Reconstruction Materials: What You Need to Know About Associated Complications
Key Takeaway
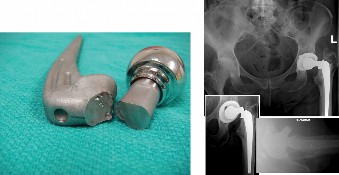
This article provides essential research regarding Reconstruction Materials: What You Need to Know About Associated Complications. A femoral neck fracture in an 82-year-old with a total hip replacement most likely occurred due to mechanical etching or laser marking on the component during production. This relates to the inherent quality of the implant's materials and associated manufacturing processes. Risk factors for such failures can include excessive body weight and inadequate proximal osseous support.
Reconstruction Materials: What You Need to Know About Associated Complications
Introduction & Epidemiology
Orthopedic reconstruction encompasses a vast array of surgical procedures aimed at restoring anatomical integrity, biomechanical function, and pain-free mobility following trauma, degenerative disease, tumor resection, or congenital deformities. Central to these endeavors is the judicious selection and application of reconstruction materials. These materials, broadly categorized as autografts, allografts, synthetic biomaterials, and prosthetic implants, each possess unique biological, mechanical, and interface properties that dictate their suitability for specific clinical scenarios.
The increasing complexity and frequency of reconstructive procedures have brought a heightened focus on the associated complications. While advancements in surgical technique, imaging, and material science have significantly improved outcomes, the inherent challenges of integrating foreign or transplanted materials into the human body persist. Complications range from acute, such as infection or neurovascular injury, to chronic issues like non-union, implant loosening, osteolysis, or immunological responses. The epidemiological landscape is dynamic, with changing patterns of material use, evolving surgical indications, and an aging, more active patient population contributing to shifts in complication profiles. A thorough understanding of these risks is paramount for informed pre-operative counseling, meticulous surgical execution, and effective post-operative management, ultimately optimizing patient outcomes and mitigating iatrogenic morbidity.
Surgical Anatomy & Biomechanics
The successful integration and long-term function of any reconstruction material are inextricably linked to the intricate surgical anatomy and biomechanical demands of the operative site. A comprehensive understanding of these factors is critical for material selection and surgical planning.
Anatomical Considerations
- Vascularity: The blood supply to the recipient bed profoundly influences graft incorporation and healing. Autografts, especially vascularized grafts, offer superior healing potential due to preserved blood flow. Allografts and synthetic materials rely on host tissue ingrowth and neovascularization, which can be compromised in poorly vascularized areas (e.g., distal tibia, scaphoid).
- Soft Tissue Envelope: Adequate soft tissue coverage is essential for protecting underlying reconstruction materials, promoting healing, and minimizing infection risk. Areas with thin soft tissue envelopes (e.g., ankle, olecranon) are particularly susceptible to exposure or breakdown, especially with prominent hardware or non-incorporating grafts.
- Innervation: Peripheral nerve pathways must be meticulously identified and protected during material harvest and implantation. Donor site morbidity from autograft harvest can include nerve injury (e.g., saphenous nerve with hamstring autograft, sural nerve with fibular autograft).
- Bone Stock & Quality: The quantity and quality of host bone directly impact the primary stability and long-term fixation of implants and the incorporation of grafts. Osteoporotic bone presents challenges for screw purchase, press-fit components, and graft integration.
- Joint Kinematics: When reconstructing periarticular structures or replacing joints, the precise anatomical configuration and kinematic profile of the joint must be restored to prevent impingement, instability, or accelerated wear.
Biomechanical Principles
-
Load Bearing vs. Load Sharing:
- Load Bearing: Materials (e.g., prosthetic implants, highly rigid plates) are designed to primarily bear the physiological loads, allowing biological healing to occur in a protected environment. Their failure often results in catastrophic loss of function.
- Load Sharing: Grafts (e.g., bone grafts, biological scaffolds) are intended to gradually integrate with host tissue, sharing loads as they heal and remodel. Their mechanical properties should ideally match the host tissue's needs during the healing cascade.
-
Mechanical Strength & Stiffness:
- The chosen material must possess sufficient initial mechanical strength to withstand physiological loads, prevent early failure, and allow for rehabilitation.
- Stiffness (elastic modulus) mismatch between the implant and host bone can lead to "stress shielding," where the implant bears too much load, reducing stress on the bone and potentially leading to bone resorption and loosening. Conversely, insufficient stiffness can lead to implant fatigue or failure.
- Fatigue Resistance: Reconstruction materials, particularly those subjected to repetitive cyclic loading (e.g., knee or hip prostheses, fracture fixation plates), must exhibit excellent fatigue resistance to ensure long-term durability.
-
Osseointegration & Biocompatibility:
- Osseointegration: The direct structural and functional connection between ordered, living bone and the surface of a load-carrying implant. This is crucial for the long-term stability of many prosthetic components. Surface treatments and material properties (e.g., titanium alloys) are designed to enhance this.
- Biocompatibility: The ability of a material to perform with an appropriate host response in a specific application. Non-biocompatible materials can provoke inflammatory or allergic reactions, leading to material degradation or host tissue rejection.
- Interface Mechanics: The interface between the reconstruction material and host tissue is critical. For bone grafts, this involves creeping substitution. For implants, it involves press-fit, cement fixation, or screw fixation, each with specific biomechanical considerations regarding stability and load transfer.
Indications & Contraindications
The selection of reconstruction materials is a nuanced decision, guided by a multitude of factors including the anatomical site, defect size, desired mechanical properties, patient comorbidities, and surgeon preference.
General Indications for Reconstruction Materials
-
Autograft:
- Small to moderate bone defects requiring osteoinductive, osteoconductive, and osteogenic properties.
- Non-unions requiring biological augmentation.
- Soft tissue reconstruction (tendon, ligament, nerve) when autologous tissue is preferred for its biological compatibility and remodeling potential.
- When allograft or synthetic options are contraindicated or have failed.
-
Allograft:
- Large bone defects (e.g., tumor resection, revision arthroplasty) where autograft harvest is impractical or insufficient.
- Ligament/tendon reconstruction (e.g., ACL reconstruction) when autograft harvest is undesirable or patient factors (multi-ligament injury, revision surgery) necessitate its use.
- Articular cartilage repair (osteochondral allografts).
- Spinal fusion.
-
Synthetic Biomaterials / Biological Scaffolds:
- Small bone defects (e.g., calcium phosphate cements, synthetic bone graft substitutes).
- Tendon/ligament augmentation or repair (e.g., synthetic meshes, xenografts, allogeneic dermal matrices).
- Articular cartilage repair (e.g., synthetic scaffolds for chondrocyte implantation).
- When a non-biological, readily available, and consistent material is desired.
-
Prosthetic Implants (Metals, Ceramics, Polymers):
- Joint replacement (arthroplasty: hip, knee, shoulder, ankle, etc.).
- Internal fixation of fractures (plates, screws, nails, wires).
- Spinal instrumentation (rods, screws, cages).
- Tumor prostheses for massive resections.
General Contraindications for Reconstruction Materials
- Active Infection: Absolute contraindication for any non-biologic implant or allograft; relative contraindication for autograft due to potential for propagation and compromised healing.
- Insufficient Host Bone/Soft Tissue: Inadequate bone stock or compromised soft tissue envelope to support fixation or promote graft incorporation.
- Severe Peripheral Vascular Disease: Compromised blood supply to the operative site, hindering graft incorporation and increasing infection risk.
-
Uncontrolled Systemic Conditions:
- Diabetes mellitus with poor glycemic control.
- Severe malnutrition.
- Immunosuppression (e.g., HIV, long-term steroid use).
- Significant tobacco use (impairs healing).
- These conditions increase the risk of infection, poor healing, and material failure.
- Patient Non-Compliance: Unrealistic expectations or inability to adhere to post-operative protocols (e.g., weight-bearing restrictions, rehabilitation).
- Allergy to Material Components: Rare but significant (e.g., nickel allergy with certain metal implants).
Table: Operative vs. Non-Operative Indications for Reconstruction
| Factor / Condition | Operative Indication for Reconstruction Material | Non-Operative Indication (Material Not Required) |
|---|---|---|
| Fracture Management | Displaced intra-articular fractures, segmental bone loss, non-unions, open fractures (external fixation/internal fixation) | Non-displaced fractures, stable fractures (conservative casting/bracing), stress fractures |
| Ligament/Tendon Injury | Complete tears with functional instability (e.g., ACL rupture, rotator cuff avulsion requiring repair/reconstruction) | Partial tears, stable injuries with adequate residual function, musculotendinous strains (PT, bracing) |
| Articular Cartilage Defect | Full-thickness defects, large defects, symptomatic defects unresponsive to conservative care (osteochondral grafting) | Small, asymptomatic defects, superficial lesions, diffuse osteoarthritis (conservative management, injections) |
| Bone Loss/Defect | Traumatic bone loss, tumor resection, severe osteomyelitis requiring debridement and reconstruction | Small contained bone cysts, minor cancellous defects amenable to natural healing |
| Joint Degeneration | End-stage osteoarthritis, inflammatory arthritis with severe joint destruction (arthroplasty) | Mild to moderate osteoarthritis (activity modification, NSAIDs, injections, PT), asymptomatic degenerative changes |
| Spinal Instability/Deformity | Significant instability, progressive deformity, neurological deficit requiring fusion/stabilization | Mild degenerative changes, stable spondylolisthesis without neurological compromise (PT, epidural injections) |
| Donor Site Morbidity Risk | Autograft harvest when benefits outweigh donor site complications. | When allograft/synthetic alternatives are suitable, or donor site risk is prohibitive (e.g., osteoporotic patient). |
| Infection Risk | Contained chronic infection following debridement (staged reconstruction), low-grade infection. | Active acute infection (requires eradication before material implantation). |
Pre-Operative Planning & Patient Positioning
Meticulous pre-operative planning and appropriate patient positioning are critical to optimize outcomes and minimize complications associated with reconstruction materials. This phase integrates material-specific considerations with general surgical principles.
Pre-Operative Planning
-
Diagnosis and Assessment:
- Thorough Clinical Evaluation: Patient history, physical examination, assessment of comorbidities, functional status, and patient expectations.
- Advanced Imaging: Radiographs, CT scans (with 3D reconstructions for complex fractures/deformities), MRI (for soft tissue and cartilage assessment), angiography (if vascular compromise suspected).
-
Material-Specific Assessment:
- Autograft: Evaluate potential donor sites for bone quality, quantity, and associated morbidity (e.g., iliac crest, fibula, hamstring tendons, patellar tendon).
- Allograft: Determine required size and morphology, ensure availability from a reputable tissue bank, confirm processing (e.g., fresh-frozen, freeze-dried, gamma-irradiated), and thaw protocols.
- Synthetic/Prosthetic: Templating for size and alignment (e.g., total joint arthroplasty), confirming inventory availability, understanding material properties (e.g., porosity, mechanical strength, biocompatibility).
-
Surgical Strategy & Material Selection:
- Defect Analysis: Precisely define the size, shape, and location of the defect requiring reconstruction.
- Biomechanical Demands: Match the mechanical properties of the chosen material to the anticipated loads and stresses at the reconstruction site.
- Biological Needs: Consider osteoinductive, osteoconductive, or osteogenic properties if bone healing is primary goal.
- Contingency Planning: Plan for potential intra-operative challenges, such as inadequate bone stock, unexpected soft tissue defects, or inability to achieve primary stability. Have alternative materials or fixation methods readily available.
-
Infection Prophylaxis:
- Pre-operative Antibiotics: Administer appropriate intravenous antibiotics 30-60 minutes prior to incision, guided by institutional protocols and patient allergies.
- Skin Preparation: Meticulous skin preparation using chlorhexidine or iodine-based solutions. Hair removal should be performed with clippers, not razors.
- Nutritional Optimization: Address any nutritional deficiencies, particularly in revision cases or patients with significant comorbidities, as poor nutrition compromises wound healing and increases infection risk.
-
Logistics:
- Confirm all necessary implants, instruments, and materials are available and sterilized.
- Coordinate with tissue banks for allograft delivery and thawing as per protocol.
- Ensure appropriate intraoperative imaging (fluoroscopy, navigation systems) is functional.
Patient Positioning
Patient positioning must achieve optimal surgical exposure, allow for safe material harvest (if applicable), permit full range of motion testing, and protect the patient from iatrogenic injury.
-
General Principles:
- Padding: All pressure points (e.g., heels, elbows, sacrum, ulnar nerve) must be meticulously padded to prevent pressure sores or nerve palsies.
- Vascular Access: Adequate IV access and arterial lines if hemodynamic monitoring is anticipated.
- Anesthesia Considerations: Close collaboration with anesthesia to manage airway, hemodynamics, and potential blood loss.
- Tourniquet: If indicated, ensure proper application and duration limits are observed.
-
Specific Positions (Examples related to material access/placement):
- Supine: Common for anterior approaches to hip, knee, and distal extremities. Allows easy access to anterior iliac crest for bone graft harvest.
- Lateral Decubitus: Used for lateral hip approaches, shoulder arthroplasty, and some spinal procedures. Care must be taken to protect the axillary nerve and ensure stable positioning.
- Prone: Typically for posterior spinal approaches, posterior hip approaches, and posterior extremity surgery. Allows access to posterior iliac crest for bone graft. Requires careful abdominal support to minimize epidural venous bleeding.
- Beach Chair: For shoulder surgery. Requires careful head positioning to prevent cervical spine injury and cerebral hypoperfusion.
- Verification: Before draping, ensure the patient is safely and securely positioned, all pressure points are protected, and the planned surgical site (and donor site, if applicable) is adequately exposed. Confirm the ability to achieve necessary intra-operative maneuvers (e.g., knee flexion/extension, joint distraction).
Detailed Surgical Approach / Technique
While "reconstruction materials" span a vast array of orthopedic procedures, the core principles of meticulous surgical technique, respecting tissue planes, achieving anatomical reduction, and ensuring stable fixation are universally applicable. This section will focus on generalized principles rather than a specific procedure.
General Principles of Surgical Technique
-
Incision and Exposure:
- Planning: Incisions should be strategically placed to provide adequate exposure, respect skin tension lines, consider future surgical approaches, and minimize damage to critical neurovascular structures.
-
Dissection:
Employ sharp dissection to minimize tissue trauma. Identify and protect major nerves and vessels. Utilize internervous planes whenever possible to reduce muscle sacrifice and denervation.
-
Example Internervous Planes:
- Thigh (anterior): Between rectus femoris (femoral nerve) and vastus lateralis (femoral nerve) for femoral shaft exposure.
- Leg (anterior): Between tibialis anterior (deep peroneal nerve) and extensor digitorum longus (deep peroneal nerve) or between tibialis anterior and gastrocnemius/soleus (sural nerve for fibular harvest).
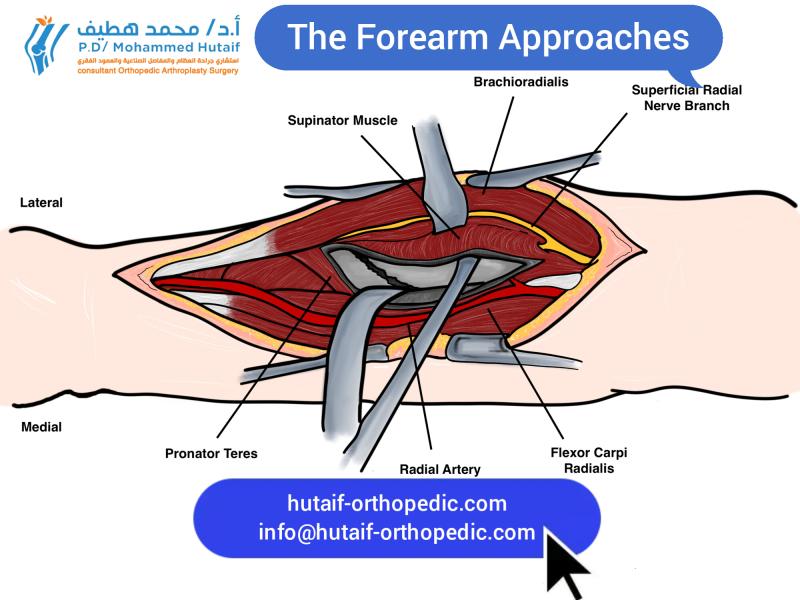
- Forearm (volar): Between flexor carpi radialis (median nerve) and palmaris longus (median nerve), or between flexor carpi ulnaris (ulnar nerve) and flexor digitorum superficialis (median nerve).
-
Example Internervous Planes:
- Soft Tissue Management: Handle soft tissues gently with retractors, avoid excessive cautery, and maintain tissue moisture to prevent desiccation.
-
Material Handling and Preparation:
-
Autografts:
- Harvesting: Meticulous technique is paramount to minimize donor site morbidity. For bone grafts (e.g., iliac crest), precise cortical windows, careful curettage, and potential use of bone graft substitutes for donor site closure. For soft tissue grafts (e.g., hamstring, patellar tendon), use appropriate strippers, harvest length and diameter carefully, and address any associated vessels/nerves.
- Preparation: Process the graft efficiently. For bone, morselize or shape as needed. For soft tissue, whipstitch ends, size for tunnels, or prepare for augmentation.
-
Allografts:
- Thawing: Follow tissue bank protocols strictly. Typically involves sterile saline baths at room temperature.
- Sizing and Shaping: Precisely size to the defect. Osteochondral allografts require specialized reamers and matching. Tendon allografts need to be prepared with appropriate tensioning and fixation points.
- Sterile Field: Maintain absolute sterility as these are foreign materials.
-
Synthetic Biomaterials / Prosthetic Implants:
- Packaging: Inspect packaging for integrity and expiration dates.
- Assembly: Assemble components as per manufacturer guidelines.
- Surface Handling: Avoid scratching or contaminating implant surfaces, particularly those intended for osseointegration. Use dedicated instruments.
-
Autografts:
-
Defect Preparation and Implant/Graft Placement:
- Debridement: Thoroughly debride all devitalized tissue, inflammatory pannus, and previous implant debris.
-
Recipient Bed Preparation:
- Bone: Decortication of cortical bone to expose cancellous bone, creating a bleeding bed for graft incorporation. Create stable recipient sites for press-fit components or graft seating.
- Soft Tissue: Freshen tendon/ligament stumps, prepare appropriate drill tunnels for graft passage.
- Reduction: Achieve anatomical or near-anatomical reduction of fractures, dislocations, or deformities. Use temporary fixation (K-wires, reduction clamps) to maintain reduction.
-
Graft/Implant Placement:
- Bone Graft: Pack graft material densely into defects, ensuring maximal contact with host bone. For structural grafts, achieve stable seating and compression.
- Soft Tissue Graft: Pass through tunnels, tension appropriately (often isometric positioning is key), and secure with interference screws, cortical buttons, or sutures.
-
Prosthetic Implants:
- Arthroplasty: Meticulous bone cuts, reaming, broaching, and trial implantation to ensure correct size, alignment, and stability. Cemented components require precise mixing and pressurization. Press-fit components require adequate bone preparation and impaction force.
- Internal Fixation: Apply plates, screws, nails according to AO principles – achieve anatomical reduction, provide stable internal fixation, preserve vascularity. Ensure proper screw length and trajectory.
-
Fixation:
- Primary Stability: The initial mechanical stability of the construct is paramount, especially for prosthetics and load-sharing grafts. This prevents micromotion, which can lead to non-union or implant loosening.
-
Material-Specific Fixation:
- Bone Grafts: Often augmented with internal fixation (plates, screws) to provide initial stability.
- Tendon/Ligament Grafts: Secured with various methods including interference screws, cortical suspensory devices, staples, or direct suture.
- Prosthetic Implants: Achieved through cementation, press-fit osseointegration, or screw fixation.
-
Closure:
- Irrigation: Thorough irrigation of the wound with saline to remove debris and reduce bacterial load.
- Hemostasis: Achieve meticulous hemostasis to prevent hematoma formation, which can compromise graft healing or increase infection risk.
- Drainage: Consider drains in cases of large dead space or anticipated significant bleeding, removing them when drainage is minimal.
- Layered Closure: Close deep fascial layers securely, followed by subcutaneous tissue and skin, respecting anatomical planes to minimize tension and promote healing.

Complications & Management
Complications associated with reconstruction materials are diverse, ranging from general surgical risks to material-specific issues, significantly impacting patient outcomes. Proactive identification and appropriate management are crucial.
General Complications
-
Infection:
- Incidence: Highly variable (0.5% to 5% for primary arthroplasty, higher for open fractures, revisions, or tumor surgery). Foreign bodies significantly increase susceptibility.
- Mechanism: Contamination during surgery, hematogenous spread, wound breakdown. Biofilm formation is a critical factor for implant-related infections.
-
Management:
- Acute (within 3-6 weeks): Irrigation and debridement (I&D) with component retention (DAIR – debridement, antibiotics, implant retention) if stable and patient is otherwise healthy; intravenous antibiotics.
- Chronic: Often requires single-stage or two-stage revision (explantation of all components, debridement, antibiotic spacer, reimplantation). Long-term suppressive antibiotics.
-
Hematoma/Seroma:
- Incidence: Common (up to 10-15%), particularly in areas with significant dead space.
- Mechanism: Inadequate hemostasis, lymphatic fluid accumulation. Can lead to increased pressure, wound dehiscence, and predisposition to infection.
- Management: Compression dressings, aspiration (sterile technique), wound drains, surgical evacuation if large or symptomatic.
-
Neurovascular Injury:
- Incidence: Rare but devastating (0.1% to 1%). Higher risk during complex dissections, revision surgery, or specific donor site harvests.
- Mechanism: Direct transection, traction injury, compression from hematoma, thermal injury.
- Management: Immediate surgical exploration and repair. Post-operative monitoring, nerve protection, and rehabilitation.
-
Wound Dehiscence/Skin Necrosis:
- Incidence: Variable, increased by poor soft tissue envelope, tension, infection, or comorbidities.
- Mechanism: Poor wound healing, infection, excessive tension on suture lines, hematoma.
- Management: Local wound care, débridement, negative pressure wound therapy, wound closure (primary or secondary intention), local or free flap coverage in severe cases.
Material-Specific Complications
-
Autografts (Bone and Soft Tissue):
-
Donor Site Morbidity:
- Incidence: Significant (up to 30-40% for iliac crest bone graft).
- Mechanism: Pain, infection, hematoma, nerve injury (e.g., lateral femoral cutaneous nerve), fracture (e.g., fibula, patella), cosmetic deformity.
- Management: Pain management, physical therapy, nerve blocks, surgical repair of fractures/hernias, careful harvest technique.
-
Graft Non-Union/Mal-union:
- Incidence: Varies by site and patient factors (5-15%).
- Mechanism: Inadequate vascularity, mechanical instability, infection, poor host bone quality.
- Management: Revision surgery with further grafting, biological augmentation (e.g., PRP, bone marrow aspirate), improved fixation.
-
Graft Resorption/Collapse:
- Incidence: Variable. More common with cancellous grafts under compressive load or in hostile environments.
- Mechanism: Excessive mechanical load, poor biological integration, infection.
- Management: Protective weight-bearing, revision surgery if symptomatic and persistent.
-
Donor Site Morbidity:
-
Allografts (Bone and Soft Tissue):
-
Disease Transmission:
- Incidence: Extremely low due to stringent donor screening and processing (<1:1,000,000).
- Mechanism: Inadequate donor screening, viral or bacterial contamination.
- Management: Prevention through strict tissue banking protocols.
-
Immunological Response/Rejection:
- Incidence: Subclinical responses are common; overt rejection leading to failure is rare, especially with processed grafts (low antigenicity).
- Mechanism: Host immune system recognition of allogeneic antigens, leading to inflammatory response and graft degradation.
- Management: Largely preventable by appropriate processing. If failure occurs, revision with autograft or prosthetic.
-
Graft Failure (Structural Collapse/Non-Incorporation):
- Incidence: Higher than autografts, especially in large structural allografts (up to 20-30% for massive allografts).
- Mechanism: Avascularity, slower incorporation, mechanical fatigue, infection.
- Management: Protected weight-bearing, biological augmentation, revision with autograft or prosthetic replacement.
-
Disease Transmission:
-
Synthetic Biomaterials / Prosthetic Implants (Metals, Ceramics, Polymers):
-
Aseptic Loosening:
- Incidence: A leading cause of long-term failure in arthroplasty (1-2% per year for certain components).
- Mechanism: Stress shielding, osteolysis from wear particles (polyethylene, cement), micro-motion at the implant-bone interface, biological reaction to debris.
- Management: Revision arthroplasty with or without bone grafting.
-
Wear and Osteolysis:
- Incidence: Variable depending on material couple and patient activity, decreasing with newer materials.
- Mechanism: Generation of microscopic particles from articulating surfaces (e.g., polyethylene, metal-on-metal), initiating a host inflammatory response leading to bone resorption.
- Management: Close monitoring, activity modification, revision surgery if symptomatic or progressive.
-
Implant Fracture/Fatigue Failure:
- Incidence: Rare for modern implants, but can occur with prolonged loading, malalignment, or material defects. Higher in non-union cases or with compromised bone.
- Mechanism: Exceeding the fatigue strength of the material, stress concentration.
- Management: Revision surgery with removal of fractured component and reimplantation, often with stronger or different fixation.
-
Corrosion/Metallosis:
- Incidence: Primarily with certain metal-on-metal implants or retained hardware.
- Mechanism: Electrochemical reaction between metal implants and body fluids, leading to metal ion release and local tissue reaction.
- Management: Monitoring metal ion levels, revision surgery with removal of offending implants and debridement.
-
Periprosthetic Fracture:
- Incidence: 1-5% in arthroplasty, often related to trauma, stress risers, or osteolysis.
- Mechanism: Trauma, bone loss, stress shielding, implant notching.
- Management: Depends on fracture location, stability, and implant loosening; can range from non-operative management to revision arthroplasty with extended fixation.
-
Allergic Reaction:
- Incidence: Rare (e.g., nickel allergy, up to 1% of population sensitive), often localized.
- Mechanism: Hypersensitivity reaction to implant constituents.
- Management: Implant removal and replacement with an inert alternative (e.g., titanium, ceramics).
-
Aseptic Loosening:
Table: Common Complications, Incidence, and Salvage Strategies
| Complication | Material Type Primarily Affected | Incidence (Approx.) | Salvage Strategy |
|---|---|---|---|
| Infection | All materials | 0.5-10% (site/case-dependent) | DAIR, single/two-stage revision, chronic suppressive antibiotics, implant removal, amputation (rare) |
| Donor Site Morbidity | Autografts | 10-40% (site-dependent) | Pain management, physical therapy, nerve blocks, surgical repair of fracture/hernia, prophylactic fixation |
| Allograft Structural Failure | Allografts (especially massive) | 5-30% | Protected weight-bearing, biological augmentation, revision with autograft/prosthetic replacement |
| Aseptic Loosening | Prosthetic Implants | 1-2% per year (THA/TKA) | Revision arthroplasty (single/two-stage), bone grafting, specialized long-stem implants |
| Wear/Osteolysis | Prosthetic Implants (Polyethylene) | 0.5-1% per year (variable) | Component exchange, revision arthroplasty, biological augmentation, material selection for newer bearings |
| Implant Fracture/Fatigue | Prosthetic Implants, Fixation | <1% (rare) | Revision surgery with removal of failed implant, reimplantation with stronger/different construct |
| Graft Non-Union/Mal-union | All grafts | 5-15% | Revision with biological augmentation (autograft, BMPs), improved fixation, physiotherapy, external fixation |
| Neurovascular Injury | All (iatrogenic) | <1% | Immediate surgical exploration and repair, nerve grafting, tendon transfers |
| Wound Complications | All (general surgical) | 5-15% | Local wound care, débridement, negative pressure wound therapy, flap coverage, antibiotics |
| Periprosthetic Fracture | Prosthetic Implants | 1-5% | ORIF, revision arthroplasty (often with extended fixation), non-operative management (rare) |
| Immunological Response | Allografts, some biomaterials | Rare (clinical) | Prevention via processing, revision with alternative material |
| Metallosis/Corrosion | Metal Implants | Rare-variable | Implant removal and replacement with non-metal components, debridement of affected tissue |
Post-Operative Rehabilitation Protocols
Post-operative rehabilitation is an integral component of successful orthopedic reconstruction, directly influencing the incorporation, remodeling, and functional outcome of the chosen materials while mitigating complications. Protocols must be tailored to the specific material, anatomical site, and patient factors.
General Principles
- Protection of Reconstruction: Initial phase focuses on protecting the surgical site from excessive stress, shear forces, or early weight-bearing that could compromise healing or fixation.
- Pain and Swelling Management: Aggressive pain control (multimodal analgesia) and edema reduction (RICE protocol, elevation, compression) facilitate early participation in therapy.
- Early Mobilization (Controlled): Gentle, controlled range of motion (ROM) exercises often begin early to prevent stiffness, promote cartilage health, and reduce adhesions, while respecting the limits of the reconstruction.
- Progressive Loading: A gradual increase in weight-bearing, resistance, and functional activities, guided by the biological healing timeline of the material and mechanical stability of the construct.
- Patient Education: Crucial for adherence to protocols, understanding precautions, and recognizing warning signs of complications.
Material-Specific Considerations
-
Autografts and Allografts (Bone):
-
Phase 1 (Protection & Early Motion, 0-6 weeks):
- Weight-Bearing: Often non-weight bearing or touch-down weight-bearing for lower extremity grafts, protected in a brace or cast. Upper extremity grafts may allow light functional use.
- Motion: Gentle, pain-free ROM within protected limits.
- Focus: Minimize swelling, pain control, maintain adjacent joint mobility.
-
Phase 2 (Progressive Loading & Strengthening, 6-12+ weeks):
- Weight-Bearing: Gradual progression to full weight-bearing as radiographic evidence of graft incorporation (callus formation, blurring of graft-host interface) and clinical stability improves.
- Motion: Increase active and passive ROM, gentle stretching.
- Focus: Initiate isometric and then isotonic strengthening exercises, core stability.
-
Phase 3 (Return to Function, 3-6+ months):
- Focus: Advanced strengthening, proprioceptive training, sport-specific drills. Return to unrestricted activity contingent on full graft incorporation and strength restoration (often 6-12 months or longer).
- Caution: Risk of re-fracture or collapse during remodeling phase.
-
Phase 1 (Protection & Early Motion, 0-6 weeks):
-
Autografts and Allografts (Soft Tissue - Tendon/Ligament):
-
Phase 1 (Protection & Immobilization, 0-2 weeks):
- Weight-Bearing: Dependent on injury site (e.g., NWB for Achilles repair, protected WB for ACL).
- Motion: Often restricted or protected in a brace/splint to prevent excessive stress on the graft.
- Focus: Wound healing, pain/swelling control, gentle muscle activation (isometric).
-
Phase 2 (Controlled Motion & Early Strengthening, 2-12 weeks):
- Weight-Bearing: Gradual increase.
- Motion: Progressive increase in ROM, often guided by therapist and surgeon.
- Focus: Restore full passive and active ROM, light resistance exercises, neuromuscular re-education.
-
Phase 3 (Progressive Strengthening & Functional Training, 3-6 months):
- Focus: Increase strength, endurance, power. Proprioception and balance training. Advanced functional drills.
- Caution: Graft remodeling and maturation continue for up to 1-2 years; avoid aggressive activities until adequate strength and stability are achieved.
-
Phase 1 (Protection & Immobilization, 0-2 weeks):
-
Prosthetic Implants (Arthroplasty):
-
Phase 1 (Immediate Post-Op & Early Mobilization, Day 1 - 2 weeks):
- Weight-Bearing: Often immediate full weight-bearing for cemented components, protected for press-fit (unless stable).
- Motion: Immediate active and passive ROM (e.g., CPM for knee, pendulum exercises for shoulder).
- Focus: Prevent DVT/PE, pain control, initiate gait training, achieve functional ROM, manage precautions (e.g., hip precautions for THA).
-
Phase 2 (Functional Restoration, 2-6 weeks):
- Focus: Improve strength, endurance, balance. Discontinue assistive devices as able. Address specific functional deficits.
-
Phase 3 (Return to Activity, 6+ weeks):
- Focus: Advanced strengthening, impact activities (low-impact only for joint replacements). Education on long-term implant care and activity modification.
-
Phase 1 (Immediate Post-Op & Early Mobilization, Day 1 - 2 weeks):
-
Internal Fixation (Plates, Screws, Nails):
-
Phase 1 (Immobilization & Protection):
- Weight-Bearing: Strict non-weight bearing or protected weight-bearing determined by fracture stability and bone quality.
- Motion: Immobilization of the fractured segment, active ROM of adjacent joints.
-
Phase 2 (Gradual Loading & Motion):
- Weight-Bearing: Progressive loading as radiographic healing advances.
- Motion: Gradual increase in ROM, often starting with passive, then active.
-
Phase 3 (Strength & Conditioning):
- Focus: Restore full strength and function. Implant removal considered once fracture is fully healed and asymptomatic, based on age, activity level, and implant location.
-
Phase 1 (Immobilization & Protection):
Monitoring and Adjustments
- Clinical Assessment: Regular evaluation for pain, swelling, wound healing, neurological status, and functional milestones.
- Radiographic Assessment: Serial radiographs to monitor graft incorporation, implant stability, and bone healing.
- Therapist Feedback: Close collaboration with physical and occupational therapists to adjust protocols based on individual patient progress and tolerance.
- Complication Vigilance: Monitor for signs of infection, DVT, neurological deficits, or implant failure and address promptly.
Summary of Key Literature / Guidelines
The landscape of reconstruction materials is continually evolving, driven by advancements in biomaterials science, surgical techniques, and a deepening understanding of biological healing processes. Evidence-based medicine forms the cornerstone of material selection and complication management.
Key Themes in the Literature
-
Autograft vs. Allograft Debate:
- Bone Grafts: Autogenous bone graft remains the "gold standard" due to its osteoinductive, osteoconductive, and osteogenic properties, making it superior for non-unions and small-to-moderate defects (Roberts et al., 2011). However, donor site morbidity (Goulet et al., 2007) limits its widespread use for large defects, where structural allografts are often necessary. The use of demineralized bone matrix (DBM) and bone morphogenetic proteins (BMPs) as allograft extenders or standalone options continues to be studied, with debates surrounding their efficacy and cost-effectiveness (Einhorn et al., 2015).
- Soft Tissue Grafts: For ACL reconstruction, patellar tendon and hamstring autografts remain the most common and robust choices, demonstrating superior long-term outcomes compared to allografts, especially in younger, active patients (Poehling et al., 2005). Allografts are often reserved for revision cases, multi-ligament injuries, or less active patients due to concerns regarding slower incorporation, higher rates of rupture, and the theoretical risk of disease transmission (Barber et al., 2018).
-
Advancements in Prosthetic Materials and Design:
- Arthroplasty: Ongoing research focuses on improving wear characteristics, reducing osteolysis, and enhancing osseointegration. Highly cross-linked polyethylene has significantly reduced wear rates in total hip arthroplasty (THA) compared to conventional polyethylene (D'Antonio et al., 2007). Ceramic-on-ceramic bearings offer excellent wear properties but carry a small risk of fracture and "squeaking" (Walter et al., 2012). Titanium alloys with porous or textured coatings promote osseointegration for cementless components, aiming for long-term stability and reduced aseptic loosening (Jasty et al., 1990).
- Fixation Hardware: Bioabsorbable implants (e.g., PLA, PGA) have emerged for certain applications (e.g., ankle fractures, ACL interference screws) to avoid stress shielding and the need for hardware removal (Böstman et al., 2007). However, concerns remain regarding inflammatory reactions and unpredictable degradation rates.
-
Infection Prevention and Management:
- Prophylaxis: Strict adherence to perioperative antibiotic protocols (e.g., first or second-generation cephalosporins), meticulous surgical technique, and laminar flow operating rooms remain crucial (Branche et al., 2013).
- Biofilm: The understanding of bacterial biofilm formation on implant surfaces has revolutionized treatment strategies. Two-stage revision arthroplasty for periprosthetic joint infection (PJI) is the standard for chronic infections (Parvizi et al., 2008), with increasing evidence for single-stage revisions in selected cases (Grammatopoulos et al., 2017). Antibiotic-loaded bone cement and coatings (e.g., silver) are adjunctive strategies.
-
Role of Biological Modifiers:
- Growth Factors and Stem Cells: The use of recombinant BMP-2 and BMP-7 for non-union treatment has demonstrated efficacy but also carries risks such as heterotopic ossification and soft tissue swelling (Govender et al., 2002). Autologous platelet-rich plasma (PRP) and bone marrow aspirate concentrate (BMAC) contain growth factors and progenitor cells, but their clinical efficacy in accelerating bone or soft tissue healing remains largely inconsistent across studies, requiring more robust research (Filardo et al., 2013).
Key Guidelines and Consensus Statements
- AAOS Clinical Practice Guidelines: The American Academy of Orthopaedic Surgeons (AAOS) regularly publishes evidence-based clinical practice guidelines on various topics, including the management of non-unions, periprosthetic joint infection, and arthroplasty techniques, often providing recommendations on material choices.
- International Consensus Group on Periprosthetic Joint Infection (ICM PJI): These consensus meetings provide comprehensive, evidence-based definitions, diagnostic criteria, and treatment algorithms for PJI, significantly influencing global practice (Parvizi et al., 2018).
- AO Foundation Principles: The AO (Arbeitsgemeinschaft für Osteosynthesefragen) principles of fracture management, emphasizing anatomical reduction, stable fixation, preservation of blood supply, and early functional mobilization, guide the use of internal fixation materials.
In conclusion, continuous engagement with the scientific literature, participation in academic discourse, and adherence to established guidelines are essential for orthopedic surgeons to navigate the complexities of reconstruction materials and their associated complications, ensuring the highest standard of patient care.
You Might Also Like