Mangled Extremity: A Comprehensive Clinical Case Presentation
Key Takeaway
Mangled extremity assessment prioritizes ATLS, systemic stabilization, and hemorrhage control. A detailed limb examination evaluates vascular integrity, nerve function, bone damage, and soft tissue viability. These critical findings guide urgent decisions on limb salvage, surgical interventions, and multidisciplinary trauma management strategies.
Patient Presentation & History
A 35-year-old male was presented to the emergency department after being involved in a high-energy motor vehicle accident. The mechanism of injury involved him being struck by a car that subsequently crushed and pinned his left lower extremity against a solid wall for an undetermined period before extrication by emergency services. Pre-hospital reports indicated significant external hemorrhage and gross deformity of the left leg. Paramedics applied a tourniquet proximally on the left thigh at the scene and initiated intravenous fluid resuscitation en route.
The patient has no significant past medical history, no known allergies, and takes no regular medications. He is an otherwise healthy non-smoker with no history of peripheral vascular disease or diabetes, which are crucial considerations in assessing limb viability.
Upon arrival, the patient was conscious but agitated, Glasgow Coma Scale (GCS) 14 (E4V4M6). Initial ATLS primary survey revealed:
*
Airway:
Patent, protected.
*
Breathing:
Spontaneous, symmetric chest expansion, clear breath sounds bilaterally. Respiratory rate 22 breaths/min, SaO2 97% on 10L non-rebreather mask.
*
Circulation:
Tachycardic (HR 120 bpm), hypotensive (BP 90/60 mmHg). Peripheral pulses (right lower limb, bilateral upper limbs) were weak but present. Left lower limb pulses were not palpable distal to the injury, despite the tourniquet. Visible active hemorrhage from the left leg wound requiring continued direct pressure in addition to the tourniquet. Two large-bore intravenous cannulae were established and rapid infusion of crystalloids initiated, followed by packed red blood cells as per institutional massive transfusion protocol (MTP) activation.
*
Disability:
GCS 14. Pupils equal and reactive to light. No focal neurological deficits in the upper limbs or right lower limb.
*
Exposure:
Full body examination revealed contusions and abrasions to the trunk and right upper extremity, but no other obvious major fractures or life-threatening injuries. The left lower extremity was obviously severely traumatized.
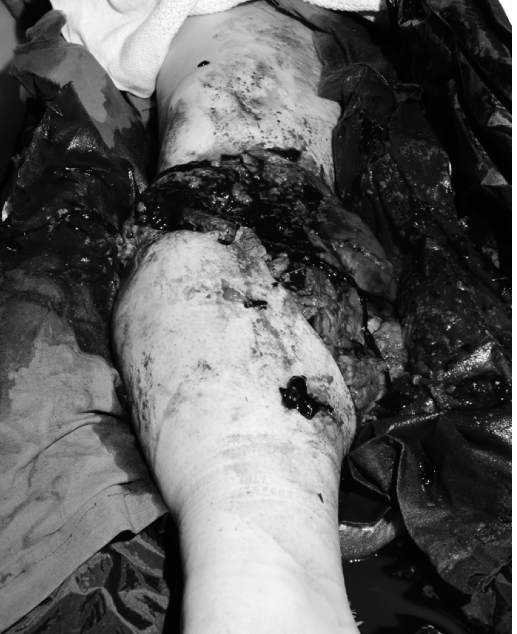
The initial clinical photograph of the patient's left lower extremity upon arrival in the emergency department depicts the severity of the trauma:

This image demonstrates an obviously mangled and severely deformed left lower leg. There are extensive, ragged, and contaminated open wounds with significant soft tissue avulsion and degloving. The exposed muscle appears dusky and non-viable in areas, and there is clear evidence of segmental bone loss with gross displacement and comminution of the tibia and fibula. The overall appearance is consistent with a high-energy crush injury, likely Gustilo-Anderson Type IIIB or IIIC, given the visible bone loss and probable vascular compromise.
Immediate interventions in the ED included:
* Continuation of crystalloid and blood product resuscitation.
* Activation of Major Haemorrhage Protocol.
* Administration of broad-spectrum intravenous antibiotics (e.g., Cefazolin 2g IV, Gentamicin 5mg/kg IV, Metronidazole 500mg IV).
* Tetanus toxoid administration.
* Intravenous analgesia (e.g., Fentanyl).
* Initial gross contamination removed with saline-soaked gauze. The wound was then covered with sterile saline-soaked dressings and a soft bandage.
* Urgent plain radiographs of the left lower extremity were ordered.
* Immediate consultation with Orthopedic Trauma, Vascular, and Plastic Surgery services was initiated.
Clinical Examination
Following initial resuscitation and stabilization, a detailed secondary survey and focused limb examination were performed.
General Appearance & Systemic Assessment
The patient's hemodynamics stabilized after 2 units of packed red blood cells and 1 liter of crystalloid, with BP 110/70 mmHg and HR 90 bpm. He remained agitated but was more cooperative. No signs of head injury, chest trauma (clear breath sounds, no crepitus), or abdominal injury (soft, non-tender, no guarding). Pelvis was stable to compression. Spine was non-tender to palpation.
Local Limb Examination (Left Lower Extremity)
-
Inspection:
- Deformity: Gross angulation and rotation of the mid-diaphyseal tibia and fibula. Significant shortening of the left lower limb relative to the right.
-
Skin & Soft Tissues:
As seen in
, there were multiple large, irregular lacerations on the anterior and medial aspects of the leg, consistent with a degloving injury. The wounds extended from just below the knee to the ankle. Significant soft tissue loss was apparent, exposing bone fragments and muscle. The exposed muscle bundles appeared dusky, non-bleeding, and frankly necrotic in several areas. Large areas of skin were avulsed and discolored, indicating doubtful viability. Gross contamination with dirt, clothing fragments, and pavement debris was present within the wound. There was significant swelling and tension in the calf compartments, even with the extensive open wounds. - Bleeding: The tourniquet was temporarily deflated for assessment; there was brisk, pulsatile arterial bleeding from a deep medial wound, confirming major vascular injury. The tourniquet was immediately reinflated.
- Compartment Assessment: The calf compartments felt tense and firm despite the open nature of the injury, suggesting potential compartment syndrome development or severe muscle contusion.
-
Palpation:
- Tenderness: Diffuse tenderness throughout the left lower leg.
- Crepitus: Palpable crepitus confirming extensive comminution and free bone fragments.
- Pulses: Femoral pulse was strong. Popliteal pulse was not palpable. Dorsalis pedis and posterior tibial pulses were absent by palpation and Doppler, even with the tourniquet temporarily released. Ankle-Brachial Index (ABI) was not measurable on the left due to absent distal pulses.
- Range of Motion: Grossly restricted and excruciatingly painful at the ankle and knee due to the severe deformity and instability. No attempt was made to force movement.
-
Neurological Assessment:
- Motor Function: Inability to dorsiflex, plantarflex, invert, or evert the foot. Patient unable to actively move toes. This could be due to pain, compartment syndrome, direct nerve injury, or muscle necrosis.
- Sensory Function: Complete anesthesia of the foot and ankle, encompassing the deep peroneal, superficial peroneal, saphenous, sural, and tibial nerve distributions. The extent of sensory loss raised high suspicion for significant neurological compromise.
The combined findings of absent distal pulses, extensive soft tissue loss with devitalized muscle, exposed comminuted bone, complete motor and sensory paralysis, and the high-energy crush mechanism strongly indicated a Gustilo-Anderson Type IIIC open fracture of the tibia and fibula, with severe mangling of the extremity.
Imaging & Diagnostics
Plain Radiographs
AP and lateral views of the left tibia/fibula, knee, and ankle were obtained.
*
Tibia/Fibula:
Revealed a highly comminuted, segmental open fracture of the mid-diaphyseal tibia with significant bone loss (approximately 5 cm defect). The fibula was also comminuted and fractured at multiple levels. Gross displacement and angulation were noted. Multiple small, radio-opaque foreign bodies (consistent with gravel/metal fragments) were embedded within the soft tissues.
*
Knee and Ankle:
No associated knee dislocation or obvious ankle fracture, but severe soft tissue swelling around these joints was evident. Projections were carefully reviewed for subtle articular involvement that might be masked by the severe diaphyseal injury.
CT Scan
A CT scan of the left lower extremity was performed following initial radiographs and patient stabilization.
*
Bony Anatomy:
Provided a more precise understanding of the fracture pattern, extent of comminution, and quantification of the bone defect (confirming a 5-6 cm segmental tibial defect). 3D reconstructions were invaluable for pre-operative planning, especially regarding length restoration and potential areas for internal fixation.
*
Soft Tissue Assessment:
Detailed the extent of muscle damage, presence of hematoma, and foreign bodies. Confirmed significant muscle necrosis within the anterior and deep posterior compartments.
*
CT Angiography (CTA):
Given the absent distal pulses and brisk arterial bleeding, a CTA of the left lower extremity was critical. It demonstrated a complete transection of the popliteal artery just distal to the genicular arteries, with significant retraction of both ends. The anterior and posterior tibial arteries, and the peroneal artery, were all disrupted distally. This confirmed a multi-vessel arterial injury consistent with a Gustilo-Anderson Type IIIC classification.
MRI
An MRI was not performed in the acute setting due to the patient's instability, presence of metallic foreign bodies, and the urgency of operative intervention. Its utility in this acute phase is limited compared to CTA and surgical exploration. However, it might be considered in later stages to assess nerve integrity, muscle viability after debridements, or for evaluation of chronic osteomyelitis if infection develops.
Laboratory Investigations
- Full Blood Count (FBC): Initial Hb 9.2 g/dL, Hct 28%, WCC 18.5 x 10^9/L (leukocytosis secondary to trauma and stress). Platelets 250 x 10^9/L.
- Electrolytes & Renal Function: Na 138 mmol/L, K 4.1 mmol/L, Cr 105 µmol/L, Urea 6.2 mmol/L. Close monitoring of renal function was critical due to the risk of rhabdomyolysis and acute kidney injury from crush syndrome.
- Coagulation Profile: INR 1.3, PTT 35 seconds. Trending towards coagulopathy requiring correction during massive transfusion.
- Creatine Kinase (CK): Initial CK 15,000 U/L. This significantly elevated level confirmed extensive muscle damage and indicated a high risk of rhabdomyolysis. Serial CK levels were planned.
- Arterial Blood Gas (ABG): pH 7.28, pCO2 38 mmHg, pO2 95 mmHg, HCO3 18 mmol/L, Lactate 4.8 mmol/L. Mild metabolic acidosis and elevated lactate consistent with shock and tissue hypoperfusion.
- Group and Cross-Match: O positive, cross-matched for 6 units of PRBCs.
- Urine Dipstick: Positive for myoglobin.
Differential Diagnosis
The primary diagnosis for this patient is a mangled left lower extremity, likely Gustilo-Anderson Type IIIC open fracture of the tibia and fibula with significant soft tissue loss and multi-vessel arterial transection. The following table provides a differential diagnostic perspective focusing on similar high-energy lower limb trauma that may present with varying degrees of severity and implications for limb salvage.
| Feature | Mangled Lower Extremity (Severe Open Fracture/Crush - Case Presentation) | Gustilo-Anderson Type IIIB Open Tibia Fracture (Isolated) | Severe Degloving Injury (e.g., Morel-Lavallée lesion with open wound) |
|---|---|---|---|
| Mechanism | High-energy, direct crush, avulsion, impalement (car vs. wall). Often pinned/trapped. | High-energy (e.g., MVC, pedestrian vs. car), direct blunt trauma. Less crush/avulsion. | High-energy tangential shear force (e.g., pedestrian vs. car, industrial accident). |
| Bone Involvement | Extensive comminution, segmental defects (5-6cm tibial loss), periosteal stripping, exposed bone. Tibia and fibula. | Significant comminution, periosteal stripping, bone exposure. Less severe bone loss or segmental defect. Usually tibia only. | May have underlying simple fracture, but primary injury is soft tissue separation. |
| Soft Tissue Injury |
Extensive, devitalized tissue, degloving (anterior/medial), avulsion, muscle necrosis (anterior/deep posterior), gross contamination, large skin loss, dusky/necrotic appearance.

|
Large skin laceration (>10cm), significant soft tissue damage, exposed bone, periosteal stripping. Less muscle necrosis or extensive degloving. | Large, open or closed degloving defect, potential for extensive fluid collection (hematoma/seroma), skin necrosis. Subcutaneous fat liquefaction common. |
| Vascular Injury | Confirmed multi-vessel transection (popliteal, anterior/posterior tibial arteries) requiring urgent repair. Gustilo IIIC. | Possible, but not the defining characteristic. Pulses usually present or easily restored. No requirement for arterial repair to be IIIB. | Less likely to have primary major arterial transection unless associated with significant bone trauma or direct vessel injury. Capillary refill may be slow due to swelling. |
| Neurological Injury | High likelihood of complete motor and sensory paralysis distally. Likely multi-nerve injury (peroneal, tibial). | Possible nerve involvement (e.g., common peroneal nerve palsy with fibular fracture). Less likely complete paralysis. | Less common unless associated with significant compartment syndrome or direct nerve contusion. |
| Compartment Syndrome | Very high risk due to crush component, reperfusion injury post-vascular repair, and muscle edema. Clinical signs masked by open wounds/nerve injury. | Moderate to high risk. | Moderate risk if associated with significant hematoma or muscle injury. |
| Limb Salvage Score | MESS, LSI, PSI scores typically high (MESS >7-10 range), indicating high probability for primary amputation. | Usually indicative of salvage, but with high complication rates. | Usually salvageable with meticulous soft tissue management. |
| Prognosis | Guarded limb salvage; high complication rate (infection, non-union, chronic pain, significant functional deficit). Primary amputation a strong consideration. | High risk of infection, non-union, delayed union. Functional outcome often good if salvaged, but prolonged recovery. | Good prognosis if managed appropriately, but can lead to chronic pain, seroma, skin necrosis, scarring. |
| Initial Management | ATLS, aggressive resuscitation, haemorrhage control, early debridement, provisional external fixation, urgent vascular repair, fasciotomies, serial debridements, staged reconstruction. | ATLS, debridement, antibiotics, wound irrigation, external fixation, definitive fixation, soft tissue cover (often local/regional flap). | ATLS, wound debridement, compression, drainage (surgical/percutaneous), skin grafting/flaps if skin loss. |
Surgical Decision Making & Classification
The decision-making process for a mangled extremity is inherently complex, balancing potential for limb salvage against risks of prolonged morbidity and potential for a non-functional limb.
Limb Salvage vs. Primary Amputation
For this patient, the critical factors pointing towards primary amputation included:
*
Absolute Indicators:
Complete multi-vessel arterial transection (popliteal, anterior/posterior tibial arteries), extensive bone loss (5-6 cm tibial segment), complete motor and sensory paralysis of the foot (highly suggestive of irreparable tibial and common peroneal nerve injury), and profound muscle necrosis in multiple compartments.
*
Relative Indicators:
High-energy crush mechanism, significant contamination, requirement for prolonged complex reconstruction, and the patient's initial physiological instability.
The
Mangled Extremity Severity Score (MESS)
was calculated for this patient:
*
Skeletal/Soft-tissue Injury:
High (4 points for high-energy blunt trauma, severe crushing, significant bone loss)
*
Limb Ischemia:
>6 hours warm ischemia time due to transected popliteal artery (3 points, doubled for >6 hours = 6 points)
*
Shock:
Systolic BP <90 mmHg persistently (2 points)
*
Age:
Patient is 35 (0 points for <30 years, 1 point for 30-50 years, 2 points for >50 years). So, 1 point.
Total MESS Score = 4 + 6 + 2 + 1 = 13.
A MESS score of ≥ 7 is generally considered an indication for primary amputation. Our patient's score of 13 strongly supported primary amputation.
Despite the high MESS score and the severity of injury, a discussion was held with the patient and his family in the emergency setting. Given his young age, previously healthy status, and explicit desire to attempt limb salvage, the multidisciplinary team opted for a trial of salvage approach, emphasizing the high risk of failure and the potential need for secondary amputation. This decision required the immediate availability of a vascular surgeon for revascularization and an orthopedic trauma surgeon for damage control stabilization and extensive debridement.
Open Fracture Classification
This injury unequivocally falls into the
Gustilo-Anderson Classification Type IIIC
.
*
Type IIIC:
Characterized by an open fracture with an associated arterial injury requiring repair, regardless of the extent of soft tissue damage. The patient's transected popliteal artery and extensive tibial segment loss cemented this classification. The significant soft tissue loss and contamination would independently place it at least as a Gustilo-Anderson Type IIIB.
Staged Surgical Approach
A staged damage control orthopedics approach was deemed essential:
1.
Stage 1 (Emergent):
Resuscitation, haemorrhage control, gross decontamination, urgent vascular repair, four-compartment fasciotomies, and provisional skeletal stabilization with an external fixator.
2.
Stage 2 (Early Definitive):
Serial debridements every 24-48 hours until a clean wound bed is achieved, followed by definitive soft tissue coverage (likely free flap).
3.
Stage 3 (Later Reconstruction):
Definitive bony reconstruction (e.g., Masquelet technique, bone transport, or vascularized fibula graft) and potential nerve repair/reconstruction.
Surgical Technique / Intervention
The surgical intervention for this mangled extremity was a multi-stage, multidisciplinary effort involving Orthopedic Trauma, Vascular, and Plastic Surgery teams.
Operative Phase 1: Emergent Debridement, Vascular Repair, Fasciotomies, and Provisional Stabilization
The patient was transferred emergently to the operating theatre.
-
Patient Positioning and Preparation:
- Supine position on a radiolucent table, allowing C-arm access from both sides.
- Left lower extremity draped free for full mobility and access for both orthopedic and vascular teams. Contralateral leg also prepped and draped for potential autologous vein graft harvest (Great Saphenous Vein - GSV).
- Tourniquet remained inflated on the left thigh to control bleeding during initial setup.
-
Haemorrhage Control and Vascular Repair (Vascular Surgeon):
- The existing tourniquet was used for initial control.
- A generous medial incision was extended to expose the transected popliteal artery and distal tibial arteries. The ends of the popliteal artery were identified and found to have significant intimal damage and retraction.
- After heparinization, the contralateral great saphenous vein was harvested (4-5 cm segment).
- The damaged arterial ends were resected back to healthy vessel. An interposition reverse saphenous vein graft was performed to reconstruct the popliteal artery, restoring flow.
- The anterior and posterior tibial arteries were also identified, severely contused, and thrombosed distally. Given the extensive soft tissue damage and the re-established popliteal flow, primary repair of these distal vessels was deemed impractical and unlikely to improve overall limb viability. The focus remained on restoring primary inflow through the popliteal artery.
- Post-repair, flow was confirmed with excellent pulsatile Doppler signals in the foot and brisk back-bleeding. The tourniquet was released.
-
Debridement and Fasciotomies (Orthopedic Trauma Surgeon):
- Immediately after reperfusion, a systematic and aggressive debridement commenced.
- Irrigation: Copious lavage was performed with 9 liters of sterile normal saline using bulb syringe, ensuring meticulous cleaning of all recesses of the wound.
- Skin: All discolored, avulsed, and non-bleeding skin edges were sharply excised. The initial degloving injury was extended to ensure full visualization of the deep structures.
- Subcutaneous Tissue: All devitalized subcutaneous fat was removed.
- Muscle: This was the most critical step. The "four Cs" (Color, Consistency, Contractility, Capillary bleed) were strictly applied. All dusky, non-contractile, non-bleeding muscle was sharply excised. Significant portions of the anterior tibialis, extensor digitorum longus, and deep posterior compartment muscles (flexor digitorum longus, posterior tibialis) were debrided. The extent of muscle necrosis was profound.
- Bone: All grossly contaminated, unattached bone fragments were removed. The periosteum was stripped in large areas. The 5-6 cm segmental tibial defect was confirmed.
- Foreign Bodies: Meticulous search and removal of all foreign debris (dirt, gravel, clothing, pavement fragments).
- Fasciotomies: Given the crush injury, prolonged ischemia, and post-revascularization swelling, extensive four-compartment fasciotomies were performed in the lateral, anterior, deep posterior, and superficial posterior compartments through generous skin incisions. This was critical to prevent secondary compartment syndrome.
-
Provisional Skeletal Stabilization:
- An anterior spanning external fixator was applied to the tibia, bridging the segmental defect and the knee and ankle joints. Proximal pins were placed in the distal femur, and distal pins in the talus/calcaneus. This provided immediate rigid stabilization, restored length, reduced deformity, and maintained access to the extensive soft tissue wounds for subsequent debridements. Pins were placed carefully to avoid the revascularized popliteal artery and future flap donor sites.
The wound was packed with saline-soaked sterile gauze and covered with a sterile dressing. Negative pressure wound therapy (NPWT) was then applied over the entire wound and surrounding skin, set at continuous negative pressure (-125 mmHg).
Operative Phase 2: Serial Debridements and Soft Tissue Coverage
- Serial Debridements: The patient returned to the operating room for serial debridements every 24-48 hours for a total of four sessions. Each session involved re-assessment of tissue viability, further excision of any newly necrotic tissue, and copious irrigation. NPWT was re-applied between sessions. This iterative process ensured a clean, healthy, granulating wound bed.
-
Soft Tissue Coverage (Plastic Surgeon):
After the fourth debridement, a clean wound with healthy, viable muscle and granulation tissue was achieved. Given the large composite tissue defect, a
free anterolateral thigh (ALT) flap
was harvested from the contralateral thigh and microsurgically transferred to cover the extensive anterior and medial leg defect. The recipient vessels were branches of the anterior tibial artery (if viable) or veins from the flap anastomosed to a recipient vein. An additional split-thickness skin graft (STSG) was harvested from the ipsilateral thigh to cover the remaining smaller defects and the donor site of the ALT flap. The application of
would illustrate a general concept of staged reconstruction, perhaps depicting the sequence of debridement, temporary coverage (NPWT), and definitive flap application.
Operative Phase 3: Definitive Skeletal Reconstruction
- Timing: Following successful flap take and stability of the soft tissue envelope (approximately 3 weeks post-injury), the patient underwent definitive bony reconstruction.
-
Masquelet Technique:
Due to the large tibial segmental defect, the induced membrane technique (Masquelet technique) was chosen.
- The external fixator was removed.
- A custom-made polymethylmethacrylate (PMMA) cement spacer was placed precisely into the tibial bone defect, maintaining length and alignment. The soft tissues were carefully closed over the spacer. This allowed for the formation of a biological membrane around the spacer.
- Second Stage Masquelet (6-8 weeks later): The patient returned to theatre. The PMMA spacer was removed, revealing a healthy, vascularized synovial-like membrane. This membrane was preserved, and the defect was filled with copious autologous cancellous bone graft harvested from the ipsilateral iliac crest.
- Definitive Internal Fixation: A locked intramedullary nail was then inserted antegrade into the tibia, spanning the reconstructed segment, providing robust internal fixation. This protected the bone graft and allowed for early weight-bearing.
Post-Operative Protocol & Rehabilitation
The post-operative and rehabilitation phases for this patient were prolonged, intensive, and multidisciplinary.
Immediate Post-Operative Period (Hospital Stay)
- Critical Care Management: Initial days in ICU for close monitoring of flap viability (clinical checks, handheld Doppler), systemic stability, and early signs of infection.
- Pain Management: Epidural analgesia for the first few days, followed by a multimodal regimen including opioids and NSAIDs.
- Antibiotic Prophylaxis: Continued IV antibiotics for at least 72 hours post-flap surgery, guided by intraoperative cultures. Oral antibiotics then transitioned to for 6 weeks, given the severity of the open fracture and reconstruction.
- Wound and Flap Care: Meticulous observation of the ALT flap for color, turgor, capillary refill, and Doppler signals to detect any signs of vascular compromise. Careful dressing changes to protect the flap and skin grafts.
- DVT Prophylaxis: Low molecular weight heparin (LMWH) initiated on post-operative day 1, continued until full mobilization.
- Nutritional Support: Early involvement of dietitians to ensure adequate protein and caloric intake for wound healing and overall recovery.
- Early Mobilization: Gentle passive and active range of motion exercises for the knee and ankle (avoiding excessive stress on the flap and incision lines) initiated by physical therapists as soon as stability allowed. Bedside exercises for the upper extremities and contralateral limb.
Intermediate Rehabilitation (Weeks to Months)
-
Weight-Bearing Progression:
- Initially non-weight-bearing (NWB) for 6-8 weeks to allow for initial bone graft incorporation and soft tissue healing.
- After the second stage of the Masquelet procedure and intramedullary nail fixation, gradual touch-down weight-bearing (TDWB) was initiated, progressing to partial weight-bearing (PWB) based on radiographic signs of healing and clinical tolerance.
- Full weight-bearing (FWB) was anticipated at 3-6 months post-definitive fixation, depending on bone consolidation.
-
Physical Therapy (PT):
- Intensive, structured program focused on regaining joint range of motion, improving muscle strength, endurance, and proprioception.
- Emphasis on ankle dorsiflexion and plantarflexion, as well as knee flexion and extension.
- Gait training with assistive devices (walker, crutches) progressing to independent ambulation.
- Occupational Therapy (OT): Assessment and training for activities of daily living (ADLs), adaptive equipment, and home modifications.
- Scar Management: Silicone sheeting, massage, and compression garments to minimize hypertrophic scarring and contractures, particularly around the flap site and fascia incisions.
- Psychological Support: Continued counseling and support for the patient and family, addressing post-traumatic stress, body image issues, and the emotional burden of prolonged recovery.
Long-Term Rehabilitation and Functional Restoration (Months to Years)
-
Addressing Complications:
- Infection: Vigilant monitoring for signs of chronic osteomyelitis. Should it occur, further debridements, prolonged targeted antibiotic therapy, and potentially revision bone reconstruction would be required.
- Non-union/Delayed Union: Radiographic follow-up every 4-6 weeks to assess bone healing. Non-union would necessitate further surgical intervention (e.g., re-grafting, revision fixation, bone stimulators).
- Nerve Recovery: The complete paralysis made spontaneous functional recovery of the tibial and peroneal nerves unlikely. Follow-up with a neurosurgeon/peripheral nerve specialist was arranged for potential secondary nerve reconstruction (e.g., nerve grafting, tendon transfers for foot drop) if no improvement was noted by 6-9 months.
- Contractures and Joint Stiffness: Aggressive physical therapy, serial casting, or surgical release may be needed.
- Chronic Pain: Multidisciplinary pain clinic referral for management of neuropathic pain and chronic musculoskeletal pain.
- Functional Training: High-level functional activities, balance training, and sport-specific rehabilitation if desired.
- Vocational Rehabilitation: Assessment for return to work or retraining for a new career path, given the potential long-term functional limitations.
- Limb Length Discrepancy: Regular assessment. If significant, a shoe lift or future limb lengthening procedure may be considered.
Pearls & Pitfalls (Crucial for FRCS/Board Exams)
Pearls
- Life Over Limb: Always adhere to ATLS principles. A mangled extremity in an unstable patient necessitates stabilization before definitive limb interventions. Manage the patient, not just the limb.
- Early & Aggressive Debridement: The "King" of open fracture management. Serial debridement (every 24-72 hours) is critical until a visibly clean and viable wound bed is achieved. "The solution to pollution is dilution" – copious irrigation.
- Damage Control Orthopedics (DCO): For mangled extremities, initial external fixation is paramount. It provides rapid stabilization, allows access for serial debridements, and prevents further soft tissue stripping.
- Vascular Repair - Time is Muscle: For Gustilo IIIC injuries, revascularization is an emergency. Prioritize reperfusion within 6 hours of warm ischemia time to salvage muscle viability.
- Low Threshold for Fasciotomy: Always perform fasciotomies in crush injuries, after prolonged ischemia, after vascular repair, or when compartment syndrome is suspected.
- Multidisciplinary Team Approach: Essential for optimal outcomes. Orthopedic trauma, vascular, plastic surgery, anesthesia, critical care, infectious disease, and rehabilitation teams must collaborate.
- Timely Soft Tissue Coverage: Achieve wound closure or definitive coverage (e.g., local, regional, or free flap) within 5-7 days of injury (the 'golden window') to minimize infection risk and promote healing.
- Bone Management (Masquelet/Transport): For significant bone loss, plan for advanced techniques like the Masquelet induced membrane technique or bone transport with circular frames. Acute shortening can be considered if limb length discrepancy is acceptable and facilitates soft tissue closure.
- Nutrition and Prophylaxis: Aggressive nutritional support aids healing. Appropriate antibiotic prophylaxis and DVT prophylaxis are non-negotiable.
-
Documentation:
Meticulous recording of findings, debridement specifics, classifications, and decision-making processes is vital.
(A well-structured clinical pathway or decision algorithm for mangled extremity management could be represented by such an image, highlighting critical steps and inter-departmental handoffs.) - Patient Expectations: Manage patient and family expectations early and realistically regarding the arduous course, potential complications, and functional outcomes.
Pitfalls
- Missing Systemic Injuries: Focusing solely on the limb and overlooking other life-threatening injuries (head, chest, abdomen, pelvis).
- Inadequate Debridement: The primary reason for infection and failed salvage. Hesitation to remove all non-viable tissue.
- Delayed Vascular Repair: Prolonged ischemia leads to irreversible muscle necrosis and increases the likelihood of amputation or severe functional deficit.
- Missed Compartment Syndrome: Failure to diagnose and treat compartment syndrome expeditiously leads to irreversible muscle and nerve damage (e.g., Volkmann's contracture). Clinical signs can be challenging in a mangled limb or anesthetized patient.
- Premature Definitive Internal Fixation: Placing plates or nails in a contaminated, unstable soft tissue envelope leads to devastating deep infections and hardware failure. External fixation is generally preferred initially.
- Delayed Soft Tissue Coverage: Leaving large open wounds for too long leads to desiccation, increased bacterial colonization, and higher rates of infection.
- Inappropriate Antibiotic Regimen: Incorrect choice, duration, or failure to adjust based on culture results and clinical response can lead to resistant infections.
- Lack of Multidisciplinary Coordination: Poor communication and planning between orthopedic, vascular, and plastic surgery teams result in fragmented care and suboptimal outcomes.
- Ignoring Nerve Injuries: Failing to assess nerve function or to consider nerve reconstruction in the long term leads to significant functional deficits.
- Over-reliance on Limb Salvage Scores: While helpful, MESS and other scores are guides. Clinical judgment, patient's physiological status, and patient preference are paramount.
- Insufficient Rehabilitation: Early immobilization, inadequate physical therapy, and lack of long-term functional training can lead to stiffness, weakness, and poor overall outcome, even with successful surgical salvage.
You Might Also Like