Mastering the Diagnosis and Surgical Management of Musculoskeletal Infections
Key Takeaway
The diagnosis of musculoskeletal infections requires a high index of suspicion, combining clinical evaluation with targeted laboratory and imaging modalities. While the classic triad of fever, swelling, and localized pain is well-known, indolent presentations are common. Definitive diagnosis relies on a synthesis of inflammatory markers, synovial fluid analysis, advanced imaging, and intraoperative tissue sampling to guide subsequent surgical debridement and targeted antimicrobial therapy.
DIAGNOSIS OF MUSCULOSKELETAL INFECTIONS
The diagnosis of musculoskeletal infection—encompassing acute and chronic osteomyelitis, septic arthritis, and periprosthetic joint infections—remains one of the most challenging and critical domains in orthopaedic surgery. The clinical presentation may range from glaringly obvious to deceptively obscure. Signs and symptoms vary significantly depending on the virulence of the infecting organism, the host's immune status, and the anatomical extent of bone and joint involvement.
While characteristic features such as fever, chills, nausea, vomiting, malaise, erythema, swelling, and localized tenderness are frequently taught, they are not universally present. The classic triad of fever, swelling, and tenderness or pain is highly suggestive, yet pain is arguably the most consistent and common symptom, driven by periosteal stretching and elevated intraosseous pressure. Fever is notoriously inconsistent, particularly in immunocompromised hosts, the elderly, or those partially treated with oral antibiotics.
Furthermore, musculoskeletal infections may present with an indolent course, manifesting merely as progressive mechanical backache (in cases of vertebral osteomyelitis or discitis) or a subtle decrease in the functional capacity of an extremity. Because no single clinical sign or non-invasive test serves as a definitive, standalone indicator of infection, the orthopaedic surgeon must synthesize clinical, laboratory, and imaging data to formulate an accurate diagnosis and subsequent surgical plan.
Clinical Pearl: Always maintain a high index of suspicion for atypical presentations. In neonates, a pseudoparalysis of a limb or a refusal to bear weight in a toddler may be the only clinical manifestation of a deep-seated osteomyelitis or septic arthritis.
LABORATORY STUDIES AND BIOMARKERS
A comprehensive laboratory evaluation is mandatory during the initial workup of suspected bone and joint infections. The standard panel must include a complete blood count (CBC) with differential, erythrocyte sedimentation rate (ESR), and C-reactive protein (CRP).
The Complete Blood Count (CBC)
The total white blood cell (WBC) count is a notoriously unreliable indicator of musculoskeletal infection. It is frequently within normal limits even in the presence of gross purulence. However, the WBC differential is of higher diagnostic utility; an acute bacterial infection typically demonstrates a "left shift" with an absolute increase in polymorphonuclear neutrophils (PMNs).
Erythrocyte Sedimentation Rate (ESR)
The ESR is a measure of the acute phase response, driven primarily by elevated fibrinogen levels causing rouleaux formation of red blood cells. While it becomes elevated in the presence of infection, it is highly non-specific.
* Confounding Factors: Fractures, recent surgery, autoimmune diseases, and malignancies can all cause significant ESR elevations.
* False Negatives: The ESR is unreliable in neonates, patients with sickle cell disease, patients on systemic corticosteroids, and in hyper-acute presentations (symptoms present for less than 48 hours).
* Kinetics: Peak elevation of the ESR occurs 3 to 5 days following the onset of infection. With adequate surgical and medical treatment, it slowly returns to baseline over approximately 3 weeks.
C-Reactive Protein (CRP)
Synthesized by the liver in direct response to inflammatory cytokines (specifically IL-6), CRP is a far superior biomarker for monitoring the therapeutic response of an infection.
* Kinetics: CRP levels increase rapidly within 6 hours of the infectious onset, reach peak elevation at 48 hours, and, crucially, return to normal within 1 week after adequate surgical debridement and appropriate antibiotic therapy have been initiated.
Surgical Warning: A secondary spike in CRP following an initial postoperative decline is highly indicative of a recurrent infection, inadequate surgical debridement, or a secondary hematoma/seroma formation.
Synovial Fluid Analysis
In cases of suspected septic arthritis, joint aspiration (arthrocentesis) is the definitive diagnostic step. Material obtained must be sent immediately for cell count, differential, Gram stain, and aerobic/anaerobic cultures.
Table 15-2: Synovial Fluid Analysis Differentiation
| Disease State | Leukocytes (WBC/mm³) | Neutrophils (%) |
|---|---|---|
| Normal | < 200 | < 25% |
| Traumatic | < 5,000 | < 25% |
| Toxic Synovitis | 5,000 - 15,000 | < 25% |
| Acute Rheumatic Fever | 10,000 - 15,000 | ~ 50% |
| Juvenile Rheumatoid Arthritis | 15,000 - 80,000 | ~ 75% |
| Septic Arthritis | > 80,000 | > 75% |
Note: In septic arthritis, the cell count is typically greater than 80,000 (often >100,000), with a marked neutrophilic predominance (>75%).
Gram stains successfully identify the organism (gram-positive or gram-negative) in only about 33% of bone and joint aspirates, meaning a negative Gram stain never rules out a septic joint.
Intraoperative Frozen Section
When the diagnosis remains equivocal during surgical exploration, intraoperative frozen sections of periprosthetic or periarticular tissue are invaluable. A finding of greater than 10 WBCs per high-power field (HPF) is highly indicative of acute infection, whereas a count of less than 5 WBCs/HPF effectively excludes acute bacterial infection.
ADVANCED IMAGING STUDIES
Radiographic and advanced imaging modalities are critical not only for diagnosis but for preoperative anatomical mapping and surgical planning.
Plain Radiography
Plain radiographs are the mandatory first step in imaging but are notoriously insensitive in the acute phase. Early findings may only show soft-tissue swelling or obliteration of normal fat planes.
* Bone Destruction: Bony changes (periosteal reaction, osteopenia, or focal lysis) are not apparent on plain films until the infection has been present for 10 to 21 days.
* Matrix Loss: Furthermore, 30% to 50% of the regional bone matrix must be destroyed before a lytic lesion becomes radiographically visible.

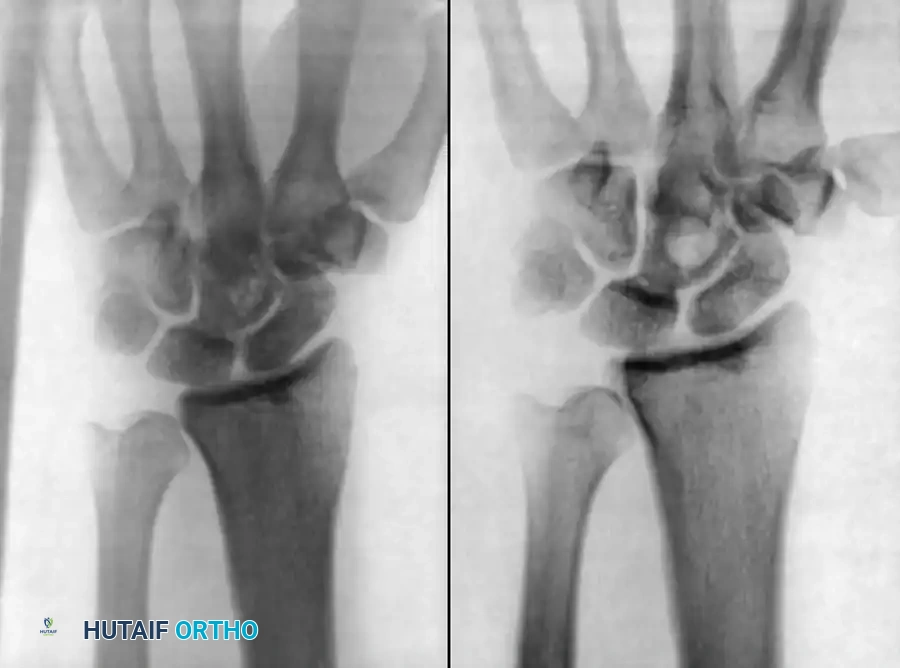
Fig. 15-1 Anteroposterior (A) and lateral (B) radiographs demonstrating advanced bone destruction and periosteal reaction typical of subacute to chronic osteomyelitis.
While fewer than 5% of plain radiographs are abnormal at initial presentation, and fewer than 30% at 1 week, up to 90% will demonstrate abnormalities by 3 to 4 weeks. Arthrography can be utilized to document proper intra-articular needle placement during aspiration; however, contrast dye should only be injected after synovial fluid is obtained, as the bactericidal properties of iodinated contrast can yield false-negative cultures.
Computed Tomography (CT)
CT scanning is highly effective for determining the extent of cortical destruction and medullary involvement. Pus within the medullary cavity replaces normal marrow fat, resulting in increased radiodensity (higher Hounsfield units) on the CT scan.
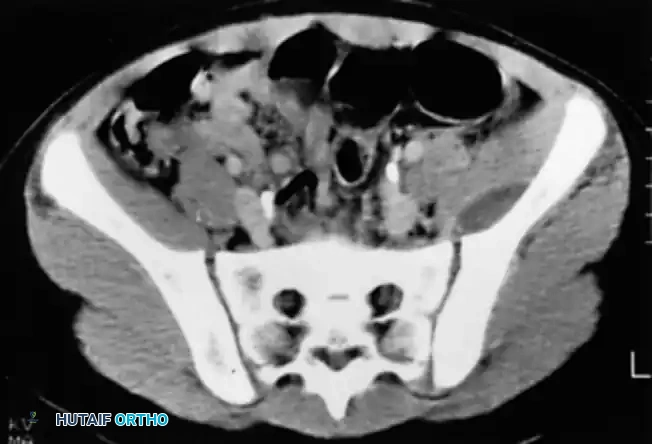
Fig. 15-2 Pelvic abscess in a child as seen on CT scan, demonstrating a large, loculated fluid collection adjacent to the ilium.
CT is the gold standard for identifying a sequestrum (a segment of necrotic bone separated from living bone by granulation tissue) and an involucrum (a layer of new bone growth outside existing bone) in chronic osteomyelitis. It is also superior for evaluating anatomically complex areas such as the sternoclavicular joint, sacroiliac joint, and spine.
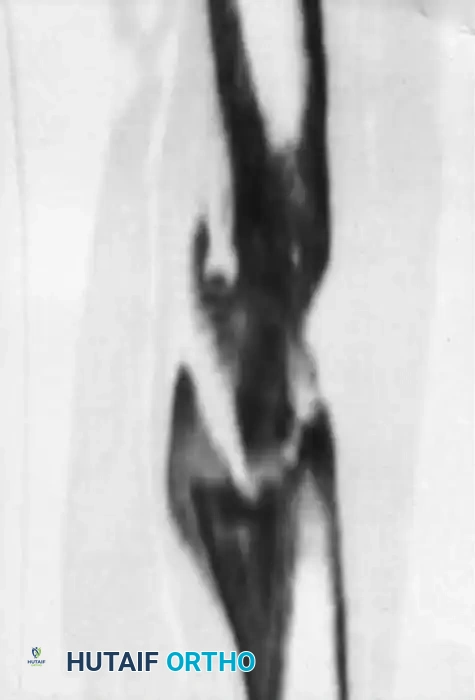
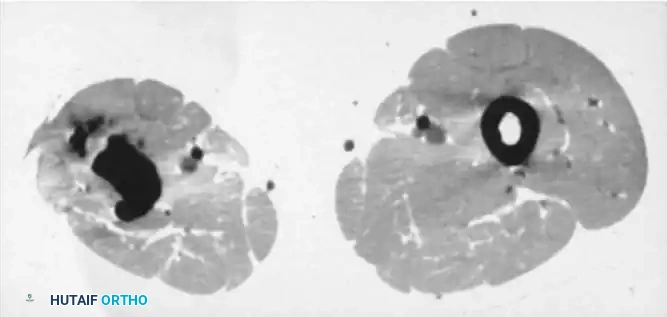
Fig. 15-3 A and B, CT scans showing dense, isolated sequestra within the medullary canal, surrounded by a sclerotic involucrum.
Radionuclide Scintigraphy
While radiographs and CT provide structural data, radionuclide scanning provides a physiological map of osteoblastic activity and regional blood flow. It is particularly useful in patients with metallic implants where MRI is contraindicated or obscured by artifact.
Technetium-99m Phosphate:
The most commonly used isotope, capable of detecting osteomyelitis within 48 hours of clinical onset. The standard technique is the Three-Phase Bone Scan:
1. Flow Phase (Radionuclide Angiogram): Demonstrates regional perfusion.
2. Equilibrium/Blood Pool Phase: Shows relative vascular flow and extracellular distribution (tissue hyperemia).
3. Delayed Phase (2-4 hours post-injection): Reflects osteoblastic activity.
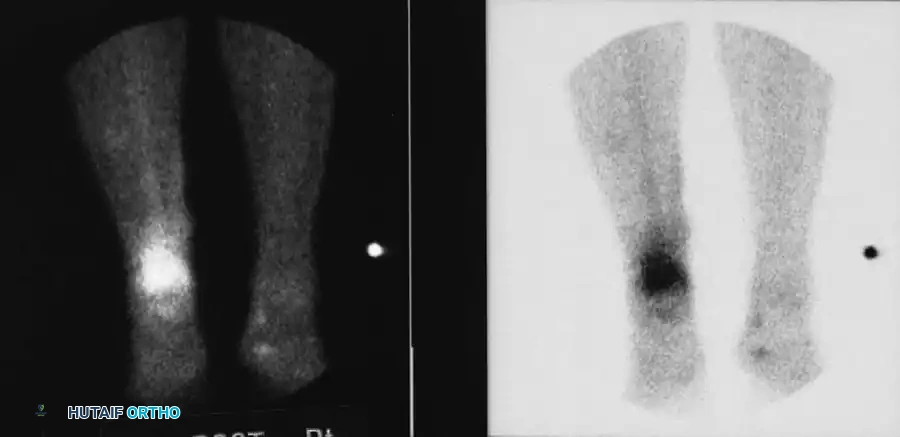
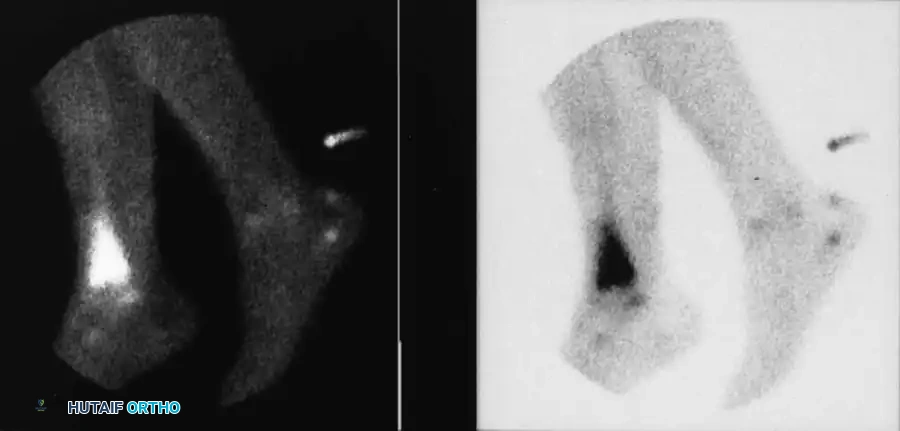
Fig. 15-4 Three-phase bone scan showing intense, focal uptake characteristic of osteomyelitis.
Table 15-3: Three-Phase Bone Scan Uptake Patterns
| Pathology | Flow Phase | Equilibrium Phase | Delayed Phase |
|---|---|---|---|
| Osteomyelitis | Increased (↑) | Increased (↑) | Increased (↑) |
| Cellulitis | Increased (↑) | Increased (↑) | Normal/Decreased (↔/↓) |
| Osteoarthritis | Normal (↔) | Normal (↔) | Increased (↑) |
Pitfall: A "cold" scan can occur in acute osteomyelitis if regional blood flow is completely occluded by subperiosteal pus, severe vasospasm, or massive soft-tissue swelling. Furthermore, Tc-99m scans are unreliable in neonates (< 6 weeks old), yielding false negatives in up to 60% of cases.
Gallium-67 Citrate & Indium-111 Leukocytes:
Gallium localizes to inflammatory lesions via direct leukocyte and bacterial uptake. When combined with Tc-99m, it helps differentiate reactive bone formation (where Tc-99m uptake > Gallium) from active infection (where Gallium uptake > Tc-99m).
Indium-111-labeled leukocytes involve in vitro labeling of the patient's autologous white blood cells. It is highly sensitive for acute osteomyelitis but less reliable for chronic osteomyelitis (which is lymphocyte-predominant) or for differentiating aseptic from septic loosening in arthroplasty.
Magnetic Resonance Imaging (MRI)
MRI is the most sensitive and specific imaging modality for evaluating bone and joint infections. It detects the increase in water content resulting from marrow edema long before cortical destruction occurs.
* T1-Weighted Images: Normal marrow fat appears bright. In osteomyelitis, this bright signal is replaced by a dark, low-intensity signal due to edema and cellular infiltration.
* T2-Weighted Images: Fluid and edema appear bright.
* STIR (Short Tau Inversion Recovery): Suppresses the fat signal, making the high-intensity fluid/edema signal of osteomyelitis highly conspicuous. STIR sequences boast a negative predictive value approaching 100% for osteomyelitis.
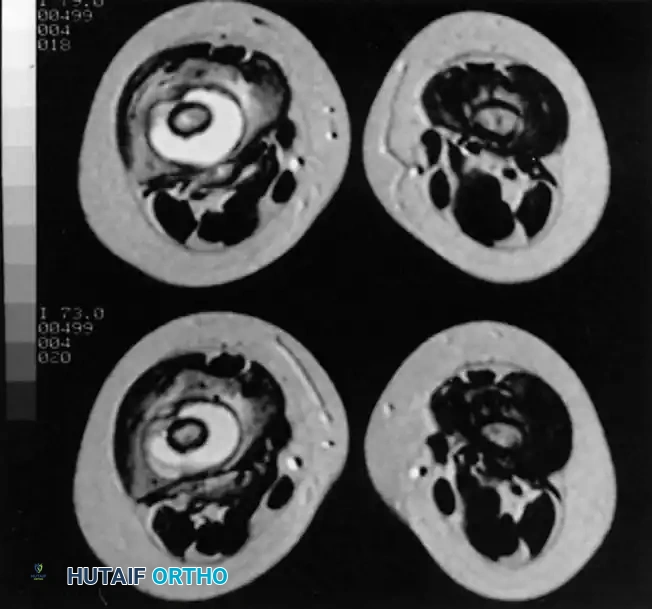
Axial MRI slices demonstrating extensive marrow edema, cortical disruption, and surrounding soft-tissue abscess formation typical of advanced musculoskeletal infection.
SURGICAL DECISION-MAKING AND OPERATIVE PRINCIPLES
The transition from diagnosis to surgical intervention must be swift. The primary goals of surgery are to obtain definitive microbiological tissue cultures, debride all necrotic and infected tissue, decompress the bone or joint, and manage the resulting dead space.
Indications for Surgery
- Aspiration-proven septic arthritis.
- Presence of a subperiosteal or intraosseous abscess on imaging.
- Failure of clinical improvement after 48-72 hours of targeted intravenous antibiotic therapy.
- Chronic osteomyelitis with identifiable sequestra or draining sinus tracts.
Patient Positioning and Preparation
- Positioning: The patient should be positioned on a radiolucent table to allow for unhindered intraoperative fluoroscopy. For lower extremity infections, the supine position with a bump under the ipsilateral hip (to internally rotate the leg) is standard.
- Tourniquet Use: A pneumatic tourniquet is highly recommended to maintain a bloodless field during the delicate dissection and identification of neurovascular structures. However, the tourniquet must be deflated prior to final debridement to accurately assess tissue viability (the "Paprika sign" – punctate bleeding from viable bone).
- Antibiotics: Crucially, prophylactic intravenous antibiotics should be withheld until deep tissue cultures are obtained, unless the patient is in florid septic shock.
Step-by-Step Surgical Approach (Example: Tibial Diaphyseal Osteomyelitis)
- Incision and Approach: Utilize an extensile longitudinal incision. Excise any previous surgical scars or sinus tracts in an elliptical fashion. Carry the dissection down to the fascia, avoiding the creation of large subcutaneous flaps which can compromise skin vascularity.
- Tissue Sampling: Before administering antibiotics, obtain a minimum of 3 to 5 distinct deep tissue samples using separate, sterile instruments to avoid cross-contamination. Send these for aerobic, anaerobic, fungal, and acid-fast bacilli (AFB) cultures, as well as histopathology.
- Cortical Windowing: For intramedullary infections, use a high-speed burr or osteotome to create a cortical window. The window should be oval to minimize stress risers and subsequent fracture risk.
- Radical Debridement: Evacuate all gross purulence. Use curettes to aggressively debride the medullary canal until healthy, bleeding cortical and cancellous bone is encountered. Remove all sequestra.
- Irrigation: Perform copious pulsatile lavage with 6 to 9 liters of normal saline. The mechanical washout of planktonic bacteria and debris is more critical than the type of fluid used.
- Dead Space Management: The resulting bony void must be managed to prevent hematoma formation and recurrent infection. This is typically achieved by packing the defect with antibiotic-impregnated polymethylmethacrylate (PMMA) beads (e.g., Tobramycin or Vancomycin). This provides massive local antibiotic concentrations without systemic toxicity.
- Closure: Achieve tension-free soft tissue closure. If the wound cannot be closed primarily without tension, leave it open and apply a negative pressure wound therapy (NPWT) device, planning for a delayed primary closure or a rotational/free muscle flap in conjunction with plastic surgery.
Postoperative Protocols
Postoperatively, the limb is typically immobilized in a well-padded splint to protect the bone, especially if a large cortical window was created. The patient is started on broad-spectrum intravenous antibiotics immediately following intraoperative culture acquisition. Once final culture sensitivities are finalized (typically 3-5 days post-op), the antibiotic regimen is narrowed and optimized in consultation with an Infectious Disease specialist. Intravenous therapy is generally continued for 4 to 6 weeks, monitored closely by serial CRP and ESR measurements.
You Might Also Like