Vascularized Bone Grafting and Capitate Shortening Osteotomy for Treatment of Kienböck Disease
Introduction & Epidemiology
Kienböck disease, the eponymous term for avascular necrosis (AVN) of the lunate bone, represents a progressive and debilitating condition characterized by pain, stiffness, and ultimately, carpal collapse and radiocarpal arthrosis. First described in detail by Robert Kienböck in 1910, its precise etiology remains multifactorial, often involving a complex interplay of anatomical, mechanical, and vascular factors. While its exact incidence is challenging to ascertain due to varied diagnostic criteria and presentation, it predominantly affects young to middle-aged adults, particularly males, with manual occupations.
The pathophysiology is believed to involve compromised blood supply to the lunate, often exacerbated by repetitive microtrauma and abnormal biomechanical loading. Anatomical variants such as negative ulnar variance, where the ulna is shorter relative to the radius, are strongly implicated as they lead to increased compressive forces across the radioscaphoid and radiolunate articulations. This elevated pressure can further impair the already precarious vascular supply of the lunate, which typically relies on a series of dorsal and volar intraosseous anastomoses.
Treatment strategies for Kienböck disease are largely dictated by its stage, commonly classified using the Lichtman system:
*
Stage I:
Normal radiographs, MRI showing decreased signal intensity on T1-weighted images.
*
Stage II:
Sclerosis of the lunate on plain radiographs, no collapse.
*
Stage IIIA:
Lunate collapse with preserved carpal height, intact articular surfaces.
*
Stage IIIB:
Lunate collapse with loss of carpal height, fixed scaphoid flexion.
*
Stage IV:
Pancarpal degenerative changes.
This comprehensive guide focuses on the combined approach of vascularized bone grafting (VBG) and capitate shortening osteotomy (CSO). This dual-pronged strategy aims to address both the underlying vascular insufficiency of the lunate through revascularization provided by the VBG and to reduce the mechanical load on the lunate by shortening the capitate, thereby correcting or ameliorating negative ulnar variance. This combination is particularly indicated in Lichtman Stages II and IIIA, where lunate architecture is largely preserved, and there is a reasonable expectation for revascularization and functional restoration.
Surgical Anatomy & Biomechanics
Successful management of Kienböck disease necessitates a profound understanding of the intricate anatomy and biomechanics of the wrist.
Lunate Anatomy and Vascularity
The lunate, situated centrally in the proximal carpal row, articulates with the radius proximally, the capitate and hamate distally, and the scaphoid and triquetrum laterally. Its unique trapezoidal shape and central position make it a critical link in carpal kinematics. Unlike many other carpal bones, the lunate lacks muscular attachments, relying solely on capsular ligaments for stability.
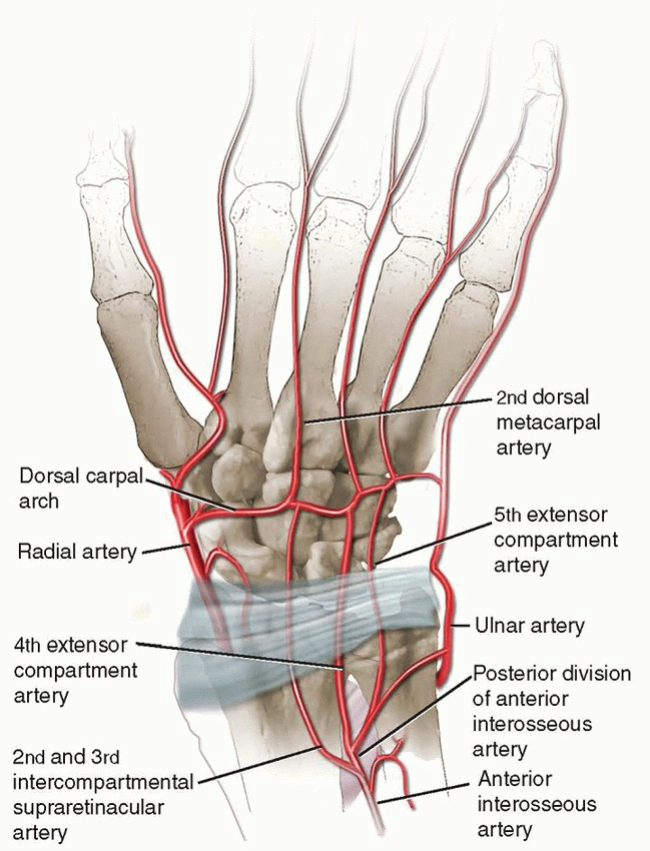
The vascular supply to the lunate is notoriously precarious and highly variable. It typically arises from branches of the dorsal and volar radiocarpal arches, primarily derived from the radial and ulnar arteries. These vessels penetrate the lunate at its dorsal and volar poles. Historically, studies have demonstrated that the lunate commonly receives its blood supply through one or two primary vascular pedicles, usually dorsally and/or volarly. Critically, the intraosseous vascular network within the lunate is often characterized by a "Y" or "U" pattern, with limited anastomoses. This inherent vascular vulnerability, coupled with its role as a fulcrum in wrist motion and its exposure to significant compressive loads, renders it susceptible to ischemic necrosis. The presence of single-entry vessels, rather than multiple, more robust ones, increases the risk of avascular events.
Wrist Biomechanics and Ulnar Variance
The wrist functions as a complex system of articulating bones, transmitting forces between the forearm and hand. Load transmission across the wrist is predominantly through the radiocarpal joint, with the radius bearing approximately 80% and the ulna 20% of the axial load in neutral ulnar variance.
Ulnar Variance
refers to the relative length of the ulna compared to the radius at the distal radioulnar joint.
*
Neutral Ulnar Variance:
Ulna and radius are at the same length.
*
Ulnar Positive Variance:
Ulna is longer than the radius.
*
Ulnar Negative Variance:
Ulna is shorter than the radius.
Negative ulnar variance is a significant predisposing factor for Kienböck disease, found in 70-80% of affected individuals. A shorter ulna results in increased load transmission across the lunate fossa of the radius, leading to chronic impaction and elevated stress on the lunate. This repetitive mechanical stress is hypothesized to further compromise the lunate's already tenuous blood supply.
Capitate Anatomy and Biomechanics
The capitate is the largest carpal bone, located centrally in the distal carpal row. It articulates proximally with the lunate and scaphoid, distally with the second, third, and fourth metacarpals, and radially with the trapezoid and ulnarly with the hamate. Its strong articulation with the lunate forms the midcarpal joint, a critical pivot point for wrist motion. Shortening the capitate reduces the height of the midcarpal column, effectively offloading the lunate and potentially converting a negative ulnar variance to neutral or even positive relative to the lunate. This shift in load distribution aims to create a more favorable mechanical environment for lunate revascularization and recovery.
Vascularized Bone Graft Anatomy (Dorsal Distal Radius)
For vascularized bone grafting in Kienböck disease, several donor sites have been described, but the most commonly utilized and efficacious are pedicled grafts from the distal radius. The specific focus here is on grafts based on the fourth and fifth extensor compartment arteries, which are often terminal branches of the posterior interosseous artery or dorsal carpal arch.
-
Dorsal Distal Radius Grafts (e.g., 4+5 Extensor Compartment Arteries or 1,2-Intercompartmental Supraretinacular Artery - 1,2-ITRA):
These grafts are typically harvested from the dorsal aspect of the distal radius.
- Vascular Pedicle: The pedicle usually consists of a small artery (e.g., a branch of the dorsal carpal arterial arch or the posterior interosseous artery) and accompanying venae comitantes. These vessels are small (0.5-1.0 mm) but consistently present. The 4th and 5th extensor compartment arteries are small terminal branches that course over the dorsal aspect of the distal radius within or adjacent to the retinaculum.
- Bone Harvest: A small corticocancellous bone block, typically 1x1 cm or smaller, is harvested from the non-articulating dorsal aspect of the distal radius, taking care to avoid the Lister's tubercle and maintain wrist joint integrity. The bone block provides osteocytes and a scaffold, while the vascular pedicle ensures immediate blood supply, promoting active revascularization and potentially accelerating healing compared to non-vascularized grafts.
Understanding these anatomical and biomechanical principles is paramount for meticulous surgical planning and execution, aiming to restore both the vascularity and the mechanical equilibrium of the wrist.
Indications & Contraindications
Patient selection is a critical determinant of success for combined vascularized bone grafting and capitate shortening osteotomy in Kienböck disease. The Lichtman classification guides the decision-making process.
Indications
- Lichtman Stage II: This stage is characterized by lunate sclerosis without collapse. VBG alone or in combination with CSO (especially if negative ulnar variance is present or significant axial loading is a factor) is a strong consideration. The goal is to arrest disease progression and achieve revascularization before significant structural changes occur.
- Lichtman Stage IIIA: Lunate collapse is present, but the overall carpal height is maintained, and importantly, the articular surfaces of the lunate are still relatively intact. This stage represents a prime indication for combined VBG and CSO. The VBG aims for revascularization and structural support, while the CSO reduces compressive forces and helps to stabilize the carpus.
- Lichtman Stage IIIB: Characterized by lunate collapse with fixed scaphoid flexion and loss of carpal height. While historically considered more challenging, some authors advocate for combined VBG and CSO in selected cases where significant degenerative changes are not yet pervasive and the primary goal is pain relief and preservation of some wrist motion, especially in younger patients. Prognosis for revascularization and complete restoration of carpal height is diminished at this stage.
- Progressive Pain: Persistent and progressive wrist pain unresponsive to conservative measures in Stages II-IIIB.
- Functional Impairment: Significant functional limitations in daily activities due to pain and stiffness.
- Negative Ulnar Variance: While not an absolute prerequisite for VBG, if negative ulnar variance is present, CSO becomes a more compelling component of the combined approach.
Contraindications
- Lichtman Stage IV: Frank pancarpal degenerative arthritis is an absolute contraindication for VBG and CSO. At this stage, revascularization is unlikely to reverse the widespread articular damage. Alternative procedures such as proximal row carpectomy (PRC), total wrist fusion, or wrist arthroplasty are typically indicated.
- Active Infection: Any active infection in the wrist or surrounding tissues.
- Severe Medical Comorbidities: Patients with severe systemic illnesses that preclude major surgery or general anesthesia.
- Unrealistic Patient Expectations: Patients must understand the potential for ongoing pain, stiffness, and the possibility of further surgery.
- Severe Osteoporosis: Compromised bone quality may hinder osteotomy healing and hardware fixation.
- Smoking: While not an absolute contraindication, active smoking significantly impairs bone healing and vascular graft integration, increasing the risk of nonunion and graft failure. Patients should be counseled aggressively regarding smoking cessation.
TABLE: Operative vs. Non-Operative Indications
| Indication Type | Lichtman Stage | Clinical Presentation | Rationale |
|---|---|---|---|
| Non-Operative | Stage I | Mild, intermittent pain, minimal functional deficit | Observation, activity modification, NSAIDs, splinting. Monitor for progression. |
| Early Stage II | Mild pain, non-progressive symptoms, patient preference | Initial trial of conservative management, especially if risk factors are high. | |
| Any Stage | Severe comorbidities, unwilling to undergo surgery | Symptomatic management only. | |
| Operative | Stage II | Persistent pain, radiographic progression, negative ulnar variance | Revascularization (VBG) and/or load reduction (CSO) to prevent collapse. |
| Stage IIIA | Persistent pain, lunate collapse, preserved articular cartilage | Revascularization (VBG) and load reduction (CSO) to stabilize the carpus. | |
| Select Stage IIIB | Persistent pain, preserved motion, minimal generalized OA, younger patient | Salvage procedure to delay further degeneration, pain relief. Prognosis poorer. | |
| Failed Conservative | Failure of non-operative measures to control pain/progression | Intervention required to address underlying pathology. |
Pre-Operative Planning & Patient Positioning
Meticulous pre-operative planning is paramount for optimizing outcomes and mitigating complications in combined VBG and CSO.
Diagnostic Imaging
-
Plain Radiographs:
Standard PA, lateral, and oblique views of the wrist are essential.
- PA View: Assess lunate collapse (e.g., carpal height ratio), sclerosis, and fragmentation. Critical for determining ulnar variance (using the perpendicular line method from the distal ulna and radius).
- Lateral View: Evaluate lunate tilt and fixed scaphoid flexion (a hallmark of Stage IIIB).
- Clenched Fist Views: Can exacerbate negative ulnar variance and help identify dynamic changes.
-
Magnetic Resonance Imaging (MRI):
The gold standard for assessing lunate viability and the extent of avascularity.
- T1-weighted images: Typically show low signal intensity in the necrotic areas.
- T2-weighted images and STIR sequences: May show high signal intensity in areas of edema or revascularization attempts.
- MRI also helps evaluate articular cartilage integrity, soft tissue swelling, and rule out other pathologies.
- Computed Tomography (CT) Scan: Particularly useful for precise assessment of bony architecture, fragmentation, and planning the capitate osteotomy. High-resolution CT can delineate the extent of collapse and assist in determining the exact amount of bone to resect from the capitate.
- Arteriography (Optional): Rarely performed routinely, but can be considered if the specific vascular anatomy of the donor site is unclear or in cases of suspected previous vascular injury. Often, the robust nature of the identified dorsal pedicles (e.g., 4th and 5th compartment arteries) makes formal arteriography unnecessary.
Patient Counseling
Comprehensive discussion regarding the nature of Kienböck disease, the rationale for the combined procedure, potential risks (nonunion, graft failure, infection, stiffness, pain, donor site morbidity), expected recovery timeline, and the possibility of further surgical intervention is crucial. Patients must have realistic expectations regarding pain relief and functional improvement.
Anesthesia
General anesthesia is typically employed, often combined with a regional anesthetic block (e.g., axillary or supraclavicular block) for enhanced post-operative pain control. This approach facilitates a smoother emergence from anesthesia and reduces immediate post-operative analgesic requirements.
Patient Positioning
The patient is positioned supine on the operating table. The operative arm is placed on a radiolucent hand table, allowing for full wrist motion and unimpeded fluoroscopic imaging.
*
Tourniquet:
A pneumatic tourniquet is applied to the proximal upper arm to maintain a bloodless field, which is critical for meticulous soft tissue dissection, particularly during vascularized graft harvest and microvascular pedicle preservation.
*
Padding:
All pressure points are meticulously padded to prevent neuropathies or skin breakdown.
*
Draping:
The limb is prepped and draped to allow for full sterile access to the entire forearm and hand, facilitating both the dorsal wrist approach for capitate osteotomy and lunate debridement, and the dorsal distal radial approach for graft harvest.
Instrumentation
- Standard hand and wrist surgical sets.
- Small osteotomes, curettes, a fine oscillating saw (e.g., micro-sagittal saw).
- High-speed burr.
- Headless compression screws (e.g., Acutrak, Herbert) or small plates for capitate fixation. K-wires for temporary fixation or graft stabilization.
- Microvascular instruments (fine forceps, vessel loops, micro-sutures if microanastomosis is contemplated, though typically not required for pedicled grafts).
- Fluoroscopy unit.
Detailed Surgical Approach / Technique
The combined procedure involves distinct, yet coordinated, surgical steps: vascularized bone graft harvest, capitate shortening osteotomy, lunate debridement, and graft placement. The sequence may vary based on surgeon preference, but often the graft harvest is initiated first, followed by osteotomy and lunate preparation.
I. Vascularized Bone Graft Harvest (Dorsal Distal Radius)
This approach focuses on a pedicled graft based on vessels within the fourth and fifth extensor compartments.
1. Incision and Exposure
A 5- to 6-cm longitudinal skin incision is made over the dorsal aspect of the distal radius, typically located between the fourth (extensor digitorum communis, EDC) and fifth (extensor digiti minimi, EDM) extensor compartments.

Careful dissection of the subcutaneous tissue is performed. The extensor retinaculum is identified and can be opened longitudinally over the 4th and 5th compartments, or carefully elevated. The goal is to expose the dorsal aspect of the distal radius, radial to Lister's tubercle.
2. Identification and Dissection of the Vascular Pedicle
The interval between the fourth and fifth extensor compartments is meticulously identified. The dorsal branch of the radial artery, or more commonly, perforators arising from the posterior interosseous artery or dorsal carpal arch (such as the 4th and 5th extensor compartment arteries or the 1,2-intercompartmental supraretinacular artery, 1,2-ITRA), are sought within the loose connective tissue overlying the dorsal radius. These vessels are typically small, requiring loupe magnification (2.5x to 4.5x) for precise identification and dissection.
The associated venae comitantes are identified. Careful, blunt and sharp dissection isolates the vascular pedicle, ensuring its integrity throughout the harvest. The pedicle needs to be sufficiently long to allow for tension-free transfer to the lunate.
The surrounding soft tissue is preserved around the vessels to protect them.
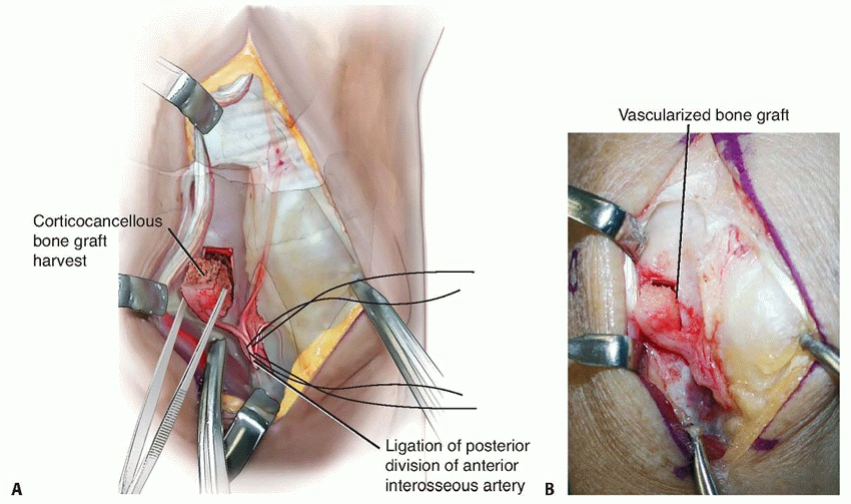
3. Bone Graft Harvest
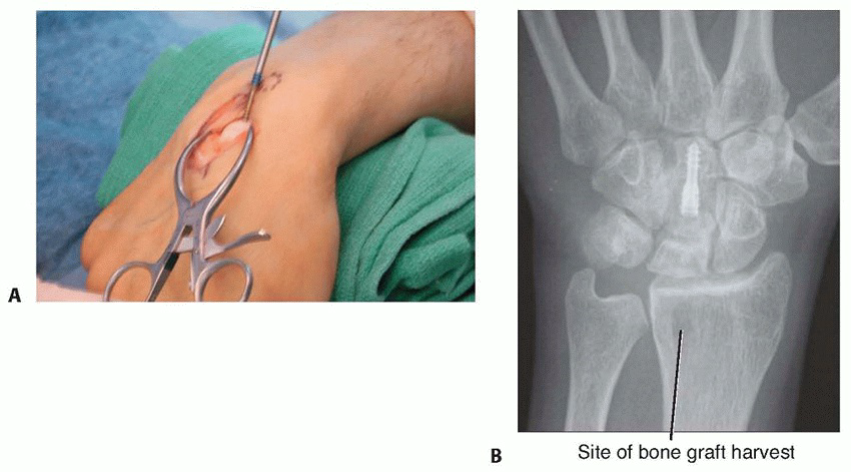
Once the vascular pedicle is isolated, a small corticocancellous bone block, typically 1.0 to 1.5 cm in length and 0.5 to 1.0 cm in width, is harvested from the non-articular dorsal aspect of the distal radius. This harvest site is distal to the joint line and usually radial to the dorsal tubercle (Lister's tubercle), ensuring no compromise to the radiocarpal articular surface. A fine oscillating saw is used to make precise osteotomies, carefully protecting the underlying vascular pedicle. The bone block is then gently elevated with its attached pedicle. The donor site can be packed with bone wax or local bone graft if needed, though often heals without additional measures.
II. Capitate Shortening Osteotomy
1. Incision and Exposure
A separate dorsal longitudinal incision, approximately 4-5 cm, is made centered over the capitate, typically between the third (extensor pollicis longus, EPL) and fourth (EDC) extensor compartments. Alternatively, a single, longer dorsal incision may be used, extending distally from the lunate.
The retinaculum is incised, and the EPL tendon is retracted radially, and the EDC tendons ulnarly. The dorsal wrist capsule is then incised longitudinally or in an H-shape to expose the dorsal aspect of the capitate. The midcarpal joint is identified.
2. Osteotomy Planning
Using pre-operative radiographs and CT scans, and intra-operative fluoroscopy, the precise amount of capitate shortening is determined. This typically ranges from 2-4 mm, aiming to restore neutral or slightly positive ulnar variance relative to the lunate, thereby offloading the lunate. The osteotomy is planned in the mid-body of the capitate, ensuring adequate proximal and distal bone stock for stable fixation.

K-wires can be used as temporary guides to mark the osteotomy cuts.
3. Osteotomy and Resection
With the aid of fluoroscopy, two parallel transverse osteotomy cuts are made in the mid-body of the capitate using a fine oscillating saw. The pre-measured segment of bone is carefully removed. It is crucial to maintain carpal alignment during the cuts to ensure proper apposition of the osteotomy fragments.
4. Reduction and Fixation
The carpus is compressed to reduce the capitate, bringing the proximal and distal fragments into close apposition. This maneuver shortens the carpal column. The osteotomy is then stabilized. Headless compression screws (e.g., Acutrak, Herbert) are commonly used due to their low profile, strong compression, and minimal soft tissue irritation.
Often, two headless screws are placed longitudinally across the osteotomy site for rotational stability and robust compression. K-wires can be used for temporary fixation prior to screw placement.
Fluoroscopy is used to confirm adequate shortening, anatomical reduction, and secure fixation. The aim is a stable construct that allows for early mobilization in the post-operative period.
III. Lunate Preparation & Graft Placement
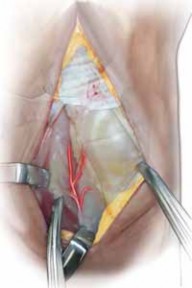
1. Lunate Exposure and Debridement
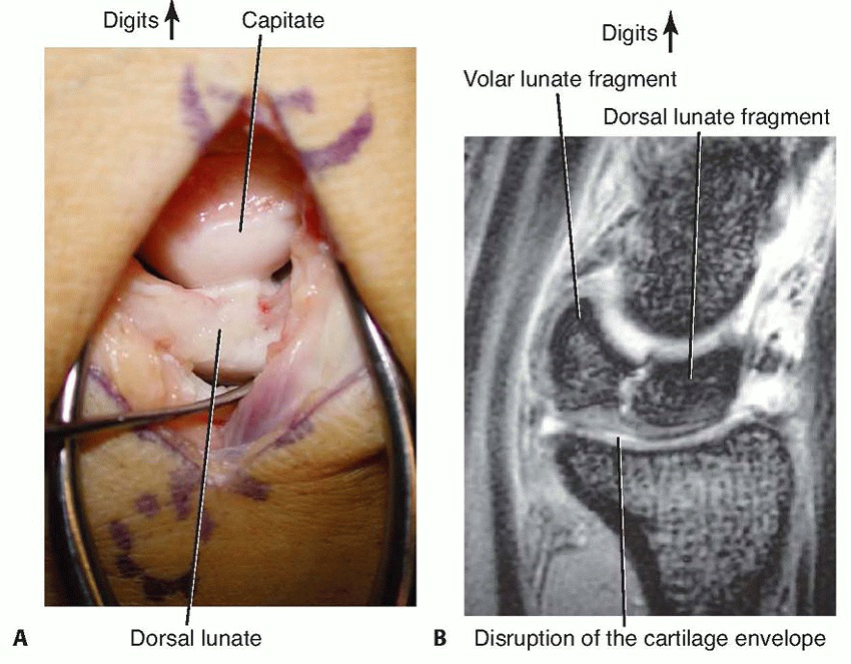
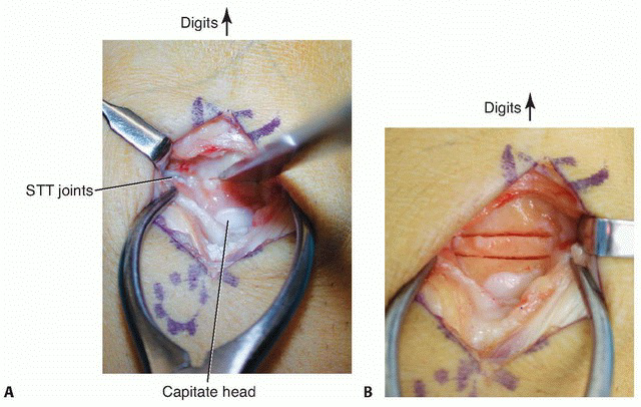
If a separate incision was made for the capitate, it may be extended, or the interval used to expose the lunate. The dorsal capsule overlying the lunate is carefully incised. The necrotic portion of the lunate is identified.
Using small curettes, osteotomes, or a high-speed burr, the necrotic, sclerotic bone of the lunate is meticulously debrided. The goal is to remove all devitalized tissue while preserving as much healthy subchondral bone and articular cartilage as possible. A cavity is created that precisely accommodates the vascularized bone graft. Hemostasis in the lunate bed is important.
2. Graft Insetting and Fixation
The previously harvested vascularized bone graft, with its intact pedicle, is now carefully transferred and inserted into the prepared lunate defect. It is critical to ensure that the pedicle is not kinked, twisted, or compressed during placement, as this could compromise its blood supply. The graft should fit snugly within the defect.
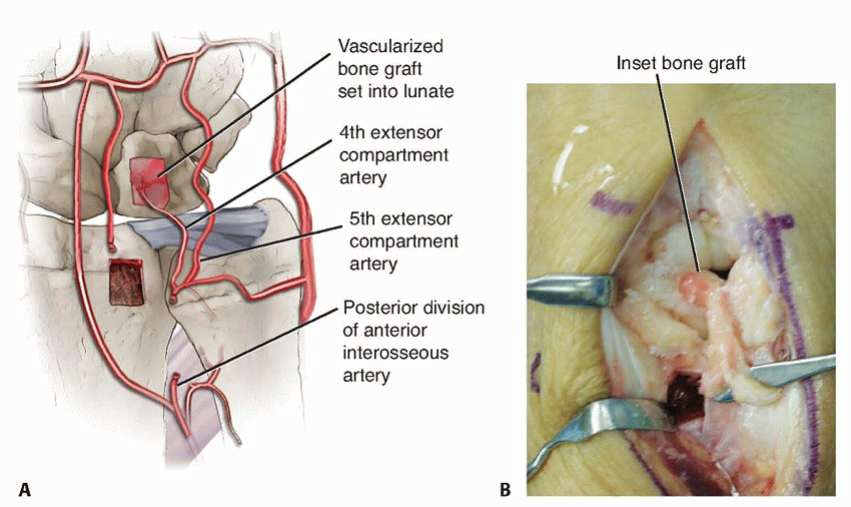
The orientation of the graft should ideally align the vascular inflow with the remaining healthy lunate vasculature, promoting maximal revascularization.
The graft is typically impacted gently into the lunate defect. In some cases, a small K-wire may be used to temporarily secure the graft within the lunate. Fibrin glue can also be utilized to help stabilize the graft.
IV. Wound Closure
Once the capitate osteotomy is fixed and the vascularized graft is securely in place within the lunate, thorough irrigation of the surgical field is performed. The dorsal wrist capsule is closed. The extensor retinaculum is repaired or left open if tension is excessive, ensuring smooth gliding of the tendons. Subcutaneous layers and skin are closed using standard surgical techniques. A sterile dressing and a sugar-tong splint or a well-molded short arm cast are applied, immobilizing the wrist in a neutral to slight extension position.
Complications & Management
Despite meticulous surgical technique, complications can arise following vascularized bone grafting and capitate shortening osteotomy. Awareness of these potential issues and strategies for management is crucial for all orthopedic surgeons.
Intraoperative Complications
- Vascular Pedicle Injury/Thrombosis: Disruption or thrombosis of the vascular pedicle during harvest or transfer. This is a critical complication as it converts a vascularized graft into a non-vascularized one. Meticulous technique, magnification, and gentle handling are paramount. If identified, microvascular repair may be attempted, but often, the graft may need to be treated as a non-vascularized graft or a new pedicled graft harvested from a different site.
- Neurovascular Injury: Damage to superficial nerves (e.g., dorsal sensory branch of the radial nerve, which crosses the dorsal wrist) or major vessels. Careful dissection and clear visualization are key.
- Fracture during Osteotomy: Unintended fracture of the capitate during osteotomy or compression. May necessitate altering the fixation strategy or abandoning the capitate shortening.
- Inadequate Fixation: Poor purchase of screws in the capitate, leading to instability of the osteotomy. Requires revision of fixation.
Early Postoperative Complications
- Hematoma: Accumulation of blood in the surgical site. May require evacuation if symptomatic or causing compartment syndrome.
- Infection: Superficial or deep surgical site infection. Requires aggressive antibiotic therapy, and potentially surgical debridement and irrigation.
- Vascular Compromise of Graft: Although less common with pedicled grafts than free flaps, graft ischemia can occur due to pedicle compression or thrombosis. Clinical signs (pain, swelling, skin changes) are often subtle. Requires immediate exploration.
- Excessive Swelling/Pain: May indicate infection, hematoma, or inadequate pain control.
Late Postoperative Complications
- Nonunion or Malunion of Capitate Osteotomy: Failure of the capitate fragments to heal or healing in an unacceptable alignment. Incidence is relatively low with rigid internal fixation. Salvage often involves revision osteotomy with re-fixation and bone grafting.
- Persistent Lunate Avascularity/Nonunion: The VBG may fail to revascularize the lunate, or the lunate may continue to collapse. This can manifest as persistent pain and radiographic progression. Salvage strategies depend on the stage of collapse and include further limited procedures (e.g., proximal row carpectomy) or total wrist fusion.
- Progression of Kienböck's Disease: Despite successful revascularization and load reduction, some cases may still progress to advanced stages of collapse and arthritis. This underscores the multifactorial nature of the disease and the limitations of current interventions.
- Wrist Stiffness/Limited Range of Motion (ROM): A common complication due to prolonged immobilization and capsular adhesions. Aggressive hand therapy is crucial. Manipulation under anesthesia may be considered.
- Hardware Irritation: Screws or plates used for capitate fixation may cause palpable irritation or impinge on tendons. Hardware removal may be necessary after osteotomy healing.
- Donor Site Morbidity: Pain, numbness (e.g., dorsal sensory branch of radial nerve irritation), or prominent scar at the graft harvest site.
- Post-traumatic Osteoarthritis: Development of degenerative changes in the radiocarpal or midcarpal joints. This is a long-term risk for any intervention for Kienböck disease, particularly in advanced stages.
TABLE: Common Complications, Incidence, and Salvage Strategies
| Complication | Incidence (Approx.) | Salvage Strategy |
|---|---|---|
| Vascular Pedicle Injury/Thrombosis | <5% | Microvascular repair, re-graft, treat as non-vascularized graft |
| Capitate Osteotomy Nonunion/Malunion | 5-10% | Revision osteotomy, re-fixation, bone grafting |
| Persistent Lunate Avascularity/Collapse | 10-20% | Proximal row carpectomy (PRC), carpal fusion, further VBG |
| Wrist Stiffness/Reduced ROM | 15-25% | Intensive hand therapy, manipulation under anesthesia |
| Hardware Irritation | 10-15% | Hardware removal (after healing) |
| Infection | <2% | Antibiotics, surgical debridement, irrigation |
| Donor Site Morbidity (Pain/Numbness) | 5-10% | Symptomatic management, nerve decompression (rare) |
| Progression to OA | Variable (Long-term) | PRC, partial/total wrist fusion, wrist arthroplasty |
Post-Operative Rehabilitation Protocols
A structured and progressive post-operative rehabilitation program is essential for maximizing functional recovery and optimizing outcomes following combined vascularized bone grafting and capitate shortening osteotomy. Close collaboration between the surgeon and a specialized hand therapist is crucial.
Initial Immobilization Phase (Weeks 0-6 to 8)
- Goal: Protect surgical sites, promote bone and graft healing, control pain and edema.
-
Immobilization:
- Immediately post-op: A well-molded sugar-tong splint or short arm cast is applied, immobilizing the wrist in neutral to slight extension and the forearm in neutral rotation.
- This immobilization period is critical for capitate osteotomy healing and graft incorporation, typically maintained for 6 to 8 weeks, potentially longer depending on radiographic evidence of healing and surgeon preference.
-
Activity:
- No weight-bearing on the affected hand.
- Strict avoidance of wrist motion.
- Encourage active range of motion (AROM) of the fingers, thumb (MCP, PIP, DIP joints), elbow, and shoulder to prevent stiffness in adjacent joints.
-
Edema Control:
- Elevation of the limb (above the heart).
- Gentle retrograde massage (proximal to distal).
- Compression garments or wraps over areas not covered by the cast/splint.
- Pain Management: Adherence to prescribed analgesics.
- Wound Care: Monitor surgical sites for signs of infection or complications.
Early Motion Phase (Weeks 8-12)
- Goal: Gradually restore passive and active wrist range of motion, initiate gentle strengthening.
-
Immobilization:
- Upon radiographic evidence of adequate capitate osteotomy healing and clinical stability, the cast/splint is discontinued.
- A removable wrist brace may be provided for protection during activity or sleep.
-
Range of Motion (ROM):
- Begin with gentle, pain-free active and active-assisted wrist flexion/extension, radial/ulnar deviation, and forearm pronation/supination.
- Passive ROM exercises may be introduced carefully by the therapist, avoiding excessive force.
- Scar massage to improve tissue mobility and reduce adhesions.
-
Strengthening:
- Isometric strengthening exercises for wrist extensors, flexors, and forearm muscles are introduced, progressing from light resistance.
- Avoid heavy gripping or forceful movements.
- Functional Activities: Gradually incorporate light, non-weight-bearing activities of daily living (ADLs), ensuring proper body mechanics and wrist protection.
Strengthening & Return to Activity Phase (Weeks 12-24 and beyond)
- Goal: Progress strength, endurance, and coordination; facilitate return to full functional activities, work, and sports.
-
Strengthening:
- Progressive resistive exercises using TheraBand, light weights, and putty.
- Focus on eccentric and concentric strengthening of all wrist and forearm musculature.
- Incorporate grip strengthening exercises.
-
Endurance and Coordination:
- Fine motor dexterity tasks.
- Proprioceptive training.
- Gradual increase in duration and intensity of activities.
-
Return to Activity:
- Gradual return to work-specific tasks, recreational activities, and sports. This process is highly individualized and depends on the patient's occupation and sport.
- Guidance on proper lifting techniques and activity modification.
- Avoidance of high-impact or repetitive loading activities for at least 6 months, and often longer.
-
Long-Term Follow-up:
- Continued clinical and radiographic assessment for at least 1-2 years to monitor lunate revascularization, capitate osteotomy healing, and progression of any degenerative changes. MRI may be repeated to assess lunate revascularization.
- Patient education regarding the chronic nature of Kienböck's disease and the potential for long-term complications or need for further intervention.
The total rehabilitation period can span 6 to 12 months, with full functional recovery potentially taking longer. Adherence to the protocol and close monitoring for signs of complications are critical throughout this phase.
Summary of Key Literature / Guidelines
The management of Kienböck disease has evolved significantly, and the combined approach of vascularized bone grafting and capitate shortening osteotomy represents a sophisticated strategy for specific stages. The body of literature supporting this approach emphasizes its role in addressing both the vascular and mechanical components of the disease.
Evidence for Combined VBG + CSO
Multiple studies have reported favorable outcomes for combined VBG and CSO, particularly in Lichtman Stages II and IIIA.
*
Revascularization:
Vascularized bone grafts have demonstrated higher rates of lunate revascularization compared to non-vascularized grafts. Histological and MRI studies often show evidence of improved vascularity and bone remodeling within the lunate post-VBG. This revascularization is crucial for halting the progression of necrosis and promoting structural integrity.
*
Pain Relief:
Consistent reports indicate significant pain reduction, which is a primary patient-reported outcome.
*
Functional Improvement:
Patients typically experience improvements in grip strength and range of motion, although complete restoration of pre-morbid function is not always achieved.
*
Radiographic Outcomes:
Studies often demonstrate stabilization of lunate height and shape, prevention of further carpal collapse, and in some cases, partial restoration of carpal height. The capitate osteotomy helps to maintain these parameters by reducing axial loading on the lunate.
*
Long-Term Durability:
While long-term follow-up remains crucial, several studies suggest that the combined procedure can provide durable results, postponing or preventing the need for more salvage procedures like proximal row carpectomy or wrist fusion in carefully selected patients.
Comparison with Other Treatment Modalities
- Radial Shortening Osteotomy: This procedure also aims to offload the lunate by shortening the radius, primarily indicated for patients with negative ulnar variance. While effective for load reduction, it does not directly address lunate revascularization. The choice between radial shortening and capitate shortening often depends on surgeon preference, the precise amount of shortening required, and potential impact on distal radioulnar joint (DRUJ) mechanics. Capitate shortening is advantageous as it does not affect the DRUJ.
- Proximal Row Carpectomy (PRC): This is a salvage procedure for Stages IIIB and IV, involving removal of the scaphoid, lunate, and triquetrum. It provides excellent pain relief and preserves a good range of motion but sacrifices carpal bones. It is generally reserved for more advanced degenerative changes where reconstructive options are less feasible.
- Partial/Total Wrist Fusion: Also salvage procedures, offering reliable pain relief and stability at the expense of motion. Reserved for end-stage arthritis or failed previous interventions.
- Non-Vascularized Bone Grafts: Historically, non-vascularized grafts were used but showed lower rates of lunate revascularization and higher failure rates, leading to the development of vascularized techniques.
Debates and Ongoing Research
- Optimal Graft Donor Site: While dorsal distal radius grafts are popular, other pedicled options (e.g., pisiform, volar distal radius) and free vascularized flaps are continually explored. The "ideal" graft remains a subject of debate, with factors such as pedicle reliability, ease of harvest, and donor site morbidity influencing choice.
- Amount of Capitate Shortening: The precise amount of capitate shortening (typically 2-4mm) is guided by pre-operative planning and intra-operative fluoroscopy, aiming to achieve neutral or slight positive ulnar variance. The optimal degree of shortening to maximize offloading while preserving carpal mechanics is still researched.
- Lichtman Stage IIIB Management: The role of VBG and CSO in Lichtman Stage IIIB remains controversial. While some advocate for its use in select cases with minimal degenerative changes, the prognosis is guarded, and many prefer salvage options.
- Role of Biologics: The integration of growth factors, stem cells, and other biologics to enhance graft survival and lunate regeneration is an active area of research.
Guidelines and Key Considerations
- Patient Selection: This is paramount. The combined VBG and CSO are best suited for younger, active patients with Lichtman Stage II or IIIA disease, where lunate collapse is minimal to moderate, and articular surfaces are preserved.
- Surgeon Experience: These are technically demanding procedures requiring proficiency in hand and wrist surgery, and often microvascular techniques (even for pedicled grafts, meticulous dissection is required).
- Long-Term Follow-up: Kienböck disease is a progressive condition. Patients require long-term clinical and radiographic monitoring to assess stability, revascularization, and the potential for late degenerative changes.
In conclusion, vascularized bone grafting combined with capitate shortening osteotomy represents a robust, evidence-based approach for the treatment of Kienböck disease in appropriate stages. It offers the unique advantage of simultaneously addressing both the vascular compromise and the abnormal biomechanical loading on the lunate, providing a viable pathway to pain relief and functional restoration in a challenging wrist pathology.
Clinical & Radiographic Imaging
You Might Also Like