Congenital Muscular Torticollis: Operative Guide
Key Takeaway
Congenital muscular torticollis (CMT) is a pediatric deformity caused by fibromatosis and subsequent contracture of the sternocleidomastoid muscle. While most cases resolve with conservative stretching during infancy, persistent deformity beyond one year of age necessitates surgical intervention. Operative management, typically via unipolar or bipolar release, aims to restore cervical range of motion and halt progressive facial asymmetry and plagiocephaly. Careful protection of the spinal accessory nerve is paramount during surgical dissection.
CONGENITAL MUSCULAR TORTICOLLIS: PATHOPHYSIOLOGY AND CLINICAL EVALUATION
Congenital muscular torticollis (CMT) is a complex pediatric musculoskeletal deformity characterized by unilateral shortening and fibrosis of the sternocleidomastoid (SCM) muscle. This contracture forces the infant's head into a pathognomonic posture: lateral flexion toward the affected side and rotation of the chin toward the contralateral shoulder.
The underlying pathology is driven by fibromatosis within the SCM muscle. Clinically, a firm, non-tender, fusiform mass (often termed a "sternomastoid tumor" or "pseudotumor") is either palpable at birth or becomes prominent during the first two weeks of life. Epidemiologically, CMT is more frequently observed on the right side. The fibrotic process may involve the muscle belly diffusely, but it is most frequently localized near the clavicular attachment.
The natural history of the SCM mass is predictable: it attains maximal size within 1 to 2 months of age and subsequently begins to regress. In the vast majority of cases, the mass diminishes and completely disappears within the first year of life. However, if spontaneous resolution fails to occur, the muscle becomes permanently fibrotic, inelastic, and contracted. This results in a permanent torticollis deformity that requires surgical intervention to prevent long-term craniofacial and cervical spine sequelae.
Etiology and the Compartment Syndrome Hypothesis
Although CMT has been documented in medical literature for centuries, its exact etiology has historically been a subject of debate. Early hypotheses included in utero fetal malposition, direct birth trauma, localized infection, and primary vascular injury. Clinical studies consistently demonstrate that infants with CMT are frequently the product of difficult deliveries (e.g., breech presentation, forceps delivery) and exhibit a significantly increased incidence of associated musculoskeletal anomalies, including metatarsus adductus, talipes equinovarus (clubfoot), and developmental dysplasia of the hip (DDH).
Clinical Pearl: There is a reported incidence of congenital dislocation of the hip or acetabular dysplasia ranging from 7% to 20% in children with CMT. Walsh and Morrissy documented an 8% rate of concurrent hip disease in their cohort. Therefore, careful clinical screening and routine radiographic or ultrasonographic examination of the hips are mandatory in every infant presenting with CMT.
Modern understanding of CMT pathophysiology has been revolutionized by the work of Davids, Wenger, and Mubarak. Their comprehensive investigations—including MRI studies, cadaveric dissections, and in vivo compartment pressure measurements—revealed that the SCM muscle is enveloped in a distinct fascial compartment. MRI of infants with CMT demonstrated signal intensities identical to those seen in extremity compartment syndromes.

Fig. 28-4 Pathophysiology of congenital muscular torticollis proposed by Davids, Wenger, and Mubarak, suggesting that CMT represents the sequela of an intrauterine or perinatal compartment syndrome.
This led to the prevailing theory that CMT is the direct sequela of an intrauterine or perinatal compartment syndrome. Engagement, flexion, bending, and rotation of the fetal neck during labor can cause compression injury to the SCM, leading to ischemia, edema, and increased intracompartmental pressure. This tamponade effect results in muscle infarction, subsequent reperfusion injury, and ultimate fibrotic replacement of the muscle fibers.
Clinical Presentation and Prognostic Imaging
When evaluating an infant with early CMT, predicting whether the fibromatosis will spontaneously resolve or progress to permanent contracture is challenging.
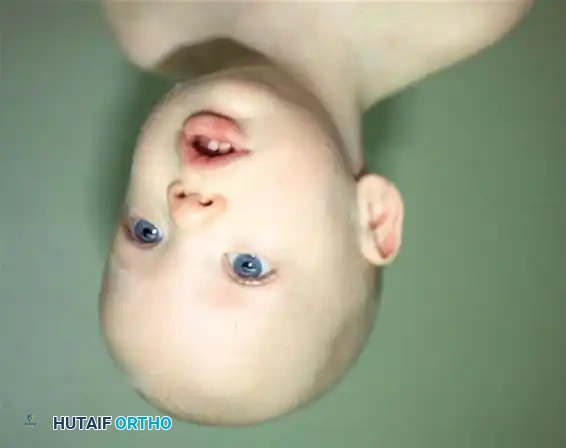
Fig. 28-3 Congenital torticollis in a 14-month-old boy demonstrating characteristic head tilt and contralateral chin rotation.
Lin and Chou demonstrated the high prognostic value of ultrasonography in predicting the need for surgical intervention. In their extensive study of 197 infants:
* Lower Third Involvement: 100% of patients (27/27) with fibrotic changes limited strictly to the lower third of the SCM recovered without surgery.
* Middle/Lower Third Involvement: Only 6% of patients (6/93) required surgery.
* Whole Muscle Involvement: 35% of patients (26/75) with diffuse, whole-muscle fibromatosis ultimately required surgical release.
NONOPERATIVE MANAGEMENT
During infancy, conservative management is the absolute standard of care. Surgical excision of the fibrotic lesion during early infancy is strictly contraindicated and medically unjustified. Surgery must be delayed until the biological evolution of the fibromatosis is entirely complete.
The cornerstone of conservative treatment is a disciplined, parent-led physical therapy regimen. Parents must be instructed on how to perform controlled, gentle manual stretching of the SCM muscle. This involves stabilizing the infant's shoulder and gently laterally flexing the head away from the affected side, followed by rotating the chin toward the affected side.
Coventry and Harris (Mayo Clinic) reported excellent results in 85% of infants treated with home-based conservative stretching. Cheng et al., in a massive prospective study of 821 patients, confirmed that controlled manual stretching is effective in approximately 95% of patients who present before 1 year of age.
Cheng divided patients into three distinct clinical groups:
1. Sternomastoid Tumor Group: Palpable mass present.
2. Muscular Group: Clinical thickening and tightness without a discrete mass.
3. Postural Torticollis Group: Postural head tilt without SCM tightness.
The presence of a palpable tumor, older age at presentation, difficult birth history, right-sided involvement, and a rotational deformity exceeding 15 degrees were all significant predictors of a prolonged treatment duration and a higher likelihood of requiring eventual surgery.
SURGICAL INDICATIONS AND TIMING
If CMT fails to resolve and persists beyond 1 year of age, the deformity becomes permanent and will progressively worsen with skeletal growth. The head becomes rigidly inclined toward the affected side, and the face rotates toward the opposite side.
Surgical Warning: Severe, untreated CMT leads to secondary structural deformities. The ipsilateral shoulder becomes elevated, and the fronto-occipital diameter of the skull decreases, resulting in severe plagiocephaly and facial asymmetry.

Fig. 28-5 Untreated torticollis (right) in a 19-year-old man; note the severely limited rotation, fixed structural deformity, and marked plagiocephaly.
Indications for Surgery:
* Persistence of SCM contracture beyond 12 to 18 months of age.
* Limitation of cervical rotation or lateral bending exceeding 30 degrees.
* Development of progressive facial asymmetry or plagiocephaly.
Timing of Surgery:
Canale, Griffin, and Hubbard (Campbell Clinic) demonstrated that nonoperative therapy after age 1 is rarely successful. Surgery performed before age 6 to 8 years is ideal, as the remaining growth potential allows for the spontaneous remodeling and correction of facial asymmetry and plagiocephaly.
While early intervention is preferred, older children and adults can still benefit. Cheng and Tang reported 81.8% excellent or good results in patients aged 10 years or older at the time of surgery. Shim et al. confirmed that while functional and cosmetic results are slightly less successful in patients who have reached skeletal maturity, marked improvement in neck motion and head tilt is still reliably achieved.
SURGICAL TECHNIQUES FOR SCM RELEASE
Several operative strategies exist for releasing the contracted SCM muscle. The choice of procedure depends on the severity of the contracture and the age of the patient.
* Unipolar Release: Division of the SCM at its distal (clavicular and sternal) attachments. Appropriate for mild to moderate deformities.
* Bipolar Release: Division of the SCM at both its distal attachments and its proximal (mastoid) attachment. Indicated for severe, recalcitrant torticollis.
* Endoscopic Release: A minimally invasive approach offering precise division of muscle fibers and an inconspicuous scar, though it requires specialized training and lacks large long-term series.
Preoperative Planning

Fig. 28-7 A, Seven-year-old boy with left congenital muscular torticollis prior to unipolar release.
Ling's review of 103 patients highlighted potential complications of open tenotomy, including tethering of the surgical scar to deep cervical structures, spontaneous reattachment of the SCM heads, and failure to correct facial asymmetry. To minimize scar tethering, which is common in infants, surgical release is generally delayed until the child is at least 1 to 4 years old.
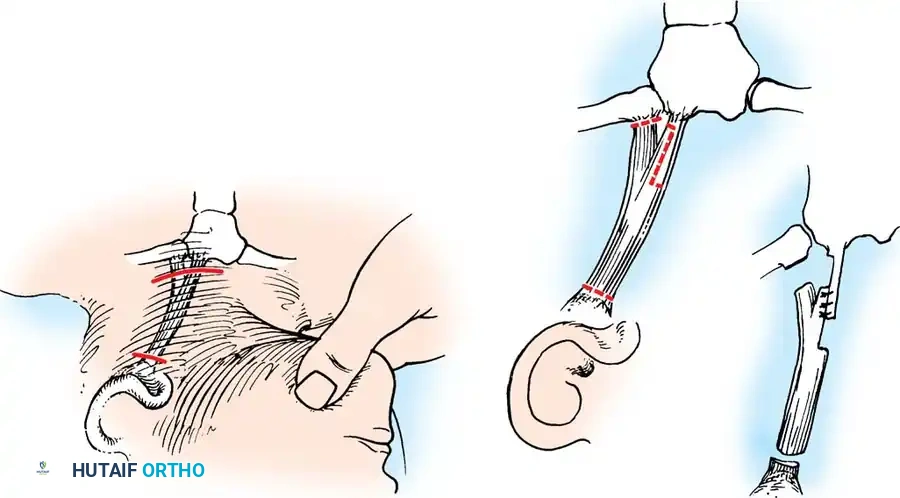
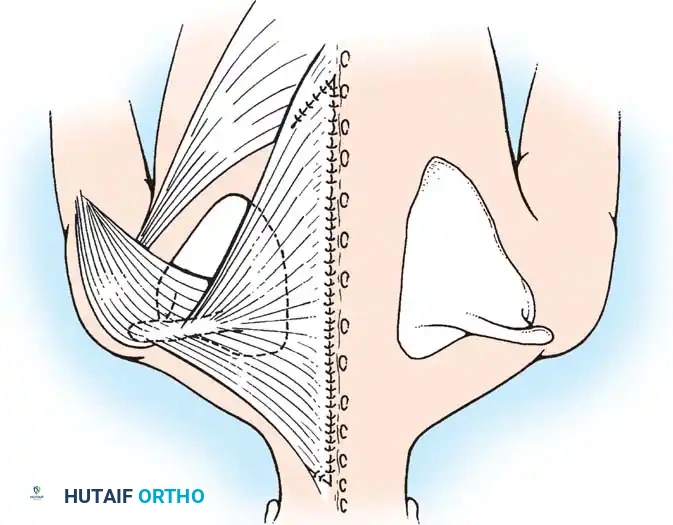
🔪 Surgical Technique 28-3: Unipolar Release
The unipolar release targets the distal insertions of the SCM. Meticulous dissection is required to protect the external jugular vein and the deeper carotid sheath structures.
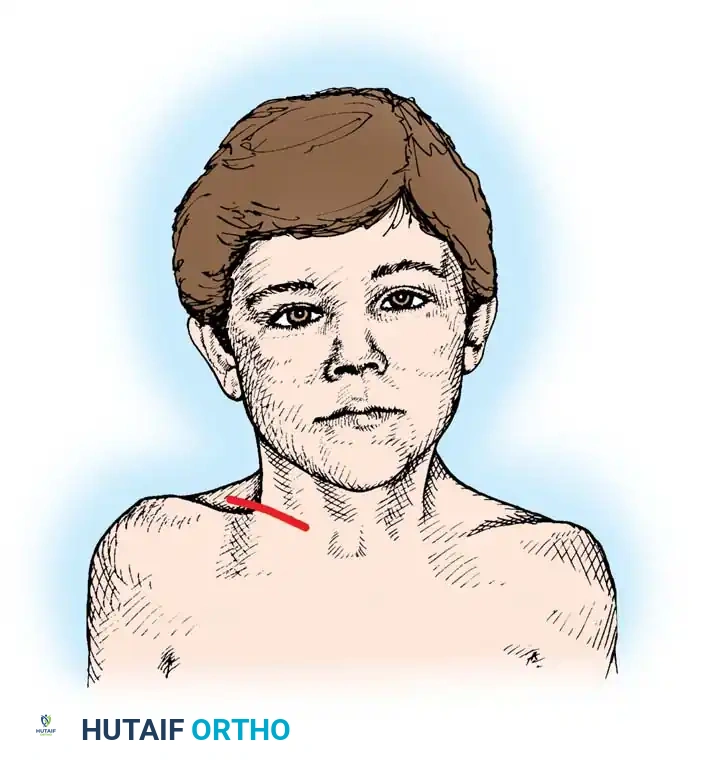
Fig. 28-6 Unipolar release for torticollis. Note the line of the skin incision parallel to the clavicle.
Step-by-Step Procedure:
1. Positioning: Place the patient supine with a transverse roll under the shoulders to extend the neck. Turn the head slightly toward the unaffected side to place the contracted SCM under maximum tension.
2. Incision: Make a transverse skin incision approximately 5 cm long, positioned just superior to and parallel to the medial end of the clavicle (Fig. 28-6). Deepen the incision through the subcutaneous tissue and platysma to expose the tendinous attachments of the sternal and clavicular heads of the SCM.
3. Tendon Isolation: Incise the SCM tendon sheath longitudinally. Carefully pass a curved hemostat or a right-angle blunt instrument posterior to the tendons. Caution: The internal jugular vein, carotid artery, and vagus nerve lie directly posterior to the SCM in the carotid sheath. Stay strictly within the SCM fascial envelope.
4. Resection: By applying traction on the hemostat, draw the tendons outside the wound. Clamp the tendons superiorly and inferiorly, and resect a 2.5 cm segment of their inferior ends.

Surgical diagram demonstrating the isolation and resection of the distal SCM tendons.
- Fascial Release: If the platysma muscle and adjacent cervical fascia remain contracted and restrict motion, divide them meticulously under direct vision.
- Dynamic Assessment: With the child's head turned maximally toward the affected side and the chin depressed, digitally explore the wound for any remaining tight bands of muscle or deep cervical fascia. Divide any residual bands until the deformity can be smoothly overcorrected.
- Conversion to Bipolar Release (If Necessary): If full overcorrection is not possible after distal release, a proximal release is mandated. Make a small transverse incision just inferior to the mastoid process. Carefully divide the proximal SCM muscle belly near the bone.
> CRITICAL PITFALL: The spinal accessory nerve (CN XI) exits the jugular foramen and pierces the deep surface of the SCM approximately 3 to 5 cm inferior to the mastoid tip. Proximal dissection must remain directly on the mastoid periosteum to avoid devastating iatrogenic denervation of the trapezius. - Closure: Achieve meticulous hemostasis. Close the platysma and skin with absorbable subcuticular sutures.
- Immobilization: Apply a bulky, soft cervical dressing that physically holds the head in the overcorrected position (lateral flexion away from the surgical side, chin rotated toward the surgical side).
Postoperative clinical appearance demonstrating restored cervical alignment and resolution of the torticollis posture.
ASSOCIATED CERVICOTHORACIC ANOMALIES: CONGENITAL ELEVATION OF THE SCAPULA (SPRENGEL DEFORMITY)
When evaluating a pediatric patient for torticollis and cervical asymmetry, the orthopedic surgeon must maintain a high index of suspicion for concurrent cervicothoracic anomalies. Congenital elevation of the scapula (Sprengel deformity) frequently presents with severe neck webbing, restricted cervical motion, and apparent torticollis due to the presence of an omovertebral bone or fibrotic bands tethering the cervical spine to the scapula.
If Sprengel deformity is identified as the primary driver of the cervicothoracic asymmetry, the Woodward operation is the procedure of choice to relocate the scapula inferiorly and restore shoulder biomechanics.
The Woodward Operation
The Woodward procedure involves the detachment, inferior translation, and reattachment of the paraspinal musculature to bring the scapula down to an anatomical level.
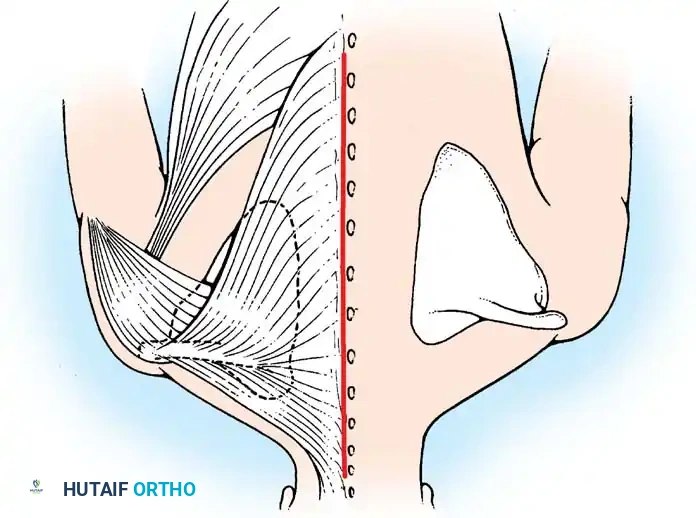
Fig. 28-2 A, Woodward operation for congenital elevation of the scapula. The elevation of the scapula, the extensive origin of the trapezius, and the planned midline skin incision are shown.
Surgical Steps of the Woodward Procedure:
1. Exposure: A midline longitudinal incision is made over the spinous processes from the upper cervical spine down to the lower thoracic spine.
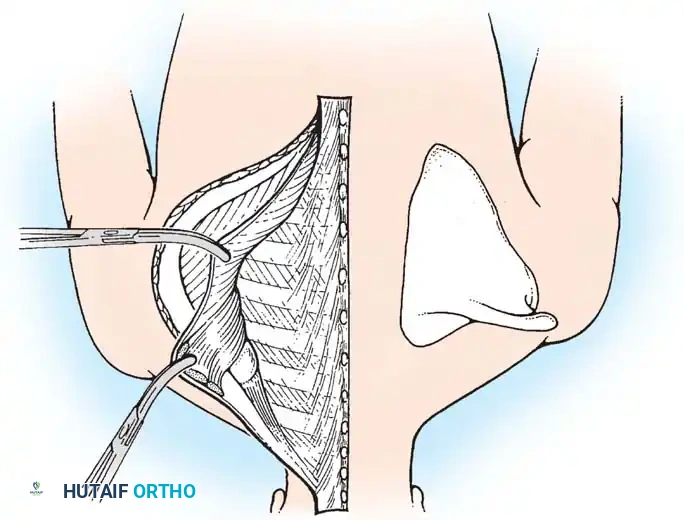
Fig. 28-2 B, The skin has been incised in the midline. The origins of the trapezius and the rhomboideus major and minor have been freed from the spinous processes and retracted laterally. The levator scapulae, any omovertebral bone, and any deformed superior angle of the scapula are identified for excision.
- Muscle Detachment: The origins of the trapezius and rhomboids are sharply detached from the spinous processes. The levator scapulae is excised. If an omovertebral bone is present (tethering the superior angle of the scapula to the cervical spine), it is resected extra-periosteally.
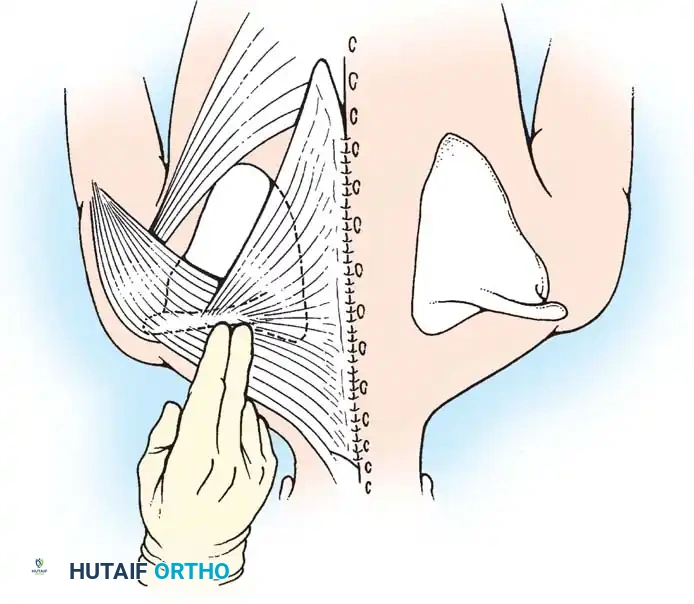
Fig. 28-2 C, The remaining narrow attachment of the trapezius superiorly has been divided at the level of C4. The scapula and its attached sheet of muscles have been displaced inferiorly. The aponeuroses of the trapezius and rhomboids are reattached to the spinous processes at a more inferior, anatomical level. This creates a redundant fold of trapezius aponeurosis inferiorly.
- Inferior Translation: With the tethering structures removed, the entire scapula and its attached muscle sheet are manually displaced inferiorly to match the level of the contralateral normal scapula.
- Reattachment: The
Associated Surgical & Radiographic Imaging
📚 Medical References
- congenital muscular torticollis, J Pediatr Orthop 26:805, 2006.
- Wada A, Jufi i T, Takamura K, et al: Pemberton osteotomy for developmental dysplasia of the hip in older children, J Pediatr Orthop 23:508, 2003.
- Wagner H: Osteotomies for congenital hip dislocation. In The hip: proceedings of the Fourth Open Scientifi c Meeting of the Hip Society, St Louis, 1976, Mosby. Wagner H: Femoral osteotomies for congenital hip dislocation. In Weil VH, ed: Progress in orthopaedic surgery, vol 2, Berlin, 1978, Springer-Verlag. Walsh JJ, Morrissy RT: Torticollis and hip dislocation, J Pediatr Orthop 18:219, 1998.
- Waters P, Kurica K, Hall J, et al: Salter innominate osteotomies in congenital dislocation of the hip, J Pediatr Orthop 8:650, 1988.
- Weinstein SL, Ponseti IV: Congenital dislocation of the hip: open reduction through a medial approach, J Bone Joint Surg 61A:119, 1979.
- Wenger DR: Congenital hip dislocation: techniques for primary open reduction including femoral shortening, Instr Course Lect 38:343, 1989.
- Wenger DR, Lee CD, Kolman B: Derotational femoral shortening for developmental dislocation of the hip: special indications and results in the child younger than 2 years, J Pediatr Orthop 15:768, 1995.
- Westberry DE, Davids JR, Pugh LI: Clubfoot and developmental dysplasia of the hip: value of screening hip radiographs in children with clubfoot, J Pediatr Orthop 23:503, 2003.
- Westin GW, Ilfeld FW, Makin M, et al: Developmental hip dislocation, Contemp Orthop 16:17, 1988.
- Willis RB: Developmental dysplasia of the hip: assessment and treatment before walking age, Instr Course Lect 50:541, 2001.
- Wynne-Davies R: Acetabular dysplasia and familial joint laxity: two etiological factors in congenital dislocation of the hip: a review of 589 patients and their families, J Bone Joint Surg 52B:704, 1970.
- Yoshitaka T, Mitani S, Aoki K, et al: Long-term follow-up of congenital subluxation of the hip, J Pediatr Orthop 21:474, 2001.
- Zadeh HG, Catterall A, Hashemi-Jejad A, et al: Test of stability as an aid to decide the need for osteotomy in association with open reduction in developmental dysplasia of the hip, J Bone Joint Surg 82B:17, 2000.
- Congenital and Developmental Coxa Vara Amstutz HC, Wilson PD Jr: Dysgenesis of the proximal femur (coxa vara) and its surgical management, J Bone Joint Surg 44A:1, 1962.
- Beals RK: Coxa vara in childhood: evaluation and management, J Am Acad Orthop Surg 6:98, 1998.
- Carroll K, Coleman S, Stevens PM: Coxa vara: surgical outcomes of valgus osteotomies, J Pediatr Orthop 17:220, 1997.
- Fisher RL, Waskowitz WJ: Familial developmental coxa vara, Clin Orthop Relat Res 86:2, 1972.
- Garrido IM, Molto FJ, Lluch DB: Distal transfer of the greater trochanter in acquired coxa vara: clinical and radiographic results, J Pediatr Orthop B 12:38, 2003.
- Kalamchi A, Cowell HR, Kim KI: Congenital defi ciency of the femur, J Pediatr Orthop 4:285, 1984.
- Kim HT, Chambers HG, Mubarak SJ, et al: Congenital coxa vara: computed tomographic analysis of femoral retroversion and the triangular metaphyseal fragment, J Pediatr Orthop 20:551, 2000.
- Ogden JA, Lee KE, Rudicel SA, et al: Proximal femoral epiphysiolysis in the neonate, J Pediatr Orthop 4:285, 1984.
- Oh CW, Thacker MM, Mackenzie WG, et al: Coxa vara: a novel measurement technique in skeletal dysplasias, Clin Orthop Relat Res 447:125, 2006.
- Pappas AM: Congenital abnormalities of the femur and related lower-extremity malformations: classifi cation and treatment, J Pediatr Orthop 3:45, 1983.
- Richie MF, Johnston CE II: Management of developmental coxa vara in cleidocranial dysostosis, Orthopedics 12:1001, 1989.
- Sabharwal S, Mittal R, Cox G: Percutaneous triplanar femoral osteotomy correction for developmental coxa vara: a new technique, J Pediatr Orthop 25:28, 2005.
- Weinstein JN, Kuo KN, Millar EA: Congenital coxa vara: a retrospective review, J Pediatr Orthop 4:70, 1984.
- Exstrophy of the Bladder Aadlen RJ, O’Phelan EH, Chisholm TC, et al: Exstrophy of the bladder: long-term results of bilateral posterior iliac osteotomies and two-stage anatomic repair, Clin Orthop Relat Res 151:193, 1980.
- Cracciolo A III, Hall CB: Bilateral iliac osteotomy: the fi rst stage in repair of exstrophy of the bladder, Clin Orthop Relat Res 68:156, 1970.
- Furnas DW, Haq MA, Somers G: One-stage reconstruction for exstrophy of the bladder in girls, Plast Reconstr Surg 56:61, 1975.
- Greene WB, Dias LS, Lindseth RE, et al: Musculoskeletal problems in association with cloacal exstrophy, J Bone Joint Surg 73A:551, 1991.
- Grotte G, Sevastikoglou JA: A modifi ed technique for pelvic reconstruction in the treatment of exstrophy of the bladder, Acta Orthop Scand 37:197, 1966.
- Gugenheim JJ, Gonzales ET Jr, Roth DR, et al: Bilateral posterior pelvic resection osteotomies in patients with exstrophy of the bladder, Clin Orthop Relat Res 364:70, 1999.
- Jani MM, Sponseller PD, Gearhart JP, et al: The hip in adults with classic
You Might Also Like