PATHOPHYSIOLOGY AND NATURAL HISTORY OF VERTEBRAL INFECTIONS
The natural history of pyogenic vertebral infections (vertebral osteomyelitis and discitis) typically involves an initial infecting source or transient bacteremic incident, followed by a progressive period of localized, intractable spinal pain. This may occur with or without significant generalized sepsis. When generalized sepsis is present, it almost universally indicates a primary source of infection distant from the spine (e.g., endocarditis, genitourinary infection, or indwelling catheters).
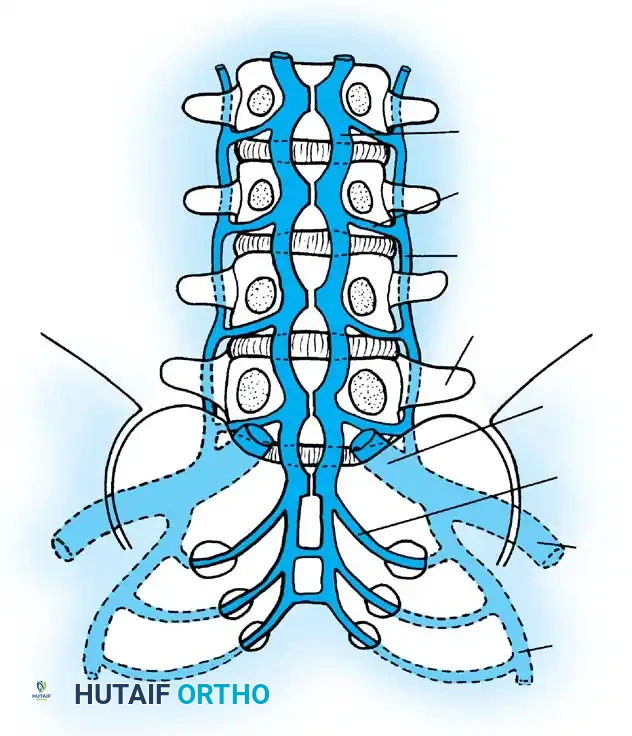
Vascular Anatomy and Hematogenous Seeding
Blood-borne spinal infections predominantly begin in the capillary loops or postcapillary venous channels located within the vertebral endplate. The unique vascular anatomy of the subchondral region in adults creates an environment conducive to bacterial stasis. Sludging of blood in these tortuous venous channels results in localized suppurative inflammation, microthrombosis, and subsequent tissue necrosis.

As the trabecular bone undergoes necrosis, bony collapse ensues, facilitating the direct spread of the infection into the adjacent, avascular intervertebral disc space. This pathophysiological cascade eventually results in endplate erosions, which represent the earliest radiographic findings visible on plain radiographs or computed tomography (CT).
Clinical Pearl: Bacterial infections rapidly attack and destroy the intervertebral disc due to the production of proteolytic enzymes. In stark contrast, tuberculous (TB) and fungal infections typically preserve the intervertebral disc until very late in the disease process. Recognizing this pathological characteristic on preoperative MRI or during surgical exploration is critical for identifying the likely infecting organism.
Patterns of Extension and Abscess Formation
Once established in the disc space and adjacent endplates, the infection can extend along multiple anatomical pathways:
* Anterior Extension: The infection breaches the anterior longitudinal ligament (ALL) to create a paravertebral abscess. In the lumbar spine, large paravertebral abscesses frequently track inferiorly along the psoas muscle fascia, potentially presenting as a groin mass or causing a psoas sign (pain with hip extension).
* Posterior Extension: The infection breaches the posterior longitudinal ligament (PLL) to cause an epidural abscess. Epidural abscesses may cross the spinal epidural space, causing direct mechanical compression of the neural elements. In severe cases, the infection may penetrate the dura to enter the meningeal space and the spinal cord itself (meningitis or myelitis).
According to Feldenzer et al., while epidural extension is a known complication, it occurs relatively late in the course of pyogenic infections and rarely occludes the spinal arteries to cause ischemic infarction of the cord.
Host Factors and Immune Response
The clinical course of a spinal infection varies drastically depending on the virulence of the infecting organism and the patient’s intrinsic immune status. Nichols postulated that the chronic infection itself may create a hypermetabolic, malnourished condition that further compromises the host's immune system, creating a vicious cycle of decline.
Hopkinson, Stevenson, and Benjamin noted that septic discitis is strongly associated with invasive procedures, underlying malignancies, and diabetes mellitus. In vulnerable populations, the mortality rate remains significant; Waldvogel and Vasey reported that death occurs in up to 10% of patients presenting with overwhelming, systemic spinal infections.
Conversely, individuals with a robust immune response may spontaneously overcome the infection without targeted antimicrobial treatment. Fraser, Osti, and Vernon-Roberts demonstrated this experimentally by injecting Staphylococcus epidermidis into the intervertebral discs of sheep. They noted that six weeks post-inoculation, only the discs injected with bacteria showed histological evidence of discitis; however, viable bacteria could no longer be cultured from those specimens. This pivotal finding indicates that there is a narrow, optimal temporal window for the bacteriological identification of disc space infections in otherwise healthy individuals, explaining the widely varying clinical presentations and the high rate of culture-negative biopsies.
NEUROLOGICAL COMPROMISE AND PARALYSIS
As the infected vertebral body softens, it loses its structural integrity and may collapse under physiological body weight and axial stress. Neurological deficits in the setting of vertebral osteomyelitis are primarily caused by two distinct mechanisms:
1. Direct Extension: Mechanical compression from an epidural abscess, or direct bacterial communication with the spinal canal leading to localized inflammation of the neural elements.
2. Secondary Compression: Retropulsion of bone fragments and disc material into the spinal canal resulting from a pathological fracture and bone softening.
Paralysis resulting from spinal infection may present acutely or insidiously. Early onset of paralysis frequently suggests the rapid epidural extension of a purulent abscess. Late-onset paralysis is more commonly mechanical, caused by the progressive development of significant focal kyphosis, vertebral body collapse with retropulsion of debris, or late abscess formation in more indolent, low-virulence infections.
Eismont's Criteria for Paralysis Risk
Eismont et al. identified four critical risk factors that indicate a significantly increased predisposition to paralysis in patients with pyogenic and fungal vertebral osteomyelitis:
1. Advanced Age: Older patients possess less physiological reserve and often have pre-existing degenerative spinal stenosis, lowering the threshold for symptomatic compression.
2. Level of Infection: Infections in the cervical spine carry a much higher risk of neurological deficit due to the smaller canal-to-cord ratio compared to the lumbar spine.
3. Debilitating Disease: The presence of systemic comorbidities such as diabetes mellitus, rheumatoid arthritis, or chronic corticosteroid usage impairs tissue healing and immune response.
4. Infecting Organism: Staphylococcus aureus infections are highly virulent, causing rapid tissue destruction and abscess formation.
Surgical Warning: Paralysis resulting from spinal tuberculosis (Pott's disease) does not strictly correlate with Eismont's four factors. TB spine often presents with profound deformity and large, cold abscesses that can cause insidious neurological decline regardless of the patient's age or comorbidities.
SURGICAL INDICATIONS AND BIOMECHANICS
While many cases of pyogenic vertebral osteomyelitis can be managed successfully with prolonged intravenous antibiotic therapy and rigid orthosis, surgical intervention becomes mandatory under specific clinical scenarios.
Indications for Operative Management
- Neurological Deficit: Progressive motor weakness, myelopathy, or cauda equina syndrome secondary to epidural abscess or bony retropulsion.
- Spinal Instability: Extensive destruction of the anterior column leading to progressive kyphotic deformity.
- Intractable Pain: Severe, mechanical back pain that fails to improve despite appropriate bracing and medical therapy, indicating gross instability.
- Medical Failure: Persistent elevation of inflammatory markers (ESR, CRP), ongoing sepsis, or enlarging abscesses despite 3 to 4 weeks of culture-directed intravenous antibiotics.
- Diagnostic Uncertainty: Need for open biopsy when multiple percutaneous image-guided biopsies yield negative cultures, especially in the setting of suspected atypical infections or malignancy.
Biomechanical Considerations
The anterior column of the spine (vertebral bodies and intervertebral discs) supports approximately 80% of physiological axial loads. Pyogenic infections preferentially destroy this anterior load-bearing structure. When the anterior column collapses, the axis of rotation shifts anteriorly, subjecting the posterior tension band (ligaments and facet joints) to immense tensile forces.
Surgical reconstruction must therefore adhere to the tension-band principle: the anterior column must be structurally reconstituted (via strut grafting or titanium cages) to bear axial loads, while the posterior column must be stabilized (via pedicle screw instrumentation) to resist flexion and rotational forces.
PREOPERATIVE PLANNING AND POSITIONING
Patient Optimization
Patients with spinal infections are often systemically unwell, malnourished, and septic. Preoperative optimization requires a multidisciplinary approach involving infectious disease specialists, intensivists, and nutritionists.
* Hemodynamic Resuscitation: Ensure adequate perfusion and correct electrolyte imbalances.
* Nutritional Support: Assess albumin and prealbumin levels. Enteral or parenteral nutrition may be required to optimize postoperative wound healing.
* Imaging: A preoperative MRI with and without gadolinium contrast is the gold standard for defining the extent of the epidural abscess and neural compression. A fine-cut CT scan is essential for evaluating bone destruction and planning instrumentation trajectories.
Surgical Positioning
Positioning depends entirely on the chosen surgical approach:
* Anterior Approaches: For cervical pathology, the patient is positioned supine with the neck slightly extended. For thoracic or lumbar anterior approaches (thoracotomy or retroperitoneal), the patient is placed in the lateral decubitus position.
* Posterior Approaches: The patient is positioned prone on a radiolucent Jackson table. Care must be taken to pad all bony prominences and ensure the abdomen hangs free to decrease intra-abdominal pressure, thereby reducing epidural venous bleeding.
SURGICAL APPROACHES: STEP-BY-STEP GUIDE
The gold standard surgical treatment for complex pyogenic vertebral osteomyelitis with anterior destruction is Anterior Radical Debridement and Strut Grafting, followed by Posterior Instrumented Fusion.
Step 1: Anterior Exposure and Radical Debridement
The anterior approach allows direct access to the primary site of infection, enabling thorough evacuation of the paravertebral and epidural abscesses without manipulating the already compromised neural elements.
- Exposure: Depending on the level, utilize a standard Smith-Robinson approach (cervical), transthoracic approach (thoracic), or retroperitoneal approach (lumbar).
- Vascular Control: Identify and mobilize the great vessels. Note: Doita et al. reported cases of contiguous infection spread to the aorta, requiring prosthetic aortic replacement in addition to spinal debridement. Extreme caution is required when mobilizing inflamed, adherent vessels.
- Corpectomy and Discectomy: Incise the ALL and annulus fibrosus. Send multiple tissue and fluid samples for aerobic, anaerobic, fungal, and mycobacterial cultures, as well as histopathology.
- Debridement: Use rongeurs, curettes, and a high-speed burr to resect all necrotic, infected bone until healthy, bleeding cancellous bone is encountered (the "paprika sign").
- Neural Decompression: Carefully resect the PLL to expose the dura and evacuate any ventral epidural purulence.
Step 2: Anterior Column Reconstruction
Once the infection has been radically debrided, the structural void must be filled to restore sagittal alignment and bear axial loads.
1. Graft Selection: Autologous structural bone graft (e.g., tricortical iliac crest or fibula) is highly resistant to infection and remains the gold standard. Alternatively, titanium mesh cages filled with local autograft or allograft can be used. Titanium is preferred over PEEK in the setting of active infection due to its ability to integrate and its lower risk of biofilm formation.
2. Insertion: Gently distract the spine to restore height and correct kyphosis. Impact the strut graft or cage into the defect, ensuring it is countersunk beneath the anterior cortical margin to prevent vascular impingement.
Step 3: Posterior Instrumentation and Fusion
While anterior instrumentation can be used, posterior pedicle screw fixation provides superior biomechanical stability, especially when the anterior bone quality is compromised by infection.
1. Exposure: Perform a standard midline posterior approach, exposing the anatomical landmarks for pedicle screw insertion.
2. Instrumentation: Place pedicle screws at least one, preferably two, levels above and below the infected segment. In cases of severe osteoporosis or extensive destruction, cement-augmented screws may be considered.
3. Deformity Correction: Apply rods and utilize compression/distraction maneuvers to finalize the correction of the kyphotic deformity.
4. Posterior Fusion: Decorticate the lamina and transverse processes. Apply bone graft to facilitate a robust posterolateral fusion.
Pitfall: Avoid placing bone graft directly into an area of active posterior purulence. If a posterior epidural abscess was drained via laminectomy, thoroughly irrigate the site and rely on the anterior strut and posterior instrumentation for stability until the infection clears.
POSTOPERATIVE PROTOCOLS AND COMPLICATIONS
Antimicrobial Management
Postoperative antibiotic therapy must be strictly guided by intraoperative culture results and managed in conjunction with an infectious disease specialist.
* Duration: A minimum of 6 weeks of targeted intravenous antibiotics is standard, often followed by an extended course of oral suppressive therapy depending on the organism and host immune status.
* Monitoring: Serial monitoring of C-reactive protein (CRP) and Erythrocyte Sedimentation Rate (ESR) is crucial. CRP typically normalizes within 2 to 3 weeks of successful treatment, whereas ESR may remain elevated for months.
Mobilization and Rehabilitation
- Bracing: Depending on the rigidity of the internal fixation and the patient's bone quality, a rigid orthosis (TLSO or cervical collar) may be prescribed for 6 to 12 weeks postoperatively to restrict micromotion and promote arthrodesis.
- Physical Therapy: Early mobilization is encouraged to prevent complications of prolonged bed rest, such as deep vein thrombosis, pulmonary embolism, and deconditioning.
Severe Complications
Surgeons must remain vigilant for life-threatening complications unique to spinal infections. Odom et al. reported cases of toxic shock syndrome occurring immediately after the surgical drainage of a vertebral abscess, driven by the sudden systemic release of bacterial exotoxins. Patients exhibiting unexplained postoperative hypotension, rash, and multiorgan dysfunction must be aggressively resuscitated and evaluated for toxic shock. Furthermore, failure of the construct, pseudoarthrosis, and recurrent infection remain significant risks, particularly in malnourished or immunocompromised hosts, necessitating meticulous long-term clinical and radiographic follow-up.
📚 Medical References
- vertebral osteomyelitis, J Bone Joint Surg 41B:796, 1959.
- Wilkinson MC: Partial synovectomy and curettage in the treatment of tuberculosis in the hip, J Bone Joint Surg 39B:66, 1957.
- Wilkinson MC: Partial synovectomy in the treatment of tuberculosis of the knee, J Bone Joint Surg 44B:34, 1962.
- Yao DC, Sartoris DJ: Musculoskeletal tuberculosis, Radiol Clin North Am 33:679, 1995.
- Yip KM, Lin J, Leung PC: Cystic tuberculosis of the bone mimicking osteogenic sarcoma, Tuber Lung Dis 77:566, 1996.
- Other Unusual Infections Ajal M, Turner J, Fagan P, et al: Actinomycosis oto-mastoiditis, J Laryngol Otol 111:1069, 1997.
- Albert MC, Zachary SV, Alter S: Blastomycosis of the forearm synovium in a child, Clin Orthop Relat Res 317:223, 1995.
- Al-Shahed MS, Sharif HS, Haddad MC, et al: Imaging features of musculoskeletal brucellosis, Radiographics 14:333, 1994.
- Barrack RL, Edmunds JO Jr, Skinner HB: Blastomycosis osteomyelitis, Orthop Rev 12:59, 1983.
- Bayer AS, Guze LB: Fungal arthritis: II. coccidioidal synovitis, Semin Arthritis Rheum 8:200, 1979.
- Benjamin B, Khan MRH: Hip involvement in childhood brucellosis, J Bone Joint Surg 76B:544, 1994.
- Bosch X, Ramón R, Font J, et al: Bilateral cryptococcosis of the hip, J Bone Joint Surg 76A:1234, 1994.
- Brennan PJ, DeGirolamo MP: Musculoskeletal infections in immunocompromised hosts, Orthop Clin North Am 22:389, 1991.
- Bryan CS: Vertebral osteomyelitis due to Cryptococcus neoformans : case report, J Bone Joint Surg 59A:275, 1977.
- Care SB, Lacey SH: Recurrent histoplasmosis of the wrist: a case report, J Hand Surg 23:1112, 1998.
- Carr MM, Fielding JC, Sibbald G, et al: Sporotrichosis of the hand: an urban experience, J Hand Surg 20A:66, 1995.
- Chelboun J, Nade S: Skeletal cryptococcosis, J Bone Joint Surg 59A:509, 1977.
- Colmenero JD, Reguera JM, Martos F, et al: Complications associated with Brucella melitensis infection: a study of 530 cases, Medicine 75:195, 1996.
- Crout JE, Brewer NS, Tompkins RB: Sporotrichosis arthritis: clinical features in seven patients, Ann Intern Med 86:294, 1977.
- Dattwyler RJ, Halperin JJ, Pass H, et al: Ceftriaxone as effective therapy in refractory Lyme disease, J Infect Dis 155:1322, 1987.
- Deresinski SC: Coccidioidomycosis of bone and joint. In Stevens DA, ed: Coccidioidomycosis: a text, New York, 1980, Plenum Medical. Diamond RD: Cryptococcus neoformans . In Mandell GL, Douglas RG, Bennett JE, eds: Principles and practices of infectious diseases, 2nd ed, New York, 1985, John Wiley & Sons. Dismukes W: NIAID Collaborative Antifungal Study Group: Treatment of systemic mycosis with ketoconazole: emphasis on toxicity and clinical response in 52 patients, Ann Intern Med 98:13, 1983.
- Dismukes W: NIAID Mycoses Study Group: Treatment of blastomycosis and histoplasmosis with ketoconazole: results of a prospective randomized clinical trial, Ann Intern Med 103:861, 1985.
- Drutz DJ, Catanzaro A: Coccidioidomycosis, Am Rev Respir Dis 117:559, 1978.
- El-Desouki MI, Benjamin RS: Diagnostic value of quantitative sacroiliac joint scintigraphy in brucellosis, Clin Nucl Med 24:756, 1999.
- Erasmus JJ, McAdams HP, Farrell MA, et al: Pulmonary nontuberculous mycobacterial infection: radiologic manifestations, Radiographics 19:1487, 1999.
- Fialk MA, Marcove RC, Armstrong D: Cryptococcal bone disease: a manifestation of disseminated cryptococcosis, Clin Orthop Relat Res 158:219, 1981.
- Gasem MH, Dolmans WM, Isbandrio BB, et al: Culture of Salmonella typhi and Salmonellaparatyphi from blood and bone marrow in suspected typhoid fever, Trop Geogr Med 47:164, 1995.
- Goodwin RA, Shapiro JL, Thurman GH, et al: Disseminated histoplasmosis: clinical and pathologic correlations, Medicine 59:1, 1980.
- Griffi ths DL: Tuberculosis of the spine: a review, Adv Tuberc Res 20:92, 1980.
- Gustke KA, Wu KK: Torulopsis glabrata osteomyelitis: report of a case, Clin Orthop 154:197, 1981.
- Hadjipavlou AG, Mader JT, Nauta HJ, et al: Blastomycosis of the lumbar spine: case report and review of the literature, with emphasis on diagnostic laboratory tools and management, Eur Spine J 7:416, 1998.
- Hall BB, Fitzgerald RH Jr, Rosenblatt JE: Anaerobic osteomyelitis, J Bone Joint Surg 65A:30, 1983.
- Hayasaka K, Aburano T, Tanaka Y: MR imaging in alveolar echinococcosis of bone, Radiat Med 13:179, 1995.
- Hughes LO, Aronson J: Skeletal infections in children, Curr Opin Pediatr 6:90, 1994.
- Kumar R, van der Smissen E, Jorizzo J: Systemic sporotrichosis with osteomyelitis, J Can Assoc Radiol 35:83, 1984.
- Levin TL, Schulman M, Zieba P, et al: Absence of lower extremity ossifi cation centers in term infants with congenital syphilis, J Perinatol 14:106, 1994.
- Lifeso RM, Weaver P, Harder EH: Tuberculous spondylitis in adults, J Bone Joint Surg 67A:1405, 1985.
- Marcus NA, Grace TG, Hodgin UG: Osteomyelitis of the sacrum and sepsis of the hip complicating pelvic actinomycosis, Orthopedics 4:645, 1981.
- Mee AP: Paramyxoviruses and Paget’s disease: the affi rmative view, Bone 24:19, 1999.
- Meier JL: Mycobacterial and fungal infections of bone and joints, Curr Opin Rheumatol 6:408, 1994.
- Molstad B, Stron R: Multiarticular sporotrichosis, JAMA 240:556, 1978.
- Moore RM, Green NE: Blastomycosis of bone: a report of six cases, J Bone Joint Surg 64A:1097, 1982.
- Morgan MA, Cockerill FR, Cortese DA, et al: Disseminated sporotrichosis with Sporothrix schenckii fungemia, Diagn Microbiol Infect Dis 2:151, 1984.
- Moser SA: Laboratory diagnosis of blastomycosis, Clin Microbiol Newsletter 7:53, 1985.
- Norden C, Gillespie WJ, Nade S, eds: Infections in bones and joints, Oxford, 1994, Blackwell Scientifi c. O’Hara M: Histopathologic diagnosis of fungal disease, Infect Control 7:78, 1986.
- Onwuasoigwe O, Gugnani HC: African histoplasmosis: osteomyelitis of the radius, Mycoses 41:105, 1998.
- [Pappagianis D: Epidemiology of coccidioidomycosis. In Stevens DA, ed: Coccidioidomycosis: a text, New York, 1980, Plenum Medical.
Patange V, Cesani F, Phillpott J, et al: Three-phase bone and Ga-67 scintigraphy in disseminated sporotrichosis, Clin Nucl Med 20:909, 1995.](https://pubmed.ncbi.nlm.nih.gov/?term=Pappagianis%20D%3A%20Epidemiology%20of%20coccidioidomycosis.%20In%20Stevens%20DA%2C%20ed%3A%20Coccidioidomycosis%3A%20a%20text%2C%20New%20York%2C%201980%2C%20Plenum%20Medical.%0A%0APatange%20V%2C%20Cesani%20F%2C%20Phillpott%20J%2C%20et%20al%3A%20Three-phase%20bone%20and%20Ga-67%20scintigraphy%20in%20disseminated%20sporotrichosis%2C%20Clin%20Nucl%20Med%2020%3A909%2C%201995.)
- Pathmanathan R, Soon ST: Cryptococcosis in the University Hospital, Kuala Lumpur, and a review of published cases, Trans R Soc Trop Med Hyg 76:21, 1982.
- Pfaller MA, Kyriakos M, Weeks PM, Kobayashi GS: Disseminated histoplasmosis presenting as an acute tenosynovitis, Diagn Microbiol Infect Dis 3:241, 1985.
- Poliner JR: Localized osseous cryptococcosis, J Pediatr 94:597, 1979.
- Rajapakse CNA: Bacterial infections: osteoarticular brucellosis, Baillieres Clin Rheumatol 9:161, 1995.
- Ralston SH, Helfrich MH: Are paramyxoviruses involved in Paget’s disease? A negative view, Bone 24:17S, 1999.
- Rippon JW: Medical mycology, 2nd ed, Philadelphia, 1982, Saunders. Rolston KVI, LeFrock JL, Berman AT, et al: Treatment of osseous cryptococcosis: report of a case and review of the literature, Orthopedics 5:1610, 1982.
- Saccente M, Abernathy RS, Pappas PG, et al: Vertebral blastomycosis with paravertebral abscess: report of eight cases and review of the literature, Clin Infect Dis 26:413, 1998.
- Sankaran-Kutty M, Marwah S, Kutty MK: The skeletal manifestations of brucellosis, Int Orthop 15:17, 1991.
- Schasfoort RA, Marck KW, Houtman PM: Histoplasmosis of the wrist, J Hand Surg 24B:625, 1999.
- Schoenwetter WF, Williams DNB: Diagnosis of blastomycosis, N Engl J Med 315:762, 1986 (letter). Schwarz J: What’s new in mycotic bone and joint diseases? Pathol Res Pract 178:617, 1984.
- Shaw CJ, Thomason AJS, Spencer JD: Fungal osteomyelitis of the foot: a report of an unusual case, J Bone Joint Surg 76B:137, 1994.
- Steere AC, Bartenhagen NH, Craft JE, et al: The early clinical manifestations of Lyme disease, Ann Intern Med 99:76, 1983.
- Steere AC, Green J, Schoen RT, et al: Successful parenteral penicillin therapy of established Lyme arthritis, N Engl J Med 213:869, 1985.
- Steere AC, Malawista SE: Cases of Lyme disease in the United States: locations correlated with distribution of Ixodes dammini, Ann Intern Med 91:730, 1979.
- Steere AC, Malawista SE: Lyme disease. In McCarty DJ, ed: Arthritis and allied conditions, 10th ed, Philadelphia, 1985, Lea & Febiger. Steere AC, Malawista SE, Hardin JA, et al: Erythema chronicum migrans and Lyme arthritis: the enlarging clinical spectrum, Ann Intern Med 86:685, 1977.
- Stillwell WT, Rubin BD, Axelrod JL: Chrysosporium, a new causative agent in osteomyelitis: a case report, Clin Orthop Relat Res 184:190, 1984.
- Tasdan Y, Alikasifglu M, Midilli K, et al: Brucella osteomyelitis of the calcaneus, Pediatr Infect Dis J 17:664, 1998.
- Tekkök IH, Berker M, Özcan OE, et al: Brucellosis of the spine, Neurosurgery 33:838, 1993.
- Voloshin DK, Lacomis D, McMahon D: Disseminated histoplasmosis presenting as myositis and fasciitis in a patient with dermatomyositis, Muscle Nerve 18:531, 1995.
- Wheat LJ, Stein L, Corya BC, et al: Pericarditis as a manifestation of histoplasmosis during two large urban outbreaks, Medicine 62:110, 1983.
- Winn RE, Anderson J, Piper J, et al: Systemic sporotrichosis treated with itraconazole, Clin Infect Dis 17:210, 1993.
- Wirbel RJ, Mues PE, Mutschler WE, et al: Hydatid disease of the pelvis and the femur, Acta Orthop Scand 66:440, 1995.
- Wolinsky E: Mycobacterial diseases other than tuberculosis, Clin Infect Dis 15:1, 1992.
- Zach TL, Penn RG: Localized cryptococcal osteomyelitis in an immunocompetent host, Pediatr Infect Dis 5:601, 1986.
- Zajc I, Orihovac Z, Bagatin M: Temporal actinomycosis: report of a case, J Oral Maxillofac Surg 57:1370, 1999.
- Zaks N, Sukenik S, Alkan M, et al: Musculoskeletal manifestations of brucellosis: a study of 90 cases in Israel, Semin Arthritis Rheum 25:97, 1995.
Detailed Chapters & Topics
Dive deeper into specialized chapters regarding natural-history
