy, and the precise, step-by-step intraoperative execution. Critical pearls, potential pitfalls, and comprehensive postoperative rehabilitation strategies are emphasized to optimize outcomes for this challenging condition.
Introduction and Epidemiology
Kienböck disease, formally defined as idiopathic avascular necrosis (AVN) of the carpal lunate, is a progressively debilitating orthopaedic condition that culminates in radiocarpal collapse, carpal instability, and end-stage arthrosis if left untreated. Originally characterized by Robert Kienböck in 1910 via rudimentary radiographic observations, the contemporary understanding of its etiology points toward a multifactorial pathogenesis. It represents a complex convergence of tenuous intraosseous vascularity, adverse radiocarpal biomechanics, and repetitive microtraumatic loading. Epidemiologically, Kienböck disease exhibits a strong predilection for adults between 20 and 40 years of age, predominantly affecting males engaged in heavy manual labor, though it is increasingly recognized in diverse demographic cohorts.
The precise pathophysiologic cascade is widely debated but is generally accepted to originate from an ischemic event—either arterial insufficiency or venous hypertension—within the lunate. This ischemia is mechanically exacerbated by anatomical predispositions, most notably negative ulnar variance. In negative ulnar variance, the relatively shortened distal ulna fails to bear its physiologic share of axial loads, disproportionately shifting compressive forces across the radiolunate articulation. This concentrated stress induces subchondral microfractures, further compromising the already fragile vascular sinusoids within the lunate body.
Therapeutic decision-making is heavily reliant on the Lichtman classification system, which categorizes the disease based on radiographic and advanced imaging parameters:
* Stage I: Normal plain radiographs; MRI demonstrates diffuse loss of T1 signal intensity indicating ischemia.
* Stage II: Radiographic sclerosis of the lunate without alteration of its anatomic shape or dimensions.
* Stage IIIA: Lunate structural collapse and fragmentation, but preservation of overall carpal height and neutral scaphoid alignment.
* Stage IIIB: Lunate collapse accompanied by loss of carpal height, fixed scaphoid palmar flexion (rotatory subluxation), and disruption of the carpal arcs.
* Stage IV: Advanced pancarpal degenerative joint disease involving the radiocarpal and midcarpal articulations.
This comprehensive academic review delineates the combined surgical intervention utilizing Vascularized Bone Grafting (VBG) and Capitate Shortening Osteotomy (CSO). This dual-modality approach is synergistically designed to address both the biological deficit (via revascularization from the VBG) and the mechanical overload (via central column offloading from the CSO). It is considered a premier joint-preserving strategy, particularly indicated for patients presenting with Lichtman Stages II and IIIA Kienböck disease.
Surgical Anatomy and Biomechanics
A profound mastery of carpal kinematics, intraosseous vascular architecture, and radiocarpal load distribution is requisite for the successful execution of joint-preserving Kienböck procedures.
Lunate Vascularity and Pathoanatomy
The lunate occupies a critical keystone position within the proximal carpal row, articulating with the radius, capitate, scaphoid, triquetrum, and hamate. It is uniquely devoid of direct tendinous insertions, relying entirely on intrinsic and extrinsic capsuloligamentous complexes for stability.
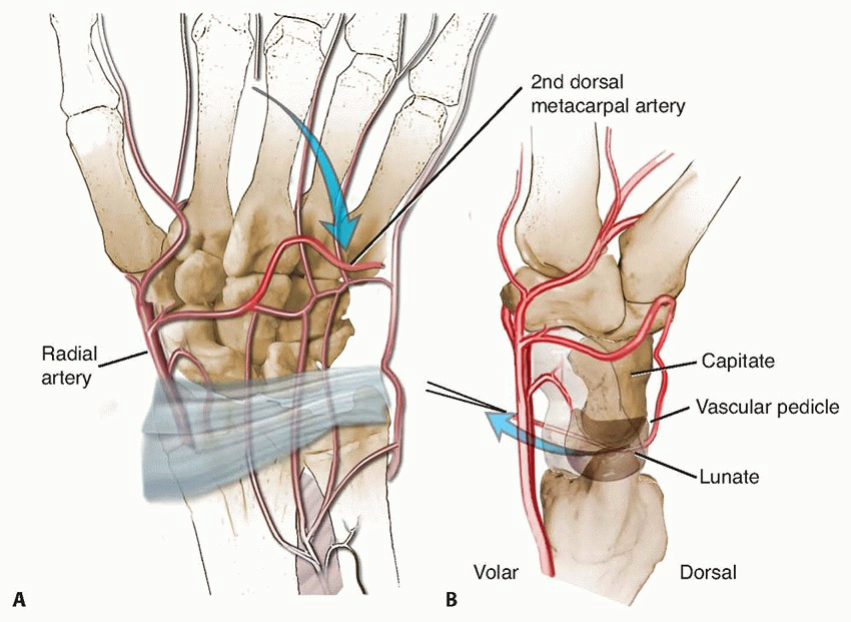
Seminal microangiographic studies by Gelberman et al. elucidated the precarious nature of lunate blood supply. The lunate receives its vascular inflow via branches from the dorsal and volar radiocarpal arches. Crucially, approximately 20% of the population possesses a single dominant volar or dorsal vessel supplying the entire intraosseous network, lacking robust intraosseous anastomoses (often presenting as a singular "Y" or "I" pattern). This anatomical variant significantly heightens the susceptibility to global lunate ischemia following localized trauma or elevated intra-articular pressure.
Radiocarpal Biomechanics and Ulnar Variance
The distal radioulnar joint (DRUJ) and radiocarpal joint function in tandem to transmit axial loads from the hand to the forearm. In a state of neutral ulnar variance, the distal radius absorbs approximately 80% of the axial load, while the distal ulna absorbs 20%.
Negative ulnar variance—defined as a proximal position of the distal ulnar articular surface relative to the distal radial articular surface—is observed in up to 80% of patients with Kienböck disease. Biomechanical studies by Palmer and Werner demonstrated that even a 2.5 mm negative ulnar variance shifts the load distribution dramatically, forcing the radius to bear up to 95% of the axial load. This massive force concentration occurs directly over the lunate fossa, subjecting the lunate to chronic, repetitive sheer and compressive forces that precipitate trabecular failure and vascular compromise.
Capitate Biomechanics and Rationale for Shortening
The capitate is the largest carpal bone and serves as the primary longitudinal axis for the wrist. It articulates directly with the distal lunate concavity. The biomechanical rationale for a Capitate Shortening Osteotomy (CSO) is to functionally decompress the central carpal column. By resecting a precise 2 to 4 mm segment of the capitate body, the surgeon effectively unloads the radiolunate joint. This converts the mechanical environment from one of excessive compression to one conducive to angiogenesis and graft incorporation. Notably, unlike radial shortening osteotomy, CSO achieves lunate decompression without altering the kinematics of the distal radioulnar joint (DRUJ), making it an excellent option regardless of ulnar variance, though it is particularly useful in ulnar negative or neutral wrists.
Donor Site Anatomy: Dorsal Distal Radius
The dorsal distal radius provides highly reliable pedicled vascularized bone grafts. The two most frequently utilized pedicles are:
1. 1,2-Intercompartmental Supraretinacular Artery (1,2-ICSRA): Courses over the first and second extensor compartments.
2. 4+5 Extensor Compartment Artery (ECA): Arises from the posterior interosseous artery or the dorsal carpal arch, coursing between the fourth (EDC) and fifth (EDM) extensor compartments.
The 4+5 ECA graft is particularly favored for Kienböck disease due to its anatomic proximity to the lunate, allowing for a shorter, more direct transposition arc. The pedicle consists of a small artery (typically 0.5–1.0 mm in diameter) and accompanying venae comitantes, supplying a block of corticocancellous bone harvested proximal to the radiocarpal articular surface.
Indications and Contraindications
Patient selection is the most critical determinant of clinical success. The combined VBG and CSO procedure is a joint-preserving operation; therefore, it is predicated on the presence of viable articular cartilage and the absence of fixed carpal collapse.
Indications
- Lichtman Stage II: Sclerotic lunate without collapse. The combined procedure is highly indicated to arrest progression, particularly if negative ulnar variance or high mechanical demand is present.
- Lichtman Stage IIIA: Lunate collapse with preserved carpal height and a neutral scaphoid. This is the classic indication for combined VBG and CSO. The VBG restores biological viability, while the CSO protects the graft from mechanical crushing during the revascularization phase.
- Select Lichtman Stage IIIB: While controversial, younger patients with Stage IIIB disease who maintain functional range of motion and lack widespread arthrosis may be considered for this procedure as a salvage effort to delay total wrist arthrodesis or proximal row carpectomy (PRC).
- Refractory Symptoms: Progressive, debilitating radiocarpal pain that has failed exhaustive conservative management (immobilization, NSAIDs, activity modification).
Contraindications
- Absolute Contraindications:
- Lichtman Stage IV: Presence of pancarpal or radiocarpal osteoarthritis. Revascularization cannot reverse established cartilage destruction.
- Coronal Split Fractures of the Lunate: Severe fragmentation precludes secure seating and containment of the vascularized graft.
- Active Infection: Local or systemic.
- Relative Contraindications:
- Tobacco Use: Active smoking profoundly impairs microvascular flow and osteogenesis, exponentially increasing the risk of graft thrombosis and nonunion.
- Advanced Age / Low Functional Demand: Older patients with lower demands may be better served by definitive salvage procedures (e.g., PRC) that require less prolonged rehabilitation.
- Severe Osteopenia/Osteoporosis: Poor bone stock compromises the rigid internal fixation required for the capitate osteotomy.
Pre Operative Planning and Patient Positioning
Rigorous preoperative assessment ensures accurate osteotomy execution and optimal graft placement.
Diagnostic Imaging Protocol
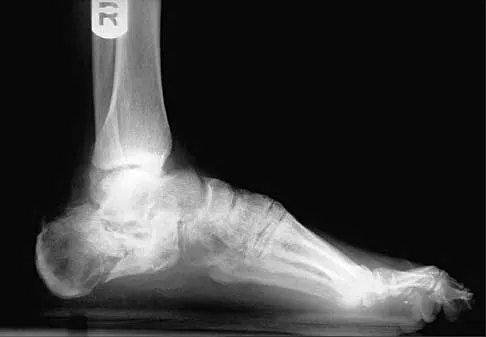
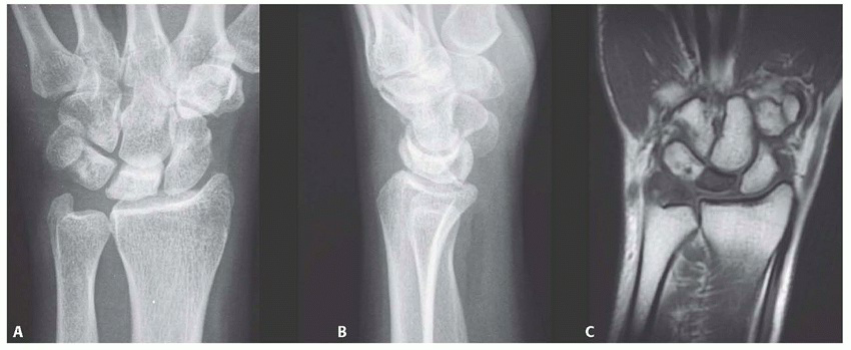
- High-Quality Plain Radiography: Zero-rotation PA, true lateral, and oblique views. The PA view is utilized to calculate ulnar variance (using the method of perpendiculars), carpal height ratio (revised method of Nattrass), and Stahl's index. The lateral view is critical for assessing the radioscaphoid angle to differentiate Stage IIIA from IIIB.
- Magnetic Resonance Imaging (MRI): The gold standard for assessing lunate viability. T1-weighted sequences demonstrate the extent of ischemia (low signal). T2/STIR sequences identify reactive edema or early fragmentation. MRI is also vital for evaluating the integrity of the lunate articular cartilage.
- Computed Tomography (CT): Thin-slice sagittal and coronal CT reconstruction is highly recommended to assess the precise 3D geometry of lunate collapse, cortical breaks, and to template the capitate osteotomy.
Anesthesia and Patient Positioning
- Anesthesia: General anesthesia is standard, typically augmented with a regional supraclavicular or axillary brachial plexus block to provide profound intraoperative vasodilation (beneficial for microvascular pedicle harvest) and robust postoperative analgesia.
- Positioning: The patient is placed supine. The operative extremity is abducted onto a radiolucent hand table.
- Tourniquet: A well-padded pneumatic upper arm tourniquet is applied. Exsanguination is performed via gravity elevation rather than an Esmarch bandage to prevent complete emptying of the microvasculature, which aids in the visualization of the tiny venae comitantes during pedicle dissection.
- Equipment: Loupe magnification (minimum 3.5x to 4.5x) or an operating microscope is mandatory. A micro-sagittal saw, high-speed burr, and headless compression screws (e.g., 2.5 mm or 3.0 mm) must be available.
Detailed Surgical Approach and Technique
The combined procedure is a highly choreographed sequence involving graft harvest, central column shortening, lunate debridement, and microvascular insetting.
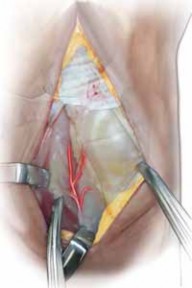
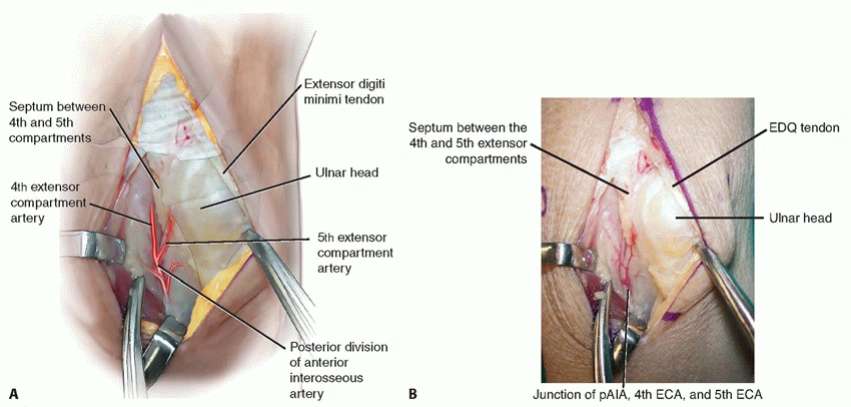
I. Vascularized Bone Graft Harvest (4+5 ECA)
1. Incision and Exposure
A 5- to 6-cm longitudinal dorsal incision is centered over the distal radius, spanning the interval between the fourth and fifth extensor compartments.
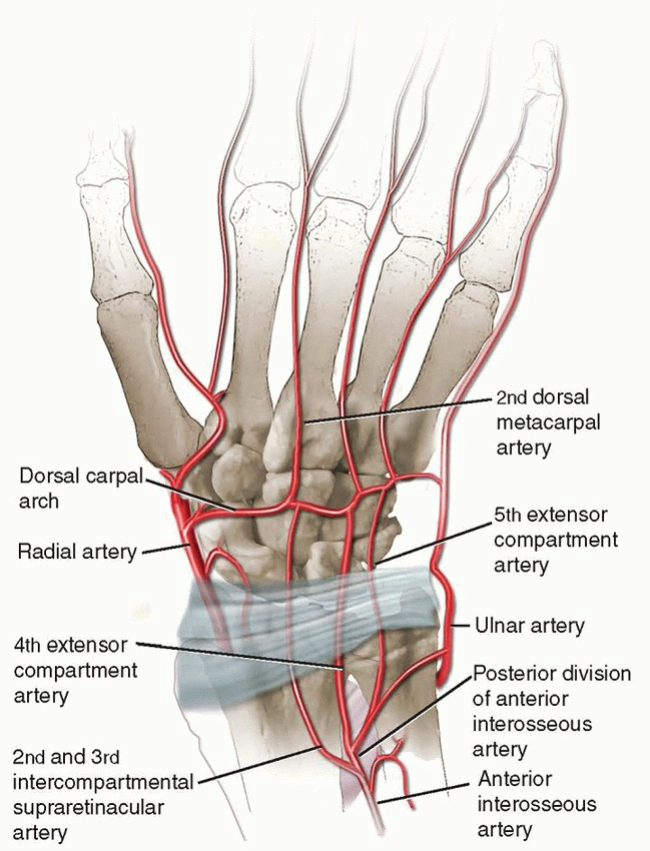
Subcutaneous tissues are bluntly dissected to protect superficial sensory branches of the radial and ulnar nerves. The extensor retinaculum is identified.
2. Pedicle Identification and Dissection
Using loupe magnification, the interval between the Extensor Digitorum Communis (EDC) and Extensor Digiti Minimi (EDM) is developed. The 4th and 5th extensor compartment artery (ECA) and its paired venae comitantes are identified within the loose areolar tissue overlying the radiocarpal capsule.
Meticulous sharp and blunt dissection is utilized to mobilize the pedicle proximally to its origin, ensuring an adequate transposition arc to reach the lunate without tension. A cuff of perivascular tissue is preserved to prevent vasospasm and protect the delicate venous drainage.
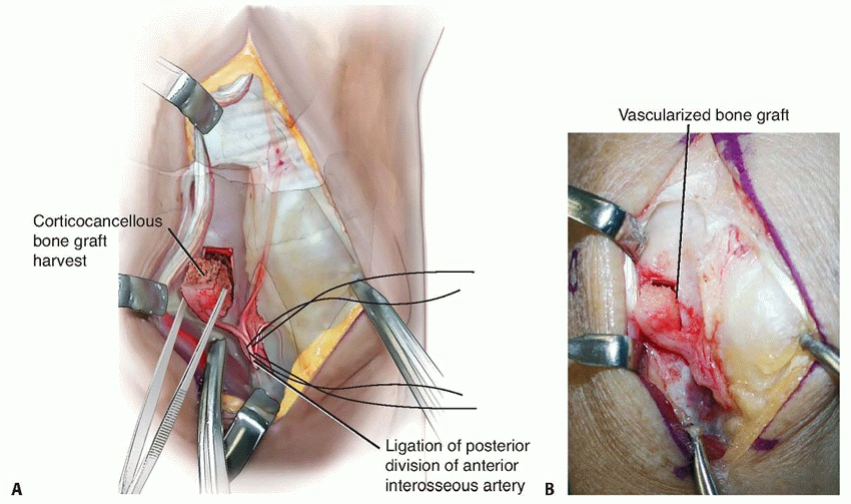
3. Bone Block Harvest
A 1.0 cm x 1.0 cm block of corticocancellous bone is mapped out on the dorsal metaphysis of the distal radius, incorporating the distal insertion of the pedicle. Using a micro-sagittal saw under continuous saline irrigation (to prevent thermal necrosis), the bone block is osteotomized. The graft is gently elevated with a fine osteotome, confirming active punctate bleeding from the cancellous surface upon temporary tourniquet deflation.
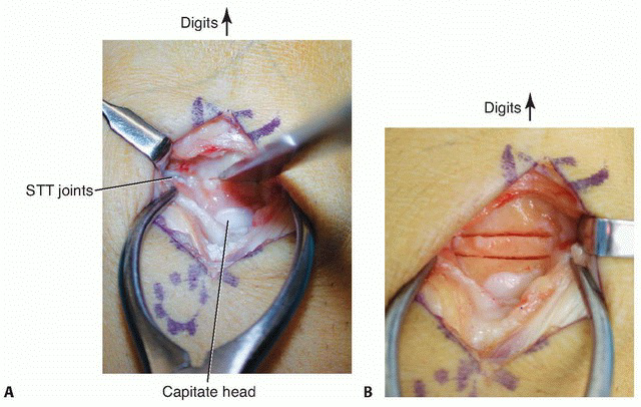
II. Capitate Shortening Osteotomy
1. Exposure and Osteotomy Planning
The dorsal incision is extended distally, or a separate longitudinal incision is made over the capitate. The third compartment is opened, and the Extensor Pollicis Longus (EPL) is transposed radially. A capsulotomy exposes the midcarpal joint. Using preoperative CT templates and intraoperative fluoroscopy, the osteotomy site is marked at the capitate waist.

2. Osteotomy Execution
Two parallel transverse cuts are made with a micro-sagittal saw, resecting a precise 2 to 3 mm wafer of bone from the capitate body. Care is taken to maintain the sagittal alignment of the capitate to prevent creating a destabilizing angular deformity.
3. Reduction and Rigid Fixation
The distal pole of the capitate is translated proximally to close the osteotomy gap, effectively shortening the central column. Rigid internal fixation is achieved using two headless compression screws placed in a retrograde or antegrade fashion.
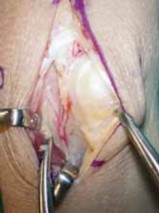
Alternatively, or as provisional fixation, parallel K-wires can be utilized.
Fluoroscopy confirms anatomic reduction, complete closure of the osteotomy site, and appropriate hardware placement without intra-articular penetration.
III. Lunate Preparation and Graft Placement
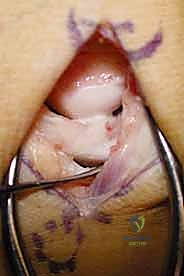
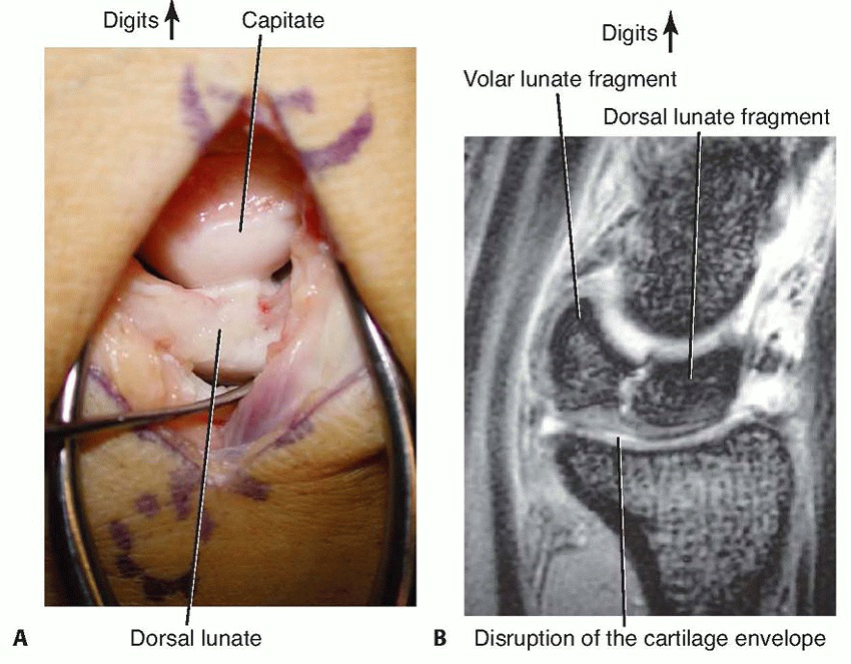
1. Lunate Core Decompression
The dorsal capsule over the lunate is sharply incised. A high-speed burr and micro-curettes are utilized to excavate the necrotic, sclerotic cancellous bone from the dorsal aspect of the lunate. It is imperative to preserve the subchondral bone shell and the volar/dorsal ligamentous attachments to maintain carpal stability. The defect is shaped to precisely match the dimensions of the harvested radial bone graft.
2. Graft Insetting
The vascularized bone graft is transposed into the lunate defect. The surgeon must meticulously verify that the pedicle follows a smooth, sweeping curve and is free from any kinks, twists, or external compression.
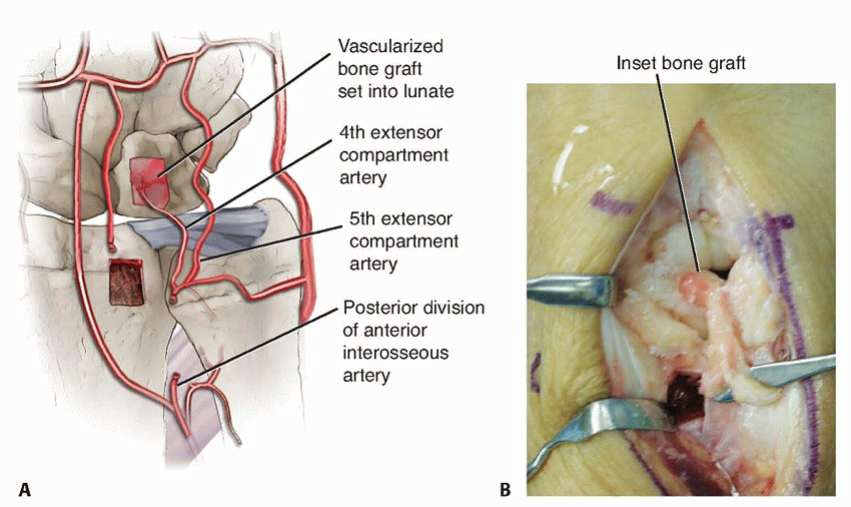
The graft is oriented such that its cancellous surface interfaces directly with the remaining viable lunate bone bed, promoting maximal osteointegration.
The graft is press-fit into the defect. If necessary, supplementary fixation with a micro K-wire or the application of fibrin tissue adhesive can be used to prevent micromotion.
IV. Closure and Immobilization
The tourniquet is deflated, and hemostasis is meticulously obtained. The viability of the graft is visually confirmed. The dorsal capsule is closed loosely to avoid compressing the vascular pedicle. The extensor retinaculum is repaired, leaving the EPL transposed subcutaneously. The skin is closed in layers. A bulky, sterile dressing and a rigid sugar-tong splint are applied with the wrist in neutral alignment.
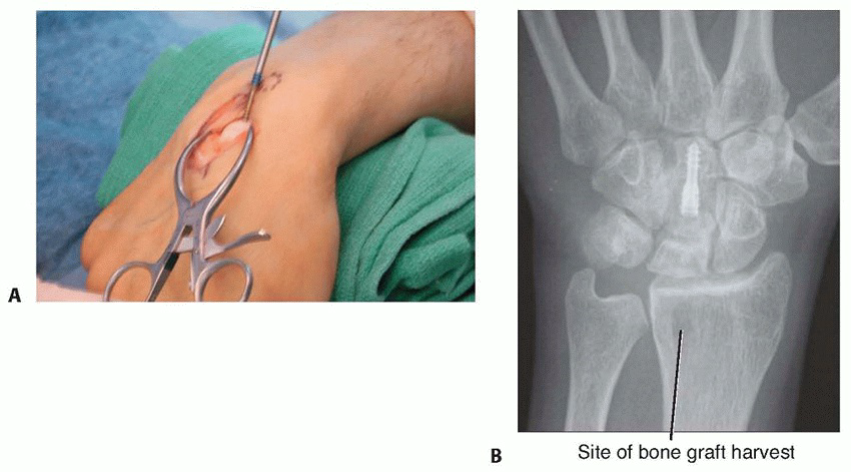
Complications and Management
Surgical intervention for Kienböck disease carries inherent risks. Anticipation and rapid management of these complications are critical for preserving wrist function.
Intraoperative Complications
- Pedicle Avulsion or Thrombosis: The most devastating technical error. Excessive traction or thermal injury can thrombose the delicate 4+5 ECA. Management: If recognized intraoperatively, the graft must be treated as a non-vascularized block, or an alternative pedicle (e.g., 1,2-ICSRA or a free medial femoral condyle flap) must be harvested.
- Iatrogenic Articular Damage: Over-penetration of the saw blade during capitate osteotomy or lunate debridement can damage the midcarpal or radiocarpal joints.
- Inadequate Capitate Fixation: Poor screw purchase can lead to osteotomy displacement. Management: Immediate revision with larger diameter headless screws or supplementary K-wire fixation.
Postoperative Complications
- Capitate Nonunion: Occurs in 5-10% of cases, often secondary to thermal necrosis during the osteotomy or inadequate rigid compression. Management: Prolonged immobilization, bone stimulators, or surgical revision with structural bone grafting and re-plating.
- Progressive Lunate Collapse (Failure of Revascularization): Despite a technically successful VBG, the lunate may continue to fragment, particularly in Stage IIIA/IIIB patients. Management: Conversion to a salvage procedure (Proximal Row Carpectomy or Four-Corner Fusion).
- Hardware Prominence: Headless screws backing out of the capitate can cause extensor tendon irritation or rupture. Management: Hardware removal after definitive radiographic union is achieved.
- Wrist Stiffness: Arthrofibrosis is ubiquitous following carpal surgery and prolonged immobilization. Management: Aggressive, protocol-driven hand therapy.
Complication Summary Table
| Complication | Estimated Incidence | Etiology / Risk Factors | Academic Salvage Strategy |
|---|---|---|---|
| Vascular Pedicle Failure | < 5% | Traction injury, poor dissection, smoking | Alternative VBG, treat as non-vascularized graft |
| Capitate Nonunion | 5 - 10% | Poor compression, thermal necrosis, smoking | Revision osteotomy, iliac crest bone graft, rigid plate fixation |
| Progressive AVN / Collapse | 10 - 20% | Advanced preoperative stage (IIIB), biologic failure | Proximal Row Carpectomy (PRC), Total Wrist Arthrodesis |
| Arthrofibrosis / Stiffness | 20 - 30% | Prolonged casting, capsular scarring | Intensive OT/PT, arthroscopic capsular release (late) |
| Hardware Irritation | 10 - 15% | Screw back-out, prominent trailing threads | Elective hardware removal post-union |
Post Operative Rehabilitation Protocols
A highly structured, multiphasic rehabilitation protocol is mandatory to ensure osteotomy union, protect the microvascular pedicle, and restore functional carpal kinematics.
Phase I: Maximum Protection (Weeks 0 to 6)
- Objective: Protect the capitate osteotomy and fragile vascular pedicle; control postoperative edema.
- Immobilization: The patient is maintained in a rigid short-arm cast or custom thermoplastic splint with the wrist strictly immobilized in neutral.
- Therapy: Absolute non-weight-bearing. Active Range of Motion (AROM) of the digits, thumb, elbow, and shoulder is initiated immediately to prevent extrinsic tendon adhesions and proximal joint stiffness.
- Monitoring: Radiographs are obtained at 2 and 6 weeks to assess capitate osteotomy alignment and hardware position.
Phase II: Controlled Mobilization (Weeks 6 to 12)
- Objective: Initiate safe radiocarpal and midcarpal motion; confirm clinical and radiographic union of the capitate.
- Immobilization: Upon radiographic evidence of bridging trabeculae at the capitate osteotomy site (typically 6-8 weeks), rigid immobilization is discontinued. A removable resting splint is utilized between therapy sessions and at night.
- Therapy: Supervised Active Range of Motion (AROM) and Active-Assisted Range of Motion (AAROM) of the wrist in flexion, extension, and radioulnar deviation. Passive stretching is strictly avoided to prevent sheer stress on the revascularizing lunate.
- Strengthening: Gentle isometric strengthening is introduced at week 10 if clinically stable.
Phase III: Strengthening and Functional Return (Weeks 12 to 24+)
- Objective: Maximize grip strength, endurance, and return to occupational/recreational baseline.
- Therapy: Progressive resistive exercises (PREs) targeting the forearm musculature. Dynamic splinting may be considered if persistent flexion/extension deficits exist.
- Clearance: Return to heavy manual labor or high-impact sports is generally restricted until 6 months postoperatively, contingent upon complete radiographic consolidation of the capitate and MRI evidence of lunate revascularization.
- Surveillance: Serial radiographs at 6 months, 1 year, and 2 years to monitor for late carpal collapse or adjacent segment arthrosis.
Summary of Key Guidelines
The integration of Vascularized Bone Grafting and Capitate Shortening Osteotomy represents a pinnacle in the joint-preserving management of Kienböck disease. Academic consensus and peer-reviewed literature underscore several critical tenets:
Efficacy of Combined Modalities
Extensive clinical series (e.g., Moran, Mathoulin) have validated that addressing both the biological ischemia and the mechanical overload yields superior long-term outcomes compared to isolated procedures. VBG alone in the presence of negative ulnar variance is highly susceptible to mechanical failure, while isolated unloading osteotomies do not reliably reverse established lunate necrosis.
Comparative Interventions
- Radial Shortening Osteotomy: While effective for unloading the lunate in ulnar negative variance, radial shortening alters the congruency of the DRUJ and carries a higher risk of ulnocarpal impaction or DRUJ arthrosis. CSO provides equivalent central column unloading without disrupting the distal radioulnar articulation.
- Salvage Procedures (PRC and Arthrodesis): Reserved strictly for Lichtman Stage IV or failed joint-preserving attempts. While they provide reliable pain relief, they permanently alter carpal kinematics and result in a significant, irreversible loss of grip strength and range of motion.
Future Directions
Ongoing academic research is focused on optimizing graft selection (e.g., comparing dorsal pedicled grafts to free medial femoral condyle flaps for massive defects) and the integration of orthobiologics (Bone Morphogenetic Proteins, Platelet-Rich Plasma) to accelerate angiogenesis within the necrotic lunate core.
Clinical and Radiographic Imaging