Orthopaedic Management of Neuromuscular Disorders: DMD and SMA
Key Takeaway
The orthopaedic management of neuromuscular disorders, including Duchenne Muscular Dystrophy (DMD) and Spinal Muscular Atrophy (SMA), requires a multidisciplinary approach. Surgical interventions primarily address progressive spinal deformities, such as neuromuscular scoliosis, and lower extremity contractures to preserve mobility and pulmonary function. This guide details evidence-based surgical techniques, including posterior spinal fusion with pelvic fixation, tendon transfers, and the management of paralytic hip dysplasia and coxa valga.
Introduction to Neuromuscular Orthopaedics
The orthopaedic management of childhood neuromuscular disorders—encompassing conditions such as Duchenne Muscular Dystrophy (DMD), Spinal Muscular Atrophy (SMA), and Cerebral Palsy (CP)—presents a complex clinical challenge. These conditions are characterized by progressive muscle weakness, muscular imbalance, and subsequent joint contractures, spinal deformities, and hip dysplasia.
The primary goals of orthopaedic intervention are to prolong ambulatory capacity, maintain sitting balance, prevent painful joint dislocations, and optimize pulmonary function. Because these diseases are often progressive, surgical timing is critical. Interventions must be carefully coordinated with the patient's overall medical status, particularly regarding cardiopulmonary reserve and bone mineral density.
Clinical Pearl: In progressive neuromuscular disorders, the loss of ambulation is a critical inflection point. Once a patient becomes wheelchair-bound, the progression of paralytic scoliosis accelerates dramatically, and pulmonary function begins a steady decline. Proactive surgical stabilization is often required before forced vital capacity (FVC) drops below safe anesthetic thresholds.
Duchenne Muscular Dystrophy (DMD)
Duchenne Muscular Dystrophy is an X-linked recessive disorder caused by a mutation in the dystrophin gene, leading to the absence of the dystrophin protein. This absence results in progressive myofiber degeneration and replacement by fibrofatty tissue.
Medical Management and Bone Health
Historically, the natural history of DMD involved loss of ambulation by age 10 to 12, followed by severe scoliosis. The advent of corticosteroid therapy (e.g., prednisone, deflazacort) has significantly altered this trajectory, prolonging ambulation and delaying the onset of scoliosis. However, chronic steroid use introduces secondary orthopaedic complications, most notably severe osteopenia and an increased risk of long-bone and vertebral compression fractures.
Cyclic administration of bisphosphonates (e.g., pamidronate) is increasingly utilized to mitigate steroid-induced osteoporosis and stabilize the growing skeleton.
Lower Extremity Deformities and Management
As muscle weakness progresses in DMD, a predictable pattern of contractures develops, typically beginning with the gastrocnemius-soleus complex and the iliotibial band. This leads to an equinovarus foot deformity, which severely compromises the patient's ability to maintain a plantigrade foot for ambulation.
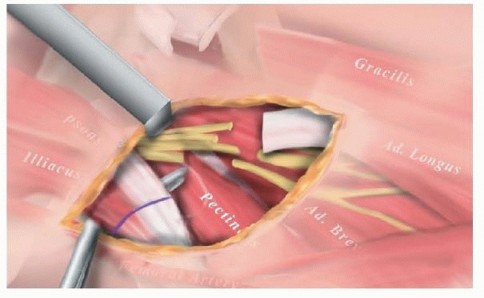
Posterior Tibial Tendon Transfer (PTTT)
To correct the equinovarus deformity and remove the deforming inversion force, a transfer of the posterior tibial tendon anteriorly through the interosseous membrane is frequently indicated.
Surgical Steps for PTTT:
1. Positioning: Supine with a bump under the ipsilateral hip to internally rotate the leg.
2. Tendon Harvest: A medial longitudinal incision is made over the navicular insertion. The posterior tibial tendon (PTT) is detached as distally as possible, maximizing length.
3. Proximal Release: A second incision is made at the medial musculotendinous junction in the distal third of the calf. The PTT is identified and pulled proximally into this wound.
4. Interosseous Routing: A third incision is made over the anterior compartment of the leg. A large window is bluntly created in the interosseous membrane. The PTT is passed from the posterior compartment to the anterior compartment through this window. Care must be taken to avoid the anterior tibial neurovascular bundle.
5. Fixation: A fourth incision is made over the dorsolateral midfoot (typically the lateral cuneiform or cuboid). The tendon is routed subcutaneously to this insertion site and secured using a biotenodesis screw or passed through a drill hole and sutured to itself under appropriate tension (foot held in neutral dorsiflexion and slight eversion).
6. Postoperative Protocol: The patient is placed in a short-leg cast for 6 weeks, non-weight-bearing, followed by an AFO (Ankle-Foot Orthosis) to protect the transfer during rehabilitation.
Spinal Deformity in DMD
Scoliosis in DMD is typically a long, sweeping, flaccid neuromuscular curve that inevitably progresses once the patient is wheelchair-bound. Associated pelvic obliquity leads to loss of sitting balance, ischial pressure sores, and further compromise of diaphragmatic excursion.
Surgical Warning: Patients with DMD are at a high risk for malignant hyperthermia-like reactions and rhabdomyolysis when exposed to depolarizing muscle relaxants (e.g., succinylcholine) or volatile inhalation anesthetics. Total Intravenous Anesthesia (TIVA) is mandatory.
Posterior Spinal Fusion with Pelvic Fixation
The gold standard for DMD scoliosis is a posterior spinal fusion (PSF) extending from the upper thoracic spine (T2 or T3) down to the pelvis. Stopping short of the pelvis inevitably leads to progressive pelvic obliquity and hardware failure.
The Galveston Technique for Pelvic Fixation:
Originally described by Allen and Ferguson, the Galveston technique remains a foundational concept for spinopelvic fixation, though modern modular iliac screws have largely replaced the classic continuous rod bending. The biomechanical principle involves anchoring the instrumentation between the inner and outer tables of the ilium.
- Exposure: A standard midline subperiosteal exposure is performed from T2 to the sacrum. The exposure is extended bilaterally over the posterior superior iliac spine (PSIS).
- Iliac Preparation: The PSIS is decorticated. A starting hole is made with an awl at the PSIS, directed toward the anterior inferior iliac spine (AIIS), staying strictly between the inner and outer cortical tables of the ilium.
- Instrumentation: Modern techniques utilize pedicle screws in the lumbar and thoracic spine. For pelvic fixation, iliac screws (or S2-alar-iliac screws) are placed into the prepared iliac pathways.
- Correction: Bilateral rods are contoured to restore physiologic sagittal alignment (lumbar lordosis and thoracic kyphosis). The rods are seated into the pedicle and iliac screws. Correction is achieved via cantilever reduction and translation.
- Arthrodesis: Meticulous facetectomies and decortication are performed. Copious autograft (from local bone) and allograft are packed along the fusion bed.
Spinal Muscular Atrophy (SMA)
Spinal Muscular Atrophy is an autosomal recessive disorder caused by a mutation in the SMN1 gene, leading to the degeneration of anterior horn cells in the spinal cord. This results in profound, symmetrical, flaccid paralysis. Unlike DMD, cognitive function is entirely spared, and sensation is intact.
Paralytic Hip Dysplasia in SMA
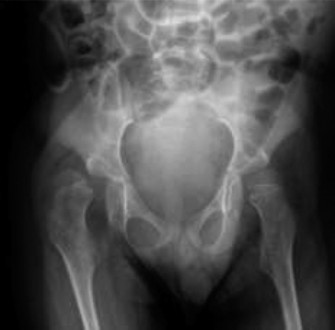
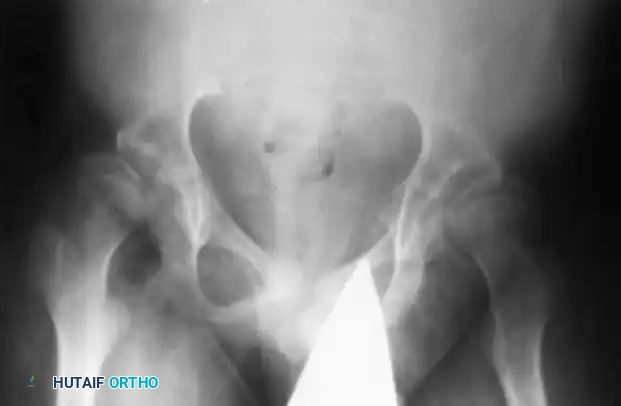
Hip instability is a hallmark of severe SMA (Types I and II). The profound weakness of the gluteal and pelvic musculature, combined with the lack of normal weight-bearing forces, leads to a failure of normal proximal femoral and acetabular development.
The classic radiographic presentation is severe coxa valga (an increased neck-shaft angle) combined with acetabular dysplasia, ultimately leading to subluxation or complete dislocation of the hip.

Fig. 32-25 Coxa valga deformity and subluxation in 12-year-old child with spinal muscular atrophy.
Management of the SMA Hip
The management of hip dislocation in SMA is highly controversial. Because these patients are non-ambulatory and lack the muscle strength to maintain joint reduction even after extensive reconstructive surgery, the recurrence rate of dislocation is exceptionally high.
- Observation: In many cases, a chronically dislocated hip in a patient with severe SMA is painless and does not interfere with sitting balance. In such scenarios, benign neglect is the most appropriate, evidence-based course of action.
- Surgical Reconstruction: If the hip is painful, or if severe pelvic obliquity is interfering with sitting or spinal alignment, surgical intervention may be considered. This typically requires a comprehensive approach:
- Soft Tissue Release: Adductor and iliopsoas tenotomies.
- Femoral Osteotomy: A Varus Derotational Osteotomy (VDRO) to correct the severe coxa valga and excessive femoral anteversion.
- Pelvic Osteotomy: A Dega or San Diego-type pericapsular osteotomy to address the acetabular deficiency and provide anterolateral coverage.
Pitfall: Performing isolated soft-tissue releases or isolated femoral osteotomies in neuromuscular hip dysplasia almost universally leads to failure. The pathology is bi-articular (acetabular and femoral), and both sides of the joint must be addressed simultaneously if surgery is undertaken.
Foot Deformities in Neuromuscular Disorders
While equinovarus is common in DMD, other neuromuscular conditions, such as Cerebral Palsy or certain peripheral neuropathies, frequently present with severe planovalgus foot deformities. The collapse of the medial longitudinal arch, combined with hindfoot valgus and forefoot abduction, leads to painful brace wear and skin breakdown over the prominent talar head.
Subtalar Arthroereisis
For flexible planovalgus deformities in young children with neuromuscular problems, subtalar stabilization via arthroereisis can be an effective joint-sparing procedure. This technique limits excessive subtalar eversion without requiring a formal arthrodesis.
Surgical Technique:
1. Incision: A small 2-3 cm oblique incision is made over the sinus tarsi, following the Langer lines.
2. Exposure: The extensor digitorum brevis muscle belly is elevated dorsally. The fibrofatty contents of the sinus tarsi are evacuated to expose the subtalar joint.
3. Trialing: The foot is manipulated into a neutral position (correcting the valgus). Sizing trials are inserted into the sinus tarsi to determine the appropriate implant size that blocks excessive eversion while allowing a few degrees of normal subtalar motion.
4. Implantation: The definitive implant (often a metallic or bioabsorbable plug, or historically a staple) is inserted.
5. Adjunctive Procedures: Arthroereisis is rarely performed in isolation. It is almost always combined with a percutaneous Achilles tendon lengthening (to address equinus contracture) and occasionally a medial column plication.
Diagnostic Modalities: Muscle Biopsy
While genetic testing has largely replaced muscle biopsy for the diagnosis of DMD and SMA, biopsies remain critical for diagnosing congenital myopathies and atypical neuromuscular presentations.
Open Muscle Biopsy Technique:
Proper surgical technique is paramount to avoid crush artifact, which renders the specimen useless for histopathological and electron microscopic evaluation.
1. Site Selection: The vastus lateralis or rectus femoris is typically chosen. The muscle should be moderately affected but not completely replaced by fat.
2. Anesthesia: Local anesthesia is infiltrated only into the skin and subcutaneous tissue. Injecting local anesthetic directly into the muscle fibers causes artifactual changes.
3. Harvest: A 3 cm longitudinal incision is made. The fascia is opened. A specialized muscle clamp (e.g., Ray clamp) is applied to a fascicle of muscle to maintain its resting length.
4. Excision: The muscle is sharply excised outside the jaws of the clamp. The specimen is immediately wrapped in saline-moistened gauze (not soaked) and transported fresh to the pathology laboratory for immediate freezing and histochemical staining.
Conclusion
The orthopaedic surgeon plays a vital role in the multidisciplinary care of patients with neuromuscular disorders. Through a deep understanding of the natural history of diseases like Duchenne Muscular Dystrophy and Spinal Muscular Atrophy, the surgeon can appropriately time interventions. Whether performing a complex spinopelvic fusion to preserve sitting balance or a tendon transfer to maintain plantigrade foot positioning, the ultimate goal remains the optimization of the patient's quality of life, functional independence, and cardiopulmonary health. Strict adherence to evidence-based protocols, meticulous surgical technique, and rigorous perioperative medical management are the cornerstones of success in this challenging patient population.
📚 Medical References
- neuromuscular disorders, J Pediatr Orthop 8:278, 1988.
- Matsuo T, Hara H, Tada S: Selective lengthening of the psoas and rectus femoris and preservation of the iliacus for fl exion deformity of the hip in cerebral palsy patients, J Pediatr Orthop 7:690, 1987.
- Matsuo T, Tada S, Hajime T: Insuffi ciency of the hip adductor after anterior obturator neurectomy in 42 children with cerebral palsy, J Pediatr Orthop 6:686, 1986.
- Mazur JM, Danko AM, Standard SC, et al: Remodeling of the proximal femur after varus osteotomy in children with cerebral palsy, Dev Med Child Neurol 46:412, 2004.
- McCarthy RE, Simon S, Douglas B, et al: Proximal femoral resection to allow adults who have severe cerebral palsy to sit, J Bone Joint Surg 70A:1011, 1988.
- McMulkin ML, Baird GO, Barr KM, et al: Proximal rectus femoris release surgery is not effective in normalizing hip and pelvic variables during gait in children with cerebral palsy, J Pediatr Orthop 25:74, 2005.
- McNerney NP, Mubarak SJ, Wenger DR: One-stage correction of the dysplastic hip in cerebral palsy with the San Diego acetabuloplasty: results and complications in 104 hips, J Pediatr Orthop 20:93, 2000.
- Miller F, Dias RC, Dabney K, et al: Soft-tissue release for spastic hip subluxation in cerebral palsy, J Pediatr Orthop 17:571, 1997.
- Miller F, Slomczykowski M, Cope R, et al: Computer modeling of the pathomechanics of spastic hip dislocation in children, J Pediatr Orthop 19:486, 1999.
- Moreau M, Cook PC, Ashton B: Adductor and psoas release for subluxation of the hip in children with spastic cerebral palsy, J Pediatr Orthop 15:672, 1995.
- Moreau M, Drummond DS, Rogala E, et al: Natural history of the dislocated hip in spastic cerebral palsy, Dev Med Child Neurol 21:749, 1979.
- Mubarak SJ, Mortensen W, Katz M: Combined pelvic (Dega) and femoral osteotomies in the treatment of paralytic hip dislocation, J Pediatr Orthop 7:493, 1987 (abstract). Murray-Weir M, Root L, Peterson M, et al: Proximal femoral varus rotation osteotomy in cerebral palsy: a prospective gait study, J Pediatr Orthop 23:321, 2003.
- Nickel VL, Perry J, Garrett A, Feiwell EN: Paralytic dislocation of the hip, J Bone Joint Surg 48A:1021, 1966.
- Noonan KJ, Walker TL, Kayes KJ, et al: Varus derotation osteotomy for the treatment of hip subluxation and dislocation in cerebral palsy: statistical analysis in 73 hips, J Pediatr Orthop B 10:279, 2001.
- Novacheck TF, Trost JP, Schwartz MH: Intramuscular psoas lengthening improves dynamic hip function in children with cerebral palsy, J Pediatr Orthop 22:158, 2002.
- Osterkamp J, Caillouette JT, Hoffer MM: Chiari osteotomy in cerebral palsy, J Pediatr Orthop 8:274, 1988.
- Pap K, Kiss S, Vizkelety T, et al: Open adductor tenotomy in the prevention of hip subluxation in cerebral palsy, Int Orthop 29:18, 2005.
- Pemberton PA: Pericapsular osteotomy of the ilium in the treatment of congenital dislocation and subluxation of the hip, J Bone Joint Surg 47A:65, 1965.
- Perry J: Kinesiology of lower-extremity bracing, Clin Orthop Relat Res 102:18, 1974.
- Perry J: Distal rectus femoris transfer, Dev Med Child Neurol 29:153, 1987.
- Perry J, Hoffer MM: Preoperative and postoperative dynamic electromyography as an aid in planning
You Might Also Like