Orthopedic Fractures: Pathology, Healing, Anatomy & Biomechanics
Key Takeaway
Orthopedic fracture pathology involves the disruption of bone integrity, classified as closed (skin intact) or open (skin breached). Healing is a complex biological process influenced by bone vascularity and biomechanics, aiming to restore alignment and stability. Understanding fracture patterns, bone structure, and epidemiological factors is crucial for effective diagnosis and treatment strategies in orthopedics.
Understanding Fracture Pathology and Healing: The Science of Repair
Introduction & Epidemiology
Fractures represent a fundamental disruption in musculoskeletal integrity, defined by a loss of continuity in the substance of a bone. This spectrum encompasses a vast range of injuries, from insidious microfractures to devastating multifragmentary or comminuted disruptions. Despite the varied presentation, the core pathological event remains consistent: the cessation of structural integrity within the osseous framework. The term "fracture" is medically equivalent to "break," though lay perception often imbues the former with greater severity.
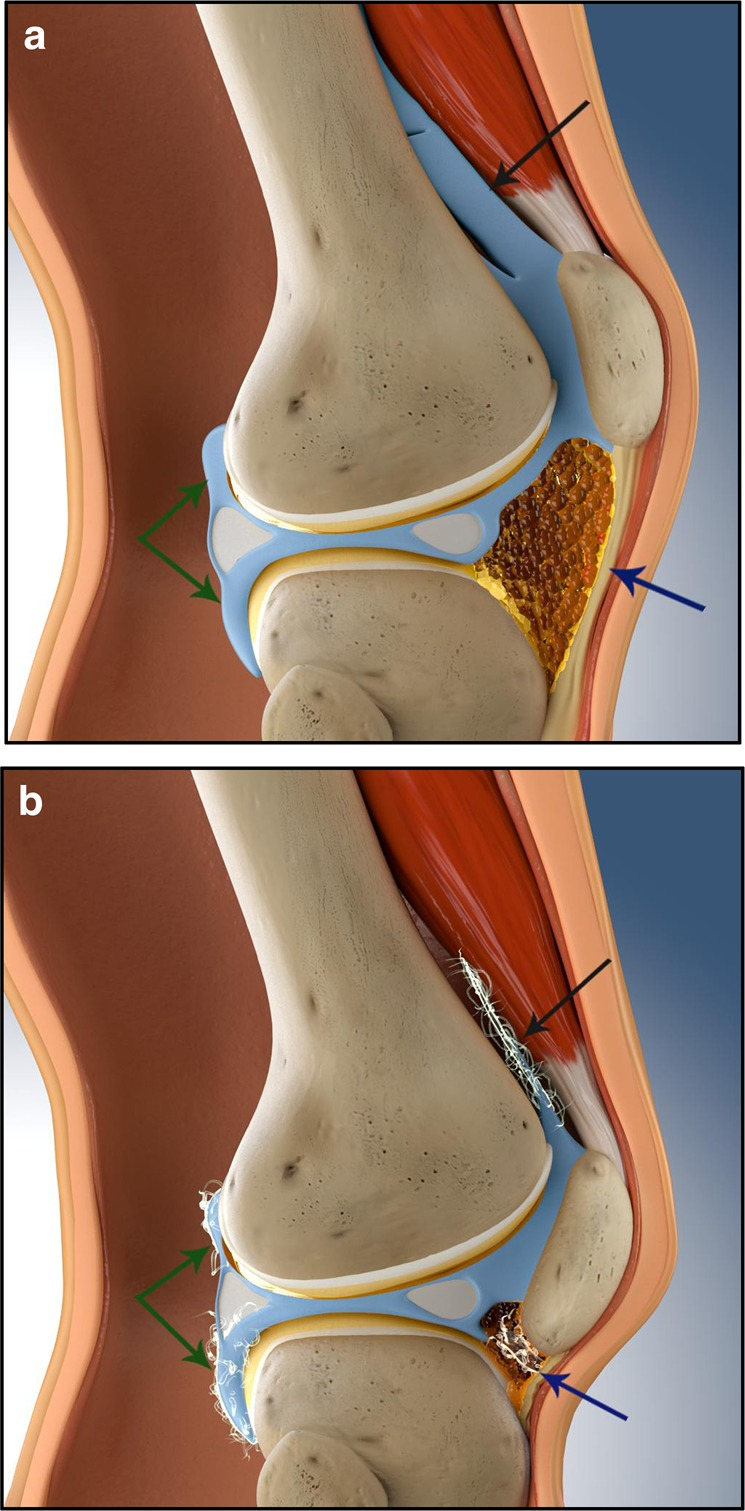
The classification of fractures is crucial for prognostic assessment and treatment planning. A primary distinction is made between closed and open fractures. In a closed fracture , the overlying skin and soft tissues remain intact, preserving the sterile environment around the bone and minimizing exogenous contamination. Conversely, an open fracture involves a wound communicating with the fracture site, establishing a direct conduit for external pathogens to colonize the compromised bone and soft tissue. This significantly elevates the risk of infection and osteomyelitis, complicating management and often leading to poorer outcomes. Furthermore, open fractures can present with substantial external hemorrhage. The historical nomenclature of "compound" for open and "simple" for closed fractures is largely obsolescent, with the latter term being particularly prone to misinterpretation regarding injury complexity.
The global epidemiology of fractures is substantial, with millions occurring annually. Factors influencing incidence include age, activity level, bone mineral density, and comorbidities. Pediatric fractures often involve physeal plates, requiring specific management to prevent growth disturbances. In adults, high-energy trauma predominates in younger populations, while low-energy fragility fractures secondary to osteoporosis are increasingly prevalent in the elderly, posing significant public health challenges due to associated morbidity, mortality, and healthcare burden. Understanding the fundamental science of fracture repair is paramount for orthopaedic surgeons, guiding both non-operative and operative strategies towards the primary goal of restoring anatomical alignment, biomechanical stability, and ultimately, functional recovery.
Surgical Anatomy & Biomechanics
A thorough understanding of surgical anatomy and biomechanics is foundational to effective fracture management. Bone, a dynamic and highly specialized connective tissue, possesses remarkable properties of strength and elasticity, which are dictated by its hierarchical structure.
Bone Structure and Vascularity
Bone tissue comprises two main types: cortical (compact) bone and cancellous (trabecular) bone.
*
Cortical bone
forms the dense outer shell of long bones, providing exceptional rigidity and resistance to bending and torsional forces. Its microstructure consists of Haversian systems (osteons) with concentric lamellae.
*
Cancellous bone
is found primarily at the metaphyses and epiphyses of long bones and within flat bones. Its porous, trabecular network provides internal structural support and dissipates compressive forces, while also housing hematopoietic marrow. The relative proportions of cortical and cancellous bone vary significantly by skeletal location, influencing fracture patterns and fixation strategies.
Bone's vascularity is critical for its viability and healing capacity. The blood supply to long bones is primarily derived from:
*
Nutrient arteries:
Penetrate the cortical diaphysis, branching into ascending and descending medullary arteries which supply the inner two-thirds of the cortex and the bone marrow.
*
Periosteal vessels:
Supply the outer one-third of the cortical bone and contribute significantly to early fracture hematoma formation and callus development, particularly in areas of extensive soft tissue stripping.
*
Metaphyseal and epiphyseal arteries:
Supply the ends of the bone, forming rich anastomoses crucial for joint health.
Disruption of this vascular network during trauma or surgical exposure directly impacts bone healing. Excessive soft tissue stripping, particularly of the periosteum, can devitalize bone fragments and impair healing, necessitating techniques that prioritize biological preservation.
Biomechanical Principles and Fracture Patterns
Fractures occur when applied stress exceeds the ultimate tensile, compressive, or shear strength of the bone. The specific loading conditions dictate the fracture morphology.
*
Tension:
Pulling forces lead to transverse fractures, particularly in cancellous bone or avulsion injuries.
*
Compression:
Crushing forces result in oblique or comminuted fractures, often impacting cancellous bone or articular surfaces.
*
Bending:
A combination of tensile and compressive forces, leading to a transverse fracture on the tension side and an oblique or butterfly fragment on the compression side.
*
Torsion:
Twisting forces produce spiral fractures, characteristic of long bones.
*
Shear:
Forces acting parallel to the bone surface, causing oblique fractures.
Understanding these load patterns aids in predicting fracture configurations and selecting appropriate implants. For instance, a long spiral fracture is amenable to intramedullary nailing, while a highly comminuted diaphyseal fracture might require bridge plating to maintain length and alignment without direct fragment reduction.
Muscle attachments and fascial envelopes also play a crucial role in fracture displacement. Specific muscle groups exert predictable deforming forces on fracture fragments, influencing the initial displacement and posing challenges to reduction. For example, in a midshaft femur fracture, the adductors tend to pull the distal fragment medially, while the iliopsoas can flex and externally rotate the proximal fragment. Recognizing these forces is integral to both closed reduction maneuvers and open surgical approaches.
The concept of stress risers is also important. Pre-existing conditions such as osteoporotic bone, metastatic lesions, or prior hardware insertion sites can act as stress risers, concentrating forces and predisposing to pathological fractures or periprosthetic fractures under loads that would typically be innocuous.
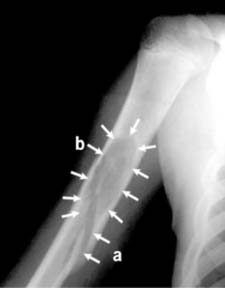
Fig. 12 illustrates complex biomechanical forces acting on bone, demonstrating how varying loads lead to distinct fracture patterns.
Indications & Contraindications
The decision-making process for fracture management, whether operative or non-operative, is multifactorial, considering patient factors, fracture characteristics, and available resources. The primary goal is to achieve stable fixation, anatomical reduction, and ultimately, functional restoration.
Indications
Non-Operative Management
is typically indicated for:
* Minimally displaced or stable fractures that are unlikely to displace further.
* Fractures in bones with high intrinsic healing potential and low functional demands (e.g., specific rib fractures, stress fractures).
* Fractures in patients with significant comorbidities rendering them unfit for surgery.
* Specific pediatric fractures, often possessing greater remodeling potential.
* Closed fractures that can be adequately reduced and held stable by external immobilization (casts, splints, braces).
Operative Management
is generally indicated for:
*
Unstable fractures:
Those prone to significant displacement, malunion, or nonunion without surgical intervention.
*
Displaced fractures:
Where anatomical reduction is necessary for optimal function or to prevent joint incongruity.
*
Open fractures:
To facilitate débridement, irrigation, infection control, and stabilization.
*
Articular fractures:
Requiring anatomical reduction to restore joint congruity and prevent post-traumatic arthritis.
*
Neurovascular compromise:
When the fracture causes or threatens damage to adjacent nerves or vessels, requiring emergent reduction and stabilization.
*
Polytrauma patients:
Often requires early fracture stabilization for damage control orthopaedics and to facilitate overall patient care.
*
Pathological fractures:
Secondary to tumors or metabolic bone disease, often requiring stabilization to prevent impending fracture or treat an existing one.
*
Failure of non-operative management:
Subsequent displacement or intractable pain despite conservative treatment.
*
Specific fracture patterns:
Such as intertrochanteric hip fractures, certain displaced ankle fractures, or intra-articular elbow fractures that consistently benefit from surgical fixation.
Contraindications
Absolute Contraindications to Surgery:
*
Medical instability:
A patient too unwell to withstand anaesthesia and surgery (e.g., severe sepsis, decompensated cardiac failure, uncontrolled hemorrhage not related to the fracture itself). These situations require medical stabilization prior to any elective fracture intervention.
*
Active systemic infection:
Unless the fracture itself is the source or an emergent open fracture requires immediate débridement.
Relative Contraindications to Surgery:
*
Severe local soft tissue compromise:
Such as severe swelling, blistering, or compromised skin envelope, which may delay surgery until soft tissue conditions improve (e.g., by elevation, serial examinations). In some cases, external fixation may be used as a temporizing measure.
*
Significant comorbidities:
Patients with poorly controlled diabetes, immunosuppression, or severe peripheral vascular disease may have increased risks of infection, wound healing complications, or implant failure. These require careful risk-benefit analysis and optimization before surgery.
*
Patient refusal:
Legally and ethically, a competent patient's refusal of surgical intervention must be respected after thorough informed consent.
*
Expected poor compliance with post-operative care:
While not an absolute contraindication, it can influence the choice of implant or rehabilitation strategy.
Operative vs. Non-Operative Indications
| Factor | Operative Management | Non-Operative Management |
|---|---|---|
| Fracture Type | Open, intra-articular, unstable, significantly displaced, pathological, impending pathological, neurovascular compromise. | Closed, minimally displaced, stable, high intrinsic healing potential (e.g., isolated fibular shaft), stress fractures. |
| Anatomical Site | Joint-spanning fractures, weight-bearing joints (e.g., hip, knee, ankle), certain long bone fractures (e.g., femur, tibia shaft). | Non-weight-bearing bones (e.g., ribs, specific avulsion fractures), pediatric fractures with good remodeling potential. |
| Patient Factors | Polytrauma, young active individuals (desire for anatomical restoration), non-compliance with casting. | Medically frail patients, poor surgical candidates, children with high remodeling capacity, patient preference (after informed consent). |
| Soft Tissue | Open wound (requiring débridement), compartment syndrome. | Intact skin, no impending skin compromise. |
| Functional Need | High demand (athletes, manual laborers), immediate stability for nursing care. | Low functional demand, ability to tolerate immobilization. |
| Complications | Malunion, nonunion (failed non-operative), impending neurovascular injury. | No immediate or impending complications with conservative care. |
Pre-Operative Planning & Patient Positioning
Meticulous pre-operative planning is critical to optimize surgical outcomes, minimize complications, and ensure efficient operating room time. This involves a systematic assessment of the patient, the injury, and the surgical strategy.
Pre-Operative Planning
-
Clinical Assessment:
- Patient History: Detailed mechanism of injury, associated injuries (polytrauma), comorbidities (diabetes, osteoporosis, peripheral vascular disease), medication history (anticoagulants, immunosuppressants), allergies.
- Physical Examination: Comprehensive neurovascular assessment (pulses, sensation, motor function), soft tissue envelope evaluation (swelling, abrasions, lacerations, skin turgor, blistering), assessment for compartment syndrome. In open fractures, meticulous wound documentation and appropriate immediate management (sterile dressing, tetanus prophylaxis, antibiotics) are paramount.
-
Imaging Review:
- Radiographs: Obtain orthogonal views (AP and lateral) of the injured area, including joints above and below. Stress views may be indicated for ligamentous stability assessment.
- Computed Tomography (CT): Essential for complex articular fractures, comminuted fractures, and to define fracture morphology, fragment orientation, and displacement. 3D reconstructions can be invaluable for pre-operative visualization and templating.
- Magnetic Resonance Imaging (MRI): Useful for evaluating soft tissue injuries (ligaments, menisci, cartilage), occult fractures, or bone marrow edema, though less frequently indicated primarily for fracture assessment.
- Angiography: Indicated if vascular injury is suspected.
-
Surgical Strategy & Templating:
- Fracture Classification: Utilize established classification systems (e.g., AO/OTA, Gustilo-Anderson for open fractures) to guide prognosis and treatment.
- Implant Selection: Determine appropriate implants (plates, screws, nails, external fixators) based on fracture pattern, bone quality, biomechanical principles (absolute vs. relative stability), and surgeon preference.
- Templating: Use radiographic templates (digital or physical) to estimate implant size, length, and trajectory, especially for plate fixation or joint replacement. This aids in anticipating challenges and minimizing intraoperative adjustments.
- Surgical Approach: Pre-plan the optimal surgical approach, considering internervous planes, neurovascular structures at risk, and soft tissue preservation.
- Contingency Planning: Anticipate potential intraoperative difficulties (e.g., comminution, poor bone quality, malreduction) and have alternative strategies and implants available.
-
Logistics:
- Anaesthesia Consultation: Assess patient suitability for general vs. regional anaesthesia.
- Blood Products: Type and cross-match blood if significant blood loss is anticipated.
- Antibiotic Prophylaxis: Administer appropriate prophylactic antibiotics prior to incision, especially crucial for open fractures.
- DVT Prophylaxis: Implement strategies to prevent venous thromboembolism.
Patient Positioning
Patient positioning must achieve optimal surgical access while simultaneously protecting the patient from iatrogenic injury (e.g., nerve compression, pressure sores). This requires careful attention to detail.
-
General Principles:
- Exposure: Ensure the operative field is fully exposed and draped appropriately, with sufficient sterile space for instruments and surgeon movement.
- Stability: The patient must be securely positioned to prevent movement during surgery, especially when utilizing traction tables or fracture tables.
- Accessibility: Allow for intraoperative fluoroscopy access without repositioning the patient or the C-arm.
- Physiological Monitoring: Ensure access for anaesthesia team to monitor vital signs.
- Padding: Use adequate padding at all pressure points (heels, sacrum, elbows, ulnar nerve, peroneal nerve at fibular head, eyes, ears) to prevent nerve palsies and pressure ulcers.
- Pre-existing Conditions: Account for pre-existing arthritis, cervical spine instability, or other conditions that might restrict positioning.
- Tourniquet: If used, ensure proper size and placement, and document inflation time and pressure.
-
Specific Positioning Considerations (Examples):
- Supine: Common for upper extremity fractures, anterior approaches to the lower extremity, and pelvic fractures. May require a bump for shoulder or clavicle fractures.
- Lateral Decubitus: Often used for proximal humerus, diaphyseal humerus, or hip fractures (especially with a fracture table). Careful axillary and perineal padding is essential.
- Prone: Utilized for posterior approaches to the spine, calcaneus, or posterior malleolus. Attention to abdominal breathing and pressure points (face, chest, knees) is critical.
- Fracture Table/Traction: Essential for long bone fractures (femur, tibia) to achieve and maintain reduction with controlled traction, and to facilitate intramedullary nailing. Proper sterile draping around the traction boots/pins is vital.
Detailed Surgical Approach / Technique
The surgical repair of fractures aims to restore anatomical alignment and achieve stable fixation, promoting timely bone healing and functional recovery. While specific techniques vary by fracture location and morphology, core principles guide every intervention.
Principles of Reduction and Fixation
Two primary modes of fracture healing, dictated by the stability of fixation, influence surgical technique:
*
Absolute stability (Primary bone healing):
Achieved with rigid fixation, typically through interfragmentary compression (e.g., lag screws, compression plating). This minimizes micromotion at the fracture site, allowing direct cortical remodeling without a significant callus. This is often desirable for articular fractures to restore joint congruity.
*
Relative stability (Secondary bone healing):
Achieved with flexible fixation (e.g., intramedullary nailing, bridge plating, external fixation) that permits controlled micromotion at the fracture site. This stimulates robust callus formation, which subsequently remodels into mature bone. This is the preferred method for diaphyseal fractures, preserving periosteal blood supply.
Surgical Steps: General Principles
-
Incision:
- Planned pre-operatively to align with tension lines, provide adequate exposure, and allow for potential extensions.
- Consider soft tissue envelope, previous scars, and potential for future procedures.
- Minimize undermining to preserve vascularity.
-
Dissection & Internervous Planes:
- Proceed meticulously through skin, subcutaneous tissue, and fascia.
- Identify and protect critical neurovascular structures. Knowledge of internervous planes (approaches that separate muscles supplied by different nerves) is paramount to minimize muscle damage and functional deficit. Examples include the deltopectoral approach for the shoulder (pectoralis major - medial pectoral nerve; deltoid - axillary nerve) or the interval between brachioradialis (radial nerve) and pronator teres (median nerve) for distal radius.
- Minimize soft tissue stripping from fracture fragments, especially comminuted pieces, to preserve their vascularity and promote healing (biological fixation principles).
-
Exposure:
- Gain adequate visualization of the fracture site. This may involve gentle retraction, temporary osteotomies (rare), or removal of intervening hematoma and necrotic tissue.
- For open fractures, thorough débridement of all devitalized tissue (skin, subcutaneous fat, muscle, bone) and high-volume irrigation are the initial critical steps to prevent infection.
-
Reduction:
- Indirect Reduction: Often preferred, especially for diaphyseal fractures, to preserve soft tissue attachments. Techniques include manual traction, ligamentotaxis (for periarticular fractures), use of external fixators as reduction tools, or percutaneous clamps.
- Direct Reduction: Involves directly manipulating fragments with reduction clamps, bone hooks, or joy sticks. This is necessary for articular fractures to achieve anatomical congruity.
- Maintenance of Reduction: Temporary fixation with K-wires or provisional clamps is often used to hold the reduction while definitive fixation is applied.
- Intraoperative fluoroscopy is indispensable to confirm reduction in multiple planes.
Fig. 3 conceptualizes the stages of indirect bone healing, showcasing the transition from hematoma to fibrous and cartilaginous callus, and ultimately, bony union.
-
Definitive Fixation:
-
Plates and Screws:
- Lag Screws: Provide interfragmentary compression for absolute stability, typically for oblique or spiral fractures.
- Compression Plates: Applied to compress the fracture ends, often used for transverse fractures.
- Neutralization Plates: Protect lag screws from bending or torsional forces.
- Bridge Plates: Used for comminuted diaphyseal fractures, spanning the comminution without direct reduction of intermediate fragments, promoting relative stability.
-
Locking Plates (LC-DCP, LCP):
Offer angular stability via threaded screw heads locking into the plate, creating a fixed-angle construct. Particularly useful in osteoporotic bone or complex comminuted fractures, acting as internal fixators.
Fig. 4 depicts various orthopedic implants, illustrating plates, screws, and potentially an intramedullary nail, highlighting the diverse armamentarium for internal fixation.
-
Intramedullary Nails (IMN):
- Load-sharing devices placed within the medullary canal of long bones (femur, tibia, humerus).
- Provide relative stability, preserving periosteal blood supply.
- Static (locked) nailing prevents shortening and rotation; dynamic (unlocked) nailing allows for axial micromotion.
- Reamed vs. unreamed: Reaming allows for a larger diameter nail and enhances cortical contact but potentially damages endosteal blood supply.
-
External Fixation:
- Pins inserted into bone fragments connected to an external frame.
- Provides rapid temporary stabilization, especially for open fractures, polytrauma, or severe soft tissue injuries (damage control orthopaedics).
- Can be definitive in certain situations (e.g., pelvic ring injuries, pilon fractures with severe soft tissue compromise).
- Allows for access to soft tissues and monitoring for swelling.
-
Wires (K-wires, cerclage wires):
- K-wires: Temporary fixation, tension band wiring (e.g., patella, olecranon).
- Cerclage wires: Adjunct to plates or nails, used for long spiral fractures, or to secure butterfly fragments (used judiciously to avoid devascularization).
-
Plates and Screws:
Bone Healing Mechanisms
The chosen fixation method directly influences the bone healing response.
*
Primary (Direct) Healing:
Occurs with absolute stability, characterized by direct remodeling of bone across the fracture gap via cutting cones of osteoclasts and osteoblasts. Requires a minimal gap (<100 µm) and rigid interfragmentary compression. No external callus is formed.
*
Secondary (Indirect) Healing:
Occurs with relative stability, involving a more robust and visible process of callus formation.
1.
Inflammation:
Hematoma formation at the fracture site provides a scaffold and initiates the inflammatory cascade, attracting mesenchymal stem cells.
2.
Soft Callus Formation:
Fibroblasts and chondroblasts lay down fibrous tissue and hyaline cartilage, forming a soft callus, providing initial stability.
3.
Hard Callus Formation:
Endochondral ossification replaces cartilage with woven bone, and intramembranous ossification directly forms woven bone, creating a hard bony callus. This significantly strengthens the fracture.
4.
Remodeling:
Over months to years, the woven bone of the callus is gradually replaced by mature lamellar bone, and the bone remodels in response to mechanical stresses (Wolff's Law), restoring the original contour and strength.
Fig. 5 provides a histological depiction of callus formation, showing the cellular events involved in bridging the fracture gap with new bone tissue during secondary healing.
Fig. 6 illustrates the intricate process of bone remodeling, detailing how woven bone is replaced by lamellar bone, restoring structural integrity and adapting to mechanical demands.
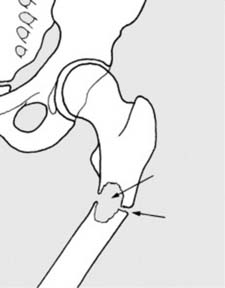
Fig. 11 provides a microscopic view or schematic of bone healing at a cellular level, emphasizing the roles of osteocytes, osteoblasts, and osteoclasts in woven bone formation and subsequent remodeling.
-
Wound Closure:
- Thorough irrigation of the surgical site.
- Hemostasis is achieved.
- Layered closure of fascia, subcutaneous tissue, and skin, minimizing tension. Drains may be utilized if significant dead space or anticipated fluid accumulation.
Complications & Management
Despite meticulous surgical technique, complications can arise following fracture repair, significantly impacting patient outcomes. Proactive identification, prevention, and timely management are paramount.
Common Complications
| Complication | Incidence | Salvage Strategies & Management |
|---|---|---|
| Infection | Open fractures: 5-50% (Gustilo-Anderson type-dependent); Closed fractures: 1-5%. | Prevention: Meticulous débridement (open fractures), prophylactic antibiotics, strict aseptic technique. Treatment: Surgical débridement, culture-guided IV antibiotics, wound vac, staged reconstruction, hardware removal (if stable union), antibiotic beads, muscle flaps, amputation (salvage failure). |
| Nonunion | Varies by bone/location (e.g., tibia 5-15%, scaphoid 10-20%). | Risk Factors: Poor vascularity, significant soft tissue damage, unstable fixation, infection, smoking, NSAID use. Treatment: Revision surgery (open reduction, rigid fixation, bone grafting – autograft or allograft), biological augmentation (BMPs, PRP), external bone stimulators. |
| Malunion | Common (up to 20% in some series), often asymptomatic. Symptomatic: <5%. | Treatment: Often non-operative if asymptomatic and functionally acceptable. Symptomatic malunion (pain, limb length discrepancy, joint incongruity, functional deficit): Corrective osteotomy and internal fixation. |
| Hardware Failure | 2-10%, often secondary to nonunion or excessive load. | Treatment: Removal of failed hardware, revision surgery with more robust fixation (stronger plate, larger nail, different fixation strategy), often combined with bone grafting if nonunion present. Addressing underlying mechanical or biological issues. |
| Neurovascular Injury | Acute: <1% (varies by fracture location); Chronic nerve entrapment: <5%. | Acute: Immediate reduction of fracture, vascular repair/bypass, nerve exploration and repair/grafting. Chronic: Nerve decompression, neurolysis, tendon transfers for persistent deficits. |
| Compartment Syndrome | Varies by injury type (e.g., tibia shaft fracture 2-9%). | Prevention: Careful monitoring of pain, paresthesia, pallor, pulselessness, paralysis, pressure. Treatment: Emergent fasciotomy. Delayed closure or skin grafting. |
| Thromboembolism (DVT/PE) | DVT: 5-60% (diagnosed with imaging); PE: 1-10%. Clinical PE: <1%. | Prevention: Mechanical (compression stockings, IPCs) and/or pharmacological prophylaxis (heparin, LMWH, oral anticoagulants) as per institutional guidelines, early mobilization. Treatment: Anticoagulation (therapeutic), IVC filter. |
| Post-Traumatic Arthritis | Common in intra-articular fractures (up to 30-50%). | Prevention: Anatomical reduction of articular surface, stable fixation, early range of motion. Treatment: Arthroscopy (débridement), osteotomy (realign joint), arthrodesis (joint fusion), arthroplasty (joint replacement). |
| Stiffness/Reduced ROM | Varies widely by joint and injury (e.g., elbow, ankle). | Prevention: Early, controlled mobilization; appropriate rehabilitation protocols. Treatment: Physical therapy, dynamic splinting, manipulation under anaesthesia, arthrolysis (surgical release). |
Fig. 7, depending on its specific content, could depict a fracture complication such as an open wound, nonunion, or post-traumatic deformity, serving as a visual reference for adverse outcomes.
Specific Considerations
- Open Fractures: High risk of infection. Gustilo-Anderson classification guides management. Immediate intravenous antibiotics, aggressive surgical débridement of devitalized tissue, high-volume irrigation, and stabilization are critical. Often requires multiple débridements.
-
Nonunion:
Defined as the failure of a fracture to heal after a time period appropriate for the location and type of fracture (typically 6-9 months), or when no further progress towards healing is observed on sequential radiographs. Can be hypertrophic (ample callus but not bridging) or atrophic (minimal callus). Atrophic nonunions often indicate poor biology, while hypertrophic nonunions suggest inadequate mechanical stability.
Fig. 8 visually represents a nonunion, possibly showing persistent fracture gap or failed callus formation, underscoring the challenge of healing arrest. - Malunion: Healing of a fracture in an anatomically unacceptable position. While some malunions are asymptomatic, others can lead to pain, functional impairment, limb length discrepancy, or accelerated articular degeneration.
Post-Operative Rehabilitation Protocols
Post-operative rehabilitation is an integral component of fracture management, aiming to restore strength, range of motion, and function while protecting the healing fracture. Protocols are tailored to the specific fracture, method of fixation, bone quality, and patient factors, but general principles apply.
Immediate Post-Operative Phase (Days 0-2 weeks)
- Pain Management: Multimodal approach using analgesics, regional blocks, and NSAIDs (judiciously, as they may impair early healing).
- Wound Care: Monitor surgical incision for signs of infection (erythema, swelling, discharge), dehiscence, or blistering. Maintain sterile dressing changes.
- Elevation & Edema Control: Elevate the injured extremity above heart level to minimize swelling. Apply compression bandages where appropriate.
-
Early Mobilization:
- Joints not immobilized by the fracture or fixation: Immediate active and passive range of motion exercises to prevent stiffness.
- Fracture site: Protected range of motion or strict immobilization as dictated by fracture stability and fixation strength. For highly stable constructs (e.g., locking plates in certain locations, stable IMN), protected weight-bearing or early range of motion of adjacent joints may be initiated.
- Weight-Bearing Restrictions: Strictly adhere to the surgeon's prescribed weight-bearing status (e.g., non-weight-bearing, toe-touch weight-bearing, partial weight-bearing, weight-bearing as tolerated). This is crucial to protect the developing callus and implant.
- DVT Prophylaxis: Continue as per pre-operative plan.
- Patient Education: Instruct on signs of complications (infection, compartment syndrome, neurovascular changes), activity restrictions, and the importance of compliance with the rehabilitation program.
Intermediate Phase (2-6 weeks)
- Progression of Range of Motion: Gradually increase active and passive range of motion exercises as tolerated and as healing progresses, guided by clinical and radiographic assessment.
- Initiation of Strengthening: Introduce isometric exercises for muscles crossing the fracture site. For non-operative cases, gentle resistive exercises for adjacent joints.
- Weight-Bearing Progression: Based on radiographic evidence of early callus formation and clinical stability, weight-bearing may be cautiously progressed. For example, from non-weight-bearing to toe-touch or partial weight-bearing.
- Gait Training: If weight-bearing is allowed, initiate gait training with appropriate assistive devices (crutches, walker) and focus on proper mechanics.
Advanced Healing & Consolidation Phase (6 weeks - 3+ months)
- Progressive Strengthening: Advance to isotonic and isokinetic exercises to rebuild muscle strength and endurance.
- Full Weight-Bearing: Once there is clear radiographic evidence of bridging callus and clinical stability, progression to full weight-bearing can be achieved. This often requires cessation of assistive devices.
- Proprioception and Balance Training: Crucial for restoring neuromuscular control, particularly for lower extremity fractures.
- Functional Activities: Gradually reintroduce activities of daily living and work-specific tasks.
- Imaging: Serial radiographs continue to monitor fracture healing, looking for callus maturation and remodeling.
Remodeling & Return to Activity (3-12+ months)
- High-Impact Activities: Gradually introduce higher impact and sport-specific activities, ensuring bone healing is complete and muscle strength, endurance, and proprioception are fully restored.
- Endurance Training: Continue to build overall physical conditioning.
- Implant Removal: Consider elective hardware removal in certain cases (e.g., prominent hardware causing irritation, infection, young active patients with IMN in specific locations), typically 12-18 months post-op after complete union. This decision is individualized based on risks and benefits.
Fig. 9 might illustrate a rehabilitation exercise or a patient demonstrating functional recovery, visually reinforcing the objectives of post-operative care.
Fig. 10 could be a radiographic series showing progressive fracture healing over time, from initial injury to consolidation, demonstrating the radiographic milestones in rehabilitation.
Throughout all phases, close communication between the surgeon, physical therapist, and patient is essential to ensure a safe and effective rehabilitation progression. Overly aggressive rehabilitation can jeopardize fixation and healing, while overly conservative approaches can lead to stiffness and delayed functional recovery.
Summary of Key Literature / Guidelines
The management of fractures is continuously informed by a robust body of evidence-based literature and clinical guidelines. Adherence to established principles significantly improves patient outcomes and minimizes complications.
Foundational Principles
- AO Principles: The Arbeitsgemeinschaft für Osteosynthesefragen (AO Foundation) has been instrumental in codifying basic principles of fracture management since the 1960s. These principles emphasize anatomical reduction (especially for articular fractures), stable internal fixation, preservation of blood supply, and early active mobilization. The evolution from absolute stability (interfragmentary compression) to biological fixation (relative stability with minimal soft tissue disruption) reflects an enhanced understanding of bone healing biology.
- ATLS (Advanced Trauma Life Support): For polytrauma patients, ATLS guidelines dictate a standardized, systematic approach to initial assessment and management, prioritizing life-threatening injuries before addressing musculoskeletal trauma. Early fracture stabilization in polytrauma (damage control orthopaedics) is recognized for its role in reducing systemic inflammatory response and improving survival.
Key Areas of Evidence
-
Open Fractures:
- Antibiotic Prophylaxis: Strong evidence supports early administration of broad-spectrum intravenous antibiotics (e.g., cephalosporins, with aminoglycosides added for Gustilo-Anderson Type III injuries) within 3 hours of injury to reduce infection rates.
- Débridement: Urgent and thorough surgical débridement of all devitalized tissue remains the cornerstone of open fracture management. Multiple débridements are often necessary. The "six-hour rule" is now understood as a guideline, with urgency taking precedence over a strict time cutoff, especially for Gustilo Type III injuries.
- Timing of Fixation: While immediate fixation is often ideal after débridement, delayed definitive fixation (up to 7-10 days) with temporary external fixation has shown comparable outcomes in some severe open fractures, allowing for soft tissue recovery.
-
Fracture Fixation Modalities:
- Intramedullary Nailing: Considered the gold standard for many long bone diaphyseal fractures (femur, tibia), offering load-sharing, biological fixation, and early mobilization. Reamed vs. unreamed nailing remains a debated topic, with reamed nails often demonstrating higher union rates but potentially greater heat-related endosteal damage.
- Plating: Locking plates have revolutionized the management of metaphyseal and periarticular fractures, offering angular stability independent of bone-plate contact, which is advantageous in osteoporotic bone and comminuted fractures. Bridge plating for comminuted diaphyseal fractures preserves biology.
- External Fixation: Essential for damage control orthopaedics in polytrauma, open fractures with severe contamination, and fractures with significant soft tissue compromise. Can be definitive for certain pelvic and tibial pilon fractures.
-
Bone Healing Augmentation:
- Bone Grafting: Autogenous bone graft (e.g., iliac crest) remains the gold standard for nonunion and defects due to its osteoconductive, osteoinductive, and osteogenic properties. Allograft and synthetic grafts are alternatives.
- Bone Morphogenetic Proteins (BMPs): Recombinant human BMP-2 and BMP-7 have demonstrated efficacy in accelerating healing in challenging fractures, particularly recalcitrant nonunions and open tibial fractures, though cost and potential adverse effects warrant careful consideration.
- Physical Stimuli: Pulsed electromagnetic fields (PEMF) and low-intensity pulsed ultrasound (LIPUS) are non-invasive modalities that can be used adjunctively for delayed unions and nonunions, with varying levels of evidence.
-
Prevention of Venous Thromboembolism (VTE):
- Guidelines from organizations like the American Academy of Orthopaedic Surgeons (AAOS) and American College of Chest Physicians (ACCP) recommend individualized VTE prophylaxis based on patient and injury risk factors. Both mechanical (intermittent pneumatic compression, elastic stockings) and pharmacological (LMWH, oral anticoagulants) methods are employed.
-
Post-Traumatic Arthritis:
- Anatomical reduction of articular surfaces and rigid internal fixation remain the best strategies for preventing post-traumatic arthritis in intra-articular fractures. Early, protected motion is also crucial.
The field of fracture pathology and healing continues to evolve with ongoing research into biomaterials, biological augmentation, and advanced imaging. Remaining current with key literature and adhering to established evidence-based guidelines ensures optimal patient care in this complex domain.
You Might Also Like