Acute Hematogenous Osteomyelitis: Comprehensive Diagnosis and Surgical Management
Key Takeaway
Acute hematogenous osteomyelitis is a severe bone infection primarily affecting the rapidly growing metaphyses of long bones in children. Prompt diagnosis via clinical evaluation, inflammatory markers, and advanced imaging is critical. While early stages may respond to targeted intravenous antibiotics, the presence of a subperiosteal or intramedullary abscess necessitates emergent surgical decompression and debridement to prevent irreversible ischemic necrosis, extensive sequestrum formation, and chronic osteomyelitis.
Introduction and Epidemiology
Acute hematogenous osteomyelitis represents the most common manifestation of bone infection, predominantly afflicting the pediatric population. Over the past several decades, the incidence of acute hematogenous osteomyelitis has decreased dramatically in developed nations. Historical data, such as the seminal study by Blyth et al., reported a 70% decrease in the incidence of acute hematogenous osteomyelitis within the Greater Glasgow Health Board from 1970 to 1997. This epidemiological shift is largely attributed to elevated standards of living, improved systemic hygiene, and the widespread implementation of routine childhood immunizations.
Despite this decline, the condition remains a critical orthopaedic emergency. It exhibits a distinct male predominance across all affected age groups. The primary etiology is transient bacteremia—a frequent occurrence in childhood secondary to minor trauma, respiratory infections, or dental procedures. While bacteremia is the inciting event, the bacteriological seeding of bone is typically multifactorial, often associated with localized blunt trauma, chronic illness, malnutrition, or an immunocompromised state. In a significant proportion of cases, the exact precipitating event remains idiopathic.
The age distribution of acute hematogenous osteomyelitis in children is classically bimodal, with peak incidences occurring in infants younger than 2 years and in pre-adolescent children aged 8 to 12 years. Understanding this demographic distribution is paramount, as the anatomical and vascular variations across these age groups dictate the pathophysiology, clinical presentation, and ultimate surgical management of the disease.
Pathophysiology and Vascular Biomechanics
The pathogenesis of acute hematogenous osteomyelitis is intricately linked to the unique vascular anatomy of the developing skeleton. In children, the infection almost exclusively targets the metaphyses of rapidly growing long bones (e.g., distal femur, proximal tibia, proximal humerus).
The terminal branches of the nutrient artery form acute hairpin loops adjacent to the physis before emptying into a network of large venous sinusoids. This architectural arrangement results in sluggish, turbulent blood flow. Furthermore, the metaphyseal region possesses a relative paucity of active phagocytic cells compared to the diaphysis. This combination of vascular stasis and localized relative immunodeficiency creates an optimal microenvironment for bacterial inoculation and proliferation.
The Ischemic Cascade and Abscess Formation
Following bacterial seeding, a robust inflammatory cascade is initiated. The localized host response generates an inflammatory exudate within the rigid, unyielding confines of the medullary cavity.
⚠️ Surgical Warning: The Compartment Syndrome of Bone
The medullary cavity acts as a closed compartment. As the inflammatory exudate accumulates, intramedullary pressure rises exponentially. This pressure rapidly exceeds capillary perfusion pressure, leading to localized ischemic necrosis of the trabecular bone and subsequent abscess formation.
As the intramedullary abscess enlarges, the purulent material seeks the path of least resistance. It tracks through the haversian and Volkmann canals, escaping through the relatively porous metaphyseal cortex into the subperiosteal space, thereby elevating the periosteum and forming a subperiosteal abscess.
If left untreated, the periosteal stripping disrupts the extrinsic blood supply to the cortical bone. The combination of endosteal ischemia (from intramedullary pressure) and periosteal ischemia (from periosteal stripping) results in massive cortical necrosis. This necrotic bone segment, isolated from its blood supply, is termed a sequestrum. Concurrently, the elevated periosteum retains its osteogenic potential and begins to lay down new reactive woven bone around the sequestrum, forming an involucrum. This marks the irreversible transition from acute to chronic osteomyelitis.

Fig. 16-2 Pathological fracture after destruction of cortical bone by infection. Extensive cortical necrosis and structural compromise can lead to catastrophic mechanical failure if surgical decompression is delayed.
Age-Specific Anatomical Variations
The clinical trajectory of osteomyelitis is heavily dictated by the patient's age and the corresponding vascular anatomy of the physis:
- Infants (Younger than 2 Years): In neonates and infants, fetal vascular channels persist, allowing blood vessels to cross the cartilaginous physis. This transphyseal vascularity permits a metaphyseal abscess to spread directly into the epiphysis. Consequently, infants are at a profoundly high risk for concomitant septic arthritis, irreversible physeal damage, limb shortening, and severe angular deformities. The hip joint is the most commonly affected articulation in this cohort.
- Children (2 to 12 Years): As the child matures, the transphyseal vessels involute. The physis effectively acts as an impenetrable mechanical and vascular barrier, preventing the direct spread of a metaphyseal abscess into the epiphysis. However, because the metaphyseal cortex in older children is thicker and more robust, the purulent exudate is more likely to spread longitudinally down the medullary canal into the diaphysis. This jeopardizes the endosteal blood supply of the entire shaft, increasing the risk of extensive diaphyseal sequestration.
- Adults (Closed Physes): Once the physes close, the metaphyseal-epiphyseal vascular anastomosis is re-established. Acute hematogenous osteomyelitis is exceedingly rare in healthy adults and is typically confined to compromised hosts (e.g., diabetes, immunosuppression). When it does occur, it frequently targets the highly vascularized vertebral bodies rather than long bones. Abscesses spread indolently, and large sequestra rarely form.
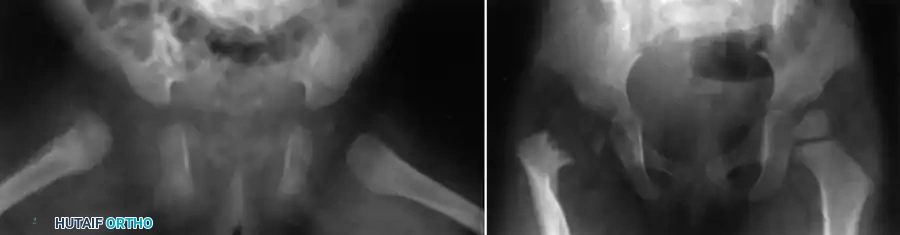
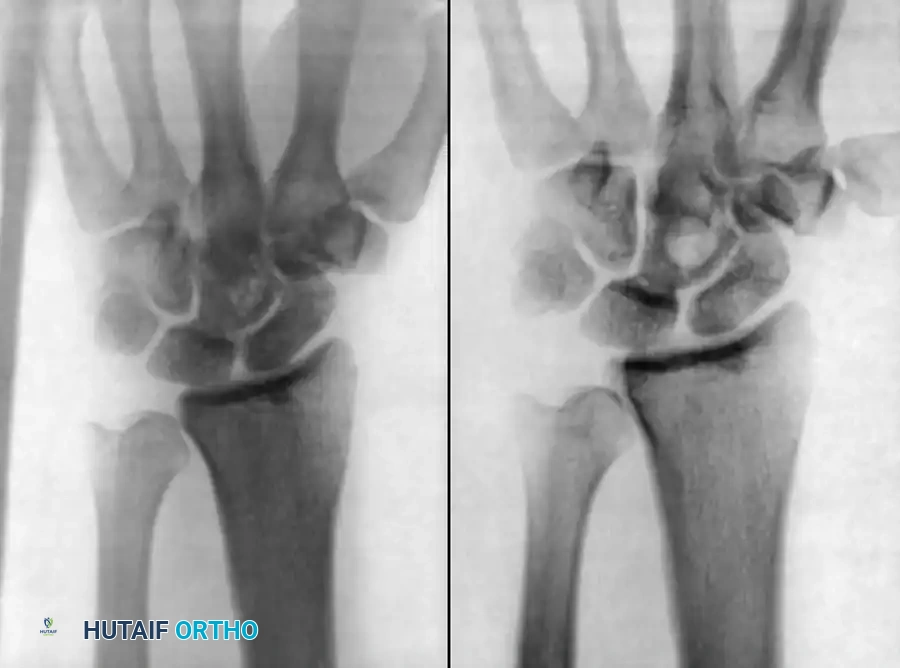
Fig. 16-3 Epiphyseal separation caused by infection in a young child. Note the catastrophic joint involvement secondary to transphyseal spread.
Microbiology and Pathogen Profiling
The bacteriological profile of acute hematogenous osteomyelitis varies significantly based on patient age, vaccination status, and underlying systemic comorbidities.
- Staphylococcus aureus: Remains the most common infecting organism across all age groups, responsible for 70-90% of cases in older children and adults. The rising prevalence of Methicillin-Resistant S. aureus (MRSA) necessitates aggressive empirical coverage in endemic regions.
- Group B Streptococcus & Gram-Negative Coliforms: Frequently isolated in neonates and premature infants, particularly those managed in neonatal intensive care units. Multifocal involvement occurs in over 40% of these cases.
- Haemophilus influenzae type b (Hib): Historically a primary pathogen in children aged 6 months to 4 years. Its incidence has been virtually eradicated in developed nations due to routine Hib immunization.
- Salmonella species: Pathognomonic association with sickle cell disease (SS or SC hemoglobinopathies). In these patients, functional asplenia and microvascular thrombosis in the bowel wall allow Salmonella translocation. The infection classically presents as diaphyseal rather than metaphyseal osteomyelitis.
- Pseudomonas aeruginosa: The most common organism isolated in intravenous drug abusers, frequently localizing to the sternoclavicular joint, symphysis pubis, or vertebral column.
- Fungal Pathogens: Increasingly identified in chronically ill, neutropenic, or prolonged total parenteral nutrition (TPN) patients.
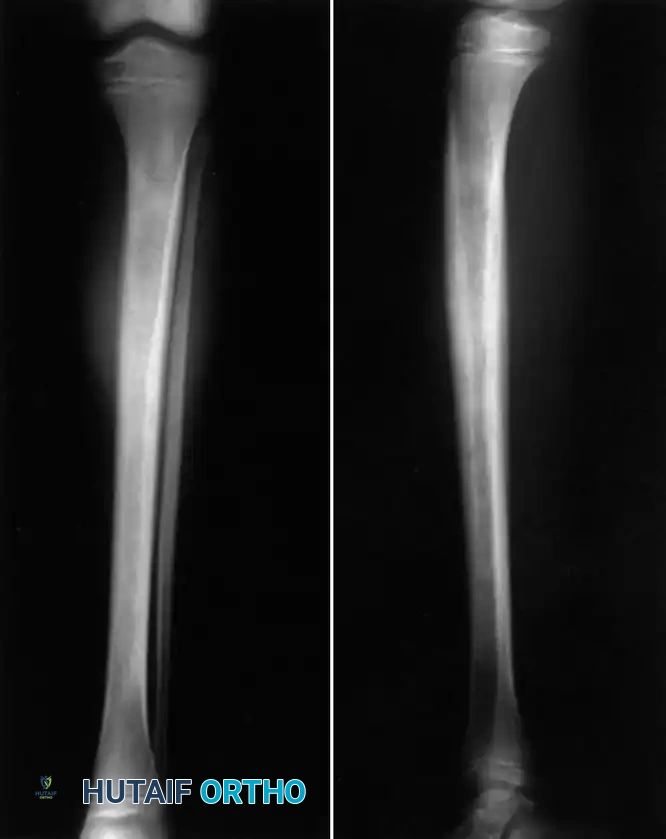
Fig. 16-4 Osteomyelitis of the tibia in a sickle cell patient. Note the extensive diaphyseal involvement characteristic of Salmonella osteomyelitis.
Clinical Evaluation and Diagnosis
The diagnosis of acute hematogenous osteomyelitis requires a high index of suspicion, as early clinical findings can be subtle, particularly in non-verbal infants or immunocompromised adults.
History and Physical Examination
Patients typically present with a history of acute onset localized pain, refusal to bear weight (pseudoparalysis in infants), and systemic symptoms such as fever, chills, and malaise. However, in neonates, the only signs may be irritability, poor feeding, and a lack of spontaneous limb movement.
Physical examination reveals exquisite point tenderness over the affected metaphysis. Erythema, calor, and induration are late signs, indicating that the infection has already breached the cortex and formed a subperiosteal abscess. In severe cases, the massive inflammatory swelling can precipitate a secondary acute compartment syndrome.
Laboratory Investigations
- White Blood Cell (WBC) Count: Often normal or only mildly elevated; a left shift may be present.
- Erythrocyte Sedimentation Rate (ESR): Typically elevated but peaks late (3-5 days) and normalizes slowly (3-4 weeks).
- C-Reactive Protein (CRP): The most sensitive and specific acute-phase reactant for osteomyelitis. It rises rapidly within 6-8 hours, peaks at 48 hours, and normalizes quickly with appropriate treatment. Serial CRP measurements are the gold standard for monitoring therapeutic response.
- Blood Cultures: Positive in approximately 50% of cases. Blood cultures must be drawn prior to the administration of empirical antibiotics.
Diagnostic Imaging
- Plain Radiographs: Standard anteroposterior and lateral radiographs are mandatory to rule out fractures or tumors. However, they are notoriously insensitive for early osteomyelitis. Deep soft-tissue swelling and obliteration of fascial planes may be seen early, but osseous changes (periosteal reaction, osteopenia, cortical destruction) require 30-50% bone mineral loss and typically do not appear until 10 to 14 days after the onset of infection.
- Technetium-99m Bone Scans: Highly sensitive (90-95%) and can confirm the diagnosis within 24 to 48 hours of symptom onset. It demonstrates focally increased radiotracer uptake in all three phases (blood flow, blood pool, and delayed osseous phase).
- Magnetic Resonance Imaging (MRI): The imaging modality of choice. MRI provides unparalleled anatomical detail, differentiating between isolated bone marrow edema, subperiosteal abscess, and concurrent soft-tissue extension. It is highly sensitive for detecting early inflammatory changes within 24 hours of onset.
- Ultrasound: Highly effective in infants for detecting subperiosteal fluid collections and guiding diagnostic aspiration.
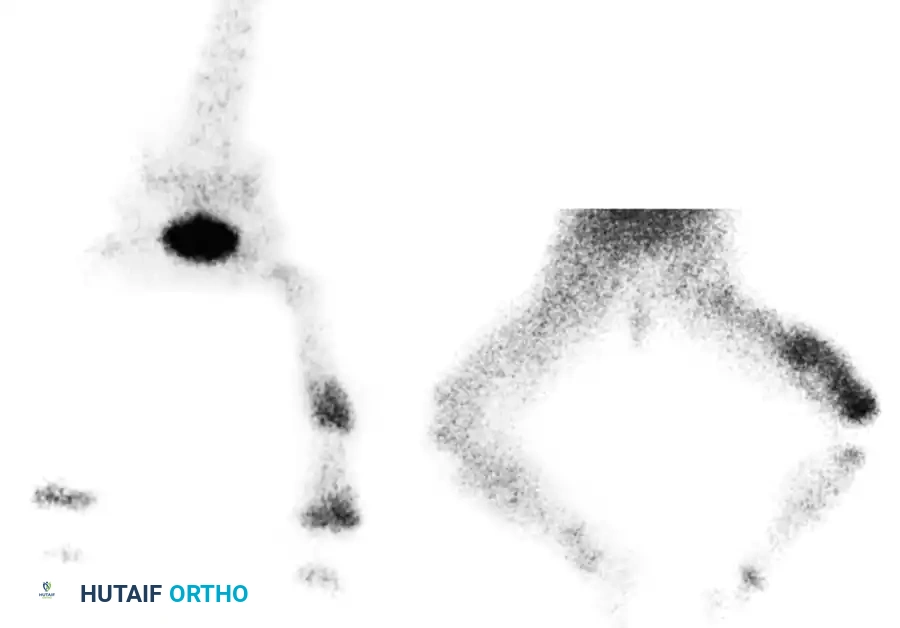
Fig. 16-6 Technetium-99m Bone scan showing intensely increased focal uptake in the metaphyseal area, diagnostic of acute osteomyelitis.
Diagnostic Aspiration
Aspiration is the definitive step for bacteriological diagnosis and should be performed under strict aseptic conditions, often utilizing fluoroscopic or ultrasound guidance.
1. A 16-gauge or 18-gauge spinal needle is advanced into the area of maximal swelling.
2. The subperiosteal space is aspirated first.
3. If no purulence is encountered, the needle is advanced through the cortex (often softened by hyperemia) into the medullary canal to obtain a marrow aspirate.
4. Samples are immediately sent for Gram stain, aerobic/anaerobic cultures, and antibiotic sensitivities.
Principles of Management
The management of acute hematogenous osteomyelitis is a delicate interplay between aggressive antimicrobial therapy and timely surgical intervention. In 1983, Nade proposed five foundational principles that remain the cornerstone of modern treatment:
- An appropriate antibiotic is effective before pus formation.
- Antibiotics cannot sterilize avascular tissues or established abscesses; such areas mandate surgical extirpation.
- If surgical removal of necrotic tissue is effective, antibiotics should prevent reformation, allowing for safe primary wound closure.
- Surgical intervention must be meticulous to avoid further damage to already ischemic bone and soft tissue.
- Antibiotics must be continued postoperatively to eradicate residual microscopic disease.
Antibiotic Therapy
Upon securing appropriate cultures (blood and aspirate), empirical intravenous antibiotics are initiated immediately. The regimen must cover S. aureus (including MRSA, depending on local antibiograms) and other age-appropriate pathogens.
If the patient demonstrates a rapid clinical response (defervescence, decreasing CRP, improved pain) within 24 to 48 hours, non-operative management with a prolonged course of antibiotics (intravenous transitioning to oral) may be sufficient.
Surgical Management: Masterclass Technique
Surgical intervention is not a failure of medical management; it is a necessary mechanical solution to a mechanical problem (abscess and ischemia).
Indications for Surgery
- The presence of a documented subperiosteal or intramedullary abscess (via MRI, ultrasound, or aspiration).
- Failure of the patient to improve clinically or biochemically (stagnant or rising CRP) after 36 to 48 hours of appropriate intravenous antibiotic therapy.
- Concomitant septic arthritis (especially in the hip or shoulder).
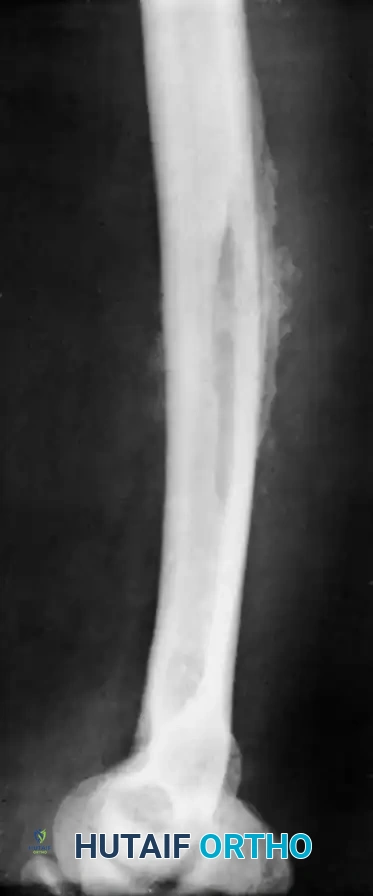
Preoperative planning must account for the exact anatomical location of the abscess to minimize iatrogenic trauma during surgical exposure.
Preoperative Planning and Positioning
- Anesthesia: General anesthesia with profound muscle relaxation.
- Positioning: The patient is positioned supine on a radiolucent table to allow for intraoperative fluoroscopy. The affected limb is elevated and prepped circumferentially.
- Tourniquet: A pneumatic tourniquet is highly recommended to provide a bloodless surgical field, which is critical for identifying necrotic versus viable bone.
🛑 Surgical Pitfall: Exsanguination in Infection
NEVER exsanguinate an infected limb using an Esmarch or elastic bandage. Forcing blood out of the limb can systematically embolize purulent material and bacteria, precipitating severe sepsis or septic shock. Instead, elevate the extremity for 3 to 5 minutes to allow gravity drainage before inflating the tourniquet.
Surgical Technique 16-1: Drainage of Acute Tibial Osteomyelitis
The following technique describes the decompression of the proximal tibia, but the biomechanical principles apply universally to all long bones.
Step 1: Surgical Approach
* Make a longitudinal anteromedial incision, 5 to 7.5 cm in length, centered over the area of maximal fluctuance or the radiographically identified metaphyseal lesion.
* Deepen the incision through the subcutaneous tissue, taking care to protect the long saphenous vein and saphenous nerve medially.
Step 2: Periosteal Elevation and Subperiosteal Decompression
* Incise the deep fascia and identify the periosteum. In the presence of a subperiosteal abscess, the periosteum will be thickened, edematous, and elevated off the cortex.
* Incise the periosteum longitudinally. Evacuate the subperiosteal purulence and send a swab for definitive culture.
* Gently elevate the periosteum using a Cobb elevator. Crucial: Limit periosteal stripping only to the area of infection to prevent further devascularization of the underlying cortex.
Step 3: Cortical Decompression (Drilling)
* Even if a subperiosteal abscess is found and evacuated, the medullary canal must be decompressed, as the primary pathology originates endosteally.
* Using a 3.2 mm or 4.5 mm drill bit, create several unicortical drill holes through the anteromedial metaphyseal cortex into the medullary canal.
* Observe the drill holes. The extrusion of frank pus or lipid-laden inflammatory fluid confirms intramedullary involvement.
Step 4: Cortical Windowing and Debridement
* If intramedullary pus is encountered, the drill holes must be connected to create a formal cortical window.
* Use a sharp osteotome or a high-speed burr to remove a small rectangular window of bone (approximately 1 cm x 2 cm).
* Biomechanical Pearl: Ensure the corners of the cortical window are rounded (ovoid shape) rather than sharp right angles. Sharp corners act as severe stress risers, significantly increasing the risk of postoperative pathological fractures.
* Introduce a small curette into the medullary canal and gently debride necrotic cancellous bone and inflammatory debris. Avoid aggressive curettage near the physis to prevent iatrogenic growth arrest.
Step 5: Irrigation and Closure
* Perform copious pulsatile lavage of the medullary canal and subperiosteal space using 3 to 6 liters of sterile normal saline.
* Place a medium-sized suction drain (e.g., Hemovac) deep into the wound, adjacent to the cortical window.
* The wound management depends on the severity of the infection. In acute cases with healthy surrounding tissue, the skin and subcutaneous tissues can be closed loosely over the drain. If there is massive contamination or questionable tissue viability, the wound should be packed open with sterile dressings for a delayed primary closure or secondary intention healing.
Postoperative Protocol and Rehabilitation
- Immobilization: The structural integrity of the bone is compromised by both the infection and the surgical cortical window. The limb must be strictly immobilized in a well-padded bivalved cast or rigid splint for 4 to 6 weeks to prevent pathological fracture.
- Weight-Bearing: Strict non-weight-bearing status is maintained until radiographic evidence of bone consolidation and cortical healing is observed.
- Antimicrobial Therapy: Intravenous antibiotics are continued postoperatively. The transition to highly bioavailable oral antibiotics is considered only when the patient is afebrile for 48 hours, local signs of inflammation have resolved, and the CRP has normalized or is rapidly trending toward baseline. The total duration of therapy (typically 4 to 6 weeks) should be managed collaboratively with an infectious disease specialist.
- Monitoring: Serial clinical examinations, weekly CRP levels, and follow-up radiographs are mandatory to ensure eradication of the infection and to monitor for late complications such as chronic osteomyelitis, physeal arrest, or angular deformity.
📚 Medical References
- Acute hematogenous osteomyelitis: a model with trauma as an etiology, J Pediatr Orthop 9:447, 1989.
- Namias N, Harvill S, Ball S, et al: Cost and morbidity associated with antibiotic prophylaxis in the ICU, J Am Coll Surg 188:225, 1999.
- Nelson CL: Prevention of infection. In Evarts CM, ed: Surgery of the musculoskeletal system, 2nd ed, New York, 1990, Churchill Livingstone. Norden CW: Antibiotic prophylaxis in orthopaedic surgery, Rev Infect Dis 13:13, 1991.
- Nossal GJV: The basic components of the immune system, N Engl J Med 316:1320, 1987.
- Oishi CS, Carrion WV, Hoaglund FT: Use of parenteral prophylactic antibiotics in clean orthopaedic surgery, Clin Orthop Relat Res 296:249, 1993.
- O’Shaughnessy M, O’Malley VP, Corbett G, et al: Optimum duration of surgical scrub-time. Br J Surg 78:685, 1991.
- Parienti JJ, Thibon P, Heller R, et al: Hand-rubbing with an aqueous alcoholic vs. traditional surgical hand-scrubbing and 30-day surgical site infection rates: a randomized equivalence study, JAMA 288:722, 2002.
- Peterson PK: Host defense abnormalities predisposing the patient to infection, Am J Med 76:2, 1984.
- Puskarich CL, Nelson CL, Nusbickel FR, et al: The use of two nutritional indicators in identifying long bone fracture patients who do and do not develop infections, J Orthop Res 8:799, 1990.
- Rainey-McDonald CG, Holliday RL, Wells GA, et al: Validity of a two-variable nutritional
You Might Also Like