ACL Reconstruction Surgery: Prepare for Success & Full Recovery
Key Takeaway
Here are the crucial details you must know about ACL Reconstruction Surgery: Prepare for Success & Full Recovery. ACL reconstruction surgery is a procedure to repair a torn anterior cruciate ligament (ACL), a critical knee stabilizer. It is performed to restore knee stability, address buckling, and improve the ability to walk, run, or play sports after an ACL tear. This surgery involves replacing the damaged ligament to prevent the tibia from sliding excessively forward.
Introduction & Epidemiology
Anterior Cruciate Ligament (ACL) injury represents a significant and frequently encountered pathology within sports medicine and orthopedic surgery. The ACL is a primary static stabilizer of the knee, critical for restricting anterior tibial translation and controlling rotational stability. Its rupture results in functional instability, often precluding participation in pivoting sports and potentially predisposing the knee to secondary meniscal tears, chondral damage, and ultimately, accelerated osteoarthritis.
The annual incidence of ACL tears in the general population is estimated between 68 and 147 per 100,000 person-years, with a disproportionately high occurrence in young, active individuals, particularly those engaged in high-risk sports such as soccer, basketball, skiing, and American football. Female athletes demonstrate a 2-8 fold higher incidence of non-contact ACL injuries compared to their male counterparts, attributed to a multifactorial interplay of anatomical (e.g., Q-angle, notch dimensions), hormonal, biomechanical (e.g., landing mechanics, muscle firing patterns), and neuromuscular factors.
The mechanism of injury is predominantly non-contact (70-80%), often involving sudden deceleration, hyperextension, or a cutting/pivoting maneuver with a valgus collapse moment. Contact injuries typically involve a direct blow to the lateral knee, forcing it into valgus with internal or external rotation. Regardless of the mechanism, the primary goal of ACL reconstruction is to restore knee stability, protect meniscal and chondral integrity, and facilitate a safe return to pre-injury activity levels. The evolution of ACL reconstructive techniques has progressed from early open repairs and extra-articular procedures to modern arthroscopically-assisted, anatomically-based reconstructions, aiming to replicate the native ligament's footprint and biomechanical function.
Surgical Anatomy & Biomechanics
Surgical Anatomy
The ACL is an intra-articular, extrasynovial ligament that originates from the posteromedial aspect of the lateral femoral condyle within the intercondylar notch. Its femoral footprint is crescent-shaped, situated obliquely along the posterior aspect of the medial wall of the lateral femoral condyle. It inserts onto a broad, saddle-shaped area on the tibia, anterior and lateral to the medial tibial spine, between the tibial tubercles.
The ACL is conventionally described as comprising two functional bundles:
*
Anteromedial (AM) bundle:
Taut in flexion, looser in extension, primarily resists anterior tibial translation.
*
Posterolateral (PL) bundle:
Taut in extension, looser in flexion, primarily resists rotational loads and anterior tibial translation near extension.
These bundles are not anatomically distinct but represent a continuum of fibers with differential tensioning patterns throughout the range of motion. A comprehensive understanding of their relative positions and functions is paramount for anatomical tunnel placement.
Associated anatomical structures critically important during ACL reconstruction include:
*
Menisci:
Medial and lateral menisci, often concurrently injured (especially the lateral meniscus in acute tears, medial meniscus in chronic tears).
*
Collateral Ligaments:
Medial collateral ligament (MCL) and Lateral collateral ligament (LCL) complexes, which provide valgus and varus stability, respectively. Concurrent MCL injuries are common and often managed non-operatively initially.
*
Articular Cartilage:
Chondral lesions or osteochondral fractures, particularly of the lateral femoral condyle and lateral tibial plateau (bone bruise pattern), are frequently associated with acute ACL tears.
*
Neurovascular Structures:
The popliteal artery and tibial/peroneal nerves are located posteriorly to the knee joint capsule, requiring careful intraoperative awareness, especially during posterior tunnel drilling or screw insertion. The infrapatellar branch of the saphenous nerve is vulnerable during medial incisions for hamstring harvest.
Biomechanics
The ACL is the primary restraint to anterior tibial translation (85%) and a significant secondary restraint to internal and external tibial rotation, particularly near extension. Its contribution to knee stability is complex and interdependent with the menisci, secondary ligaments (MCL, LCL, PCL), and muscle dynamic stabilizers.
Key biomechanical concepts pertinent to ACL reconstruction include:
*
Knee Kinematics:
The ACL contributes to the "screw-home mechanism" during terminal knee extension, which involves external rotation of the tibia on the femur. ACL deficiency disrupts this intricate kinematic pattern.
*
Isometricity:
While a truly isometric graft cannot be achieved, the goal is to place tunnels in locations that minimize graft length changes throughout the range of motion. Modern anatomical reconstructions aim for near-isometric placement, primarily by respecting the native femoral and tibial footprints.
*
Graft Tensioning:
Appropriate graft tension is crucial. Over-tensioning can lead to joint stiffness, loss of motion, and increased graft stress, while under-tensioning can result in persistent laxity.
*
Ligamentization:
This biological process describes the transformation of the autograft/allograft into a structure resembling the native ACL. It involves initial avascular necrosis followed by revascularization, cellular repopulation, and collagen remodeling. This process can take 12-24 months and influences rehabilitation timelines.
*
Rotational Stability:
While single-bundle ACL reconstruction effectively restores anterior-posterior stability, some residual rotational laxity (e.g., pivot shift) may persist, especially in high-risk patients. This has led to renewed interest in anatomical double-bundle reconstruction and lateral extra-articular tenodesis (LEAT) or anterolateral ligament (ALL) reconstruction for enhanced rotational control.
Indications & Contraindications
The decision for ACL reconstruction is individualized, considering patient age, activity level, functional instability, presence of associated injuries, and overall health status.
Operative Indications
- Symptomatic Instability: Recurrent episodes of "giving way" or buckling during activities of daily living, sport-specific maneuvers, or ambulation, often exacerbated by pivoting or cutting. This is the most compelling indication.
- Young, Active Patients: Individuals planning to return to high-demand, pivoting, or contact sports (e.g., football, soccer, basketball, skiing).
- Associated Meniscal Tears: Particularly reparable meniscal tears (e.g., longitudinal tears in the red-red zone, bucket-handle tears) that require simultaneous stabilization for healing. Unstable meniscal tears in an ACL-deficient knee are at high risk of re-tear or failure.
- Associated Ligamentous Injuries: Combined ACL and MCL/LCL injuries, particularly Grade III injuries with persistent instability, may necessitate a staged or combined reconstructive approach.
- Chronic ACL Deficiency with Progressive Instability: Patients who initially pursued non-operative management but develop increasing instability or secondary meniscal/chondral pathology.
- Failure of Non-Operative Management: Persistent symptomatic instability despite dedicated rehabilitation and activity modification.
Contraindications
Absolute Contraindications:
*
Active Infection:
Uncontrolled local or systemic infection.
*
Uncorrected Severe Lower Extremity Malalignment:
Significant varus or valgus malalignment (e.g., >5 degrees) should be addressed with an osteotomy prior to or concurrent with ACL reconstruction, as it can predispose to graft failure and continued instability.
*
Severe Medical Comorbidities:
Patients with medical conditions precluding safe anesthesia and surgery.
*
Extremely Poor Patient Compliance/Motivation:
The success of ACL reconstruction is highly dependent on patient adherence to a rigorous and lengthy rehabilitation protocol.
Relative Contraindications:
*
Skeletally Immature Patients:
Risk of physeal injury and growth disturbance. Physeal-sparing techniques (e.g., epiphyseal tunnels, all-epiphyseal, over-the-top, transphyseal with small tunnels) or non-operative management until skeletal maturity may be considered based on Risser stage, remaining growth, and activity level.
*
Advanced Degenerative Joint Disease (OA):
In knees with significant chondral loss and established osteoarthritis, ACL reconstruction alone may not provide satisfactory outcomes. Concurrent or staged osteotomy or consideration of arthroplasty might be more appropriate.
*
Sedentary Lifestyle:
Patients with a sedentary lifestyle who experience minimal or no functional instability may be candidates for non-operative management, provided they are counselled on the risks of future meniscal or chondral damage.
*
Limited Range of Motion (Arthrofibrosis):
Pre-existing arthrofibrosis or significant stiffness should be addressed prior to or concomitantly with ACL reconstruction to optimize postoperative motion.
Table: Operative vs. Non-Operative Indications for ACL Tears
| Parameter | Operative Indication | Non-Operative Indication |
|---|---|---|
| Age | Young (<30-40 years) | Older (>40-50 years) |
| Activity Level | High-demand, pivoting/contact sports, manual labor | Low-demand, sedentary, straight-line activities only |
| Instability Symptoms | Recurrent "giving way" during ADLs or sports | No or minimal instability, occasional minor symptoms |
| Associated Injuries | Reparable meniscal tears, Grade III MCL/LCL tears | Isolated ACL tear, stable meniscal tears, Grade I/II MCL |
| Physical Exam | Gross instability (e.g., 2+/3+ Lachman, positive Pivot Shift) | Mild laxity, negative/trace Pivot Shift |
| Motivation/Compliance | High motivation for rigorous rehabilitation | Poor motivation, inability to commit to rehab |
| Degenerative Changes | Minimal to none | Advanced osteoarthritis |
| Risk of Secondary Injury | High (e.g., young athlete with unstable knee) | Lower (e.g., older, sedentary patient) |
Pre-Operative Planning & Patient Positioning
Thorough pre-operative planning is crucial for optimizing outcomes and minimizing complications.
Pre-Operative Planning
-
History and Physical Examination:
- Detailed history of injury mechanism, symptoms (pain, swelling, giving way), previous knee injuries, and activity goals.
- Systematic knee examination: Inspection (effusion, atrophy), palpation, range of motion, and stability testing (Lachman test, Anterior Drawer test, Pivot Shift test – often under anesthesia, sag test for PCL). Assessment of meniscal pathology (McMurray's, joint line tenderness) and collateral ligaments.
-
Imaging:
- Radiographs: AP, lateral, Merchant (patellofemoral) views, Rosenberg view (weight-bearing 45° flexion) to assess for degenerative changes, effusions, and rule out fractures. Notch osteophytes or tunnel widening from previous surgery should be noted.
- Magnetic Resonance Imaging (MRI): Confirms ACL rupture, delineates tear pattern, and evaluates for associated injuries (meniscal tears, chondral lesions, collateral ligament sprains, bone bruises). Also aids in assessing graft donor site integrity (e.g., hamstring scarring).
-
Graft Selection:
A critical decision, often based on patient factors (age, activity, previous surgery), surgeon preference, and graft availability.
-
Autografts:
- Bone-Patellar Tendon-Bone (BPTB): Gold standard for many, excellent strength, reliable bone-to-bone healing, good for revision. Disadvantages: Anterior knee pain, patellar fracture, extensor mechanism morbidity.
- Hamstring Tendons (Semitendinosus and Gracilis - ST/G): Less anterior knee pain, smaller incision. Disadvantages: Slower soft tissue-to-bone healing, potential for hamstring weakness, less reliable for revision.
- Quadriceps Tendon (QT): Increasing popularity, strong, versatile (bone plug or soft tissue only), less donor site morbidity than BPTB. Disadvantages: Potential for anterior knee pain, quadriceps weakness.
-
Allografts:
- Advantages: No donor site morbidity, quicker surgery, larger size options.
- Disadvantages: Risk of disease transmission (though rare with modern processing), slower incorporation, higher re-rupture rates in younger, active patients (especially <25-30 years), cost. Reserved for select cases (older, less active, revision surgery, multi-ligament injury).
-
Autografts:
- Pre-habilitation: Pre-operative physical therapy focusing on restoring full range of motion (especially extension), reducing effusion, and strengthening the quadriceps and hamstrings, is strongly recommended to improve post-operative outcomes.
- Anesthesia and DVT Prophylaxis: Routine pre-operative anesthesia consultation. DVT prophylaxis (e.g., mechanical compression devices, pharmacologic agents) considered based on patient risk factors and surgical duration.
- Equipment Preparation: Ensure all necessary arthroscopy equipment, specific graft harvest kits, tunnel reamers, guide pins, fixation devices (interference screws, suspensory fixation, staples), and instruments for associated pathology (meniscal repair, chondral procedures) are readily available.
Patient Positioning
- Supine Position: Patient is placed supine on the operating table.
- Tourniquet: A high thigh tourniquet is applied, ensuring adequate padding, and inflated after exanguination of the limb.
- Leg Holder: The operative leg is positioned in a standard leg holder, allowing for full range of motion and valgus stress application. Alternatively, a pneumatic foot holder with a bolster under the ipsilateral gluteal region can be used, providing good mobility.
- Foot Support: A padded foot support or traction boot may be used for maintaining knee flexion or applying longitudinal traction.
- Contralateral Leg: The contralateral leg is often placed in a well-padded stirrup or frog-leg position to facilitate surgeon access.
- Sterile Prep and Draping: Standard sterile preparation of the entire lower extremity, from the iliac crest to the toes, including potential graft harvest sites (medial proximal tibia for hamstrings, anterior knee for BPTB/QT), is performed. The knee is typically draped to allow free manipulation and full visualization of relevant landmarks.
Detailed Surgical Approach / Technique (Hamstring Autograft Focus)
This section outlines a typical arthroscopically-assisted single-bundle ACL reconstruction using hamstring autograft. Variations exist based on surgeon preference, graft choice, and patient anatomy.
1. Arthroscopic Portal Placement
- Anterolateral (AL) Portal: Primary viewing portal, positioned approximately 1 cm superior to the lateral border of the patellar tendon, just lateral to the inferior pole of the patella.
- Anteromedial (AM) Portal: Working portal, positioned 1 cm superior to the medial border of the patellar tendon, medial to the inferior pole of the patella. Ideally, this portal allows access to the entire intercondylar notch and both femoral and tibial footprints.
- Accessory Medial Portal (AMP): An optional, more distal medial portal (accessory anteromedial or "far medial") may be created as needed for specialized tasks, such as optimal femoral tunnel drilling for anatomical footprints, meniscal repair, or removal of intermeniscal ligament pathology.
2. Diagnostic Arthroscopy
- Systematic evaluation of all knee compartments (suprapatellar pouch, patellofemoral joint, medial compartment, lateral compartment, intercondylar notch) with the arthroscope in the AL portal.
- Assess for meniscal tears (repair vs. partial meniscectomy), chondral lesions, loose bodies, and associated ligamentous injuries (MCL, LCL, PCL).
- Confirm ACL rupture, visualize the remnant, and clear any impinging soft tissue or scar tissue from the notch using a motorized shaver or radiofrequency ablator. Preserve any viable ACL remnant tissue as it may contribute to graft revascularization and proprioception.
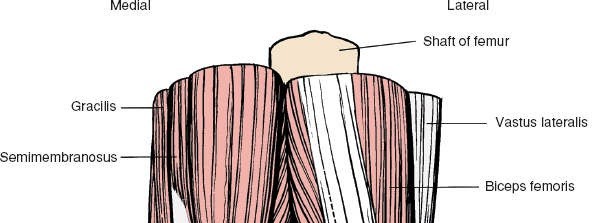
3. Graft Harvest (Hamstring Autograft)
- A 2-3 cm longitudinal or transverse incision is made over the pes anserinus insertion, approximately 2-3 cm distal and medial to the tibial tubercle.
- The sartorius fascia is incised longitudinally, revealing the semitendinosus and gracilis tendons.
- The semitendinosus tendon is identified first, freed from its surrounding adhesions, and a closed-ended tendon stripper is passed proximally to detach it from its musculotendinous junction. The same procedure is performed for the gracilis tendon.
- The harvested tendons are cleaned of muscle remnants, folded in half (typically creating a 4-strand graft), whipstitched at the ends with strong non-absorbable sutures, and then sized to determine the optimal diameter (aiming for ≥8 mm, ideally ≥8.5 mm). Length is typically 60-70 mm.
4. Femoral Tunnel Creation
- Anatomical Femoral Footprint Identification: The ACL's femoral origin is on the posteromedial aspect of the lateral femoral condyle. The goal is to place the center of the femoral tunnel within the native ACL footprint, between the AM and PL bundles.
-
Guide Pin Placement (Anteromedial Portal Technique):
- The knee is flexed to 110-130 degrees to minimize the risk of posterior wall blowout.
- The guide pin is inserted through the AM portal, aiming for the center of the femoral footprint, approximately at the 10 o'clock position (right knee) or 2 o'clock (left knee) on the condyle wall.
- Image intensifier (fluoroscopy) can be used to confirm guide pin placement.
- Reaming: A cannulated reamer matching the graft diameter is passed over the guide pin, carefully reaming the femoral tunnel. Reaming depth is typically 25-30 mm, depending on the graft length and fixation device. The posterior cortical wall must be preserved (minimum 2 mm bone bridge).
- Tunnel Chamfering: The edges of the tunnel opening are chamfered to prevent graft abrasion.
5. Tibial Tunnel Creation
- Anatomical Tibial Footprint Identification: The ACL's tibial insertion is anterior and lateral to the medial tibial spine. The goal is to place the tunnel in the "sweet spot" to avoid impingement in extension and reproduce stability throughout the range of motion.
- Tibial Guide Placement: An arthroscopic tibial guide is inserted through the AM portal. The tip is placed at the center of the native tibial footprint. This is typically located 5-7 mm anterior to the PCL and 43-49% of the anterior-posterior distance of the medial tibial plateau from the anterior edge.
- Guide Pin Drilling: A guide pin is drilled from the anteromedial aspect of the tibia, proximally and posteriorly, into the knee joint at the desired tibial footprint. The angle should prevent impingement against the intercondylar notch in full extension.
- Reaming: A cannulated reamer matching the graft diameter is passed over the guide pin, reaming the tibial tunnel. The length of the tibial tunnel should allow sufficient space for the graft and fixation device.
6. Graft Passage
- A passing suture or wire is retrieved from the femoral tunnel, passed across the joint, and out through the tibial tunnel.
- The graft is then attached to this passing suture and carefully pulled through the tibial tunnel and into the femoral tunnel. Meticulous care is taken to avoid graft damage, twisting, or impingement. The graft should fill the tunnels snugly.
7. Graft Fixation
-
Femoral Fixation:
- Suspensory Fixation (e.g., Endobutton, Cortical Button): A button is flipped on the lateral femoral cortex, suspending the graft within the tunnel. This allows for cortical load sharing and is biomechanically strong.
- Interference Screw: A bioabsorbable or metallic screw is inserted between the graft and the tunnel wall, providing robust fixation.
- Cross-Pin/TransFix: Pins are drilled perpendicular to the femoral tunnel, capturing the graft.
-
Tibial Fixation:
- Interference Screw: Similar to femoral fixation, providing strong in-tunnel fixation.
- Post Screw and Washer: Sutures from the graft are tied over a post screw and washer on the anteromedial aspect of the tibia.
- Staple/Suture Post: Less common as primary fixation, more as adjunct.
- Hybrid Fixation: Often a combination of methods (e.g., suspensory on femoral, interference on tibial) is used to maximize construct strength.
- Graft Tensioning: The graft is typically tensioned to approximately 80 N (or firm manual tension) while the knee is held in 20-30 degrees of flexion (for single-bundle reconstruction) to prevent over-constraining the knee in extension. Dynamic tensioning (cycling the knee through range of motion) can reduce creep.
- Intraoperative Assessment: After fixation, a thorough arthroscopic assessment of graft placement and tension is performed. Lachman and Pivot Shift tests are repeated to confirm stability. The graft should be free of impingement throughout the range of motion.
8. Wound Closure
- The arthroscopic portals and graft harvest site are closed in layers.
- Local anesthetic is infiltrated for post-operative pain control.
- A sterile dressing is applied, and often a hinged knee brace is placed in full extension.
Special Considerations
- Double Bundle Reconstruction: Aims to reproduce the AM and PL bundles with two separate grafts/tunnels. Offers superior rotational stability in some studies but is technically demanding with increased surgical time and potential for tunnel convergence.
- Anterolateral Ligament (ALL) Reconstruction/Lateral Extra-Articular Tenodesis (LEAT): Increasing interest in augmenting ACL reconstruction, particularly in high-risk patients (e.g., young athletes with high-grade pivot shift, revision cases) to control residual rotational laxity.
- Physeal-Sparing Techniques: In skeletally immature patients, specific techniques are employed to avoid drilling across open physes, minimizing the risk of growth disturbance. Options include all-epiphyseal tunnels, over-the-top techniques, or partial transphyseal drilling with small diameter grafts.
Complications & Management
Complications following ACL reconstruction, though relatively infrequent, can significantly impact patient outcomes. Vigilant intraoperative technique and comprehensive postoperative care are essential for prevention and timely management.
Intraoperative Complications
-
Neurovascular Injury:
- Incidence: Rare (<1%), but potentially devastating. Popliteal artery, tibial nerve, peroneal nerve.
- Mechanism: Guide pin or reamer penetration of posterior capsule, excessive traction, graft passage. Saphenous nerve injury during hamstring harvest.
- Management: Immediate recognition, angiogram/nerve conduction studies, surgical exploration and repair, compartment syndrome monitoring.
-
Fracture:
- Incidence: Rare. Tibial plateau, femoral condyle, patella (BPTB harvest).
- Mechanism: Tunnel blowout, guide pin malposition, aggressive reaming, stress riser from graft harvest.
- Management: Internal fixation, protected weight-bearing, re-assessment of fixation.
-
Graft Damage/Misplacement:
- Incidence: Varies.
- Mechanism: Abrasion during passage, tunnel impingement, wrong tunnel placement.
- Management: Graft exchange, re-drilling tunnels, notchplasty.
-
Instrument Breakage:
- Incidence: Rare.
- Mechanism: Fatigue, misuse.
- Management: Retrieval, fragment identification, potential open conversion.
Early Postoperative Complications
-
Infection (Septic Arthritis):
- Incidence: 0.1% to 1.0%.
- Presentation: Pain, swelling, redness, fever, limited ROM, elevated inflammatory markers.
- Management: Urgent arthroscopic irrigation and debridement, synovial fluid culture, broad-spectrum IV antibiotics tailored to culture results. Graft retention is often attempted but may require removal if infection persists.
-
Arthrofibrosis/Stiffness:
- Incidence: 5-10%, more common with delayed surgery, poor pre-hab.
- Presentation: Loss of flexion, extension, or both. Common causes: cyclops lesion, poor tunnel placement, over-tensioning.
- Management: Aggressive physical therapy, manipulation under anesthesia (MUA), arthroscopic lysis of adhesions (LOA) and cyclops lesion resection.
-
Hemarthrosis:
- Incidence: Common (mild), significant (less common).
- Mechanism: Postoperative bleeding.
- Management: RICE, analgesia. Aspiration for severe pain/tension, but risk of infection.
-
Deep Vein Thrombosis (DVT) / Pulmonary Embolism (PE):
- Incidence: DVT 1-10%, PE <0.5%. Higher in patients with risk factors.
- Management: DVT prophylaxis (mechanical/pharmacologic) based on risk. Diagnosis with duplex ultrasound. Anticoagulation for DVT/PE.
-
Numbness:
- Incidence: Common (minor), especially infrapatellar branch of saphenous nerve (hamstring harvest).
- Management: Reassurance, resolves partially over time. Neuropathic pain rarely requires intervention.
Late Postoperative Complications
-
Graft Failure/Re-rupture:
- Incidence: 2-10%, higher in young patients (<25 years), female athletes, and those returning to sport too early.
- Mechanism: Re-injury trauma, technical error (malposition, poor fixation), biological failure (lack of ligamentization).
- Management: Revision ACL reconstruction. Requires careful pre-operative planning to address original technical errors, tunnel widening, and graft choice.
-
Persistent Instability:
- Incidence: Varies.
- Mechanism: Incomplete restoration of kinematics, graft laxity, untreated associated laxity (e.g., ALL).
- Management: Revision surgery, potentially with augmentation (ALL/LEAT).
-
Patellofemoral Pain:
- Incidence: 10-25%, higher with BPTB grafts.
- Mechanism: Donor site morbidity, altered patellar tracking.
- Management: Physical therapy, activity modification, analgesia. Rarely surgical intervention.
-
Tunnel Widening:
- Incidence: Common radiographically, but often asymptomatic. More pronounced with soft tissue grafts and interference screw fixation.
- Mechanism: Biomechanical (micromotion, stress shielding) and biological (osteolysis, synovial fluid ingress).
- Management: Usually conservative unless symptomatic. May complicate revision surgery.
-
Post-traumatic Osteoarthritis (PTOA):
- Incidence: High (up to 50-70% within 10-20 years), regardless of successful reconstruction.
- Mechanism: Initial injury, subsequent meniscal/chondral damage, altered knee kinematics.
- Management: Symptomatic management of OA. ACL reconstruction may reduce progression but does not eliminate risk.
Table: Common Complications, Incidence, and Salvage Strategies
| Complication | Incidence (%) | Management / Salvage Strategy |
|---|---|---|
| Infection | 0.1-1.0 | Urgent arthroscopic I&D, antibiotics, ± graft removal |
| Arthrofibrosis/Stiffness | 5-10 | PT, MUA, arthroscopic LOA/cyclops resection |
| Graft Failure/Re-rupture | 2-10 | Revision ACL reconstruction (addressing original error, tunnel management) |
| Persistent Instability | Variable | Revision ACL, ± ALL/LEAT augmentation, address concomitant laxity |
| Patellofemoral Pain | 10-25 (BPTB) | PT, activity modification, analgesia |
| DVT/PE | 1-10 (DVT), <0.5 (PE) | Prophylaxis (mechanical/pharmacologic), anticoagulation |
| Neurovascular Injury | <1 | Immediate repair, compartment monitoring |
| Tunnel Widening | High (radiographic) | Conservative if asymptomatic. Bone grafting for revision. |
| Post-traumatic OA | 50-70 (long-term) | Symptomatic management, future arthroplasty consideration |
Post-Operative Rehabilitation Protocols
Post-operative rehabilitation is as critical as the surgical procedure itself for optimizing outcomes and ensuring a safe return to activity. Protocols are typically phased, guided by biological healing timelines and individualized patient progression.
Phase I: Protection & Early Motion (Weeks 0-6)
- Goals: Protect graft, minimize pain/swelling, restore full knee extension, achieve early flexion, initiate quadriceps activation.
-
Weight-Bearing:
- Initial: Partial weight-bearing (PWB) with crutches, often for 2-4 weeks.
- Progression: Full weight-bearing (FWB) as tolerated, once quadriceps control is achieved and gait is normalized.
- Bracing: Use of a hinged knee brace, locked in extension initially (for ambulation and sleep), is common but controversial. Some protocols allow for immediate ROM without bracing. If used, gradually unlock for increasing flexion.
-
Exercises:
- Range of Motion: Passive range of motion (PROM) and active-assisted range of motion (AAROM). Focus on achieving full extension immediately (heel props, prone hangs). Flexion is progressed gradually (CPM machine, heel slides).
- Muscle Activation: Quadriceps setting, straight leg raises (SLR) in multiple planes (supine, side-lying), gluteal sets, hamstring co-contractions.
- Proprioception: Gentle weight shifts, balance training on stable surfaces (if FWB).
- Cryotherapy & Compression: Consistent application for edema and pain control.
- Avoid: Open kinetic chain (OKC) resisted knee extension exercises between 0-45 degrees, which generate high anterior tibial shear forces on the healing graft.
Phase II: Strength & Neuromuscular Control (Weeks 6-12)
- Goals: Restore full pain-free range of motion, improve muscle strength and endurance, enhance neuromuscular control and proprioception, initiate light functional activities.
-
Exercises:
- Strength Training: Progression to closed kinetic chain (CKC) exercises (mini-squats, leg presses, lunges, step-ups), hamstring curls (OKC >60 degrees flexion), calf raises. Gradual introduction of OKC knee extension (e.g., from 90-45 degrees, then from 90-0 degrees cautiously at 12+ weeks).
- Cardiovascular: Stationary cycling, elliptical trainer, swimming (kickboard prohibited initially).
- Proprioception & Balance: Unstable surface training (wobble board, foam pad), single-leg stance, single-leg balance.
- Core Strengthening: Plank, bird-dog, abdominal exercises.
- Avoid: Sudden twisting motions, high impact activities, activities requiring cutting or pivoting.
Phase III: Return to Activity & Sport-Specific Training (Months 3-6)
- Goals: Progress strength, power, and agility; initiate sport-specific drills; prepare for return to advanced activities.
-
Exercises:
- Plyometrics: Low-level hopping, jumping, and landing drills.
- Agility Drills: Ladder drills, cone drills (straight line then gradual change of direction).
- Running Program: Gradual progression from light jogging to full-speed sprinting.
- Sport-Specific Drills: Drills without opposition or contact.
- Criteria for Progression: No pain or effusion, full ROM, satisfactory strength (e.g., >70-80% of contralateral limb on isokinetic testing), good dynamic stability.
Phase IV: Return to Sport (Months 6-12+)
- Goals: Safe return to competitive sport or high-demand activities.
-
Criteria for Return to Sport (RTS):
- Time: Minimum 9-12 months post-surgery for high-risk sports, due to biological graft maturation. Younger patients (e.g., <25) may benefit from longer protocols.
- Clinical: No pain, no effusion, full ROM.
- Strength: Quadriceps and hamstring strength >90% of contralateral limb on isokinetic testing.
- Functional Testing: Hop testing (single leg hop, triple hop, crossover hop, 6-meter timed hop) >90% limb symmetry index (LSI).
- Neuromuscular Control: Excellent balance, agility, and landing mechanics.
- Psychological Readiness: Assessment of fear of re-injury.
- Considerations: Brace use for RTS is controversial; some may recommend for high-risk sports, but evidence for prevention of re-injury is limited. Continued strength and conditioning program.
- Risk Factors for Re-injury: Young age, female gender, participation in pivoting/contact sports, residual quadriceps weakness, failure to meet objective RTS criteria, and return to sport before 9 months.
Summary of Key Literature / Guidelines
ACL reconstruction remains an active area of research, with continuous evolution in surgical techniques, graft choices, and rehabilitation protocols. Current evidence supports several key principles:
- Anatomical Reconstruction: Modern ACL reconstruction emphasizes anatomical tunnel placement to more closely replicate the native ligament's footprint and biomechanical function. While double-bundle reconstruction was once thought to provide superior rotational stability, systematic reviews and meta-analyses generally show no clear clinical superiority over well-performed anatomical single-bundle reconstruction, albeit with increased technical demands.
-
Graft Choice:
- Autografts (BPTB, Hamstring, Quadriceps tendon) remain the gold standard, offering superior re-rupture rates compared to allografts, especially in young, active patients.
- BPTB often provides reliable bone-to-bone healing and strong fixation, frequently favored for revision surgery, but has higher rates of anterior knee pain and kneeling pain.
- Hamstring autografts are associated with less anterior knee pain but slower soft tissue-to-bone healing and potential hamstring weakness.
- Quadriceps tendon autograft is gaining popularity due to its robust size, favorable biomechanical properties, and potentially lower donor site morbidity than BPTB.
- Allografts are generally reserved for older, less active patients, revision surgery, or multi-ligament reconstructions due to concerns about slower incorporation and higher failure rates in younger cohorts.
- Lateral Augmentation: The role of concurrent Anterolateral Ligament (ALL) reconstruction or Lateral Extra-Articular Tenodesis (LEAT) with intra-articular ACL reconstruction is a growing area of interest. Recent literature suggests that adding a lateral procedure may improve rotational stability and reduce re-rupture rates in high-risk patients (e.g., those with a high-grade pivot shift, hyperlaxity, or young athletes). Consensus statements (e.g., from ESSKA) are emerging to guide patient selection.
- Skeletally Immature Patients: Physeal-sparing techniques are recommended to minimize the risk of growth disturbance in pediatric and adolescent patients with open physes. The choice of technique depends on remaining growth potential and activity level, often involving all-epiphyseal, partial transphyseal, or over-the-top approaches.
- Rehabilitation: Evidence-based rehabilitation is crucial. Early weight-bearing and range of motion are safe and beneficial. The consensus emphasizes a progressive, criterion-based approach to return to sport, with objective testing (isokinetic strength, hop tests) and psychological readiness assessment. Current guidelines advocate for a minimum of 9-12 months post-surgery before full return to pivoting sports, particularly for young athletes, due to the protracted graft maturation process (ligamentization). Premature return to sport is a significant risk factor for re-injury.
- Prevention of Osteoarthritis: Despite successful anatomical and biomechanical restoration, ACL reconstruction does not eliminate the risk of post-traumatic osteoarthritis. Ongoing research focuses on understanding the long-term biological consequences of ACL injury and reconstruction, including the role of associated meniscal and chondral injuries, and exploring novel biological augmentation strategies to mitigate OA progression.
Organizations such as the American Academy of Orthopaedic Surgeons (AAOS), the European Society of Sports Traumatology, Knee Surgery & Arthroscopy (ESSKA), and the International Society of Arthroscopy, Knee Surgery and Orthopaedic Sports Medicine (ISAKOS) regularly publish guidelines and consensus statements that inform best practices in ACL reconstruction. The current landscape highlights a personalized approach, integrating patient factors, injury characteristics, surgical technique, and rigorous, prolonged rehabilitation to achieve the best possible outcomes.
You Might Also Like