Osteonecrosis (AVN): Pathophysiology, Epidemiology & Clinical Manifestations
Key Takeaway
Osteonecrosis (ON) is the death of bone cells due to interrupted blood supply, leading to structural failure. Key mechanisms include vascular insult, cellular dysfunction, marrow edema, and coagulation abnormalities, often affecting the femoral head. It progresses from silent bone death to subchondral collapse and severe osteoarthritis.
Introduction & Epidemiology
Osteonecrosis (ON), also referred to as avascular necrosis (AVN) or aseptic necrosis, represents a pathological condition characterized by the death of osteocytes and bone marrow cells due to an interruption of the vascular supply in the absence of infection. This cellular demise ultimately leads to structural failure of the affected bone, most notably subchondral collapse, often resulting in secondary degenerative arthritis. While it can affect any bone, the femoral head is the most frequently involved site, accounting for a significant proportion of cases requiring total hip arthroplasty in younger patient populations.
The fundamental insult in osteonecrosis is a disruption of bone viability. The assessment of bone viability is complex, but histopathological examination remains the gold standard, demonstrating empty lacunae and necrotic marrow. Biochemical markers, such as hydroxyproline levels , reflecting collagen turnover, may show alterations, though their specific diagnostic utility for early ON remains a subject of ongoing research. Elevated hydroxyproline levels, indicating increased collagen degradation, might paradoxically be seen in reparative phases, while severely ischemic areas would show a profound reduction in metabolic activity.
Epidemiologically, ON exhibits a bimodal distribution, affecting both younger and older adults. The estimated incidence of ON of the femoral head (ONFH) in the United States is approximately 10,000 to 20,000 new cases annually, with men more commonly affected than women, particularly in the 30-50 year age group. The etiology significantly influences prevalence; corticosteroid-induced ONFH and alcohol-associated ONFH are among the most common non-traumatic forms. Idiopathic ONFH accounts for a substantial percentage, highlighting the multifactorial and often cryptogenic nature of the disease. While the hip is overwhelmingly the primary site, other joints such as the knee, shoulder, ankle, and jaw (medication-related osteonecrosis of the jaw, MRONJ) can also be affected.
The pathophysiology of osteonecrosis is multifaceted, involving a complex interplay of vascular, cellular, and biomechanical factors. Key mechanisms and associated conditions include:
- Vascular Insult and Ischemia: The primary event is an interruption of blood supply. This can be macroscopic, such as following trauma (e.g., femoral neck fracture, hip dislocation, slipped capital femoral epiphysis [SCFE]), or microscopic, involving thrombosis, fat embolization, or vascular compression within the bone marrow.
-
Cellular Dysfunction:
- While less commonly cited as a primary driver, specific research has identified virus-like inclusion bodies in osteoclasts , suggesting potential abnormal function of osteoclasts. This could imply a role for dysregulated bone resorption in disease progression, contributing to structural weakening prior to or concurrent with osteocyte death. Such an observation prompts further investigation into infectious or inflammatory triggers influencing cellular processes within the bone microenvironment.
- Osteocyte death initiates a cascade of events. Dead osteocytes lose their ability to maintain bone matrix and respond to mechanical stimuli, leading to microfractures.
- Marrow Edema and Intramedullary Pressure: Osteonecrosis may be related to enlargement of space-occupying marrow fat cells, which lead to ischemia of adjacent tissues. This "fat emboli" theory suggests that adipocyte hypertrophy or necrosis within the bone marrow can cause an increase in intramedullary pressure, compressing the fragile intraosseous capillaries and leading to ischemia. This mechanism is particularly implicated in steroid-induced ON.
- Coagulation Abnormalities: A significant proportion of patients with idiopathic osteonecrosis of the femoral head and Legg-Calvé-Perthes disease (a form of pediatric ONFH) demonstrate underlying thrombophilia. This includes a deficiency of antithrombin factors protein C and protein S, and increased levels of lipoprotein(a). These hypercoagulable states predispose to microthrombosis within the small terminal arterioles supplying the bone, resulting in focal ischemia. Genetic predispositions to hypercoagulability are increasingly recognized as contributors.
-
Disease Progression and Radiographic Appearance:
The appearance of osteonecrosis on imaging studies evolves with the phase of the disease:
- Early Phase (ARCO Stage 0-I): Often asymptomatic or subtle. Histologically, osteocyte death is present. Radiographs are typically normal. MRI is critical for early diagnosis, showing bone marrow edema and characteristic low signal on T1-weighted images and high signal on T2-weighted images with a serpiginous low-signal rim.
-
Active Phase (ARCO Stage I-II):
Symptomatic, with developing radiographic changes.
- Lytic phase: Intense osteoclastic bone resorption. This represents the body's attempt at repair, where necrotic bone is removed. Radiographically, areas of lucency or osteopenia may be seen.
- Mixed phase: A combination of lytic and sclerotic changes.
- Sclerotic phase: Osteoblastic bone formation occurs in an attempt to wall off the necrotic area or repair it. This results in increased osteodensities, particularly at the reactive interface zone.
-
Later Phase (ARCO Stage III-IV):
Collapse of the subchondral bone, leading to mechanical failure.
- Subchondral collapse: Characterized by the "crescent sign" on radiographs, indicating a fracture of the subchondral bone plate.
- Articular cartilage collapse: The overlying articular cartilage loses its support, leading to flattening of the femoral head, most frequently in the anterolateral region, and eventually secondary osteoarthritis.
-
Associated Systemic Conditions:
- Steroids: The most common non-traumatic cause. High-dose, long-term corticosteroid use is a significant risk factor, likely due to fat cell hypertrophy and embolization, vasculitis, or coagulation abnormalities.
- Heavy Alcohol Use: Mechanism thought to involve fat emboli from fatty liver and hyperlipidemia, leading to vascular occlusion.
-
Blood Dyscrasias:
- Sickle Cell Disease: Vaso-occlusive crises directly compromise blood supply. Repeated sickling events in the microvasculature of bone lead to ischemia and infarction.
- Gaucher Disease: Accumulation of glucocerebroside-laden macrophages in the bone marrow causes increased intramedullary pressure and vascular compression.
- Other myeloproliferative disorders.
- Dysbarism (Caisson Disease): Seen in divers and compressed air workers. Rapid decompression leads to nitrogen bubble formation within blood vessels and tissues, causing vascular occlusion and fat emboli.
- Excessive Radiation Therapy: Damages endothelial cells and leads to progressive endarteritis and fibrosis, compromising blood supply.
- Systemic Lupus Erythematosus (SLE): Likely multifactorial, involving vasculitis, corticosteroid use, and possibly hypercoagulability.
- Organ Transplantation: Often associated with high-dose corticosteroid therapy.
- HIV Infection: Direct viral effects, immune dysregulation, and antiretroviral therapy (particularly protease inhibitors) are implicated.
- Pancreatitis: Fat embolization from damaged pancreatic tissues.
- Bisphosphonate-Related Osteonecrosis of the Jaw (BRONJ) / Medication-Related Osteonecrosis of the Jaw (MRONJ): A distinct entity linked to antiresorptive (bisphosphonates, denosumab) and antiangiogenic medications.
Understanding these diverse etiologies and pathophysiological mechanisms is crucial for appropriate diagnosis, staging, and management of osteonecrosis, particularly when considering surgical interventions.
Surgical Anatomy & Biomechanics
Successful surgical intervention for osteonecrosis, particularly of the femoral head (ONFH), necessitates an intimate understanding of the region's complex anatomy, especially its vascular supply and biomechanical properties.
The femoral head receives its blood supply primarily from the medial femoral circumflex artery (MFCA) and, to a lesser extent, the lateral femoral circumflex artery (LFCA), both branches of the profunda femoris artery. These arteries give rise to ascending cervical arteries, which pierce the capsule at its reflections and ascend along the femoral neck as retinacular vessels (superior, inferior, anterior, and posterior). The superior retinacular vessels, originating from the MFCA, are the dominant blood supply to the superolateral weight-bearing portion of the femoral head—the region most susceptible to ONFH due to its terminal arterial supply and vulnerability. The artery of the ligamentum teres, a branch of the obturator artery, provides a variable and often insufficient supply in adults but can be more significant in children.

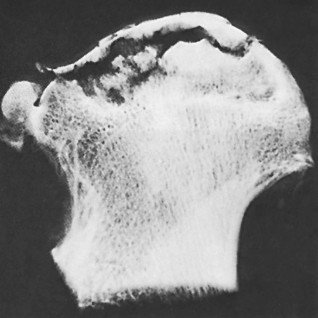
The image provided depicts a clear representation of the vascular anatomy of the femoral head, highlighting the critical role of the medial circumflex femoral artery's retinacular branches. The susceptibility of the superolateral aspect of the femoral head to ischemic necrosis becomes evident when considering this terminal vascular pattern. Disruption to these retinacular vessels, whether by trauma, increased intramedullary pressure, or microvascular occlusions, directly leads to osteocyte death in the dependent bone.
Biomechanically, the femoral head is designed to withstand significant compressive and shear forces during weight-bearing and activity. The trabecular bone within the femoral head is organized into primary compressive and tensile groups, distributing stress efficiently. The subchondral bone plate acts as a critical interface, distributing loads from the articular cartilage to the underlying trabecular bone. In ONFH, the death of osteocytes weakens the trabecular bone, leading to microfractures. As the disease progresses, the structural integrity of the subchondral bone is compromised, culminating in the "crescent sign" (subchondral fracture) and subsequent collapse of the articular surface. This collapse is particularly devastating in the anterolateral weight-bearing region of the femoral head, leading to incongruity, pain, and accelerated degenerative changes within the hip joint. The extent and location of the necrotic lesion directly impact the structural stability and predict the likelihood of collapse. Lesions involving more than 30-50% of the femoral head or affecting critical weight-bearing areas are at higher risk of progression.
Understanding the precise location of the necrotic lesion relative to the femoral head's vascularity and weight-bearing zones is paramount for surgical planning, informing decisions between joint-preserving procedures (e.g., core decompression, osteotomy, grafting) and joint-sacrificing procedures (e.g., total hip arthroplasty).
Indications & Contraindications
The management of osteonecrosis is complex and depends heavily on the stage of the disease, the size and location of the lesion, the patient's age, activity level, and the underlying etiology. The primary goals are to alleviate pain, prevent or delay articular surface collapse, and preserve hip function.
Non-Operative Indications:
Non-operative management is typically reserved for early-stage disease, small lesions, or patients with significant contraindications to surgery.
*
Asymptomatic Patients:
Rarely seen in clinic, but if diagnosed incidentally in very early stages (e.g., ARCO Stage 0-I) without pain, watchful waiting may be considered.
*
Early Stage Disease (ARCO Stage I, Ficat-Arlet Stage I/IIa):
Before subchondral collapse (crescent sign).
* Small lesions (e.g., less than 15-20% of the femoral head volume, or <15 mm length).
* Limited weight-bearing, activity modification.
* Pharmacotherapy:
*
Bisphosphonates:
May inhibit osteoclast activity, reducing bone resorption and potentially preventing collapse. Data are mixed, but some studies show promise in early stages.
*
Statins:
Proposed mechanisms include cholesterol reduction, improved endothelial function, and anti-inflammatory effects.
*
Anticoagulants:
For patients with thrombophilia, to prevent further microthrombosis.
*
Vasodilators (e.g., Iloprost):
To improve microcirculation.
*
Electrical Stimulation/PEMF:
Controversial, with limited high-quality evidence.
*
Legg-Calvé-Perthes Disease:
In children, initial management is almost always non-operative, focusing on containment and allowing for revascularization and remodeling. Surgical intervention (osteotomy) is considered if non-operative measures fail or for specific radiographic patterns.
*
Significant Medical Comorbidities:
Patients with severe cardiac, pulmonary, or other systemic diseases that preclude surgical intervention.
Operative Indications:
Surgical intervention is generally indicated for symptomatic patients, particularly those with progressive disease or risk of collapse. The choice of procedure depends on the stage.
-
Joint-Preserving Procedures (ARCO Stage I/II, Pre-collapse):
Aim to prevent collapse and promote healing.
-
Core Decompression:
- Indications: ARCO Stage I, IIa, IIb (pre-collapse), especially for small to medium-sized lesions. Patients are typically younger with good articular cartilage.
- Goal: Reduce intramedullary pressure, allow for revascularization, provide a channel for reparative cells.
-
Vascularized/Non-Vascularized Bone Grafting:
- Indications: ARCO Stage IIb/c, some early Stage III (with intact articular cartilage). Larger lesions not amenable to core decompression alone. Used to provide structural support and/or introduce viable bone with a blood supply.
- Commonly involves fibular autograft.
-
Osteotomy (e.g., Rotational Osteotomy):
- Indications: ARCO Stage IIb/c, early Stage III with a well-demarcated lesion occupying a non-weight-bearing portion after rotation. Younger patients with good bone stock and intact articular cartilage over a significant portion of the weight-bearing surface.
- Goal: Move the necrotic segment out of the primary weight-bearing area.
-
Core Decompression:
-
Joint-Sacrificing Procedures (ARCO Stage III/IV, Post-collapse):
When joint preservation is no longer feasible.
-
Total Hip Arthroplasty (THA):
- Indications: ARCO Stage III (subchondral collapse with incongruity) and Stage IV (secondary osteoarthritis). Patients with significant pain, functional limitation, and failed previous joint-preserving procedures. Generally considered the gold standard for end-stage ONFH.
- Hemiarthroplasty: Rarely indicated for ONFH due to high rates of acetabular erosion, typically reserved for very low-demand elderly patients.
-
Total Hip Arthroplasty (THA):
Contraindications:
*
Absolute Contraindications:
* Active local or systemic infection.
* Rapidly progressive joint destruction (e.g., Charcot arthropathy, severe inflammatory arthritis) unless it's a THA.
* Uncontrolled systemic illness precluding safe anesthesia and surgery.
* Active local malignancy.
*
Relative Contraindications:
* Patient non-compliance.
* Severe generalized osteoporosis (for joint-preserving procedures).
* End-stage disease for joint-preserving procedures (e.g., ARCO Stage IV).
* Ongoing uncontrolled etiologic factors (e.g., active alcohol abuse without cessation, continued high-dose steroid use without careful risk-benefit analysis).
Table 1: Operative vs. Non-Operative Indications for Femoral Head Osteonecrosis
| Treatment Modality | Primary Indications | Primary Contraindications |
|---|---|---|
| Non-Operative | - ARCO Stage 0-I (asymptomatic/mild symptoms) | - Symptomatic progression |
| (Activity Modification, | - Small lesions (<15-20% involvement, <15mm) | - Subchondral collapse (crescent sign) |
| Pharmacotherapy) | - Children with Legg-Calvé-Perthes disease | - Rapid disease progression |
| - Significant medical comorbidities precluding surgery | ||
| Operative: Joint-Preserving | ||
| Core Decompression | - ARCO Stage I, IIa, IIb (pre-collapse) | - ARCO Stage III, IV (subchondral/articular collapse) |
| - Small to medium-sized lesions | - Large lesions (>30-50% involvement) | |
| - Younger patients with good articular cartilage | - Active infection | |
| **Vascularized/Non- | - ARCO Stage IIb/c, early Stage III (intact articular cartilage) | - ARCO Stage IV (severe osteoarthritis) |
| Vascularized Bone Graft** | - Larger lesions than amenable to core decompression alone | - Active infection, severe systemic comorbidities |
| - Younger patients where joint preservation is paramount | - Inadequate recipient vessels (for vascularized graft) | |
| Rotational Osteotomy | - ARCO Stage IIb/c, early Stage III (well-demarcated lesion, intact articular cartilage in potential new weight-bearing area) | - ARCO Stage IV (severe osteoarthritis) |
| - Younger patients with good bone stock and compliance | - Diffuse ON, large cystic lesions, significant shortening | |
| - Lesion amenable to relocation from weight-bearing zone | - Inadequate bone stock for stable fixation | |
| Operative: Joint-Sacrificing | ||
| Total Hip Arthroplasty | - ARCO Stage III (subchondral collapse, painful incongruity) | - Active infection |
| - ARCO Stage IV (secondary osteoarthritis, severe pain, functional limitation) | - Uncontrolled systemic illness | |
| - Failed previous joint-preserving procedures | - Neuropathic joint (relative) |
Pre-Operative Planning & Patient Positioning
Meticulous pre-operative planning is paramount for optimizing outcomes in surgical management of osteonecrosis. This involves comprehensive patient evaluation, detailed imaging analysis, and precise surgical strategy formulation.
1. Patient Evaluation:
*
History and Physical Examination:
A thorough history should identify risk factors (steroid use, alcohol abuse, sickle cell, trauma, etc.), duration and character of pain, impact on function, and previous treatments. Physical examination includes range of motion, gait assessment, and neurovascular status.
*
Systemic Workup:
Given the frequent association with systemic conditions, a comprehensive medical workup is essential. This includes assessing cardiovascular, pulmonary, renal, and endocrine status. Coagulation profiles should be evaluated, especially in idiopathic cases, for inherited or acquired thrombophilias (Protein C/S deficiency, Factor V Leiden, lipoprotein(a) levels). This may guide perioperative prophylaxis or long-term management.
*
Informed Consent:
Detailed discussion with the patient regarding the diagnosis, disease progression, proposed surgical procedure, potential risks (infection, nerve injury, non-union, progression of ON, need for further surgery including THA), benefits, and alternative treatments. For joint-preserving procedures, the potential for failure and subsequent THA must be explicitly communicated.
2. Imaging Analysis:
*
Plain Radiographs:
Anteroposterior (AP) pelvis, AP hip, and frog-leg lateral views are standard. Radiographs are crucial for staging using systems like Ficat-Arlet (Stages 0-IV) or ARCO (Stages 0-IV). They identify early sclerotic changes, the "crescent sign" (subchondral fracture), flattening of the femoral head, and joint space narrowing. However, they are often normal in early stages.
*
Magnetic Resonance Imaging (MRI):
The most sensitive imaging modality for early diagnosis of ONFH, often detecting lesions before radiographic changes. T1-weighted images typically show low signal intensity due to marrow necrosis, while T2-weighted images often reveal a "double-line sign" (a serpiginous low-signal rim with an inner high-signal line), highly characteristic of ON. MRI accurately delineates lesion size, extent, and location, which is critical for surgical planning (e.g., determining the entry point for core decompression or the extent of fibular graft needed).
*
Computed Tomography (CT) Scan:
Useful for assessing the precise morphology of the femoral head, particularly for planning osteotomies or when complex structural grafting is considered. It provides excellent detail of the cortical and trabecular bone architecture, subchondral collapse, and any associated cysts.
*
Bone Scan (Technetium-99m):
Can show decreased uptake in the necrotic area (cold spot) in early ON, followed by increased peripheral uptake (hot spot) during the reparative phase. Less specific than MRI but can be used in cases where MRI is contraindicated.
3. Surgical Strategy Formulation:
*
Staging and Lesion Characteristics:
The ARCO staging system (Association Research Circulation Osseous) is preferred for its comprehensiveness and prognostic value, classifying ONFH based on imaging findings (MRI, X-ray) from Stage 0 (pre-clinical) to Stage IV (osteoarthritis). Lesion size (e.g., small, medium, large, based on percentage of femoral head involvement) and location (anterolateral is most common and critical) are key determinants.
*
Procedure Selection:
Based on stage, lesion characteristics, patient age, and activity. For example, ARCO Stage I/II often dictates joint-preserving strategies (core decompression, grafting, osteotomy), while ARCO Stage III/IV typically requires total hip arthroplasty.
*
Instrumentation and Implants:
Confirm availability of appropriate drills, reamers, bone graft material (autograft/allograft), osteotomy jigs, fixation devices (screws, plates), and prostheses (for THA). If vascularized grafting is planned, microsurgical instruments and an experienced microvascular team are essential.
*
Intraoperative Imaging:
C-arm fluoroscopy is indispensable for all joint-preserving procedures, ensuring accurate targeting of the necrotic lesion and confirming placement of grafts or osteotomy cuts.
4. Patient Positioning:
The specific surgical procedure dictates patient positioning to optimize exposure and access.
-
Core Decompression / Vascularized Fibular Graft / Anterior Rotational Osteotomy:
- Supine Position: Standard for anterolateral or direct lateral approaches.
- Setup: The patient is positioned supine on a radiolucent table. The affected hip is brought to the edge of the table, or placed on a fracture table to allow for hip flexion/extension and rotation for C-arm access. A bump can be placed under the ipsilateral buttock to facilitate internal rotation.
- C-arm Access: The C-arm should be positioned to allow for immediate AP and lateral views of the hip without repositioning the patient or the limb. The contralateral leg may be abducted or placed in a leg holder to allow for C-arm sweep.
- Draping: Sterile draping should allow for full range of motion of the hip and provide access to potential graft harvest sites (e.g., fibula, iliac crest) if autografting is planned.
-
Total Hip Arthroplasty (THA):
- Lateral Decubitus Position: Common for posterior or direct lateral approaches. The patient is positioned on their unaffected side, stabilized with bolsters or beanbags. The affected limb is prepped and draped to allow for full range of motion.
- Supine Position: Used for direct anterior or anterolateral approaches. Similar setup as for core decompression, but often with a specialized table (e.g., Hana table) for direct anterior approach to facilitate hip hyperextension and external rotation.
-
Posterior Rotational Osteotomy:
- Lateral Decubitus or Prone Position: May be used depending on surgeon preference and specifics of the osteotomy. Prone position offers excellent posterior exposure but is more complex to set up.
Careful attention to patient positioning is crucial to prevent pressure injuries, nerve palsies, and ensure optimal surgical exposure and intraoperative imaging.
Detailed Surgical Approach / Technique
The detailed surgical approach for osteonecrosis is highly dependent on the stage of the disease, lesion characteristics, and patient factors. This section will focus on common joint-preserving strategies for ONFH (Core Decompression, Vascularized Fibular Grafting, Rotational Osteotomy) and briefly touch upon Total Hip Arthroplasty for end-stage disease.
1. Core Decompression
Indications: ARCO Stages I, IIa, IIb (pre-collapse), small to medium lesions, typically in younger patients. Aims to relieve intramedullary pressure, allow revascularization, and provide access for biological augmentation.
Approach & Positioning:
* Patient supine on a radiolucent table, affected limb prepped and draped to allow for full range of motion.
* C-arm fluoroscopy essential for accurate targeting.
* Incision: Typically a 2-4 cm longitudinal incision over the lateral aspect of the proximal femur, distal to the greater trochanter, or an anterolateral incision. The choice of incision depends on the intended trajectory and surgeon preference.
Technique:
*
Skin Incision & Fascial Dissection:
Incision through skin and subcutaneous tissue. The fascia lata is incised longitudinally. The vastus lateralis muscle is split or incised and reflected to expose the lateral femoral cortex.
*
Entry Point Selection:
Using C-arm guidance (AP and lateral views), an entry point is marked on the lateral femoral cortex. This point is typically 1-2 cm distal to the vastus ridge (junction of the greater trochanter and femoral shaft) and slightly posterior to the midline of the femoral shaft in the lateral view to avoid damage to the piriformis fossa and greater trochanteric apophysis. The trajectory aims for the necrotic lesion, typically in the anterosuperior aspect of the femoral head.
*
Pilot Hole & Guide Wire Insertion:
A small drill bit (e.g., 3.2 mm) or an awl is used to create a pilot hole through the lateral cortex. A cannulated guide wire (e.g., 2.5 mm) is then advanced under continuous fluoroscopic guidance, aiming to reach the necrotic lesion within the femoral head. The tip of the guide wire should be approximately 5-10 mm from the articular surface to avoid penetration and cartilage damage. The anteroposterior and lateral views are crucial to confirm correct positioning.
*
Reaming/Decompression:
* Once the guide wire is optimally positioned, a cannulated drill bit (e.g., 8-10 mm diameter) is advanced over the guide wire to core out a channel through the femoral neck and into the necrotic zone. Multiple channels can be created, depending on lesion size and extent. Some surgeons prefer to use successively larger drills or a trephine.
* The goal is to remove necrotic bone and marrow, thereby decompressing the intraosseous pressure. The core specimen can be sent for histopathological analysis.
*
Biological Augmentation (Optional but Increasingly Common):
* Following core decompression, the channel can be filled with biological adjuncts to promote healing and revascularization.
*
Bone Marrow Aspirate Concentrate (BMAC):
Harvested from the ipsilateral iliac crest, concentrated, and injected into the decompression channel or mixed with a bone graft. Contains mesenchymal stem cells and growth factors.
*
Autologous Bone Graft (e.g., from iliac crest):
Provides structural support and osteoconductive scaffold.
*
Allograft (e.g., demineralized bone matrix):
Osteoconductive.
*
Synthetic Scaffolds/Growth Factors:
Experimental or emerging options.
*
Closure:
The drill hole in the cortex is often left open or plugged with bone wax. The vastus lateralis is repaired, fascia lata closed, and skin closed in layers.
2. Vascularized Fibular Grafting
Indications: Larger lesions (ARCO Stage IIc, early Stage III), younger patients, failure of core decompression, when structural support and revascularization are needed.
Approach & Positioning:
* Requires two surgical teams simultaneously: one for fibula harvest, one for hip exposure.
*
Hip Exposure:
Anterolateral approach to the hip is common. Patient supine.
* Incision: Typically a modified Hardinge approach (direct lateral) or Watson-Jones approach (anterolateral). Dissection proceeds between tensor fascia lata and gluteus medius. The anterior capsule is incised to access the femoral head.
*
Fibular Harvest:
Usually from the ipsilateral leg (to minimize overall surgical time and facilitate patient positioning).
* Incision: Longitudinal incision over the lateral aspect of the lower leg.
* Dissection: Careful dissection to identify and preserve the peroneal artery and venae comitantes, which supply the fibula. Muscles (peroneus longus and brevis, soleus) are detached from the fibula.
* Harvest: A segment of fibula (typically 12-15 cm) is osteotomized, preserving the vascular pedicle.
* Closure: Meticulous closure of the donor site to prevent hematoma and nerve injury.
Technique (Hip):
*
Femoral Head Access:
After exposing the femoral head, a channel is created from the intertrochanteric region or femoral neck, directed into the necrotic lesion. This channel needs to accommodate the fibular graft. A cannulated drill or osteotome can be used.
*
Debridement:
The necrotic bone within the channel and lesion is debrided as thoroughly as possible.
*
Graft Insertion:
The vascularized fibular graft is carefully inserted into the prepared channel in the femoral head. It should be seated firmly, preferably extending to the subchondral bone, providing structural support.
*
Microvascular Anastomosis:
This is the most critical step. The vascular pedicle of the fibular graft (peroneal artery and venae comitantes) is anastomosed to suitable recipient vessels in the hip region (e.g., branches of the lateral femoral circumflex artery or deep femoral artery). This requires microsurgical expertise.
*
Closure:
Capsule, muscle layers, and skin are closed. A drain may be placed.
3. Rotational Osteotomy
Indications: Pre-collapse (ARCO Stage IIb/c) or early collapse (ARCO Stage III) with a well-demarcated lesion, typically in younger patients with significant intact articular cartilage. The lesion must be localized such that it can be rotated out of the primary weight-bearing zone.
Approach & Positioning:
* Patient supine on a radiolucent table. C-arm is essential.
* Approach depends on the planned osteotomy (anterior, posterior, or transtrochanteric).
*
Anterior Rotational Osteotomy:
Anterolateral approach to the proximal femur.
*
Posterior Rotational Osteotomy:
Direct posterior or posterolateral approach, often requiring partial trochanteric osteotomy.
Technique (Example: Anterior Rotational Osteotomy):
*
Exposure:
Standard anterolateral approach to the proximal femur and femoral neck. The anterior capsule is partially released.
*
Osteotomy Planning:
Based on pre-operative CT and intraoperative fluoroscopy, the precise location and angle of the osteotomy are planned to rotate the necrotic segment away from the weight-bearing dome. The osteotomy is typically transtrochanteric or subcapital.
*
Osteotomy Cut:
A Gigli saw or oscillating saw is used to perform the planned osteotomy, creating a wedge or dome-shaped cut. Careful protection of soft tissues and neurovascular structures is critical.
*
Rotation:
The distal segment of the femur is rotated relative to the proximal segment, effectively moving the necrotic zone to a non-weight-bearing area and bringing healthy bone into the weight-bearing region.
*
Fixation:
The osteotomy is stabilized with appropriate internal fixation (e.g., dynamic hip screw, angled blade plate, locking plates and screws). The fixation must be robust to allow for early mobilization while protecting the osteotomy site.
*
Closure:
Layered closure of muscle, fascia, and skin.
4. Total Hip Arthroplasty (THA)
Indications: End-stage ONFH (ARCO Stage III with significant collapse and incongruity, or Stage IV with secondary osteoarthritis), severe pain, functional limitation, failure of joint-preserving procedures.
Approach & Positioning:
* Patient positioning depends on the chosen surgical approach (e.g., lateral decubitus for posterior or direct lateral approach, supine for direct anterior approach).
* Standard preparation and draping for hip arthroplasty.
Technique:
*
Exposure:
Standard surgical approach (e.g., posterior, direct lateral, direct anterior) to the hip joint.
*
Femoral Neck Osteotomy:
The femoral neck is osteotomized to remove the femoral head.
*
Acetabular Reaming:
The acetabulum is reamed to prepare for the acetabular component, ensuring proper sizing and inclination. The acetabular component (cemented or uncemented) is then impacted or screwed into place.
*
Femoral Preparation:
The femoral canal is prepared with rasps to accommodate the femoral stem. The stem (cemented or uncemented) is then inserted.
*
Trial Reduction:
Trial components are used to assess leg length, offset, stability, and range of motion.
*
Definitive Implants:
The definitive femoral head and acetabular liner are implanted.
*
Closure:
Layered closure, often with drains. Hip precautions are usually instituted post-operatively depending on the approach.
Each of these surgical techniques requires meticulous attention to detail, comprehensive anatomical knowledge, and careful intraoperative assessment to achieve optimal outcomes and minimize complications.
Complications & Management
Surgical interventions for osteonecrosis, like all orthopedic procedures, carry inherent risks of complications. Understanding these complications, their incidence, and appropriate management strategies is critical for patient counseling and optimizing outcomes.
General Complications (Applicable to most hip surgeries):
- Infection: Superficial or deep surgical site infection. Incidence varies (0.5-2%). Management involves antibiotics, debridement, and potentially implant removal in severe cases (e.g., two-stage revision for THA).
- Thromboembolic Events: Deep vein thrombosis (DVT) and pulmonary embolism (PE). Incidence varies (0.5-5%). Prophylaxis (mechanical and chemical) is essential. Management includes anticoagulation; severe cases may require thrombolysis or embolectomy.
- Neurovascular Injury: Damage to sciatic nerve, femoral nerve, lateral femoral cutaneous nerve, or major vessels. Incidence is low (<1-2%). Management involves meticulous surgical technique, intraoperative monitoring, and, if injured, observation or surgical repair.
- Hematoma Formation: Can lead to increased pain, swelling, and risk of infection. Managed with drainage.
- Persistent Pain: May occur despite technically successful surgery, due to ongoing disease, adjacent joint pathology, or neuropathic pain.
Procedure-Specific Complications:
1. Core Decompression:
*
Femoral Neck Fracture:
Incidence 1-5%. Due to weakening of the femoral neck from drilling, especially with multiple or large channels, or aggressive weight-bearing. Salvage: Internal fixation (e.g., cannulated screws) or THA if fragment non-union or collapse occurs.
*
Subchondral Collapse/Articular Cartilage Damage:
If the drill penetrates the articular cartilage or does not adequately decompress the lesion, or if the lesion progresses. Incidence can be up to 30% if the disease progresses. Salvage: Grafting, osteotomy, or THA.
*
Inadequate Decompression/Disease Progression:
The most common failure, leading to continued pain and eventual collapse. Incidence varies (20-40% at 2-5 years). Salvage: Conversion to vascularized graft, osteotomy, or THA.
*
Nerve Injury:
Lateral femoral cutaneous nerve (LFCN) dysesthesia if approach too anterior. Incidence <1%. Salvage: Observation, nerve blocks, or neurectomy for persistent symptoms.
2. Vascularized Fibular Grafting:
*
Donor Site Morbidity:
*
Fibular Fracture:
Incidence 5-10%. During harvest or post-operatively, especially if too much fibula is removed or patient weight-bears prematurely. Salvage: Internal fixation, bracing, protected weight-bearing.
*
Peroneal Nerve Injury:
Incidence <1%. During exposure of the fibula, leading to foot drop. Salvage: Observation, AFO, nerve repair.
*
Compartment Syndrome:
Rare, due to hematoma or muscle swelling. Salvage: Fasciotomy.
*
Graft Failure:
*
Vascular Thrombosis:
The most critical complication, leading to necrosis of the fibular graft. Incidence 5-15%, often within the first few days. Salvage: Immediate re-exploration and microvascular revision.
*
Non-Union of Graft:
Failure of the fibular graft to incorporate into the femoral head. Incidence 5-10%. Salvage: Revision grafting, internal fixation, or conversion to THA.
*
Graft Collapse/Progression of ON:
Despite successful vascularization, the femoral head may still collapse or the ON may progress if the lesion is too large or underlying factors are not controlled. Incidence up to 20-30% long-term. Salvage: Osteotomy or THA.
*
Infection:
Similar to core decompression, but potentially more complex due to two surgical sites.
3. Rotational Osteotomy:
*
Non-Union or Malunion:
Failure of the osteotomy to heal, or healing in an unacceptable position. Incidence 5-15%. Salvage: Revision internal fixation, revision osteotomy, or THA.
*
Avascular Necrosis of Osteotomized Segment:
If the remaining blood supply to the rotated segment is compromised. Rare, but potentially devastating. Salvage: THA.
*
Hardware Failure:
Breakage or loosening of plates/screws. Incidence 5-10%. Salvage: Revision internal fixation, often with stronger implants.
*
Sciatic Nerve Palsy:
Rare, particularly with posterior approaches due to retraction. Incidence <1%. Salvage: Observation, neurolysis.
*
Shortening/Leg Length Discrepancy:
If significant bone is resected or the osteotomy collapses. Salvage: Limb lengthening procedures or shoe lifts.
*
Progression of ON / Secondary OA:
If the initial lesion was too large, or if the disease progresses in other areas of the femoral head. Incidence up to 20-40% long-term. Salvage: THA.
4. Total Hip Arthroplasty (THA):
*
Dislocation:
Incidence 1-5%. Higher in posterior approach without proper capsular repair, or with patient non-compliance to precautions. Salvage: Closed reduction; recurrent cases may need open reduction, revision of components, or constrained liners.
*
Periprosthetic Infection:
Incidence 0.5-1.5%. A severe complication. Salvage: Debridement and implant retention (DAIR) in early cases, or two-stage revision (implant removal, spacer, antibiotics, then re-implantation) in chronic cases.
*
Periprosthetic Fracture:
Incidence 1-2%. During or after surgery. Salvage: Internal fixation, revision of components.
*
Aseptic Loosening:
Long-term failure of implant fixation to bone due to biological or mechanical factors. Incidence varies by implant type and patient activity, 0.5-1% per year. Salvage: Revision THA.
*
Leg Length Discrepancy:
Mild discrepancy is common. Significant discrepancy (>10-15mm) can lead to gait abnormalities and back pain. Salvage: Shoe lifts, or revision THA for severe cases.
*
Heterotopic Ossification:
Formation of bone in soft tissues around the joint. Incidence 1-10%. Prophylaxis with NSAIDs or radiation. Salvage: Excision if symptomatic.
*
Nerve Injury:
Sciatic or femoral nerve palsy (0.5-1%). Management: Observation, bracing, neurolysis.
Table 2: Common Complications, Incidence, and Salvage Strategies in ONFH Surgery
| Complication | Incidence (Approx.) | Primary Surgical Procedure(s) | Salvage Strategy |
|---|---|---|---|
| General Complications | |||
| Surgical Site Infection | 0.5-2% | All | Antibiotics, I&D implant removal (THA) |
| DVT/PE | 0.5-5% | All | Anticoagulation, filter, thrombolysis |
| Neurovascular Injury | <1-2% | All | Observation, nerve repair, AFO |
| Hematoma | Variable | All | Drainage, observation |
| Core Decompression | |||
| Femoral Neck Fracture | 1-5% | Core Decompression | Internal fixation (screws), THA (if non-union/collapse) |
| Subchondral Collapse | Up to 30% | Core Decompression | Vascularized graft, osteotomy, THA |
| Disease Progression | 20-40% | Core Decompression | Vascularized graft, osteotomy, THA |
| Vascularized Fibular Grafting | |||
| Fibular Fracture (Donor) | 5-10% | Fibular Graft | Internal fixation, protected weight-bearing |
| Peroneal Nerve Injury (Donor) | <1% | Fibular Graft | Observation, AFO, nerve repair |
| Graft Vascular Thrombosis | 5-15% | Fibular Graft | Immediate microvascular revision |
| Graft Non-Union / Collapse | 5-10% | Fibular Graft | Revision grafting, internal fixation, THA |
| Rotational Osteotomy | |||
| Osteotomy Non-Union / Malunion | 5-15% | Osteotomy | Revision internal fixation, revision osteotomy, THA |
| Hardware Failure | 5-10% | Osteotomy | Revision internal fixation |
| AVN of Osteotomized Segment | Rare | Osteotomy | THA |
| Total Hip Arthroplasty | |||
| Dislocation | 1-5% | THA | Closed reduction, open reduction, revision of components, constrained liners |
| Periprosthetic Infection | 0.5-1.5% | THA | DAIR, two-stage revision |
| Periprosthetic Fracture | 1-2% | THA | Internal fixation, revision of components |
| Aseptic Loosening | 0.5-1% / year | THA | Revision THA |
Thorough pre-operative assessment, careful surgical technique, and appropriate post-operative care are the cornerstones of minimizing and managing these potential complications.
Post-Operative Rehabilitation Protocols
Post-operative rehabilitation is an integral component of the overall management strategy for osteonecrosis, designed to optimize recovery, promote healing, protect the surgical repair, and restore function. Protocols vary significantly based on the specific surgical procedure performed and the extent of bone involvement.
1. Core Decompression (with or without Biological Augmentation)
Goals: Protect the decompression site, promote healing and revascularization, maintain hip range of motion (ROM), and gradually restore weight-bearing.
Phase I: Immediate Post-Operative (0-6 weeks)
*
Weight-Bearing:
Strict non-weight-bearing (NWB) or touch-down weight-bearing (TDWB) on the affected limb with crutches or a walker. This is crucial to prevent mechanical stress on the decompressed area and reduce the risk of subchondral collapse.
*
ROM:
Gentle passive and active-assisted range of motion exercises for the hip, avoiding extreme rotation or hip flexion beyond 90 degrees initially if a more anterior approach was used. Focus on flexion, extension, abduction, and adduction within pain-free limits.
*
Strengthening:
Isometric quadriceps and gluteal sets (e.g., quad sets, gluteal squeezes) to maintain muscle tone. Ankle pumps to prevent DVT.
*
Precautions:
Avoid impact loading, twisting motions.
Phase II: Early Healing & Gradual Weight-Bearing (6-12 weeks)
*
Weight-Bearing:
Progress from TDWB to partial weight-bearing (PWB) (e.g., 25-50% body weight) as tolerated, guided by pain and radiographic assessment for signs of healing. Continue using crutches.
*
ROM:
Continue with active ROM exercises. May begin gentle stretching.
*
Strengthening:
Initiate light resistance exercises for hip abductors, adductors, extensors, and flexors (e.g., resistance bands, hip flexion in supine, short arc quads). Focus on core stability.
*
Modalities:
Therapeutic ultrasound or electrical stimulation may be considered to enhance healing.
Phase III: Advanced Strengthening & Return to Activity (12 weeks - 6 months)
*
Weight-Bearing:
Progress to full weight-bearing (FWB) and discontinue crutches as tolerated.
*
Strengthening:
Advance to more challenging resistance exercises, including functional movements (e.g., step-ups, lunges, squats). Focus on proprioception and balance training.
*
Activity:
Gradually reintroduce low-impact activities (e.g., swimming, cycling). Avoid high-impact sports or activities involving repetitive jumping/running for at least 6-12 months, or until cleared by the surgeon based on imaging.
2. Vascularized Fibular Grafting
Goals: Protect the graft and microvascular anastomosis, ensure graft incorporation, and restore function. Similar to core decompression but with stricter, prolonged weight-bearing restrictions.
Phase I: Immediate Post-Operative (0-8 weeks)
*
Weight-Bearing:
Strict NWB on the operated leg. This is critical for microvascular healing and graft integration.
*
ROM:
Gentle active and passive hip ROM exercises. Avoid aggressive movements that could compromise the graft or anastomosis.
*
Strengthening:
Isometric exercises for hip and core muscles. Ankle pumps.
*
Donor Site Care:
Manage donor site pain and swelling. Monitor for complications (e.g., nerve injury, compartment syndrome). Early ankle ROM for fibula donor limb.
Phase II: Early Graft Integration & Gradual Weight-Bearing (8-16 weeks)
*
Weight-Bearing:
Progress from NWB to TDWB or very light PWB (e.g., 10-20% body weight) based on surgeon's discretion, typically after 8-12 weeks, and only if radiographic evidence suggests graft stability.
*
ROM & Strengthening:
Continue as per core decompression protocols, gradually increasing intensity. Focus on hip stability.
Phase III: Advanced Strengthening & Return to Activity (16 weeks - 9 months)
*
Weight-Bearing:
Gradual progression to FWB. Discontinue assistive devices.
*
Strengthening & Activity:
As per core decompression, but return to high-impact activities will be delayed (typically 9-12 months or longer) and carefully assessed. Regular radiographic monitoring for graft incorporation and stability.
3. Rotational Osteotomy
Goals: Achieve osteotomy union, protect the rotated segment, and restore function. Requires prolonged NWB or protected weight-bearing.
Phase I: Immediate Post-Operative (0-8 weeks)
*
Weight-Bearing:
Strict NWB. This is paramount to ensure osteotomy healing and stable fixation.
*
ROM:
Gentle passive and active-assisted ROM, avoiding rotations that stress the osteotomy site.
*
Strengthening:
Isometric hip and core exercises. Ankle pumps.
Phase II: Osteotomy Healing & Gradual Weight-Bearing (8-16 weeks)
*
Weight-Bearing:
Progression to TDWB or PWB based on radiographic evidence of osteotomy healing (callus formation). Continue using crutches.
*
ROM & Strengthening:
Gradually increase ROM. Introduce light resistance exercises for hip and core.
*
Radiographic Monitoring:
Frequent radiographs to assess union.
Phase III: Advanced Strengthening & Return to Activity (16 weeks - 9 months)
*
Weight-Bearing:
Once radiographic union is confirmed, progress to FWB and discontinue crutches.
*
Strengthening & Activity:
Progressive resistance exercises, balance, and proprioception training. Return to low-impact activities after 6 months. High-impact activities are typically discouraged or delayed significantly (9-12 months) and only after complete union and adequate muscle strength.
4. Total Hip Arthroplasty (THA)
Goals: Restore full weight-bearing, achieve excellent pain relief, and regain functional ROM, while adhering to specific hip precautions if applicable.
Phase I: Immediate Post-Operative (Days 0-7)
*
Weight-Bearing:
Full weight-bearing (FWB) as tolerated with assistive devices (walker/crutches), unless specific surgeon instructions or bone quality issues dictate otherwise.
*
ROM:
Initiate gentle active and passive ROM. Avoid extreme positions based on surgical approach (e.g., avoid hip flexion >90°, adduction past midline, internal rotation for posterior approach; avoid hip extension, external rotation for direct anterior approach).
*
Strengthening:
Isometric gluteal and quadriceps sets. Ankle pumps. Early ambulation.
*
Discharge Planning:
Focus on transfers, gait training, and home exercise program.
Phase II: Early Strengthening & Functional Mobility (1-6 weeks)
*
Weight-Bearing:
Continue FWB. Gradually wean off assistive devices.
*
ROM:
Progress ROM exercises while adhering to precautions.
*
Strengthening:
Begin light progressive resistance exercises for hip abductors, extensors, and quadriceps. Emphasis on core stability.
*
Functional Training:
Progress gait mechanics, stair climbing.
Phase III: Advanced Strengthening & Return to Full Activity (6 weeks - 3 months +)
*
Weight-Bearing:
Independent ambulation.
*
Strengthening:
Advanced resistance training, functional exercises, balance training.
*
Activity:
Gradual return to light recreational activities. Most restrictions related to hip precautions are lifted after 6-12 weeks, depending on surgeon preference and implant stability. Avoid high-impact sports, deep squats, or activities with high dislocation risk.
General Principles for All Protocols:
*
Pain Management:
Effective post-operative analgesia is crucial for participation in rehabilitation.
*
Individualization:
Protocols must be tailored to the individual patient's progress, pain levels, and surgical findings.
*
Patient Education:
Patients must be thoroughly educated on precautions, exercises, and the expected timeline for recovery.
*
Long-Term Follow-up:
Regular clinical and radiographic follow-up is essential to monitor for progression of disease, implant loosening (for THA), or late complications.
Adherence to these structured rehabilitation protocols, under the guidance of a physical therapist and close collaboration with the surgical team, is fundamental to achieving optimal long-term outcomes for patients undergoing surgical management of osteonecrosis.
Summary of Key Literature / Guidelines
The landscape of osteonecrosis research and clinical guidelines has evolved considerably, reflecting advancements in understanding its complex pathophysiology and diverse treatment modalities. While comprehensive, this summary highlights pivotal literature and consensus statements guiding current orthopedic practice.
1. Staging and Diagnosis:
The
ARCO (Association Research Circulation Osseous) classification system
remains the most widely accepted and comprehensive staging system for ONFH. It incorporates plain radiographs, MRI, and histological findings, offering a more nuanced approach than previous systems like Ficat-Arlet.
*
ARCO Guidelines (1992, updated 2018):
These guidelines emphasize MRI as the gold standard for early diagnosis (Stage 0-I) and for accurately assessing lesion size and location, which are critical prognostic factors. The double-line sign on T2-weighted MRI is highly specific.
*
Mont MA, et al. (JBJS Am, 2005):
A foundational review outlining the etiology, diagnosis, and treatment of ONFH, emphasizing the importance of early diagnosis before collapse.
2. Pathophysiology and Etiology:
A significant body of literature supports the multifactorial nature of ON, with particular emphasis on vascular compromise and cellular dysfunction.
*
Wang T, et al. (Bone Res, 2017):
Reviewed the cellular and molecular mechanisms of ON, highlighting the roles of adipogenesis, lipid metabolism, oxidative stress, and inflammatory pathways in corticosteroid-induced ON.
*
Glueck CJ, et al. (Clin Orthop Relat Res, 1997):
Pioneering work demonstrating the association between inherited and acquired thrombophilias (e.g., Factor V Leiden, protein C/S deficiency, hyperlipoproteinemia(a)) and idiopathic ONFH, underscoring the role of microthrombosis.
*
Chan SC, et al. (Int J Mol Sci, 2021):
Recent insights into the contribution of osteoclast dysfunction and osteocyte apoptosis, further linking the cellular aspects mentioned in the seed content. The "virus-like inclusion bodies in osteoclasts" (from the seed) remain a less widely replicated or fully elucidated mechanism in the broader literature, often considered within the context of generalized cellular stress or specific infectious triggers not yet universally accepted.
3. Non-Operative Management:
The efficacy of non-operative treatments is generally limited, particularly once lesions are established or large.
*
Lai KA, et al. (J Bone Joint Surg Br, 2005):
A randomized controlled trial suggesting that alendronate may prevent collapse in early-stage ONFH. This study sparked interest in bisphosphonates, though long-term outcomes and universal applicability remain debated.
*
Agarwala S, et al. (J Arthroplasty, 2015):
Systematic reviews have explored various pharmacologic interventions (statins, vasodilators), but strong evidence for consistent prevention of collapse in large, symptomatic lesions is lacking.
4. Joint-Preserving Procedures:
These procedures aim to prevent collapse in pre-collapse stages (ARCO Stage I/II) or early collapse with intact articular cartilage (early Stage III).
*
Core Decompression:
*
Koo KH, et al. (Clin Orthop Relat Res, 2002):
Demonstrated favorable outcomes for core decompression in early-stage ONFH, especially for small to medium-sized lesions. Outcomes significantly worsen once collapse occurs.
*
Hernigou P, et al. (Clin Orthop Relat Res, 2007, and later works):
Extensive research on the use of bone marrow aspirate concentrate (BMAC) combined with core decompression, showing improved outcomes compared to core decompression alone, particularly in reducing collapse rates.
*
Vascularized Fibular Grafting:
*
Urbaniak JR, et al. (Clin Orthop Relat Res, 1994):
Seminal work establishing vascularized fibular grafting as a viable option for large pre-collapse or early-collapse lesions, particularly in younger patients, offering both structural support and biological revascularization.
*
Mittal M, et al. (J Bone Joint Surg Am, 2014):
A systematic review supporting improved long-term outcomes and prevention of collapse with vascularized fibular grafting compared to non-vascularized options for specific indications.
*
Rotational Osteotomy:
*
Sugioka Y (Clin Orthop Relat Res, 1984):
Pioneer in rotational osteotomy techniques for ONFH. His long-term results showed good success in carefully selected patients with localized lesions.
*
Various studies (e.g., Kim HK, et al. J Bone Joint Surg Br, 2001):
Highlight that the success of osteotomy is highly dependent on patient selection, lesion size, and careful surgical technique to adequately reorient the weight-bearing axis away from the necrotic segment.
5. Total Hip Arthroplasty (THA):
The definitive treatment for end-stage ONFH (ARCO Stage III/IV) with articular collapse and secondary osteoarthritis.
*
Mont MA, et al. (J Arthroplasty, 1996):
Reported excellent long-term results for THA in ONFH, comparable to THA for osteoarthritis, especially with modern implant designs.
*
Kim YS, et al. (J Bone Joint Surg Am, 2009):
Showed that while THA for ONFH can be successful, younger patients and those with specific etiologies (e.g., steroid-induced) may have higher rates of revision compared to primary OA due to underlying bone quality or comorbidities, though this gap has narrowed with improved techniques.
6. Emerging Therapies and Future Directions:
Research continues into novel biological approaches and advanced imaging.
*
Stem Cell Therapies:
Autologous mesenchymal stem cells (MSCs), particularly from bone marrow, hold promise in augmenting core decompression, with ongoing clinical trials exploring optimal delivery and efficacy.
*
Growth Factors and Gene Therapy:
Investigating the use of pro-angiogenic and osteogenic growth factors (e.g., BMPs, VEGF) or gene therapy to enhance bone repair and revascularization.
*
Advanced Imaging:
Further development of quantitative MRI techniques and biomarkers for earlier diagnosis and more precise prognosis.
Conclusion:
Current guidelines for ONFH management advocate for a stepwise approach based on ARCO staging. Early diagnosis via MRI is critical. For pre-collapse stages, joint-preserving procedures like core decompression (often with biological augmentation) or vascularized fibular grafting are preferred, especially in younger patients. Rotational osteotomy remains an option for highly selected cases. Once significant subchondral collapse or secondary osteoarthritis has occurred, total hip arthroplasty offers reliable pain relief and functional restoration, with excellent long-term outcomes. Continued research into pathophysiology and biological interventions aims to further improve outcomes and potentially prevent disease progression.
You Might Also Like