Acute Traumatic Lesions of Knee Ligaments: A Master Surgical Guide
Key Takeaway
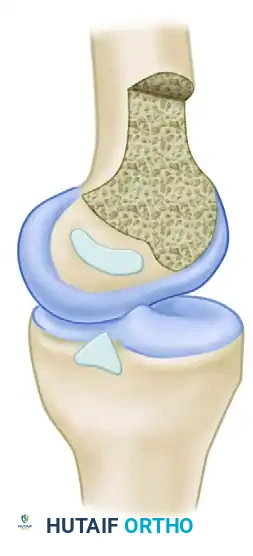
Acute traumatic lesions of knee ligaments require precise biomechanical understanding and meticulous clinical evaluation. Ligament failure occurs sequentially, with complete disruption necessitating significant joint displacement. This guide details the etiology, mechanisms of injury, and the four phases of ligament healing. It provides orthopedic surgeons with evidence-based protocols for diagnosing, classifying, and surgically managing complex multi-ligamentous knee injuries to restore anatomical stability and optimize patient outcomes.
ACUTE TRAUMATIC LESIONS OF LIGAMENTS: GENERAL CONSIDERATIONS
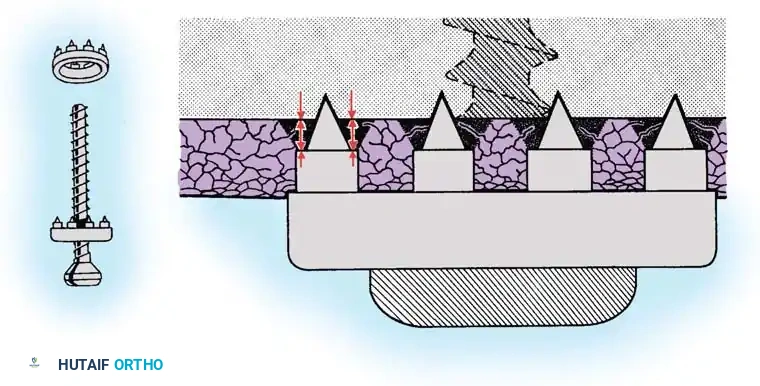
The management of acute traumatic lesions of knee ligaments remains one of the most complex and heavily debated domains in operative orthopedics. Biomechanical loading studies, scanning electron microscopy, and high-speed photography have unequivocally demonstrated that ligaments fail via a progressive, serial, and sequential mechanism of microfiber failure.
Single collagen fibers are inherently non-extensible and begin to fail at a critical threshold of 7% to 8% elongation. The absolute number of disrupted collagen fibers within the ligamentous substance dictates whether the structure is functionally or morphologically compromised. Pioneering investigators, including Kennedy and Noyes, have demonstrated that gross morphological continuity may deceptively persist in a ligament even after the yield point—the definitive indicator of complete biomechanical failure—has been surpassed.
Surgical Pitfall: Visual observation of ligament integrity at the time of surgery is a notoriously inadequate indicator of the true extent of failure. A ligament that appears macroscopically intact may have suffered catastrophic internal microfiber disruption, severe damage to its intrinsic microvascular supply, irreversible residual elongation, and a complete loss of future functional capability.
Complete disruption with a macroscopic loss of continuity requires extreme joint displacement. Consequently, the complete rupture of an isolated knee ligament is exceedingly rare without concomitant damage to secondary restraints. The extreme joint displacement required to completely disrupt a primary ligament inevitably produces at least some degree of structural failure in the surrounding capsuloligamentous supporting structures.
ETIOLOGY AND EPIDEMIOLOGY
Knee ligaments are predominantly injured during high-demand athletic activities, particularly those involving high-velocity contact, such as American football, rugby, and martial arts. Non-contact sports like skiing, ice hockey, and gymnastics also generate sufficient sudden, multi-planar stress to disrupt knee ligaments.
High-energy trauma, particularly motor vehicle accidents (MVAs) and motorcycle collisions, represents a major etiology for multi-ligamentous knee injuries. A classic example is the "dashboard injury," where a passenger’s flexed knee strikes the automobile dashboard upon impact, driving the tibia posteriorly and catastrophically tearing the posterior cruciate ligament (PCL). Furthermore, ligament disruption frequently occurs without direct contact when sudden, severe loading or tension is placed on the joint—such as when a running athlete firmly plants a foot to suddenly decelerate, pivot, or change directions.
MECHANISMS OF INJURY
Understanding the precise kinematic mechanism of injury is paramount for accurate diagnosis and surgical planning. Four primary mechanisms are capable of disrupting the ligamentous structures around the knee:
1. Abduction, Flexion, and Internal Rotation
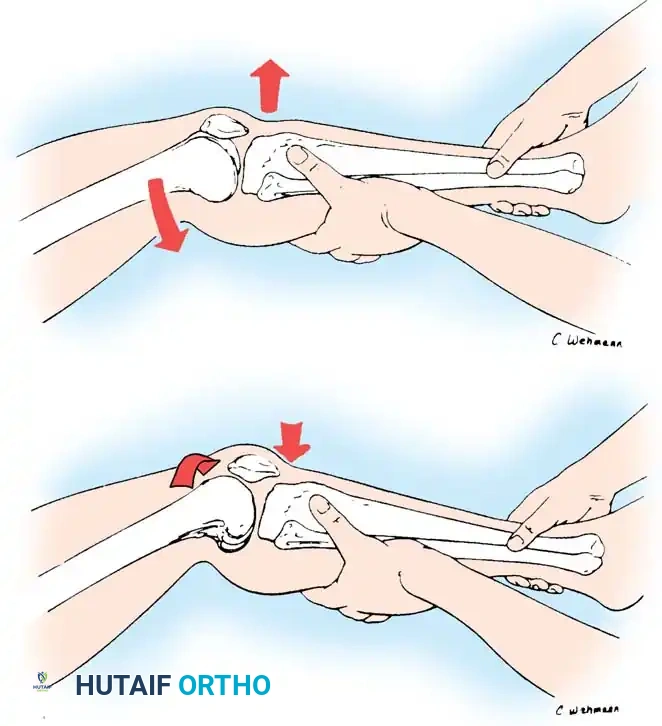
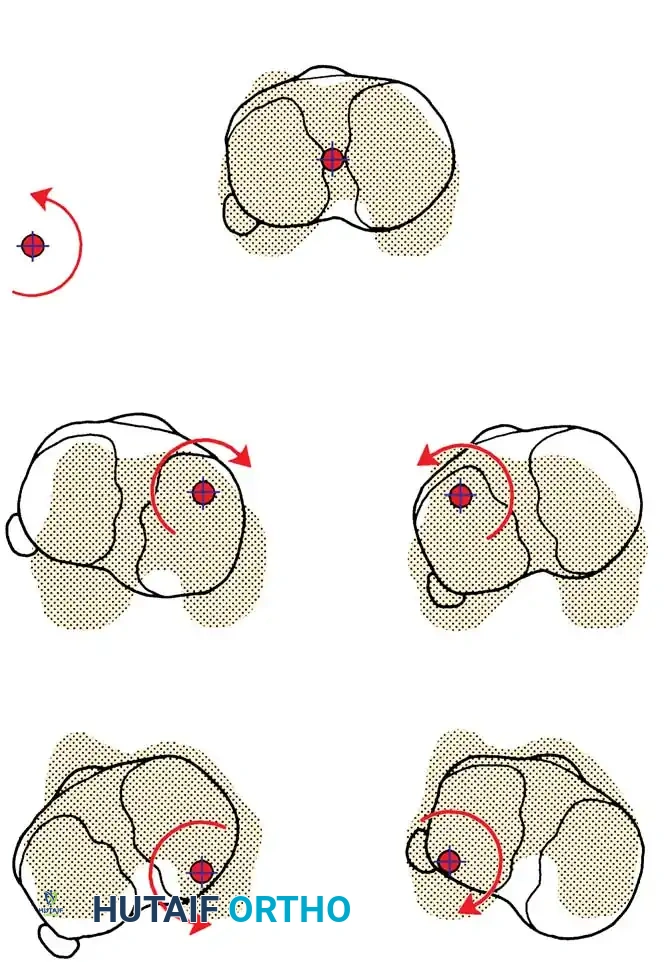
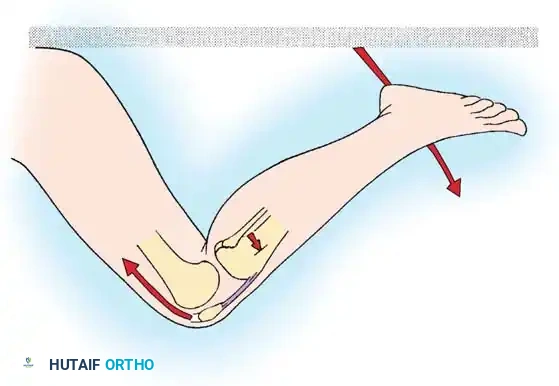
By far the most common mechanism is abduction (valgus), flexion, and internal rotation of the femur on the tibia. This typically occurs when the weight-bearing leg of an athlete is struck from the lateral aspect. The femur rotates internally due to the shift of body weight on the fixed tibia.

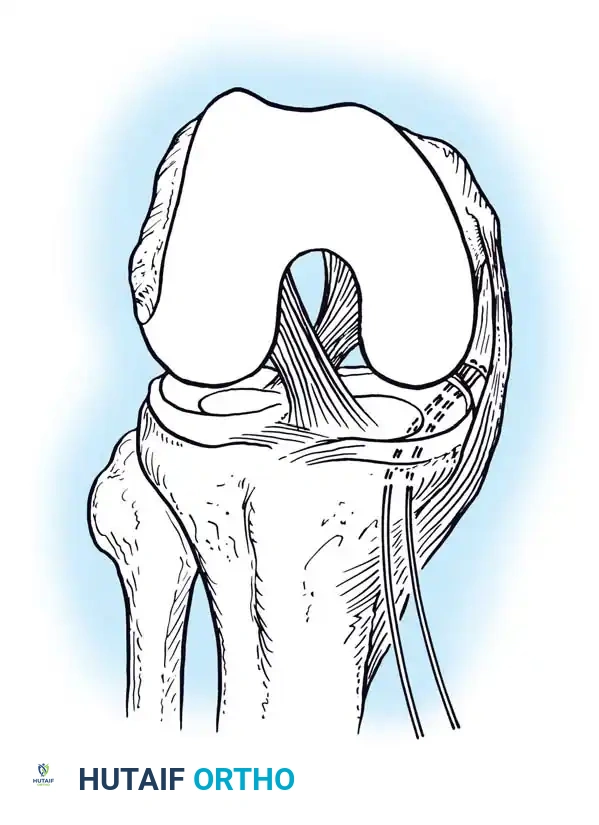
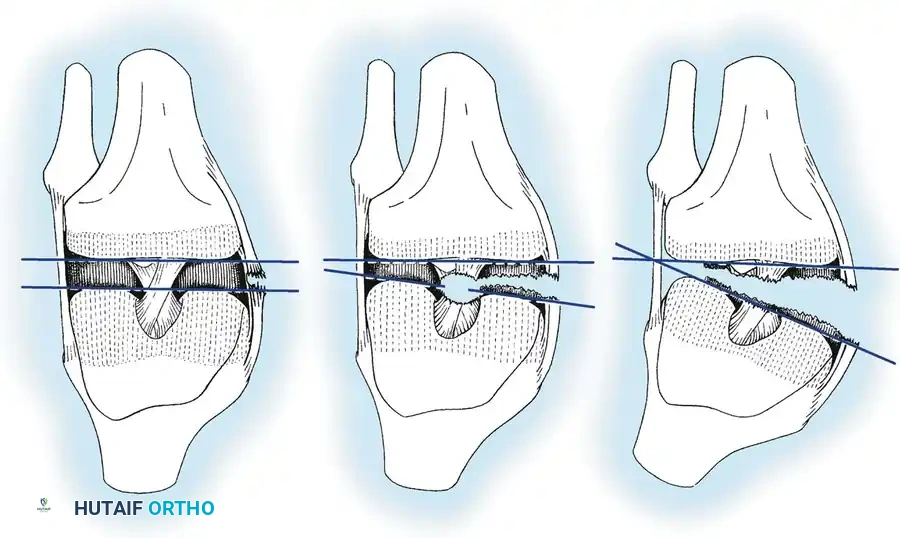
This mechanism produces a devastating cascade of injury on the medial side of the knee. The medial collateral ligament (MCL) and the deep medial capsular ligaments are the initial structures to fail. If the applied force is of sufficient magnitude, the anterior cruciate ligament (ACL) is subsequently torn. The medial meniscus may become entrapped between the medial femoral and tibial condyles, tearing at its periphery as the capsular structures avulse, producing the classic "Unhappy Triad" of O’Donoghue.
2. Adduction, Flexion, and External Rotation
This mechanism is less common but produces profound primary disruption to the lateral and posterolateral structures. The lateral collateral ligament (LCL) is usually disrupted initially. Depending on the magnitude of the trauma, the injury progresses to involve the arcuate ligament complex, the popliteus tendon, the iliotibial band, the biceps femoris, and one or both cruciate ligaments.
Surgical Warning: High-energy varus injuries carry a significant risk of traction injury to the common peroneal nerve. A thorough preoperative neurological assessment is mandatory, and surgical exploration must include careful neurolysis and protection of the nerve.
3. Hyperextension
Force directed to the anterior aspect of the extended knee forces the joint into hyperextension. This mechanism primarily injures the ACL. If the deforming force continues, it leads to stretching and eventual disruption of the posterior capsule and the PCL.
4. Anteroposterior Displacement
Pure anteroposterior forces applied to either the femur or the tibia (e.g., the aforementioned dashboard injury) cause isolated or combined injuries to the cruciate ligaments, depending strictly on the direction of tibial translation relative to the femur.
THE BIOLOGY OF LIGAMENT HEALING
The ability of a torn ligament to heal depends on its anatomical location (intra-articular vs. extra-articular), the presence of associated injuries, the selected treatment modality, and the patient's systemic biology. Extra-articular ligaments (like the MCL) have a robust healing capacity, whereas intra-articular ligaments (like the ACL) fail to heal due to synovial fluid enzymes and a lack of a stabilizing provisional scaffold.
The healing process is divided into four overlapping histological and biochemical phases:
- Hemorrhage Phase (0-72 hours): Following a mid-substance tear, blood flows into the defect created by the retracting ligament ends, forming a localized hematoma.
- Inflammatory Phase (1-14 days): Inflammatory cells and monocytes migrate into the injury site. They convert the hematoma into granulation tissue and phagocytose necrotic debris. Within two weeks, a fragile network of immature, parallel collagen fibers begins to replace the granulation tissue.
- Reparative Phase (5 days - 6 weeks): Fibroblasts proliferate rapidly, producing an extracellular matrix in a random, disorganized pattern. Neovascularization occurs to support the high metabolic demand of the healing tissue. Early collagen is predominantly Type III.
- Remodeling Phase (6 weeks - 1+ years): Collagen fibers gradually align along the long axis of the ligament in response to mechanical stress, resulting in increased maturation of the matrix. Type III collagen is slowly replaced by stronger Type I collagen.
Clinical Pearl: Although a healed ligament may grossly resemble the original structure, long-term studies demonstrate that its histological and morphological appearance remains permanently altered. The healing tissue contains an increased number of smaller-diameter collagen fibrils, abnormal "crimping" patterns, and poor collagen alignment for up to one year.
Biomechanical functional testing reveals that the ultimate load to failure of a healed ligament remains significantly lower than that of a native, uninjured ligament, even after 12 months of remodeling.
CLASSIFICATION OF LIGAMENTOUS SPRAINS
Standardized classification is critical for accurate clinical communication and treatment formulation. The American Medical Association (AMA) defines a sprain as an injury limited to ligaments, classified into three degrees of severity:
- First-Degree (Mild): Tear of a minimal number of fibers. Characterized by localized tenderness, minimal swelling, and no clinical instability.
- Second-Degree (Moderate): Disruption of a significant portion of ligamentous fibers. Characterized by moderate pain, joint effusion, loss of function, and mild-to-moderate instability with a firm endpoint.
- Third-Degree (Severe): Complete structural disruption of the ligament. Characterized by marked instability and no palpable endpoint during stress testing.
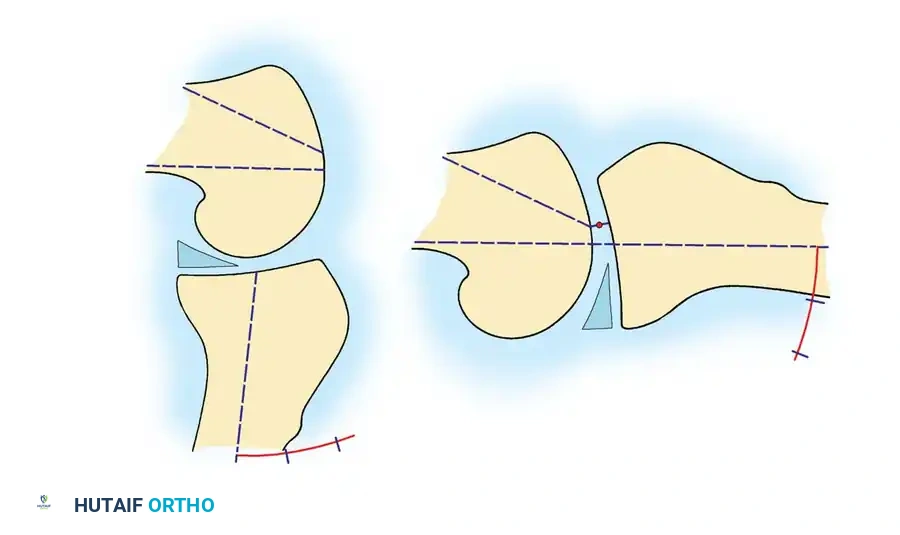
Third-degree sprains are further sub-classified based on the degree of joint separation measured during stress testing compared to the contralateral normal knee:
* 1+ Instability: 5 mm or less of separation.
* 2+ Instability: 5 to 10 mm of separation.
* 3+ Instability: 10 mm or greater of separation.
DIAGNOSTIC EVALUATION
History and Mechanism
A meticulous history often yields the diagnosis before the physical examination begins. The surgeon must ascertain the position of the knee at the time of injury, the weight-bearing status, the direction of the applied force, and the sensation of a "pop" or tearing.
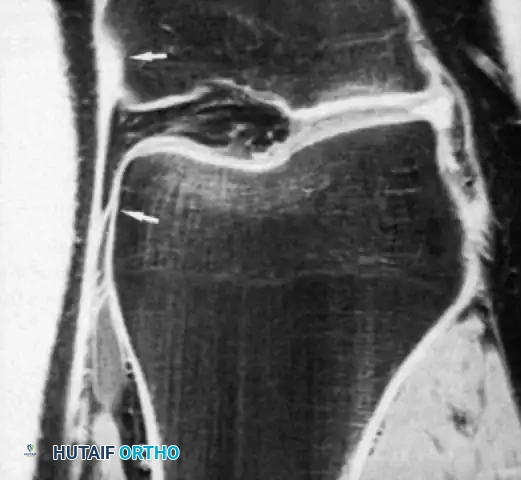
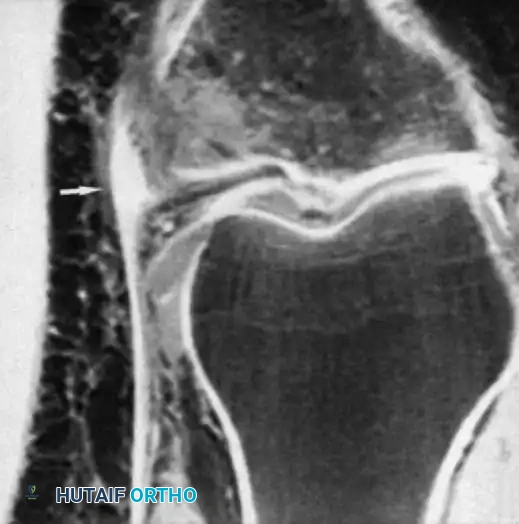
The timing of swelling is a critical diagnostic clue. Rapid intra-articular swelling (within 1 to 2 hours) strongly suggests a hemarthrosis, indicative of an ACL tear, osteochondral fracture, or peripheral meniscal tear. Conversely, swelling that develops gradually overnight usually indicates an acute traumatic synovitis.
Physical Examination
The examination must be precise, systematic, and performed as soon as possible before severe swelling and involuntary muscle guarding obscure the findings. Both lower extremities must be fully exposed for comparison.
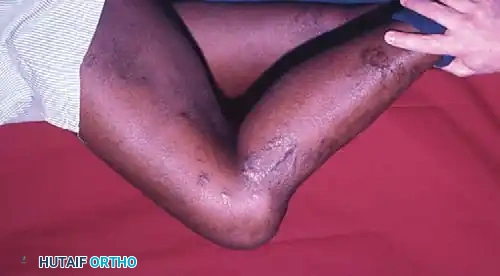
Observe for ecchymosis, abrasions, and the resting attitude of the limb. Muscle atrophy, particularly of the vastus medialis obliquus (VMO), occurs rapidly following knee trauma and should be noted.
Palpation of the collateral ligaments from their femoral origins to their tibial/fibular insertions is necessary to localize the exact site of pathology. A palpable defect is occasionally present immediately after injury, particularly at the tibial insertion of the MCL or the fibular insertion of the LCL.
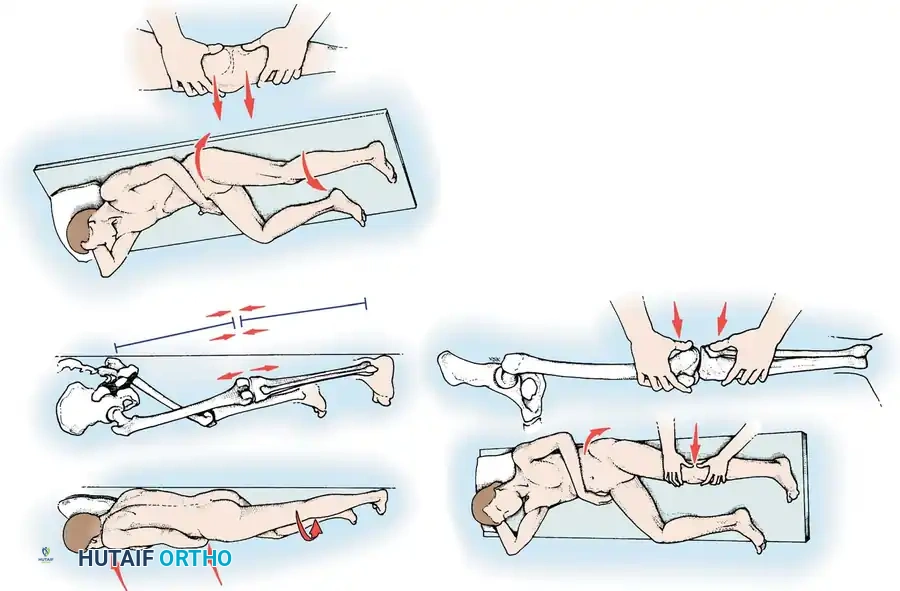
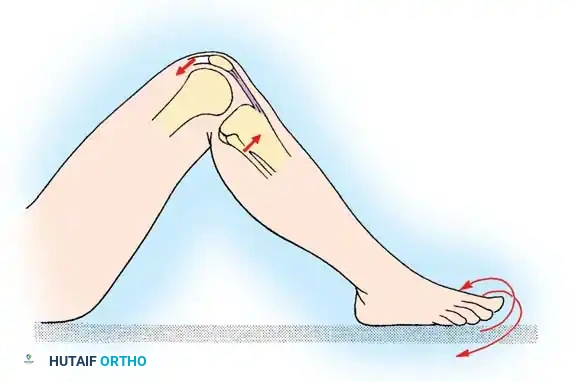
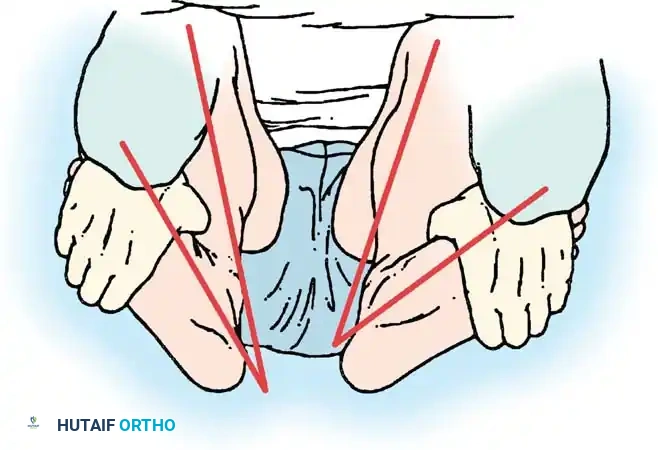
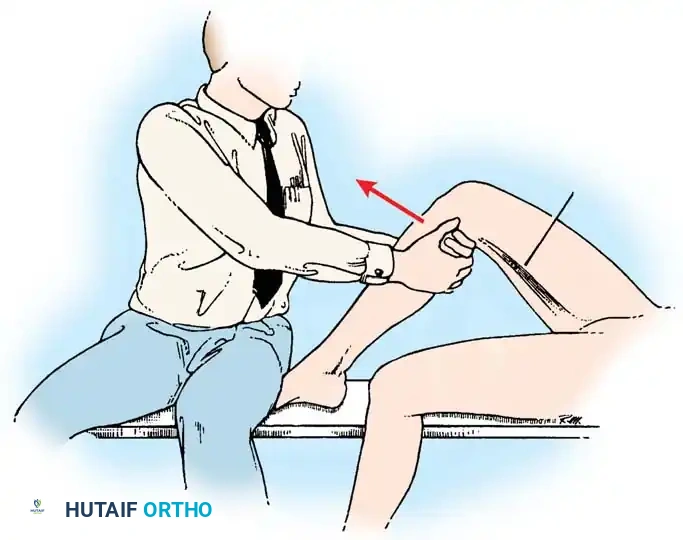
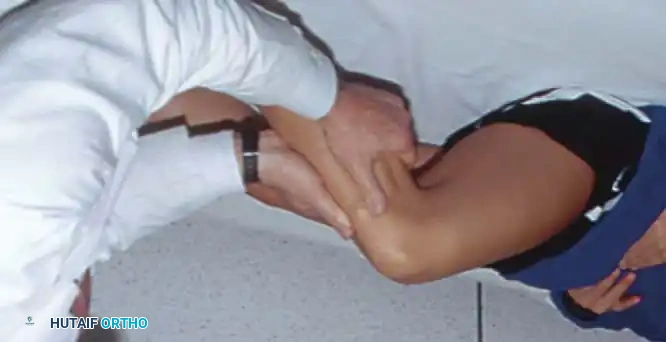
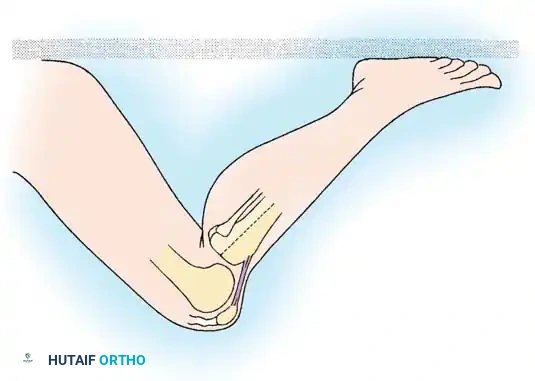
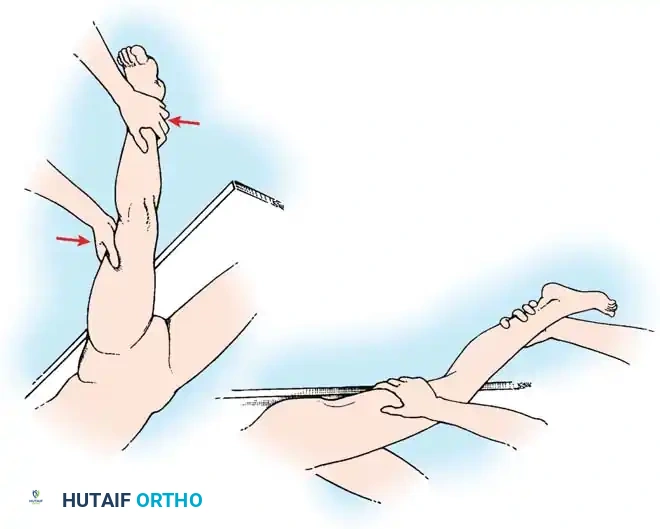
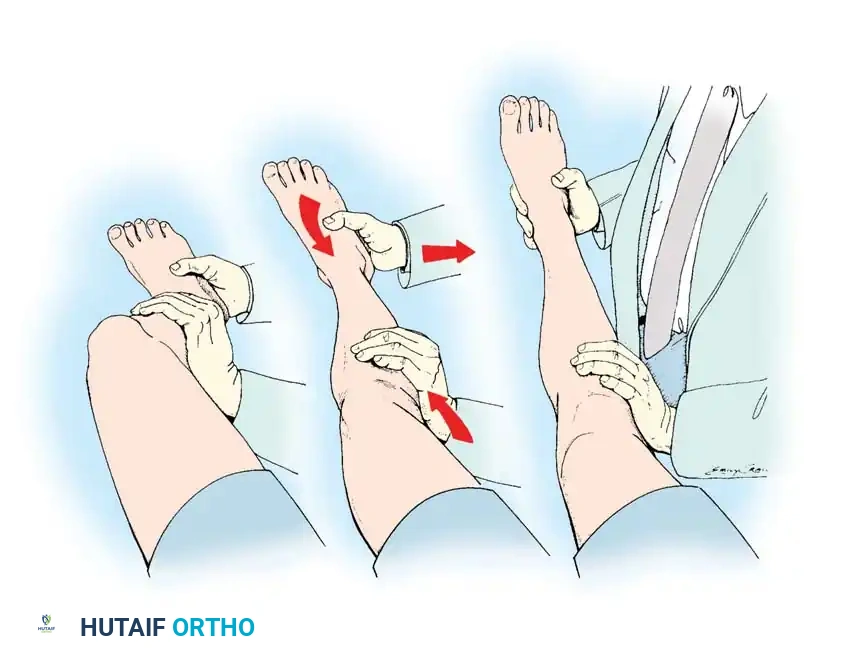
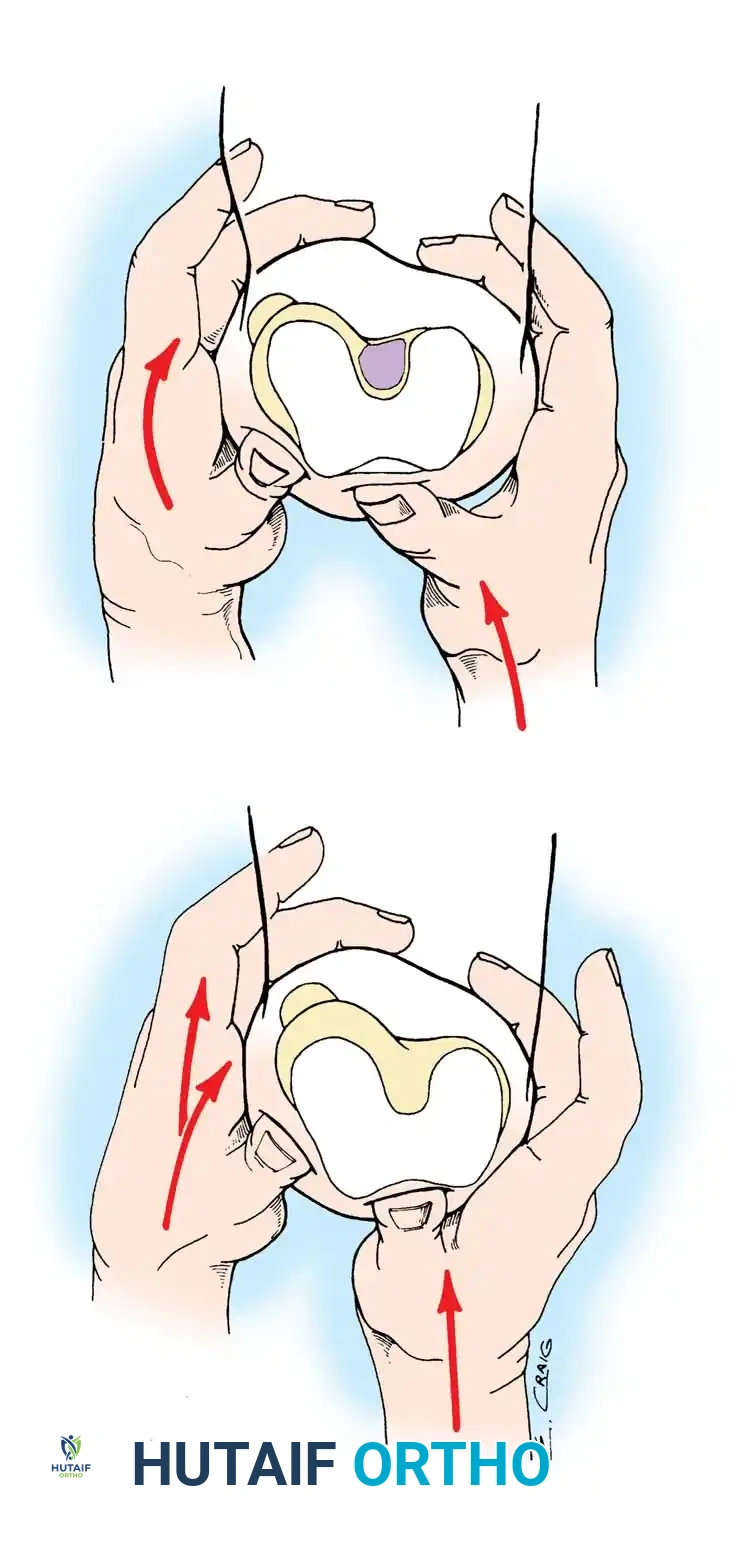
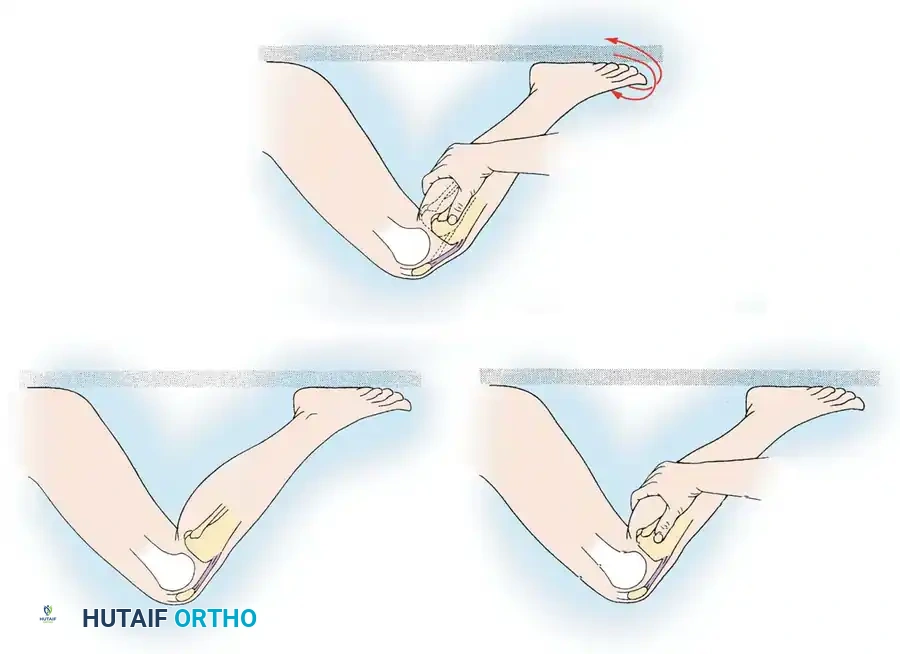
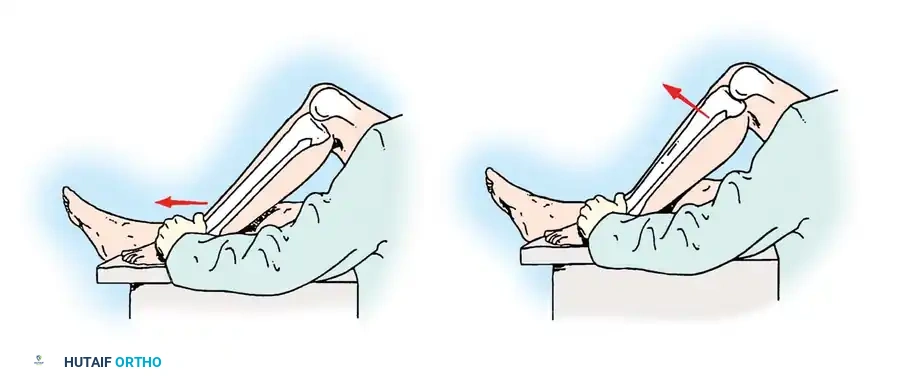
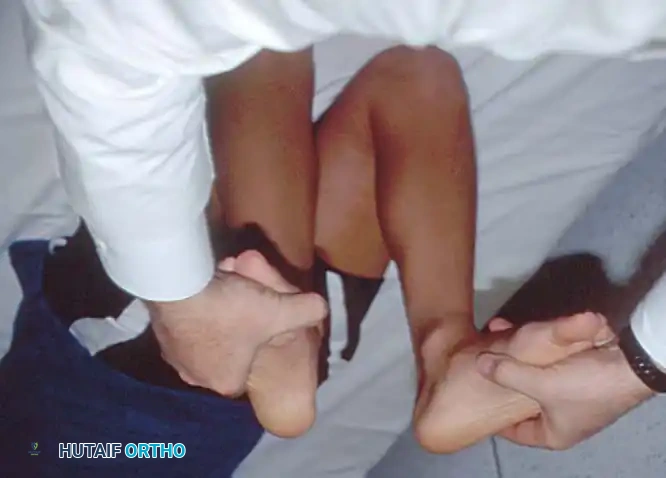
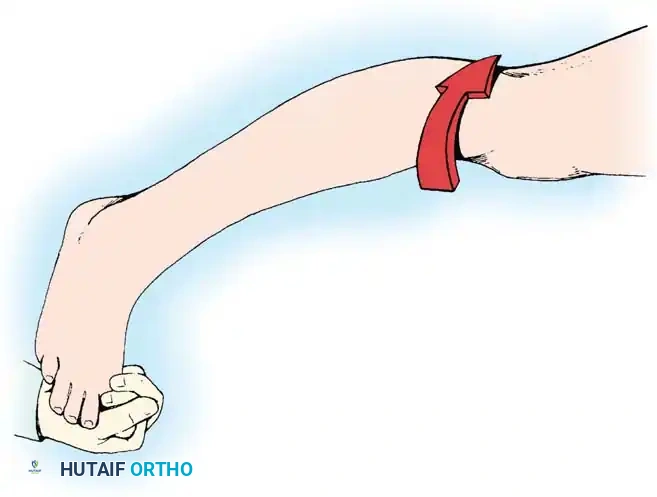
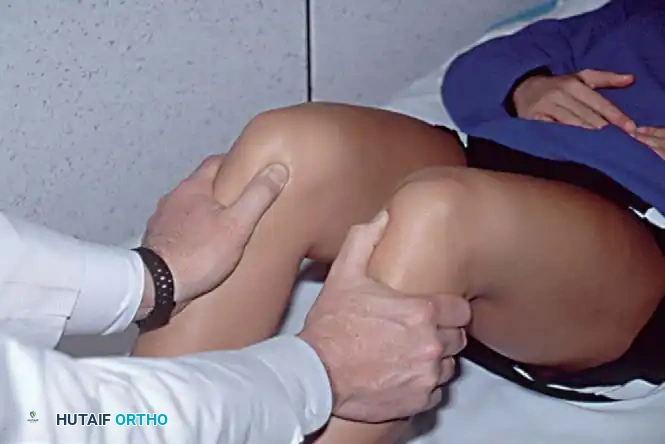
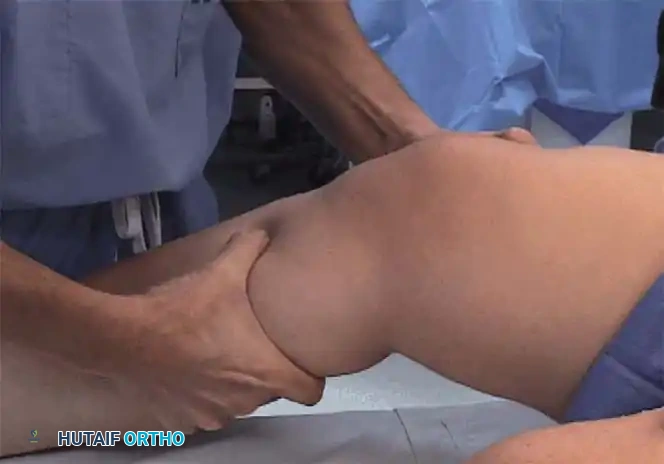
Stress testing is the cornerstone of the physical examination. Valgus and varus stress tests should be performed at both 0 degrees (testing the capsule and cruciates) and 30 degrees of flexion (isolating the collateral ligaments). The Lachman test, anterior drawer, and pivot-shift tests evaluate the ACL, while the posterior drawer and dial tests evaluate the PCL and posterolateral corner.
SURGICAL MANAGEMENT: INDICATIONS AND POSITIONING
While Grade I and II sprains are universally treated non-operatively with functional bracing and early rehabilitation, the management of Grade III injuries depends on the specific ligament involved and the presence of multi-ligamentous trauma.
Isolated Grade III MCL injuries can often be managed conservatively with excellent outcomes. However, Grade III injuries of the LCL/posterolateral corner, combined multi-ligamentous injuries (e.g., ACL + MCL, or knee dislocations), and injuries involving bony avulsions require prompt surgical intervention. The goal of surgery is the precise restoration of anatomical structure and normal isometric tension.
Patient Positioning:
The patient is placed supine on the operating table. A high thigh tourniquet is applied. A lateral post or a specialized leg holder is utilized to allow full range of motion and the application of valgus/varus stress during the procedure. The contralateral leg is placed in a well-leg holder to prevent deep vein thrombosis and allow fluoroscopic access.
SURGICAL APPROACHES AND TECHNIQUES
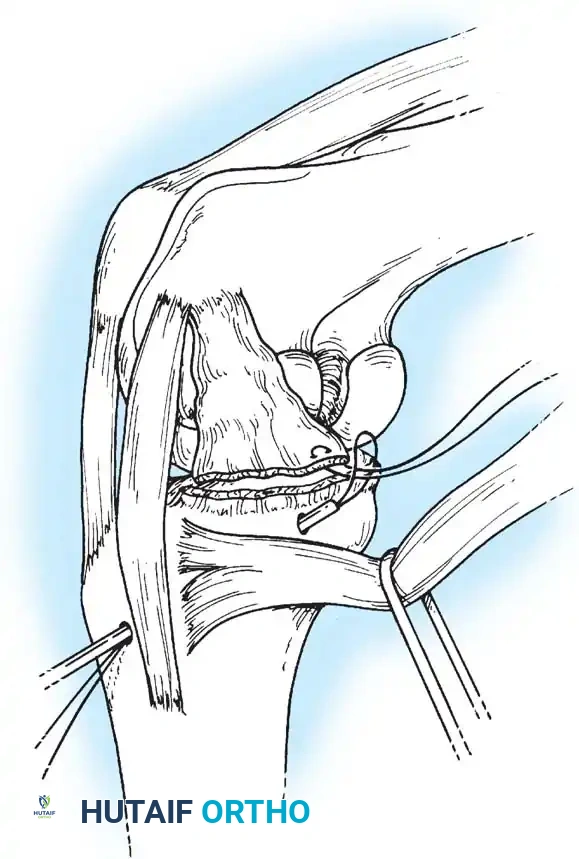
Medial Collateral and Capsular Repair
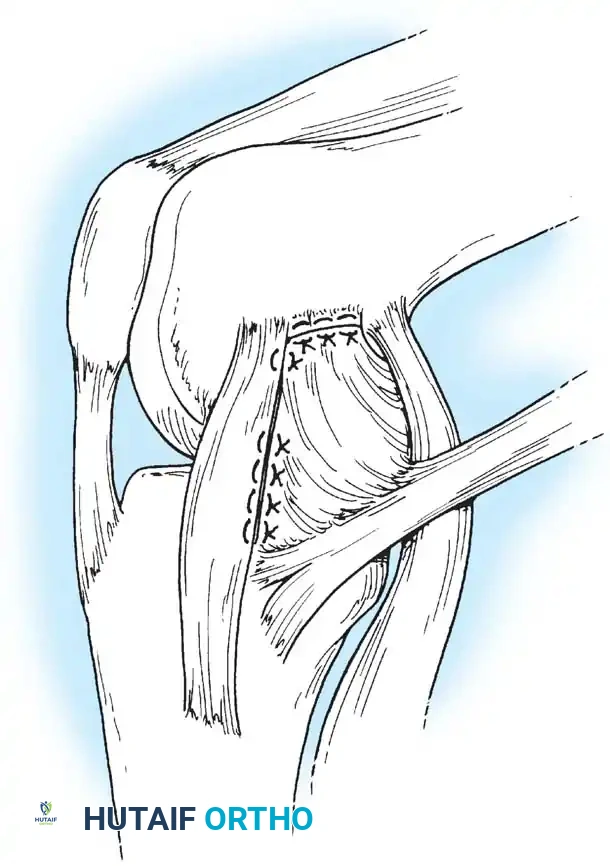
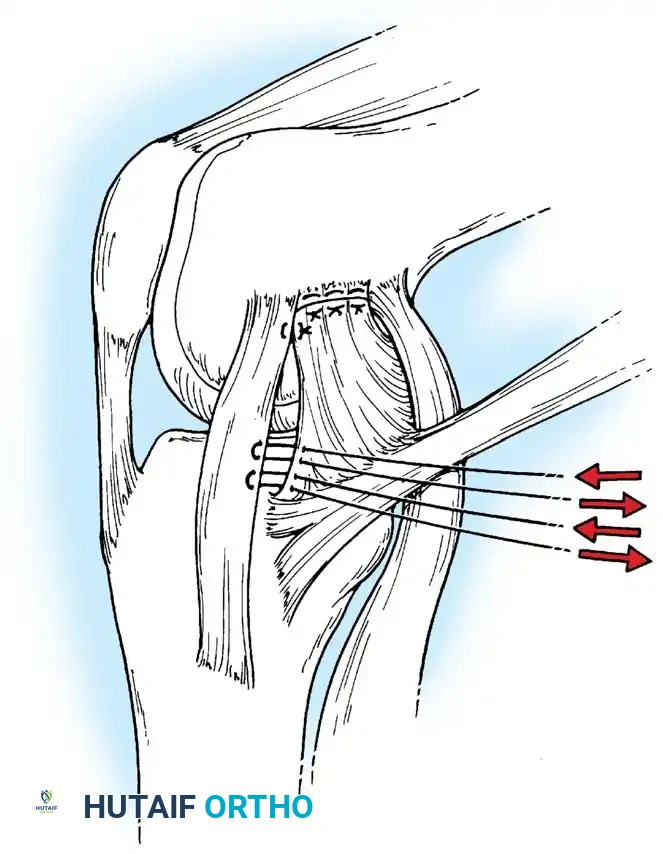
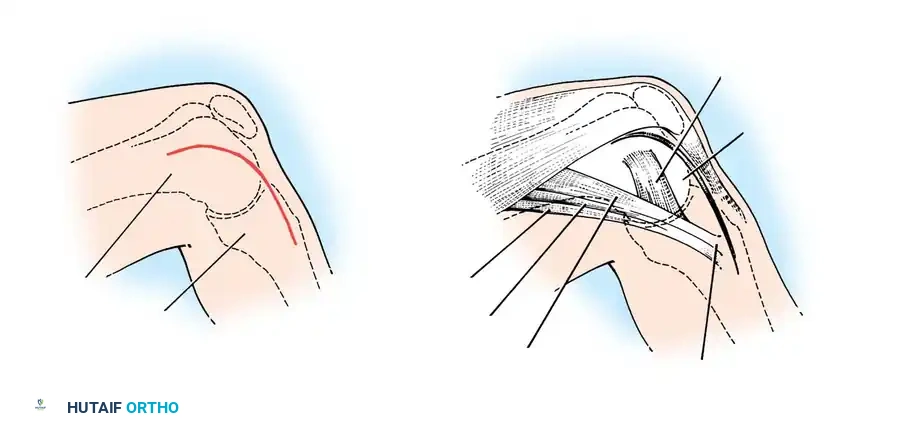
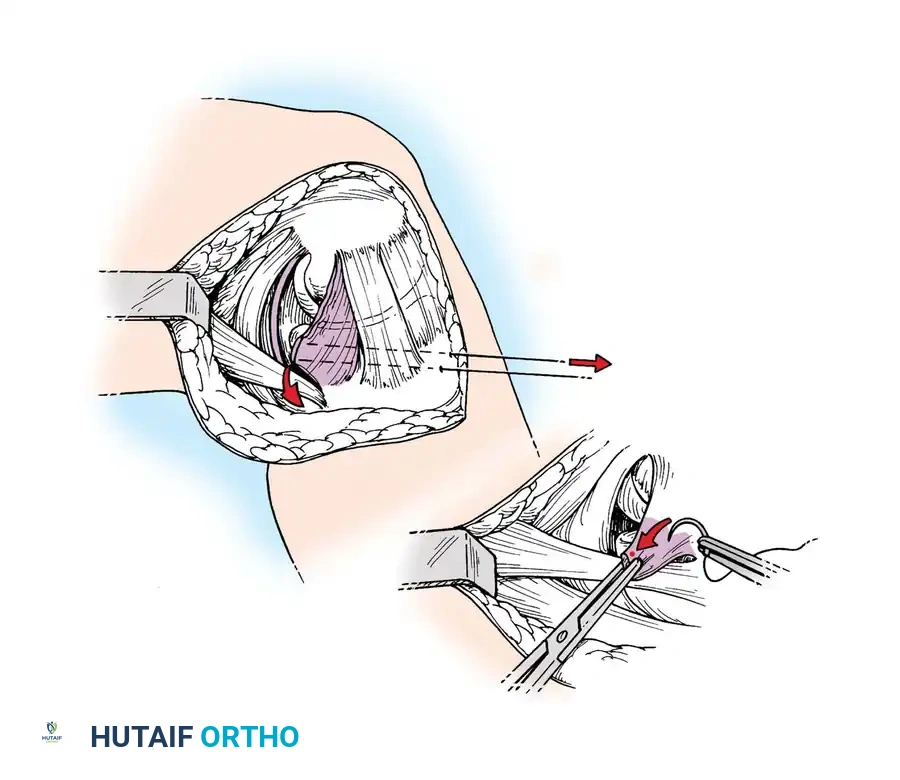
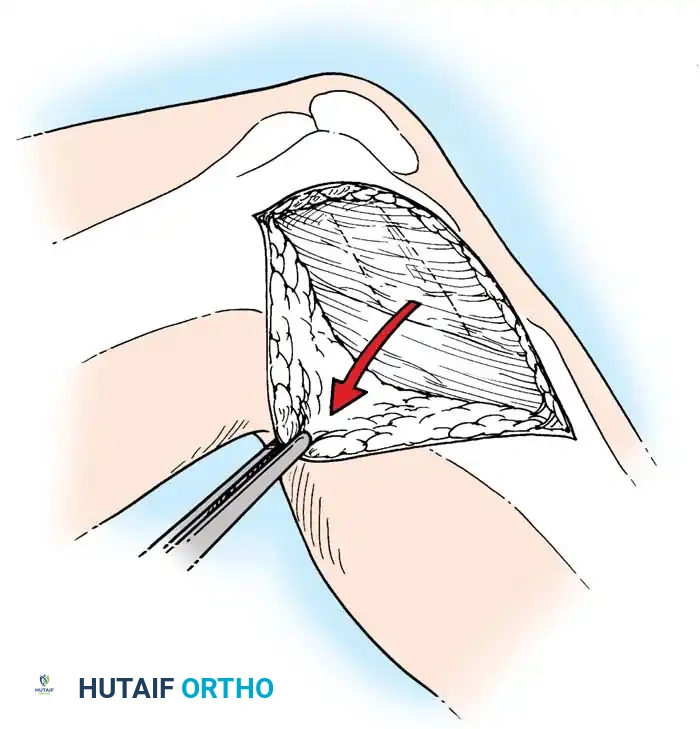
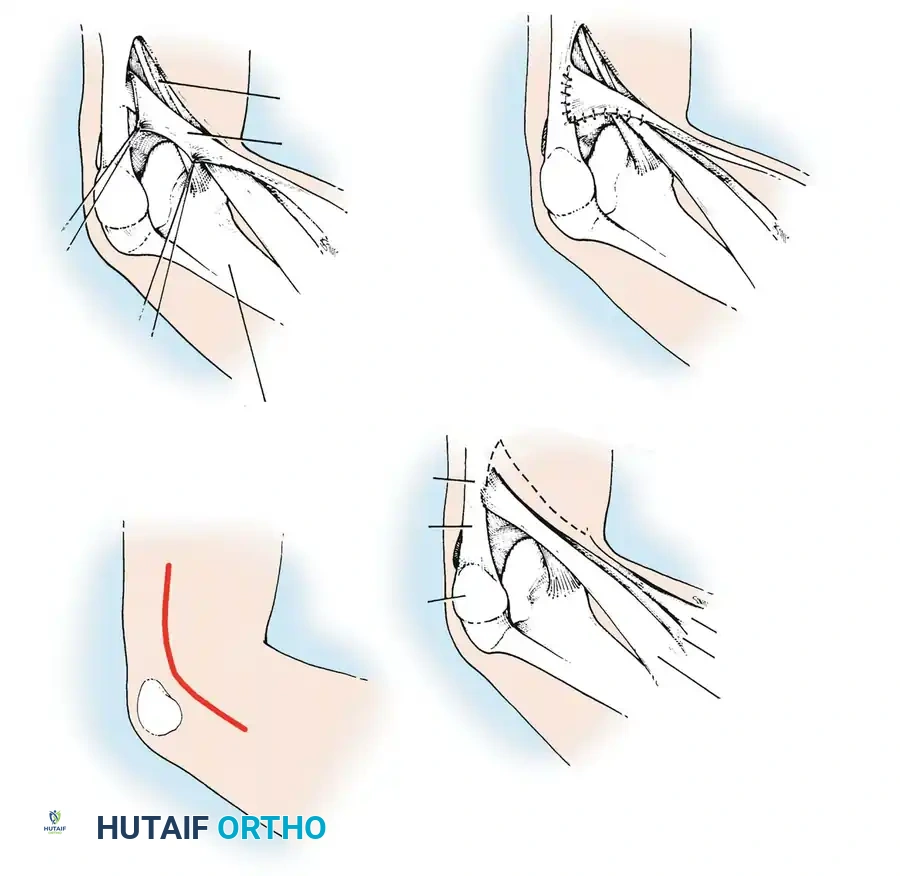
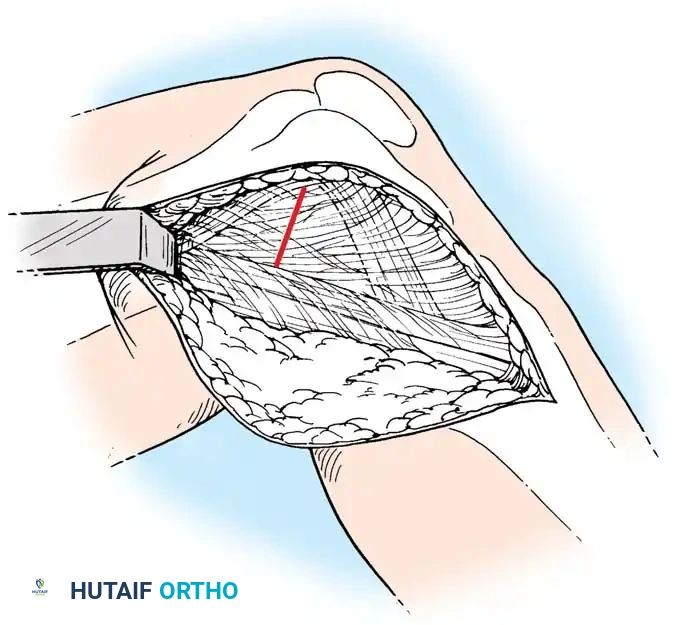
For acute medial-sided repairs, a longitudinal incision is made over the medial aspect of the knee, extending from the medial epicondyle to the anteromedial tibia.
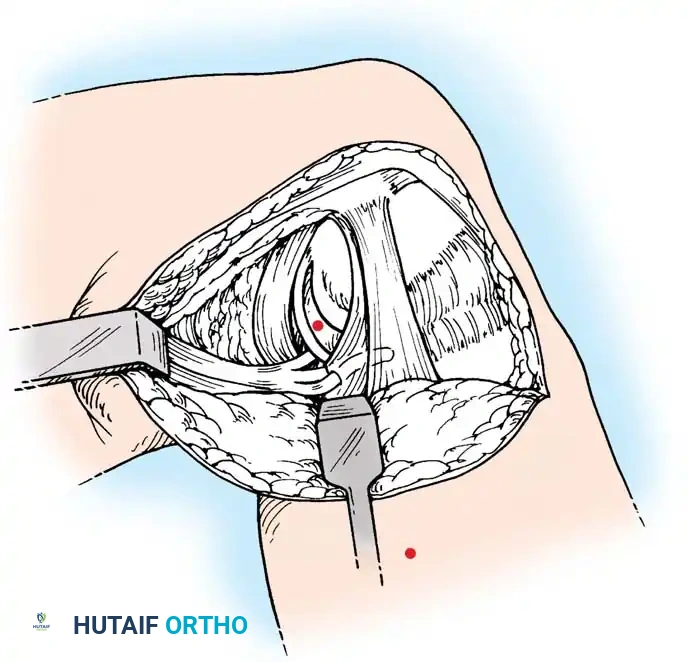
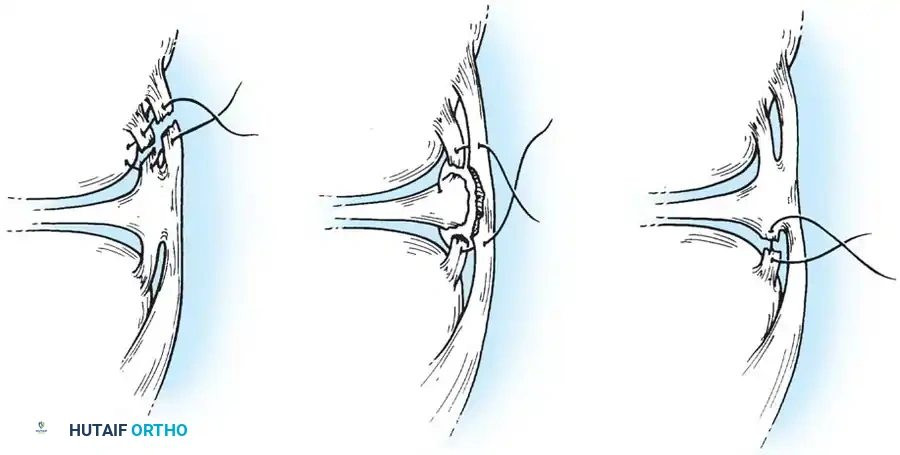
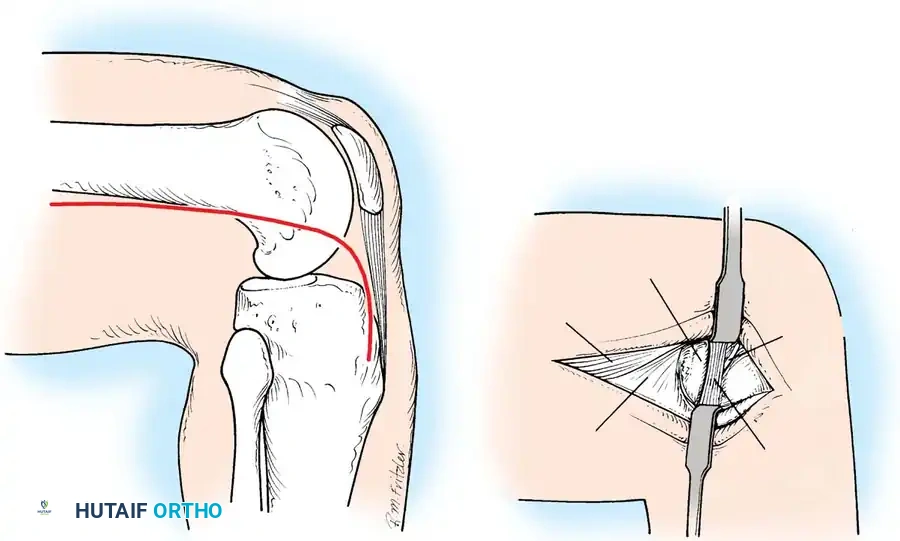
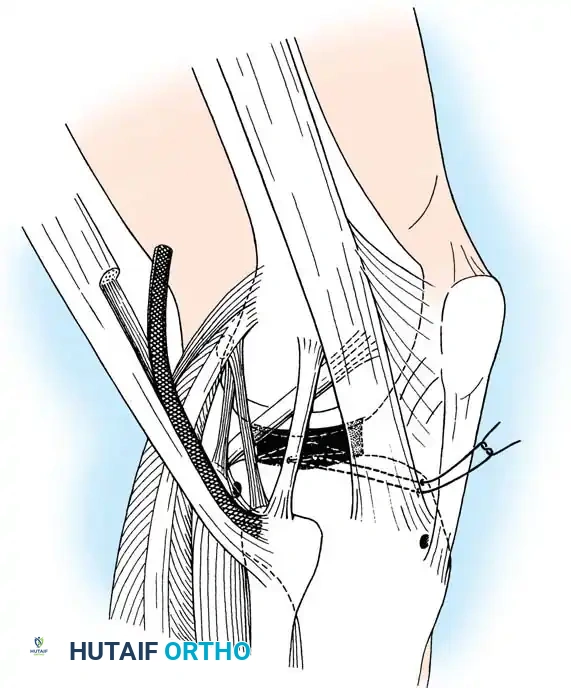
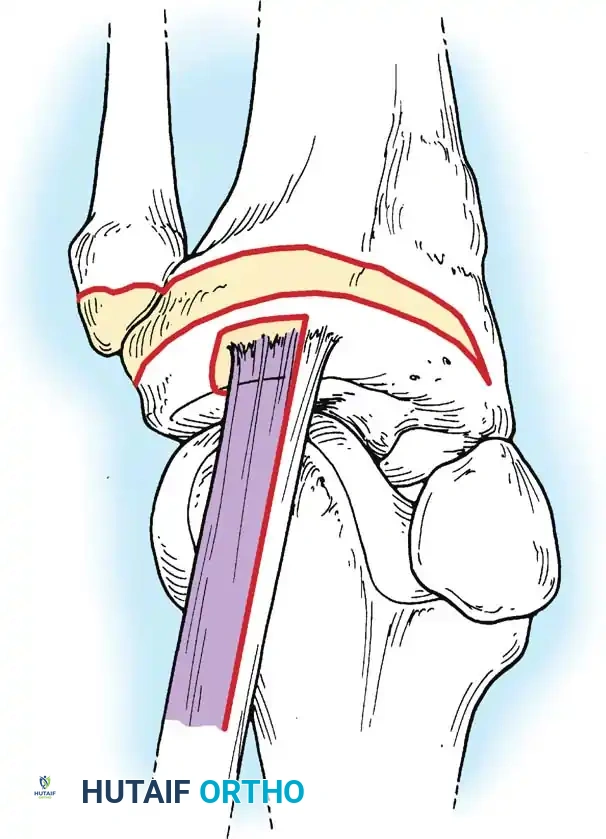
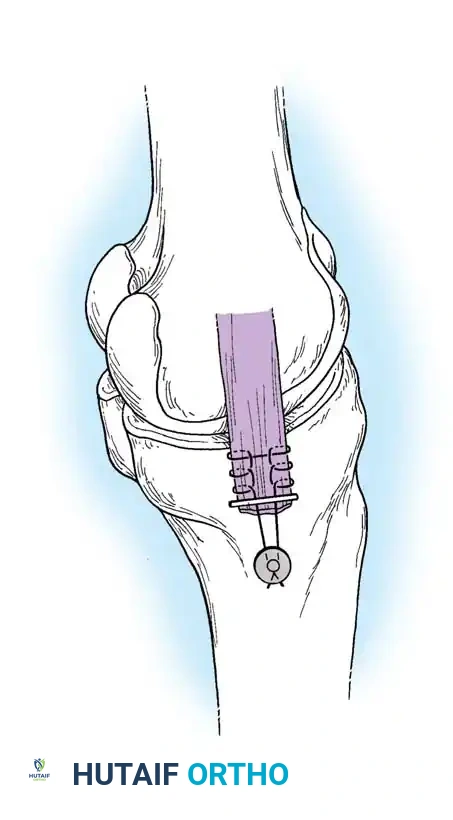
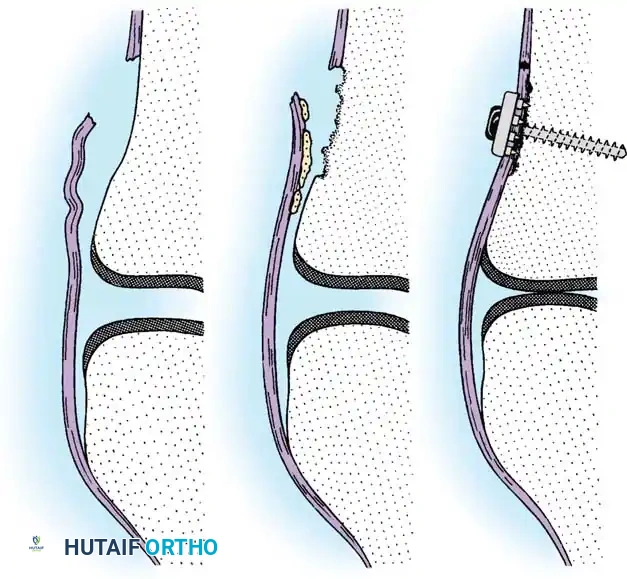
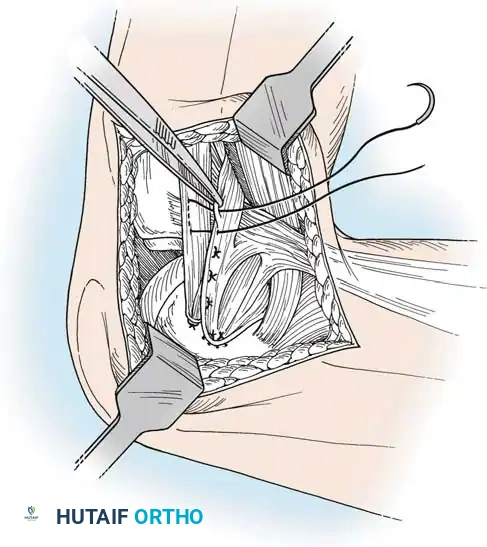
The deep fascia is incised, and the sartorius fascia is identified. The superficial MCL is exposed. If the injury is an avulsion from the femoral origin or tibial insertion, it is repaired using heavy non-absorbable sutures and suture anchors.
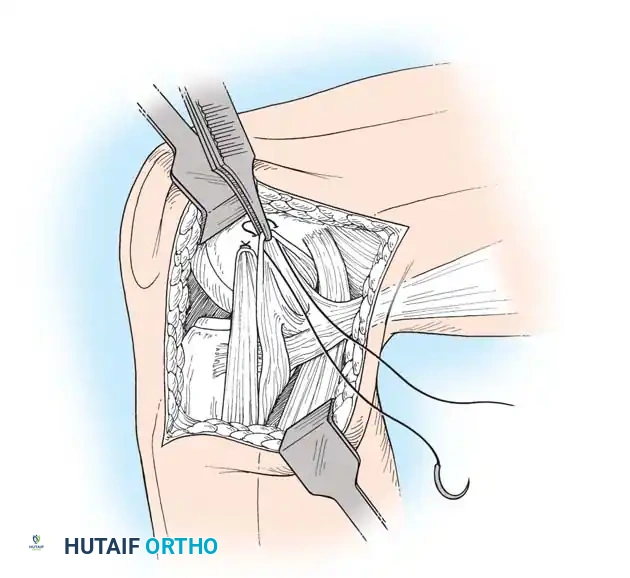
Mid-substance tears are meticulously re-approximated. The deep capsular ligaments (meniscofemoral and meniscotibial) must be inspected. If the medial meniscus is peripherally detached, it is repaired to the capsule prior to tensioning the superficial MCL.
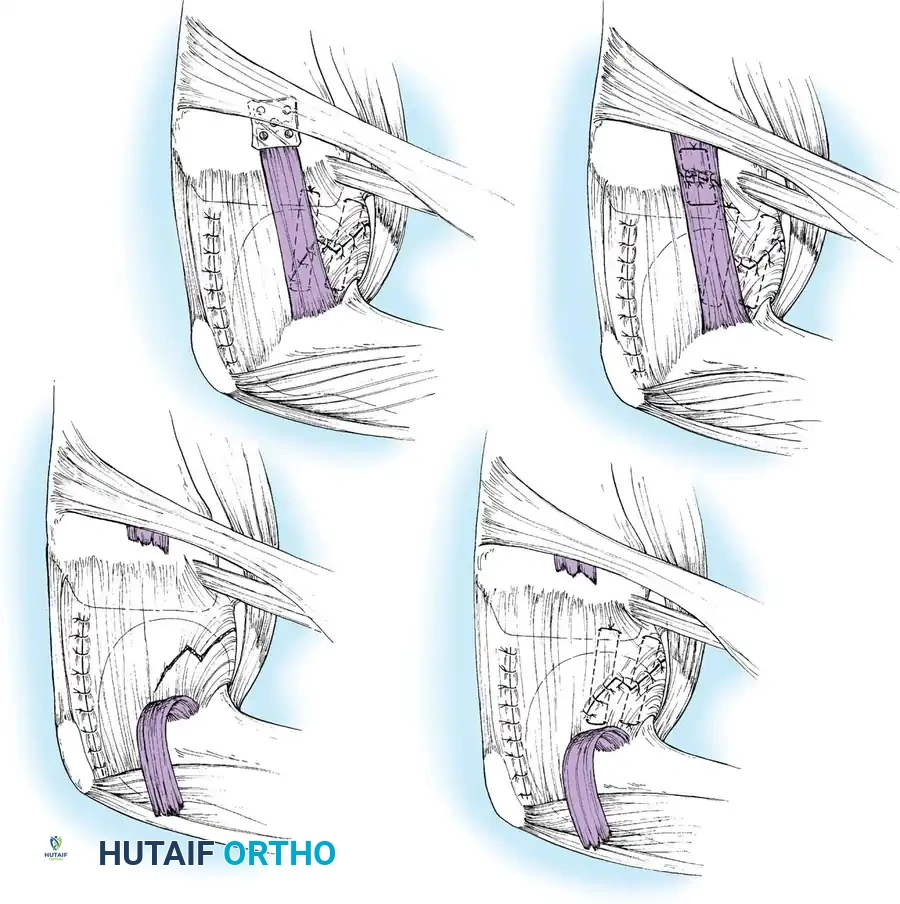
In cases of severe tissue maceration or chronic attenuation, the ligament may need to be advanced and secured to the bone using a spiked ligament washer or a cortical button to restore appropriate valgus stability.
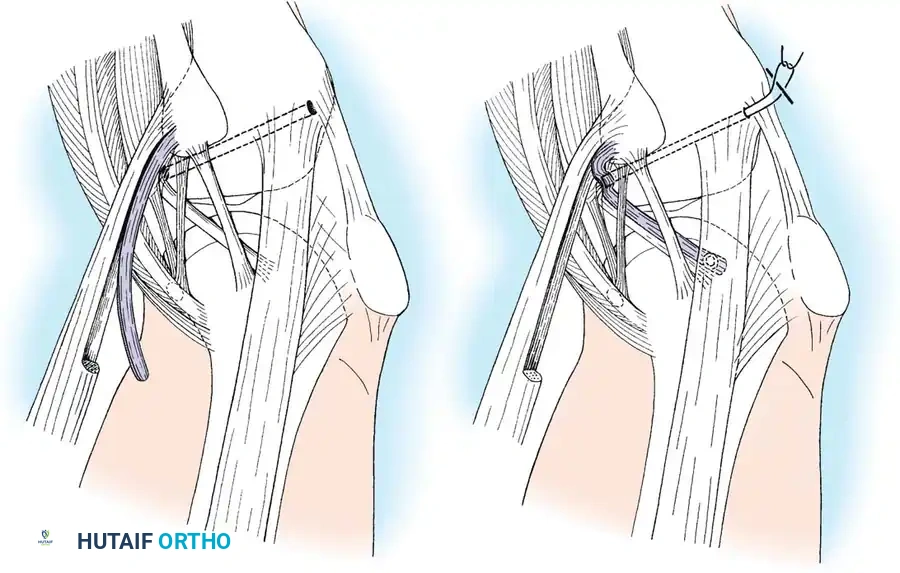
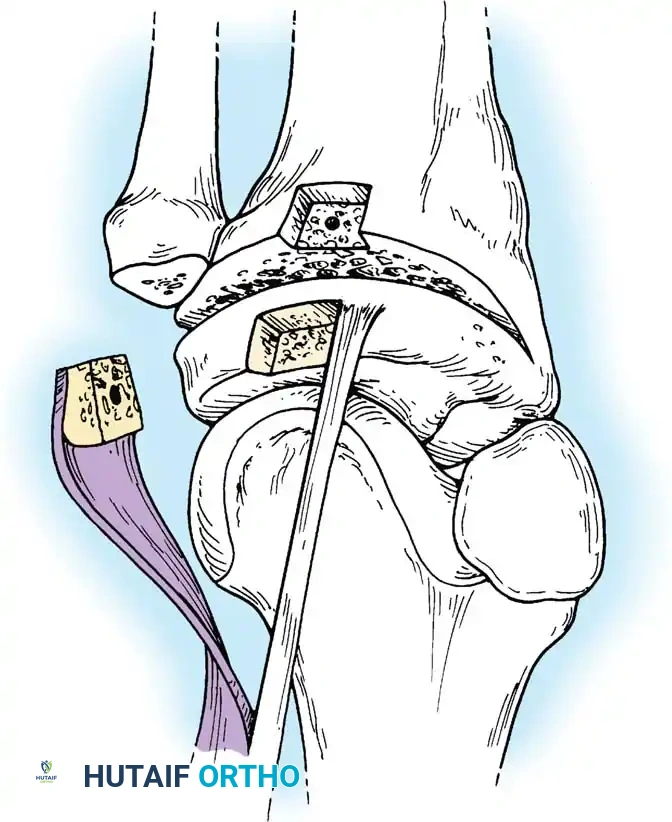
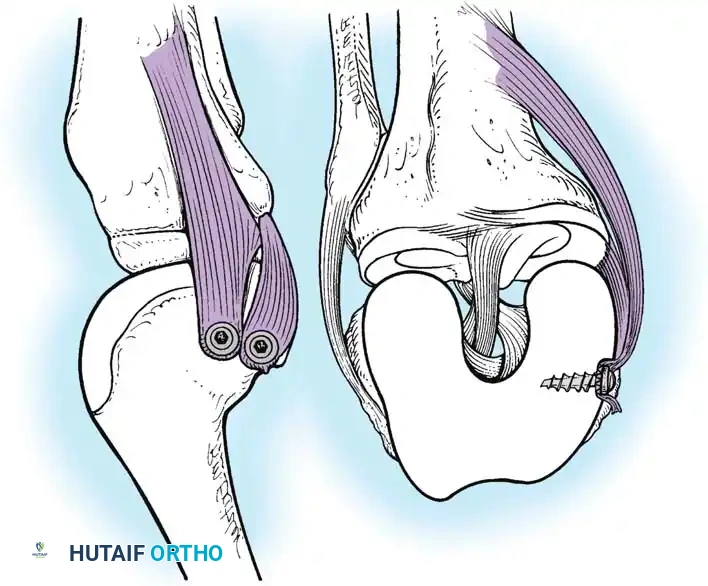
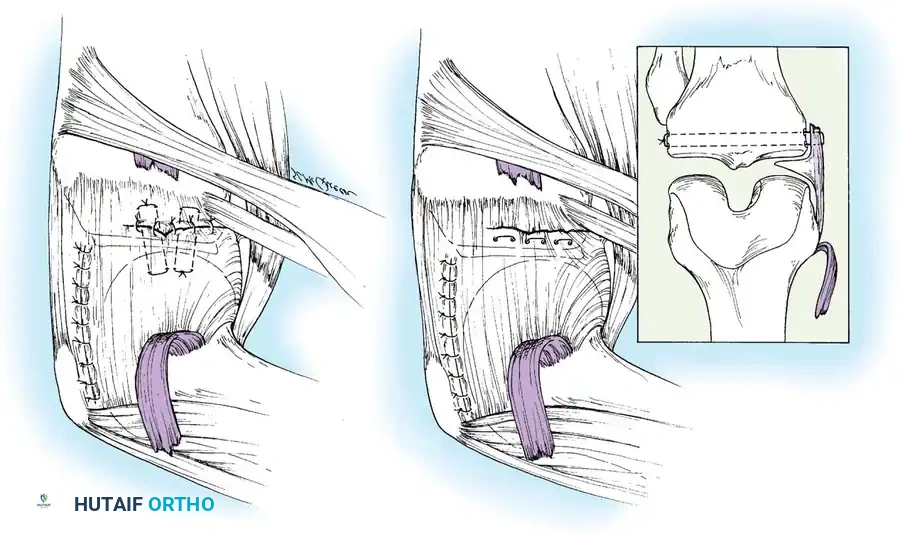
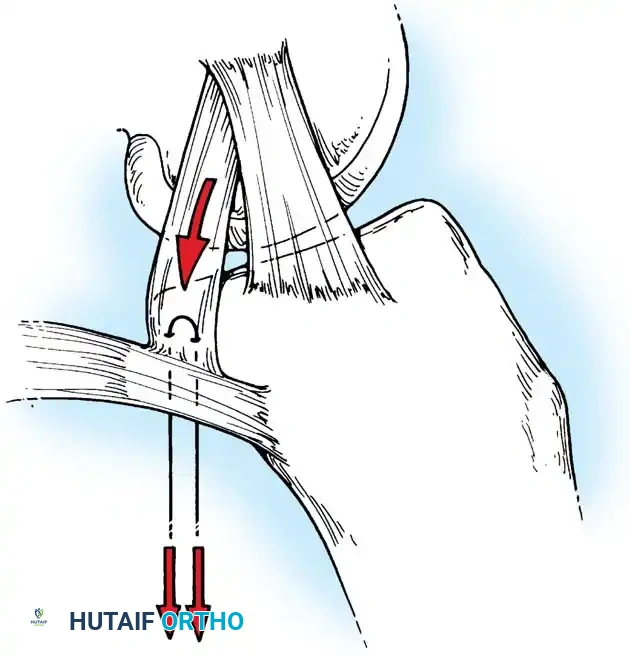
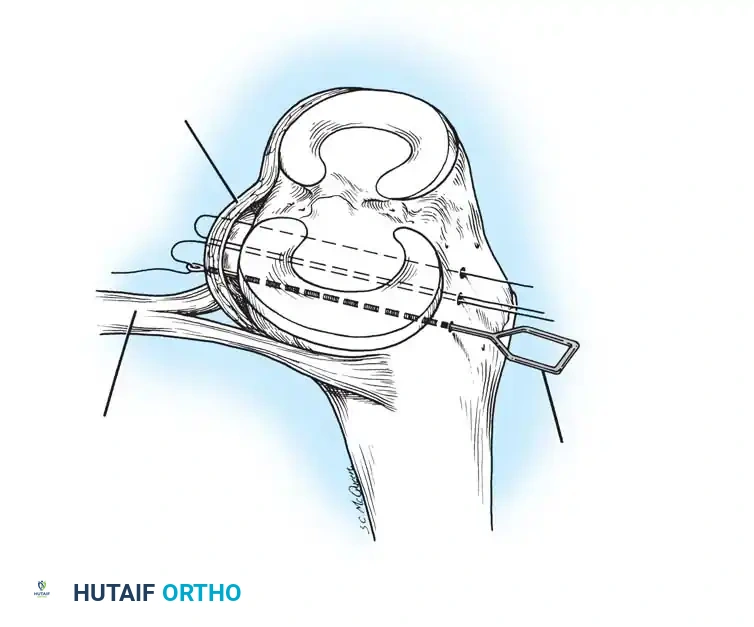
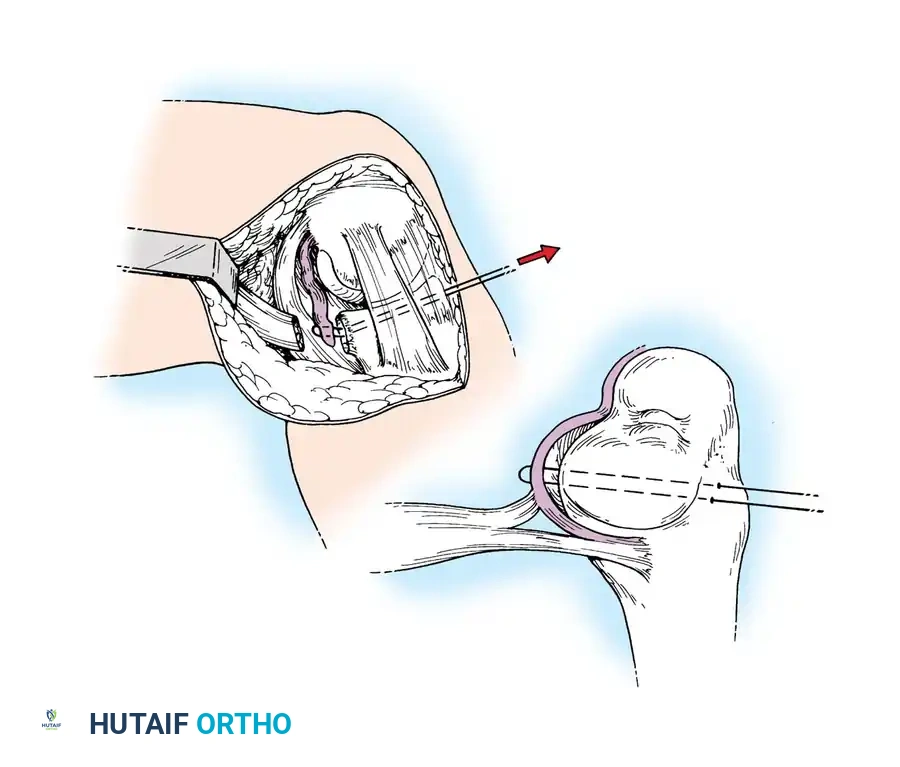
Lateral Collateral and Posterolateral Corner (PLC) Repair
Lateral-sided injuries are notoriously unforgiving and rarely heal with conservative management due to the inherent varus alignment of the lower extremity during the normal gait cycle.
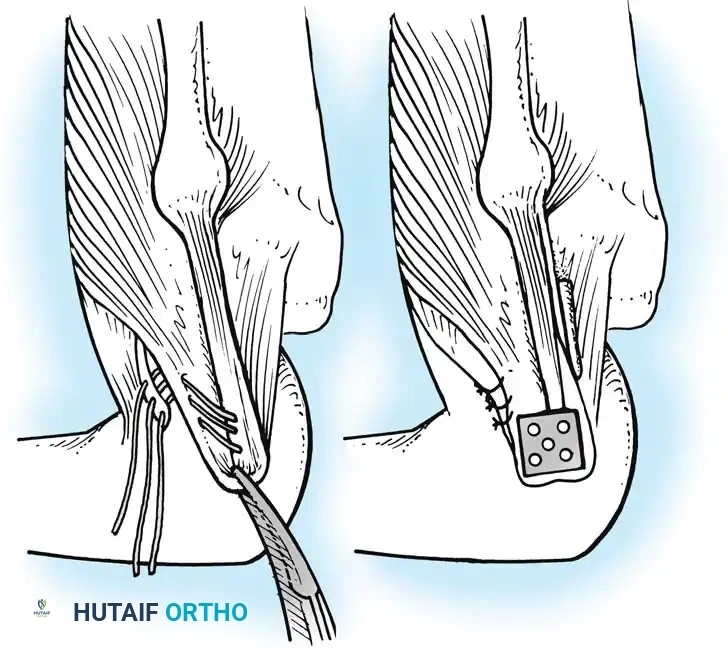
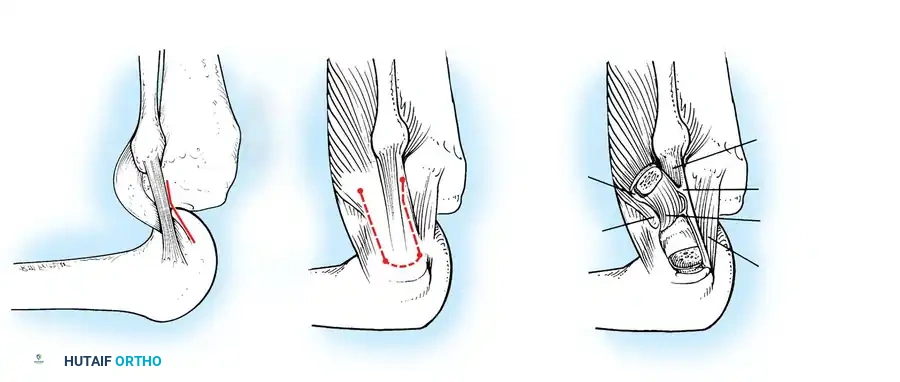
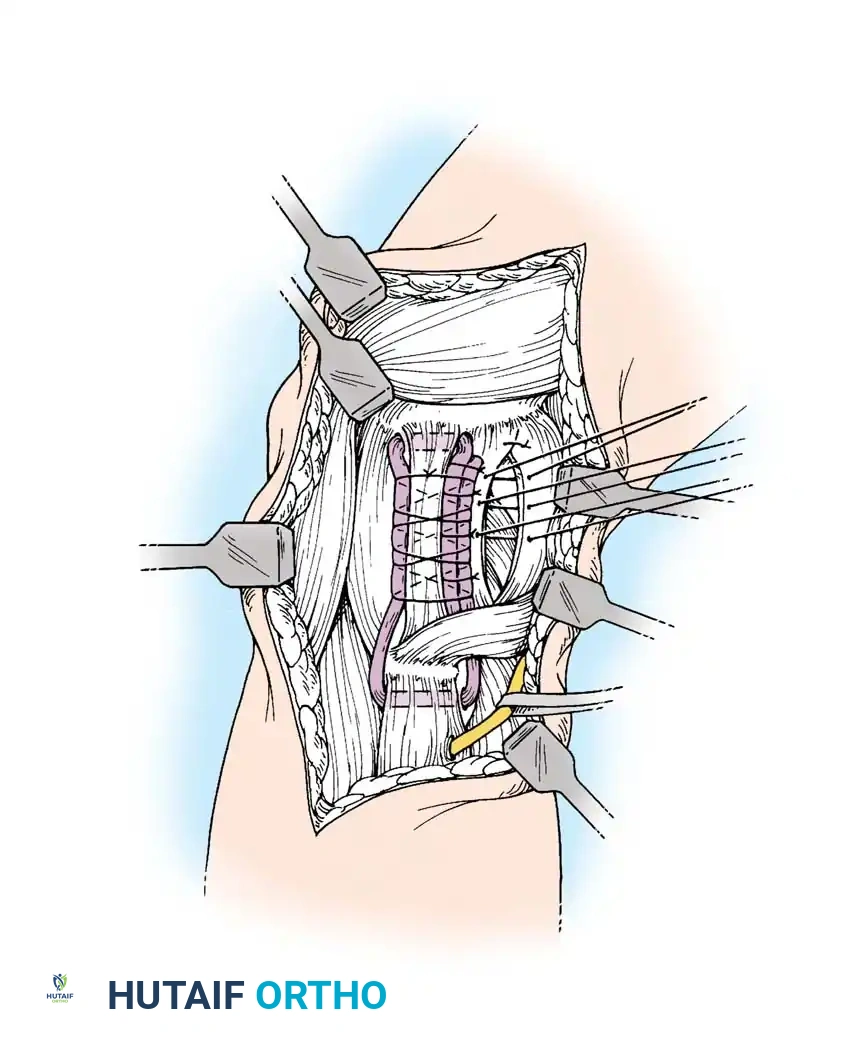
A curvilinear incision is made over the lateral aspect of the knee, incorporating the iliotibial band and the biceps femoris. The common peroneal nerve must be identified, neurolysed, and protected with a vessel loop throughout the procedure.
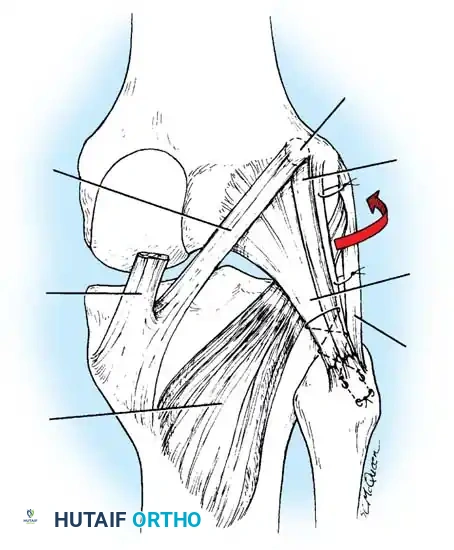
The LCL, popliteus tendon, and popliteofibular ligament are systematically evaluated. Avulsions from the fibular head are common.
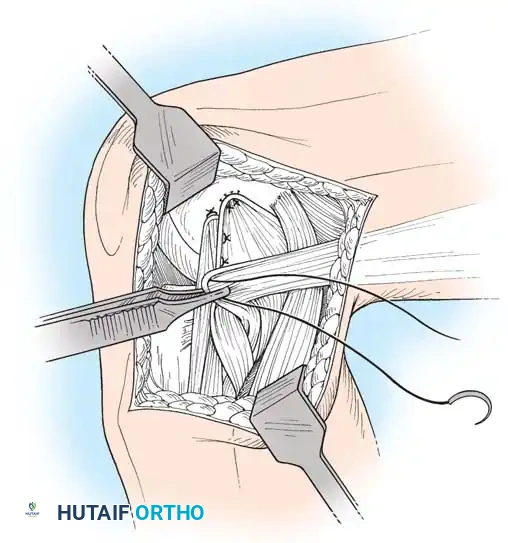
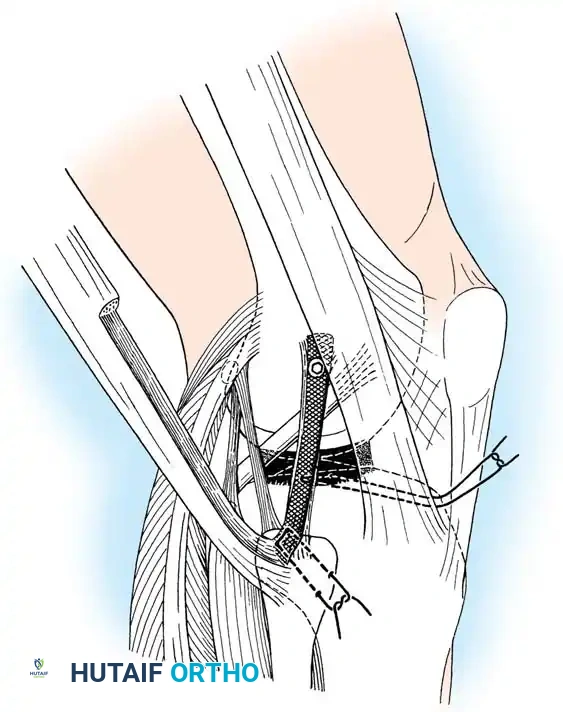
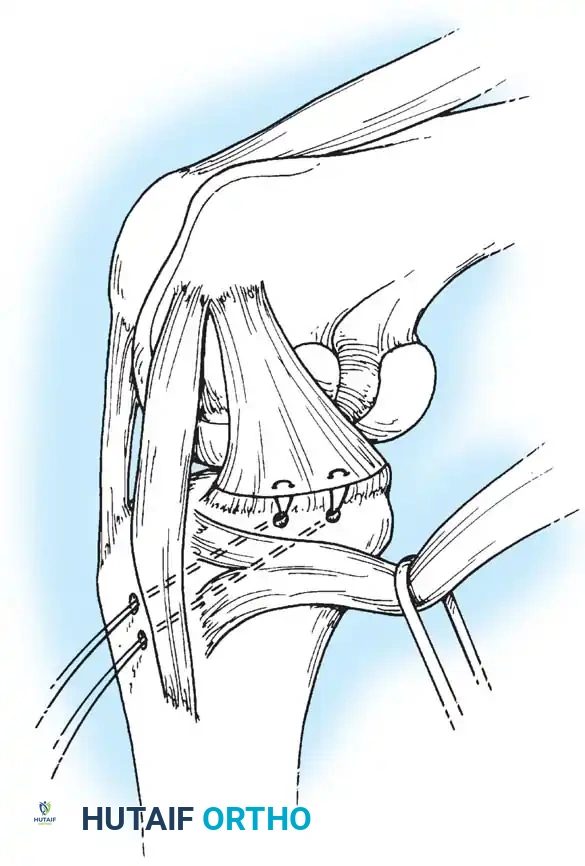
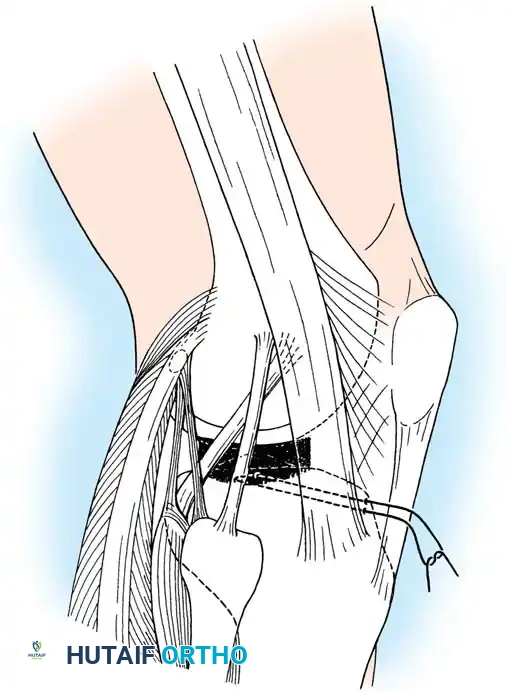
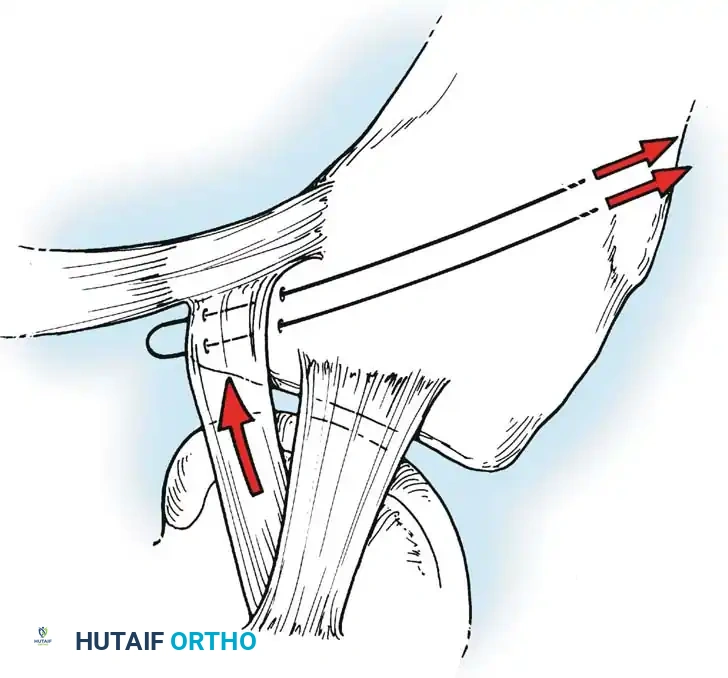
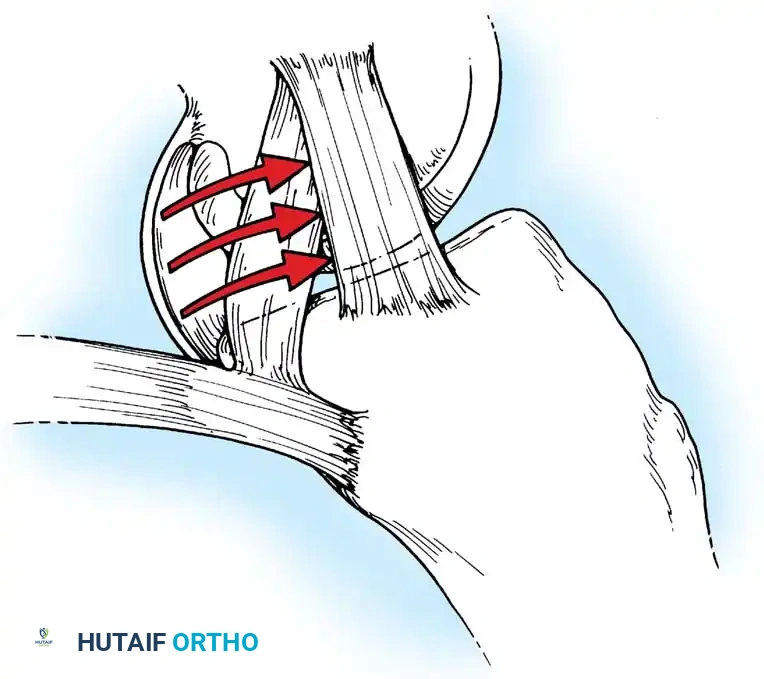
Deep capsular tears are repaired with interrupted sutures. If the LCL or popliteus is avulsed from the lateral femoral epicondyle, anatomical footprints are identified, and the structures are repaired using suture anchors. Tensioning must be performed with the knee in 30 degrees of flexion and neutral rotation.
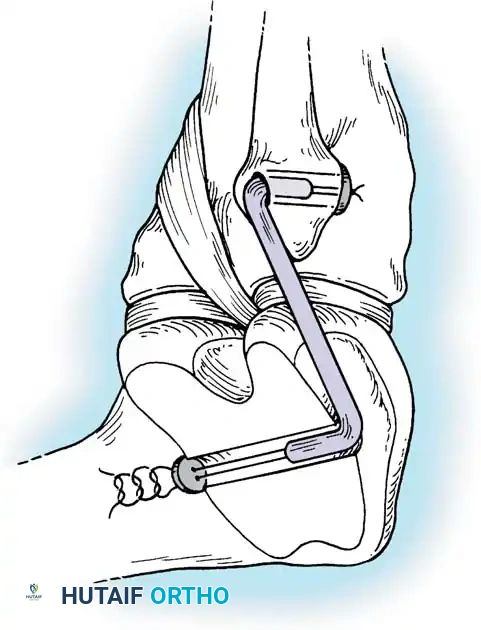
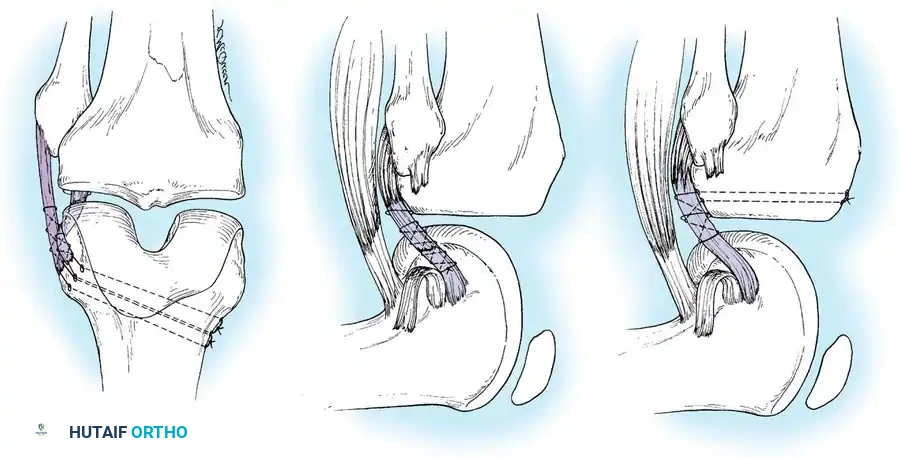
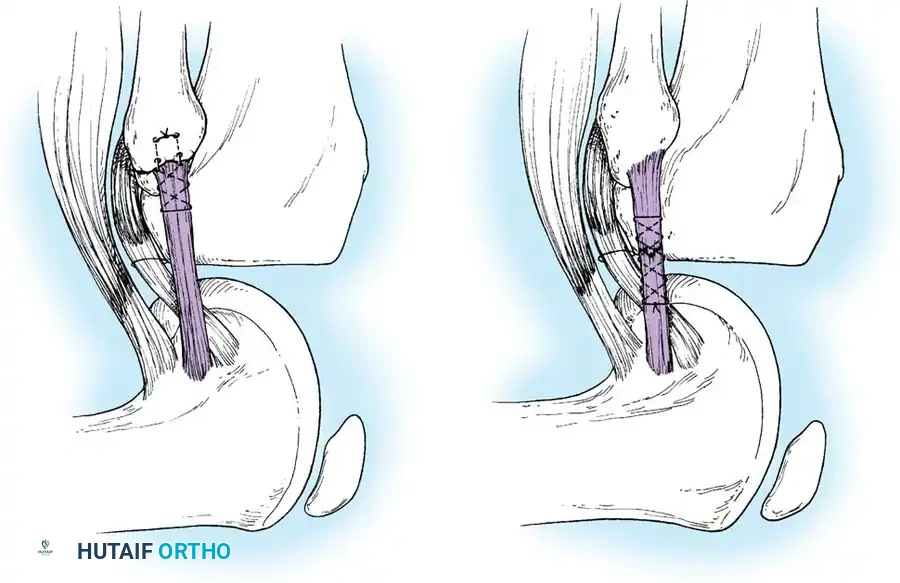
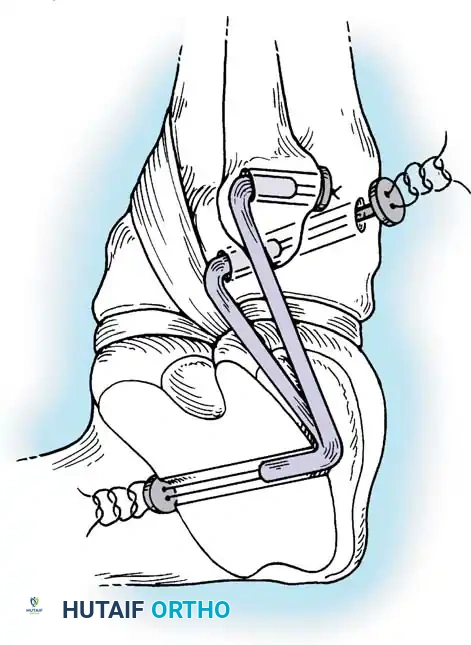
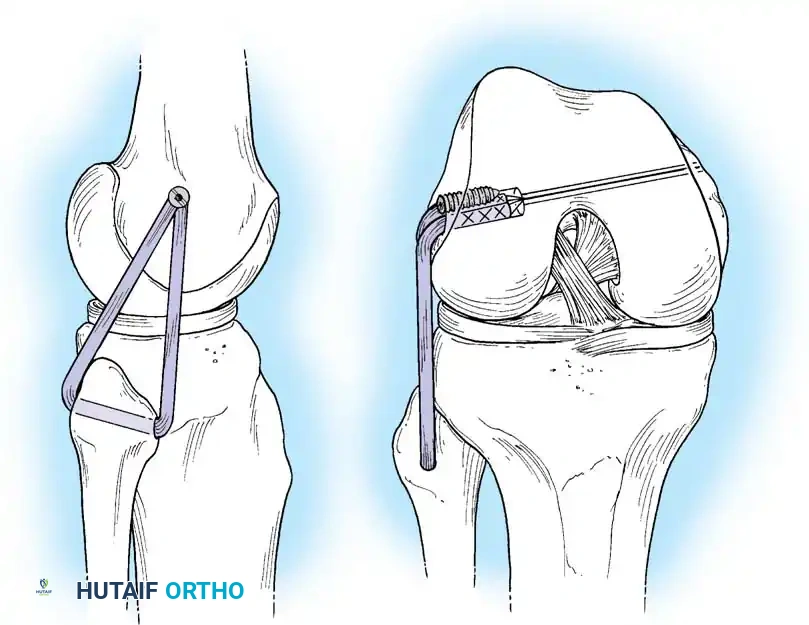
In cases where the native tissue is non-viable or the injury is subacute, an anatomical posterolateral corner reconstruction using an autograft or allograft (e.g., Achilles or semitendinosus) is mandated. The graft is routed to recreate the LCL and popliteofibular ligament.
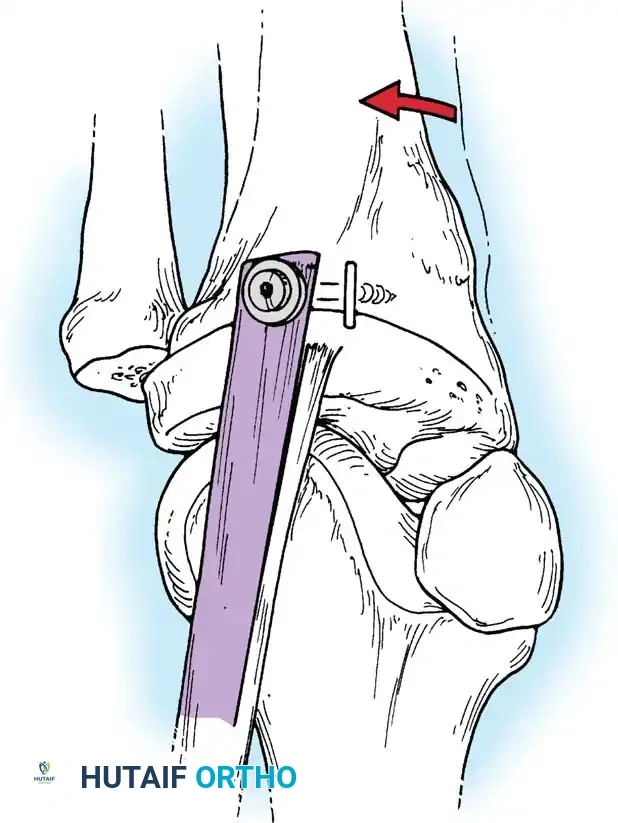
Robust fixation is achieved using interference screws or screw-and-washer constructs in the fibular head and lateral femoral condyle. The biomechanical integrity of the construct is verified by taking the knee through a full range of motion, ensuring no impingement or laxity.
POSTOPERATIVE REHABILITATION PROTOCOLS
The postoperative management of acute ligament repairs requires a delicate balance between protecting the healing tissue and preventing arthrofibrosis.
- Phase I: Protection and Early Motion (Weeks 0-4): The knee is placed in a hinged brace locked in extension for weight-bearing. Passive and active-assisted range of motion (ROM) is initiated immediately, typically restricted from 0 to 90 degrees to prevent excessive strain on the repair. Patellar mobilization is critical.
- Phase II: Controlled Loading (Weeks 4-8): Weight-bearing is progressively advanced. The brace is unlocked to allow full ROM. Closed kinetic chain exercises (e.g., mini-squats, leg presses) are introduced to stimulate collagen remodeling along lines of stress.
- Phase III: Strengthening (Weeks 8-16): Discontinuation of the brace. Focus shifts to progressive resistance training, proprioception, and neuromuscular control. Isokinetic strengthening of the quadriceps and hamstrings is emphasized.
- Phase IV: Return to Sport (Months 6-12): Progression to sport-specific drills, cutting, pivoting, and plyometrics. Return to competitive play is permitted only when the patient demonstrates >90% limb symmetry index on functional hop testing and possesses full, painless ROM with a stable clinical exam.
By adhering to strict biomechanical principles, executing meticulous surgical techniques, and guiding patients through evidence-based rehabilitation, orthopedic surgeons can successfully restore joint kinematics and return patients to their pre-injury levels of function following severe acute traumatic ligamentous lesions.
Associated Surgical & Radiographic Imaging




You Might Also Like