Advanced Orthopedic Guide: Tendon Anatomy, Biomechanics & Pathophysiology
Key Takeaway
Tendons transmit muscle forces, facilitating movement. Their unique strength derives from a hierarchical structure predominantly composed of Type I collagen (75% dry weight), water (50-60%), elastin, and proteoglycans like decorin. Tenocytes maintain this matrix, adapting the tendon to mechanical loads and resisting injuries.
Introduction & Epidemiology
Tendons, critical components of the musculoskeletal system, transmit forces from muscle to bone, facilitating joint movement and stability. Their unique hierarchical structure and viscoelastic properties enable them to withstand significant tensile loads while maintaining flexibility. Injuries to tendons, ranging from acute ruptures to chronic degenerative tendinopathies, represent a substantial burden on healthcare systems globally.
Epidemiologically, tendon injuries are prevalent across all age groups and activity levels. Acute ruptures, such as those of the Achilles, rotator cuff, and quadriceps/patellar tendons, often occur during sudden eccentric loading or forceful contractions, particularly in athletic populations. The incidence of Achilles tendon ruptures, for instance, is estimated to be 11-30 per 100,000 population, with a notable increase in middle-aged individuals engaging in recreational sports. Rotator cuff tears similarly affect a significant portion of the population, with prevalence increasing with age, reaching over 50% in those over 60 years.
Chronic tendinopathies, characterized by pain and dysfunction without overt inflammation, are even more common. Conditions like lateral epicondylitis, patellar tendinopathy (jumper's knee), and gluteal tendinopathy affect millions annually, often resulting from overuse, repetitive microtrauma, or systemic factors. These conditions frequently lead to prolonged disability, impacting quality of life and imposing significant socioeconomic costs through lost productivity and healthcare expenditures. Understanding the intricate anatomy, biomechanics, and pathophysiological responses of tendons is paramount for effective diagnosis, treatment, and rehabilitation strategies in orthopedic practice.
Surgical Anatomy & Biomechanics
The macroscopic and microscopic architecture of tendons is meticulously designed to withstand and transmit tensile forces. Tendons exhibit a hierarchical organization, beginning at the molecular level and progressing to the gross anatomical structure.
Structure and Composition
The unique mechanical properties of tendons are directly attributable to their intricate composition and structural arrangement.
- Water: Comprising approximately 50%–60% of the total tendon weight, water is crucial for maintaining viscoelasticity and facilitating nutrient transport.
-
Collagen:
This is the most abundant solid component, representing about 75% of the tendon's dry weight.
- Type I Collagen: Predominantly, 95% of collagen is type I, providing exceptional tensile strength. Its hierarchical arrangement is critical for tendon function. Collagen molecules self-assemble into microfibrils, which then form subfibrils, fibrils, and finally collagen fibers. These fibers are grouped into fascicles, which are further bundled to form the macroscopic tendon.
- Type III Collagen: Present in smaller amounts, typically 5%, but increases significantly in healing tendons and chronic tendinopathy, suggesting a role in early repair and disorganized matrix formation.
- Other minor collagens (e.g., types V, VI, XII) also exist, contributing to fibril diameter regulation and integration with the extracellular matrix (ECM).
-
Elastin:
Constituting 1%–2% of the dry weight, elastin is a highly elastic protein that allows the tendon to resume its original shape after stretching. Its presence is particularly relevant to the "toe region" of the stress-strain curve, where the initial, non-linear deformation of the tendon occurs as collagen crimp is straightened.


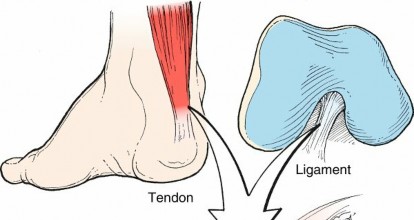
FIG. 1.44 Tendon and ligament architecture. From Brinker MR, Miller MD: Fundamentals of orthopaedics, Philadelphia, 1999, Saunders, p 15. -
Proteoglycans (PGs):
These macromolecules, consisting of a protein core with glycosaminoglycan chains, play vital roles in tendon hydration, fascicle gliding, and collagen fibrillogenesis.
- Decorin: The most predominant proteoglycan in tendons. It regulates tendon diameter by binding to collagen fibrils and provides cross-links between collagen fibers. Decorin has also been shown to possess antifibrotic properties through its inhibition of transforming growth factor-beta 1 (TGF-β1), which is implicated in scar formation.
- Aggrecan: Present at points of tendon compression, such as where tendons wrap around bony prominences, contributing to compressive resilience.
- Biglycan: Similar to decorin, biglycan also interacts with collagen fibrils and contributes to collagen organization.
- Other PGs like Lumican and Fibromodulin also play roles in collagen fibril assembly and matrix organization.
Cellular Components: Tenocytes and Tenoblasts
Tendons are sparsely cellular, primarily composed of specialized fibroblasts known as tenocytes.
-
Tenocytes:
These spindle-shaped cells are derived from the mesoderm.
- Function: Tenocytes are the primary architects and maintenance cells of the tendon. They synthesize the vast majority of the extracellular matrix components, including collagen, elastin, and proteoglycans. They also assemble early collagen fibrils and produce matrix-degrading enzymes (MMPs) and their inhibitors (TIMPs), which are critical for matrix remodeling and turnover.
- Mechanotransduction: Tenocytes are highly mechanosensitive. They detect strain during tendon loading through deflection of cell cilia and integrin-mediated signaling. This mechanotransensory function drives their metabolic activity, adapting the tendon to mechanical demands. Proper mechanical loading is essential for tenocyte health and function.
- Healing: Tenocyte proliferation and production of collagen are critical during tendon healing, directly influencing the strength and reducing the risk of repair ruptures.
- Tenoblasts: Immature tenocytes, more metabolically active and responsible for much of the ECM synthesis during development and repair.
Gross Anatomy and Connective Tissue Sheaths
Tendons are enveloped by various connective tissue sheaths that facilitate gliding and provide vascular supply:
*
Endotenon:
A thin layer of connective tissue that surrounds individual collagen fascicles, allowing them to glide past one another and housing neurovascular bundles.
*
Epitenon:
A connective tissue layer that surrounds the entire tendon, situated beneath the paratenon.
*
Paratenon:
A loose areolar connective tissue sheath surrounding tendons in areas without synovial sheaths (e.g., Achilles tendon). It reduces friction and provides a robust vascular supply.
*
Tenosynovium:
A double-layered synovial sheath found in areas of high friction (e.g., wrist, ankle, hand). The inner visceral layer adheres to the tendon, and the outer parietal layer lines the adjacent structures, with synovial fluid in between to lubricate gliding.
Vascularity and Innervation
Tendon vascularity is relatively sparse, particularly in mid-substance regions, which has implications for healing.
*
Blood Supply:
Primarily derived from three sources:
1. The musculotendinous junction (MTJ).
2. The osteotendinous junction (OTJ).
3. Vessels within the paratenon or mesotenon (a fold of synovial membrane that carries vessels to the tendon within a synovial sheath).
*
Avascular Zones:
Many tendons exhibit critical avascular zones (e.g., the mid-substance of the Achilles tendon, the "critical zone" of the supraspinatus tendon) that are more susceptible to degeneration and impaired healing.
*
Innervation:
Tendons receive both sensory and autonomic innervation. Sensory innervation is responsible for pain perception and proprioception, mediated by specialized mechanoreceptors:
*
Golgi Tendon Organs (GTOs):
Located at the MTJ, primarily monitor muscle tension.
*
Ruffini Endings and Pacinian Corpuscles:
Found in the paratenon and epitenon, providing information on stretch and pressure.
Biomechanics
Tendon biomechanics are critical to understanding injury mechanisms and developing effective repair strategies.
*
Stress-Strain Curve:
This fundamental curve illustrates the tendon's response to tensile loading.
*
Toe Region:
Initial non-linear deformation (up to ~2-3% strain) due to the straightening of collagen fibril crimp. Elastin also contributes to this region.
*
Linear Region:
As crimp is eliminated, collagen fibrils align and stretch, exhibiting a linear elastic response (up to ~4-8% strain).
*
Yield Point:
Beyond this point, microfailure of collagen fibrils begins.
*
Ultimate Tensile Strength (UTS):
The maximum stress a tendon can withstand before macroscopic failure.
*
Failure/Rupture:
Occurs when strain exceeds ~8-10%, leading to complete disruption of the tendon.
*
Viscoelasticity:
Tendons are viscoelastic, meaning their mechanical properties are time and rate-dependent.
*
Creep:
Tendons slowly deform over time under constant load.
*
Stress Relaxation:
Tendons exhibit a decrease in stress over time when held at a constant strain.
*
Hysteresis:
Energy is lost as heat during loading and unloading cycles.
*
Factors Affecting Mechanical Properties:
Age, chronic disease (e.g., diabetes), medications (e.g., corticosteroids, fluoroquinolones), and repetitive loading all influence tendon strength and elasticity.
Pathophysiology of Tendinopathy
Tendinopathy is now understood as a continuum of tendon pathology, not purely an inflammatory process. Cook and Purdam's continuum model describes three stages:
1.
Reactive Tendinopathy:
An acute, non-inflammatory proliferative response of tenocytes to sudden overload, characterized by increased proteoglycan and water content, leading to tendon swelling and thickening. This stage is potentially reversible.
2.
Tendon Dysrepair:
Progression from reactive tendinopathy, involving greater matrix disorganization, increased cellularity, and attempted but unsuccessful healing. Collagen separation and focal regions of neovascularization occur.
3.
Degenerative Tendinopathy:
Advanced pathology with extensive collagen disorganization, cell death (apoptosis), lipid deposition, calcification, and neo-innervation. This stage is largely irreversible and often associated with chronic pain and increased risk of rupture.
Histologically, degenerative tendinopathy is characterized by angiofibroblastic hyperplasia, tenocyte apoptosis, loss of collagen crimp, and matrix disorganization rather than classic inflammatory cell infiltration.

This illustration (if available in the provided image list) or a similar depiction of tendon histology showing collagen fibers and tenocytes would be relevant here.
Indications & Contraindications
The decision to pursue operative versus non-operative management for tendon injuries is multifaceted, weighing the acuity, extent of injury, patient factors, and functional demands.
General Principles
- Acute Tendon Ruptures: Complete disruptions often benefit from surgical repair, particularly in active individuals, to restore function and minimize re-rupture rates.
- Chronic Tendinopathies: The mainstay of treatment is typically non-operative, focusing on load management, eccentric exercise, and physical therapy. Surgical intervention is reserved for refractory cases with clear structural pathology.
- Avulsion Injuries: Tendon avulsions, where the tendon pulls off a piece of bone, almost always require surgical reattachment.
- Impingement Syndromes: Tendons susceptible to impingement (e.g., rotator cuff, peroneal tendons) may require surgical decompression or tenodesis/transfer in cases of persistent pain and dysfunction despite conservative measures.
Operative Indications
-
Acute Complete Ruptures:
- Achilles tendon (especially in active individuals).
- Rotator cuff tears (full-thickness, symptomatic, or traumatic in younger patients).
- Patellar and quadriceps tendon ruptures (typically requires emergent repair).
- Distal biceps tendon ruptures.
- Flexor and extensor tendon lacerations in the hand/forearm.
- Acute avulsion fractures (e.g., hamstrings, adductors, calcaneal tuberosity).
-
Chronic Tendinopathies Refractory to Non-Operative Treatment (typically 3-6 months):
- Chronic patellar tendinopathy with structural pathology (e.g., debridement, partial fasciculectomy).
- Chronic gluteal tendinopathy with tears.
- Recalcitrant lateral/medial epicondylitis with failed conservative care.
- Rotator cuff tendinopathy with partial tears causing persistent pain and dysfunction.
-
Tendon Instability/Subluxation:
- Recurrent peroneal tendon subluxation/dislocation.
- Recurrent patellar dislocation with associated soft tissue pathology.
-
Massive, Irreparable Tendon Tears:
- Rotator cuff tears requiring superior capsular reconstruction, tendon transfer (e.g., latissimus dorsi), or reverse total shoulder arthroplasty.
- Achilles tendon ruptures with large gaps requiring V-Y plasty or flexor hallucis longus (FHL) transfer.
Non-Operative Indications
-
Partial Tendon Tears:
- Most partial Achilles tendon tears.
- Small, asymptomatic rotator cuff tears.
- Many partial patellar or quadriceps tendon tears.
-
Early-Stage Tendinopathies:
- Acute reactive tendinopathy.
- Early tendon dysrepair (prior to significant degeneration).
-
Patients with High Surgical Risk:
- Significant medical comorbidities (e.g., uncontrolled diabetes, severe cardiovascular disease) precluding safe anesthesia and surgery.
-
Low Functional Demand:
- Sedentary individuals or elderly patients with acceptable function despite injury.
-
Non-compliance/Unrealistic Expectations:
- Patients unwilling or unable to adhere to rigorous post-operative rehabilitation protocols.
Contraindications
-
Absolute Contraindications:
- Active local or systemic infection.
- Unstable medical comorbidities.
- Severe, irreversible nerve injury that would preclude functional recovery even with tendon repair/reconstruction.
-
Relative Contraindications:
- Poor soft tissue envelope or compromised vascularity in the surgical field.
- Significant osteopenia or poor bone quality that may compromise anchor fixation.
- Smoking, obesity, or diabetes, which are known risk factors for impaired wound healing and increased complication rates. These may warrant aggressive pre-operative optimization.
- Unrealistic patient expectations regarding outcomes or recovery timelines.
Summary of Operative vs. Non-Operative Indications
| Condition | Operative Indications | Non-Operative Indications |
|---|---|---|
| Acute Complete Ruptures |
- Healthy, active individuals with acute full-thickness tears (Achilles, Rotator Cuff, Quadriceps/Patellar, Distal Biceps).
- Avulsion fractures requiring reattachment. - Lacerations of hand/foot tendons with functional deficit. |
- Elderly, sedentary patients with low functional demands (selected cases, e.g., Achilles).
- Significant medical comorbidities precluding surgery. - Patients unwilling to undergo surgery or prolonged rehabilitation. |
| Partial Tendon Tears |
- Symptomatic partial tears refractory to 3-6 months of conservative treatment.
- Large partial tears (>50% thickness) in active individuals. - Progressive symptoms or functional deficit despite non-operative management. - Tears causing mechanical impingement or instability. |
- Most small (<50% thickness) or asymptomatic partial tears.
- Early-stage symptomatic partial tears with appropriate response to conservative management (rest, activity modification, physical therapy, injections). |
| Chronic Tendinopathy |
- Documented structural pathology (e.g., significant degeneration, calcification, ossification) with persistent pain and functional limitation after 6 months of comprehensive non-operative treatment.
- Recalcitrant tendinopathy leading to chronic pain and disability affecting daily activities or athletic performance. - Tendon instability/dislocation. |
- First-line treatment for almost all tendinopathies.
- Reactive tendinopathy, early tendinosis. - Patients with adequate symptom control and functional status through conservative measures. - Patients with underlying systemic conditions managed medically (e.g., inflammatory arthropathies, though specific tendon issues may still require surgical intervention if severe). |
| Failed Previous Repair/Reconstruction |
- Symptomatic re-rupture after initial repair.
- Persistent instability or functional deficit after primary reconstruction. - Non-union or mal-union causing pain and dysfunction. |
- Asymptomatic re-rupture or functional tear in low-demand patients.
- Revision surgery often carries higher risks and may be less successful than primary repair. |
Pre-Operative Planning & Patient Positioning
Meticulous pre-operative planning is essential for optimizing outcomes and minimizing complications in tendon surgery.
Diagnostic Workup
- History and Physical Examination: A thorough history detailing the mechanism of injury, symptom duration, previous treatments, and functional limitations is crucial. Physical examination should precisely localize pain, assess range of motion, strength, stability, and integrity of the affected tendon.
-
Imaging Studies:
- Plain Radiographs: Primarily to rule out avulsion fractures, identify calcifications, or assess concurrent arthritic changes.
- Magnetic Resonance Imaging (MRI): Gold standard for delineating tendon pathology, including partial versus full-thickness tears, retraction, degeneration, and associated soft tissue or bone edema. Provides excellent soft tissue contrast.
- Ultrasound (US): A dynamic, cost-effective tool for assessing tendon tears, inflammation, and neovascularization. Operator-dependent but excellent for real-time assessment of tendon movement and guided injections.
- Computed Tomography (CT): Useful for assessing bony avulsions or complex fractures, particularly when planning fixation strategies.
- Pre-operative Planning: Based on imaging and clinical assessment, the surgeon determines the exact nature of the injury, feasibility of direct repair, need for augmentation (graft type: autograft vs. allograft), choice of implants (suture anchors, interference screws, transosseous tunnels), and surgical approach. Graft harvest sites are identified if autograft is planned.
Patient Preparation and Anesthesia
- Medical Optimization: Comprehensive medical evaluation to assess fitness for surgery and anesthesia. Optimization of comorbidities (e.g., diabetes control, smoking cessation).
- Anesthesia: General anesthesia is common, often supplemented with regional nerve blocks (e.g., interscalene for shoulder, popliteal for Achilles) for post-operative pain control, which can significantly reduce opioid consumption and enhance early rehabilitation.
-
Prophylaxis:
- Antibiotics: Pre-operative intravenous antibiotics, typically a first or second-generation cephalosporin, administered within one hour of incision.
- DVT Prophylaxis: Depending on patient risk factors and surgical site (e.g., lower extremity surgery often warrants chemical or mechanical prophylaxis).
Patient Positioning
Correct patient positioning is critical for optimal surgical exposure, minimizing nerve compression, and ensuring patient safety.
*
Beach Chair Position:
Commonly used for shoulder surgeries (e.g., rotator cuff repair, biceps tenodesis). Allows excellent visualization of the glenohumeral joint and subacromial space. Requires careful attention to head support, blood pressure monitoring (risk of hypotension), and proper padding to prevent nerve compression (brachial plexus, ulnar nerve).
*
Lateral Decubitus Position:
Alternative for shoulder surgery, particularly for arthroscopic cases. Provides gravity-assisted distraction of the glenohumeral joint. Requires careful padding of bony prominences and axilla.
*
Prone Position:
Standard for Achilles tendon repair, posterior ankle, and some hamstring avulsion repairs. Ensures clear posterior exposure. Requires careful padding of the chest, pelvis, and lower extremities, with particular attention to peroneal nerve at the fibular head and common femoral nerve.
*
Supine Position:
Used for patellar tendon, quadriceps tendon, distal biceps repair, and most hand/wrist tendon procedures. Allows for easy access to the anterior aspect of the limb. Limb can be draped free or placed on a leg holder/hand table.
*
Tourniquet Use:
Frequently employed for lower and upper extremity tendon surgeries to provide a bloodless field, typically inflated 250-300 mmHg for upper extremity and 300-350 mmHg for lower extremity. Inflation time should be monitored, with a general limit of 90-120 minutes.
*
Imaging during surgery:
Fluoroscopy may be necessary for precise placement of suture anchors in bone or to confirm reduction of bony avulsions.
Detailed Surgical Approach / Technique
Surgical techniques for tendon repair and reconstruction vary significantly based on the specific tendon, nature of the injury (acute vs. chronic, partial vs. complete), and patient factors. However, several fundamental principles underpin all successful interventions.
General Principles of Tendon Surgery
- Debridement: Removal of all non-viable, degenerative, or chronically inflamed tissue. In tendinopathy, this involves excising areas of angiofibroblastic hyperplasia and disorganized matrix. In ruptures, frayed tendon ends are debrided to healthy tissue to promote robust healing.
- Mobilization: The tendon ends must be mobilized adequately to achieve a tension-free repair. This may involve releasing adhesions, performing interval slides, or, in chronic retracted tears, utilizing techniques like V-Y lengthening plasty (e.g., Achilles) or margin convergence (e.g., rotator cuff).
-
Repair Techniques:
-
Direct Repair:
For acute ruptures, direct end-to-end repair is often preferred. Strong suture techniques like the modified Kessler, Krackow, or modified Bunnell stitches maximize suture-tendon purchase and provide robust strength.

This image, if an illustration of suture techniques, would be relevant here. - Tendon-to-Bone Fixation: For avulsion injuries or tears off bony insertions, suture anchors (bioabsorbable or titanium), transosseous tunnels, or interference screws are utilized. The goal is to re-establish the tendon's footprint on bone.
- Load Sharing: Repairs should aim to restore the normal mechanical load distribution across the tendon and its insertion.
-
Direct Repair:
For acute ruptures, direct end-to-end repair is often preferred. Strong suture techniques like the modified Kessler, Krackow, or modified Bunnell stitches maximize suture-tendon purchase and provide robust strength.
-
Augmentation/Reconstruction:
When direct repair is not feasible due to large gaps, poor tissue quality, or chronic retraction, augmentation or reconstruction with autograft or allograft is indicated.
- Autografts: Common choices include hamstring tendons (semitendinosus, gracilis), quadriceps tendon, or patellar tendon. Advantages include biological integration and no risk of disease transmission. Disadvantages include donor site morbidity.
- Allografts: Achilles tendon, tibialis anterior, or semitendinosus allografts can be used. Advantages include no donor site morbidity and larger graft availability. Disadvantages include potential for immunological reaction and slower incorporation.
- Synthetic Augmentation: While less common for primary repair due to concerns of mechanical failure and foreign body reaction, synthetic materials are occasionally used as scaffolds or to reinforce repairs in selected cases.
- Biological Augmentation: The role of biologics (e.g., PRP, bone marrow aspirate concentrate (BMAC), stem cells) in tendon healing is an area of ongoing research. While promising, current evidence for routine use in all tendon repairs is mixed and still evolving. They are typically used in conjunction with structural repair.
Example Surgical Approach: Arthroscopic Rotator Cuff Repair (Supraspinatus)
This approach highlights principles applicable to many tendon repairs.
- Patient Positioning: Lateral decubitus position is common, though beach chair can also be used. The arm is placed in a traction device with 10-15 lbs of weight, abducted 30 degrees, and flexed forward 15 degrees.
-
Portal Placement:
- Posterior Portal: Primary viewing portal, 2 cm inferior and 1 cm medial to the posterior-lateral corner of the acromion.
- Lateral Working Portal: 3-4 cm distal to the lateral acromial edge, providing optimal angle for suture anchor insertion and knot tying.
- Anterior Portal (Optional): For anterior labral work, biceps assessment, or additional working access.
- Diagnostic Arthroscopy: Initial assessment of the glenohumeral joint, biceps tendon (consider tenodesis/tenotomy if pathology present), labrum, and articular cartilage.
-
Bursoscopy and Subacromial Decompression:
- Transition to the subacromial space. Extensive bursectomy is performed to visualize the rotator cuff tear and the undersurface of the acromion.
- Acromioplasty (subacromial decompression) is performed if significant acromial spurring or impingement is present, using a burr to resect the anterior-inferior acromial spur and create a flat undersurface.
-
Tendon Mobilization and Preparation:
- The torn rotator cuff edges are identified. A shaver and radiofrequency ablator are used to debride non-viable tissue from the tendon edges and the bony footprint on the greater tuberosity.
- Tendon mobilization is critical. Release of adhesions from the undersurface of the deltoid and rotator interval may be necessary. Margin convergence sutures can be used for larger U-shaped tears to reduce the size of the defect before reattachment to bone.
- Bone Bed Preparation: The bony footprint on the greater tuberosity is prepared by decortication using a burr to create a bleeding, cancellous bone bed, optimizing biological healing potential.
-
Suture Anchor Insertion and Repair:
- Single-Row Repair: Suture anchors are placed along the articular margin of the greater tuberosity. Sutures are then passed through the torn tendon edge using suture passers or specialized grasping instruments and tied.
- Double-Row Repair (Suture Bridge): Considered biomechanically superior for larger tears. Medial row anchors are placed at the articular margin. Lateral row anchors are placed more laterally on the greater tuberosity, with sutures from the medial row anchors passed through the tendon and then under the lateral row anchors to create a broad compression repair (suture bridge). This construct provides a larger footprint and increased contact pressure, theoretically enhancing healing. Knotless anchors are often preferred in this setup for ease and efficiency.
- Tension Management: The repair must be tension-free to promote healing and reduce re-rupture risk. Excessive tension can lead to cutout and failure.
- Closure: Portals are closed with sterile strips or sutures. Sterile dressing applied.

This image, if an illustration of suture anchors or a surgical field, would be relevant here.
Example Surgical Approach: Open Achilles Tendon Repair
- Patient Positioning: Prone position with the foot dorsiflexed to 90 degrees and supported. A thigh tourniquet is applied.
- Incision: A posteromedial or posterolateral longitudinal incision (typically 10-15 cm) is preferred over a midline incision to avoid pressure points and minimize wound healing complications. Alternatively, a transverse incision can be used for smaller tears. For percutaneous repair, multiple small incisions are made.
- Dissection: Careful dissection through the subcutaneous tissue, identifying and protecting the sural nerve (which typically runs posterolaterally). The paratenon is incised longitudinally.
- Hematoma Evacuation and Debridement: The ruptured tendon ends are identified. Hematoma is evacuated, and any frayed, necrotic, or degenerative tissue is sharply debrided back to healthy, bleeding tendon.
-
Repair:
- For acute ruptures, a strong core suture technique (e.g., Krackow or modified Kessler stitch) is passed through both the proximal and distal tendon ends. Typically, two to four locking stitches are placed on each side.
- The foot is brought into plantarflexion to relax the gastrocnemius-soleus complex, allowing the tendon ends to be approximated without excessive tension. The sutures are then tied, creating a strong repair. The goal is to achieve tension-free apposition with the ankle in approximately 20-30 degrees of plantarflexion.
- For chronic ruptures or cases with significant gaps, augmentation may be necessary. This can involve a V-Y plasty of the proximal gastrocnemius aponeurosis, local fascial flaps, or tendon transfers such as the flexor hallucis longus (FHL) tendon. The FHL transfer involves harvesting the FHL tendon proximally and routing it into the calcaneus to augment the repair.
- Paratenon Closure: The paratenon is meticulously repaired over the tendon to promote gliding and vascularity.
- Wound Closure: Subcutaneous tissues and skin are closed in layers. A bulky dressing and a posterior splint or cast are applied, maintaining the ankle in plantarflexion.
Complications & Management
Tendon surgery, like any surgical intervention, carries inherent risks and potential complications. Awareness of these, along with strategies for prevention and management, is crucial.
General Surgical Complications
- Infection: Superficial or deep surgical site infections. Incidence varies by site and patient factors (e.g., 0.5-5%). Management involves IV antibiotics, irrigation and debridement, and potentially hardware removal.
- Deep Vein Thrombosis (DVT) / Pulmonary Embolism (PE): Risk is higher with lower extremity immobilization. Incidence for symptomatic DVT is 0.5-2%. Prophylaxis (chemical/mechanical) and early mobilization are key.
- Nerve Injury: Temporary neuropraxia or permanent nerve damage (e.g., axillary nerve in shoulder, sural nerve in Achilles, radial nerve in elbow). Incidence is typically <1%. Prevention includes careful dissection and identification of neurovascular structures. Management ranges from observation to neurolysis or repair.
- Vascular Injury: Rare but serious. Careful dissection and anatomical knowledge are paramount.
- Pain: Post-operative pain is expected; persistent or severe pain may indicate complications.
- Stiffness/Arthrofibrosis: Common after shoulder or elbow surgery, particularly if rehabilitation is delayed or inadequate. Early controlled motion and aggressive physical therapy are preventive.
- Anesthetic Complications: Related to general or regional anesthesia.
Tendon-Specific Complications
| Complication | Incidence | Salvage Strategies |
|---|---|---|
| Re-rupture / Repair Failure | Variable; 2-15% (e.g., Rotator Cuff), 3-10% (Achilles) |
- Non-operative management for asymptomatic tears or low-demand patients.
- Revision surgery: Direct repair with augmentation (allograft, autograft), tendon transfer (e.g., FHL for Achilles, Latissimus Dorsi for rotator cuff), or reverse shoulder arthroplasty for massive irreparable cuff tears. |
| Tendon Non-Healing / Non-Union | Less common for acute repairs; higher in chronic tears with poor tissue quality |
- Biologic augmentation (e.g., PRP, BMAC) if viable tissue.
- Revision surgery with debridement, augmentation, or reconstruction. - Immobilization extension. |
| Adhesions / Scarring | Variable, especially in hand flexor tendons |
- Early controlled motion protocols.
- Tenolysis (surgical release of adhesions). - Physical therapy with manual mobilization and soft tissue techniques. |
| Graft Failure / Rejection | Low, but can occur with allografts or autografts |
- Revision surgery with new graft.
- Consideration of alternative reconstruction strategies. - Immunosuppression (for rejection, rare in allografts but considered for extreme cases). |
| Suture Anchor Pullout / Failure | <5% |
- Revision surgery with new anchors in different locations or larger/stronger anchors.
- Use of transosseous sutures or alternative fixation methods. - Optimization of bone quality pre-op. |
| Persistent Tendinopathy | Variable, particularly after debridement for chronic cases |
- Continued physical therapy, eccentric loading.
- Injection therapies (e.g., corticosteroids (caution with tendon), PRP, dry needling). - Re-evaluation for alternative diagnoses or missed structural pathology. - Rarely, repeat debridement or surgical augmentation. |
| Complex Regional Pain Syndrome (CRPS) | <1% |
- Early recognition and referral to pain specialist.
- Multimodal pain management (medications, nerve blocks, physical therapy). |
| Calcification of Tendon/Soft Tissue | Variable, common after patellar/Achilles repair |
- Non-operative management (stretching, anti-inflammatories).
- Rarely, surgical excision if symptomatic and impinging. |
| Sural Nerve Neuroma (Achilles Repair) | Up to 10% in open Achilles repair |
- Non-operative management (padding, local injections).
- Surgical excision of neuroma with transposition or nerve graft if persistent and debilitating. |
Post-Operative Rehabilitation Protocols
Post-operative rehabilitation is a cornerstone of successful tendon surgery, critical for optimizing healing, restoring function, and preventing re-injury. Protocols are typically structured into phased approaches, guided by the biological healing timeline and specific tendon biomechanics.
Key Principles
- Protection: Initial phase focuses on protecting the repair from excessive stress that could lead to failure. This often involves immobilization (casts, boots, slings) or controlled range of motion (ROM) bracing.
- Progressive Loading: Tendons respond to mechanical load through mechanotransduction. Gradual, controlled loading is essential to stimulate collagen synthesis, improve collagen organization, and enhance the tendon's tensile strength.
-
Biological Healing Timelines:
Collagen maturation is a slow process, taking weeks to months to regain significant strength. Rehabilitation must respect these biological realities, avoiding premature aggressive loading.
- Inflammatory phase: Days to weeks.
- Proliferative (repair) phase: Weeks to months.
- Remodeling phase: Months to years.
- Patient Adherence: Patient education and compliance with the rehabilitation program are paramount. Non-adherence significantly increases the risk of complications.
- Multidisciplinary Approach: Collaboration between the surgeon, physical therapist, and patient is essential for individualized care and goal setting.
Phased Approach (General Framework)
Phase I: Maximal Protection / Immobilization (Typically 0-6 weeks post-op)
- Goals: Protect surgical repair, minimize pain and inflammation, prevent adhesions (where appropriate).
-
Interventions:
- Immobilization: Splint, cast, or brace, often in a position that reduces tension on the repair (e.g., plantarflexion for Achilles, internal rotation for rotator cuff).
- Early Controlled Motion: For certain repairs (e.g., some rotator cuff repairs, flexor tendon repairs in the hand), carefully prescribed passive or active-assistive ROM within protected ranges may begin early to prevent stiffness and facilitate gliding, under strict supervision.
- Pain & Edema Management: Ice, elevation, NSAIDs (with caution regarding their effect on healing), appropriate analgesics.
- Gentle Isometric Exercises: Often initiated later in this phase, without resistance, to activate muscles without stressing the repair.
- Progression Criteria: Reduced pain, minimal swelling, adequate wound healing.
Phase II: Controlled Motion / Early Strengthening (Typically 6-12 weeks post-op)
- Goals: Gradually increase ROM, initiate gentle strengthening, improve neuromuscular control.
-
Interventions:
- Gradual Weaning from Immobilization: Transition to a removable brace or sling.
- Active and Active-Assistive ROM: Progressively increase range of motion, ensuring no pain or undue stress on the repair.
- Gentle Strengthening: Low-load isometric exercises, progressing to concentric exercises with light resistance (e.g., elastic bands, light weights).
- Proprioception and Balance: Exercises to improve joint position sense and stability.
- Progression Criteria: Full passive ROM, minimal pain with active movement, no signs of repair instability.
Phase III: Progressive Strengthening / Functional Training (Typically 12-24 weeks post-op)
- Goals: Maximize strength, power, endurance, and functional capacity; prepare for return to activity.
-
Interventions:
- Advanced Strengthening: Progress to higher resistance concentric and eccentric exercises. Eccentric loading is particularly important for tendon remodeling and strength.
- Plyometric Exercises: (If appropriate for the tendon and patient goals) to improve power and dynamic control.
- Sport-Specific Drills: Introduction of activities mimicking desired functional or athletic movements, with gradual increase in intensity and complexity.
- Cardiovascular Conditioning: Maintain overall fitness.
- Progression Criteria: Symmetrical strength and ROM compared to the contralateral side, ability to perform functional tasks without pain, confidence in movement.
Phase IV: Return to Activity / Sport (Typically 6-12+ months post-op)
- Goals: Gradual, safe return to full activity or sport; injury prevention.
-
Interventions:
- Gradual Integration: Phased return to sport or occupational activities, beginning with low intensity and gradually increasing volume and intensity.
- Advanced Functional Testing: Sport-specific performance testing to ensure readiness.
- Maintenance Program: Long-term exercise program focusing on strength, flexibility, and proprioception to prevent recurrence.
- Progression Criteria: Full recovery of strength, power, and endurance; successful completion of sport-specific testing; no pain or apprehension with activity.
Variations by Tendon
- Achilles Tendon: Early plantarflexion splinting, progressing to controlled ankle motion boots. Early weight-bearing (often protected) is becoming more common. Eccentric calf strengthening is critical in later phases.
- Rotator Cuff: Initial sling immobilization (4-6 weeks) with restricted external rotation. Passive ROM progresses to active-assisted, then active ROM. Emphasis on scapular stabilization and deltoid strengthening.
- Patellar/Quadriceps Tendon: Initial knee immobilization. Gradual ROM progression, avoiding excessive knee flexion initially. Quadriceps strengthening is paramount, with a focus on eccentric loading.
- Hand/Wrist Flexor Tendons: Very early controlled active motion protocols (e.g., modified Kleinert or Duran) are crucial to prevent adhesions and improve gliding, often initiated within days of surgery, requiring diligent hand therapy.
Summary of Key Literature / Guidelines
The field of tendon surgery is continuously evolving, driven by advancements in understanding tendon biology, biomechanics, and surgical techniques. Evidence-based medicine guides current practice, with ongoing research addressing controversies and exploring novel strategies.
Evidence-Based Practice
- Rotator Cuff Repair: Numerous systematic reviews and meta-analyses support arthroscopic repair for symptomatic, full-thickness tears to reduce pain and improve function. While double-row repairs have demonstrated superior biomechanical properties in cadaveric studies and some clinical trials suggest lower re-tear rates for larger tears, long-term functional superiority over well-performed single-row repairs for all tears remains debated. The role of early versus delayed mobilization post-rotator cuff repair is also a contentious area, with many advocating for early controlled motion to prevent stiffness without compromising healing.
- Achilles Tendon Rupture: A robust body of literature supports surgical repair over non-operative management for acute complete Achilles ruptures in active individuals to reduce the risk of re-rupture. However, recent trends show growing success with functional bracing and accelerated rehabilitation protocols, even for non-operative cases. Percutaneous repair techniques offer reduced wound complication rates but may have a higher re-rupture rate in some series compared to open repair. The use of early functional rehabilitation (e.g., immediate weight-bearing in a controlled boot) after repair has been shown to be safe and may accelerate recovery without increasing re-rupture risk.
- Chronic Tendinopathy: For recalcitrant tendinopathies, surgical debridement (e.g., for lateral epicondylitis or patellar tendinopathy) is typically reserved for cases failing extensive non-operative management (often >6 months). The evidence for specific surgical techniques (e.g., release of common extensor origin, shaving of patellar tendon lesions) shows modest efficacy. The role of biologics such as Platelet-Rich Plasma (PRP) and stem cell therapies remains controversial. While some studies show promising results, particularly for patellar and Achilles tendinopathy, others demonstrate no significant advantage over placebo or standard care. High-quality, large-scale randomized controlled trials are still needed to define their precise indications and efficacy.
Controversies and Emerging Concepts
- Arthroscopic vs. Open Approaches: The debate persists for many tendon pathologies. While arthroscopic techniques offer advantages of smaller incisions, less soft tissue disruption, and potentially faster initial recovery, open techniques may provide more direct visualization and robust repairs in complex cases. Surgeon experience and the specific pathology often dictate the choice.
- Biologics in Tendon Healing: The enthusiasm for PRP, BMAC, and mesenchymal stem cells (MSCs) is high, driven by their potential to enhance the biological healing environment. However, inconsistent results in clinical trials often stem from variations in preparation protocols, cell concentrations, and patient selection. Standardized protocols and clearer indications are still under investigation.
- Optimal Rehabilitation Protocols: The balance between protecting the repair and promoting early motion/loading is a continuous challenge. Accelerated rehabilitation protocols are increasingly favored but require careful patient selection and close supervision.
- Tendon Augmentation: The use of synthetic scaffolds or acellular dermal matrices for augmenting repairs (e.g., massive rotator cuff tears, chronic Achilles gaps) is gaining traction. While they can provide structural support, their long-term integration and mechanical properties require further evaluation.
- Understanding Mechanotransduction: Deeper insights into how tenocytes respond to mechanical stimuli are paving the way for targeted interventions, potentially influencing rehabilitation strategies and developing pharmacological agents that modulate tendon healing.
Guidelines
Professional organizations such as the American Academy of Orthopaedic Surgeons (AAOS) and the American Orthopaedic Society for Sports Medicine (AOSSM) provide clinical practice guidelines (CPGs) based on systematic reviews of the literature. These guidelines offer evidence-based recommendations on diagnosis, non-operative management, and surgical indications for common tendon injuries (e.g., rotator cuff tears, Achilles tendon ruptures), serving as valuable resources for orthopedic surgeons in decision-making. Continuous engagement with the scientific literature and participation in ongoing research are essential for staying abreast of the evolving landscape of tendon management.
Clinical & Radiographic Imaging


You Might Also Like