Timing and Techniques in Peripheral Nerve Repair: A Comprehensive Surgical Guide
Key Takeaway
The timing of peripheral nerve repair significantly dictates functional recovery. Primary repair within 6 to 8 hours is indicated for clean, sharp transections, minimizing end-organ denervation and optimizing fascicular alignment. Conversely, high-energy trauma or war wounds necessitate delayed secondary repair. Successful neurorrhaphy demands meticulous preparation of nerve ends, preservation of the vasa nervorum, and a strictly tension-free coaptation, often requiring interfascicular grafting to bridge critical defects and prevent intraneural fibrosis.
INTRODUCTION TO PERIPHERAL NERVE REPAIR
The restoration of function following peripheral nerve transection remains one of the most formidable challenges in operative orthopaedics and reconstructive microsurgery. The decision-making process surrounding the timing of surgery, the management of concomitant skeletal trauma, and the meticulous preparation of the nerve ends dictates the ultimate success or failure of axonal regeneration. It has been a time-honored policy to advise primary suture whenever physiologically and anatomically possible. This recommendation is deeply rooted in the biological reality of what occurs to the distal nerve stump, motor endplates, sensory receptors, and the surrounding musculoskeletal envelope during prolonged periods of denervation.
However, the controversy concerning whether primary or secondary nerve repair yields superior outcomes requires a nuanced understanding of the mechanism of injury, the zone of trauma, and the physiological state of the patient. This comprehensive guide delineates the evidence-based protocols for the timing of peripheral nerve surgery, the biomechanics of neurorrhaphy, and the advanced microsurgical techniques required to optimize fascicular alignment and functional recovery.
TIMING OF SURGERY: STRATEGIC CLASSIFICATIONS
The timing of peripheral nerve repair is broadly categorized into primary, delayed primary, and secondary repair. The choice of timing is never arbitrary; it is dictated by the mechanism of injury, the degree of wound contamination, and the availability of appropriate microsurgical expertise and equipment.
Primary Repair (0 to 8 Hours)
Primary repair, performed within the first 6 to 8 hours following injury, is the gold standard for sharp, clean transections (e.g., glass lacerations, scalpel injuries) where there are no major complicating systemic injuries.
- Biological Advantages: Primary repair dramatically shortens the time of denervation of the end organs. It capitalizes on the lack of distal stump shrinkage and the absence of significant proximal neuroma formation.
- Anatomical Advantages: Fascicular alignment is vastly improved because minimal excision of the nerve ends is required. The topographical anatomy of the nerve is preserved, allowing for highly accurate epineurial or group fascicular coaptation.
- Prerequisites: Such repairs must be performed by an experienced microsurgeon in an institution equipped with high-resolution operating microscopes, specialized microsurgical instrumentation, and appropriate microsutures (e.g., 8-0 to 10-0 nylon).
Surgical Pearl: When performing a primary repair, utilize the superficial epineurial vascular network (vasa nervorum) as a roadmap. Aligning the longitudinal epineurial vessels is one of the most reliable methods to ensure correct rotational alignment of the fascicles.
Delayed Primary Repair (7 to 18 Days)
Delayed primary repair is indicated when the initial wound is contaminated, when the patient has life-threatening polytrauma requiring damage control orthopaedics, or when the surgical expertise/equipment is not immediately available.
During this 7 to 18-day window, the initial inflammatory phase has subsided, and the extent of nerve contusion or crush begins to demarcate. The Schwann cells in the distal stump have begun to proliferate, creating an optimal biological environment for receiving regenerating axons.
Secondary Repair (3 Weeks to 3 Months)
Secondary repair is mandatory for high-energy injuries, crush injuries, avulsions, and war wounds (e.g., high-velocity gunshot wounds or blast injuries).
- The War Wound Paradigm: In high-energy ballistic or blast trauma, primary sutures have historically compared unfavorably with early secondary suture. The zone of injury in a high-velocity wound extends far beyond the macroscopic transection. Primary repair in this setting inevitably leads to coaptation of necrotic or severely contused nerve tissue, resulting in dense intraneural fibrosis and failure of regeneration.
- Demarcation: Delaying the repair allows the longitudinal extent of the intraneural scarring to fully demarcate. The surgeon can then accurately resect the proximal neuroma and distal glioma back to healthy, pouting fascicles before performing an interfascicular graft.
Surgical Warning: Do not delay repair in anticipation of spontaneous regeneration if a definitive diagnosis of complete nerve division (neurotmesis) has been made. Only if the patient’s life or limb is seriously endangered should the operation be postponed beyond the optimal secondary window.
INFLUENCE OF CONCOMITANT INJURIES AND FRACTURES
A concomitant fracture is not a contraindication for early nerve exploration and repair; rather, it often dictates a combined, synergistic surgical approach. The management of the skeletal injury profoundly impacts the success of the neurorrhaphy.
Bone Shortening and Gap Management
Operating before the fracture becomes fully united offers distinct biomechanical advantages. If a significant nerve gap exists, resection of an ununited or partially united fracture to achieve bone shortening is a much less formidable procedure than performing an osteotomy on a fully united bone.
- Upper Extremity: In the humerus or forearm, acute bone shortening of up to 2 to 3 centimeters can often be tolerated to facilitate a primary, tension-free end-to-end nerve repair, thereby avoiding the need for nerve grafting.
- Lower Extremity: Bone shortening in the femur or tibia is less tolerated due to limb-length discrepancy issues, making nerve grafting the preferred reconstructive ladder in the lower limb.
Joint Mobilization and Stiffness
Restriction of joint motion is minimal if the nerve is repaired soon after the injury. If the repair is delayed, periarticular fibrosis and joint contractures develop. Later, motion may be so severely limited that the surgeon is prevented from flexing the joint enough to overcome a gap between the nerve ends, forcing the use of long, less favorable nerve grafts.
PATHOPHYSIOLOGY OF PROXIMAL VS. DISTAL INJURIES
The level of the nerve injury along the axis of the limb plays a critical role in the prognosis. Proximal injuries (e.g., brachial plexus, proximal sciatic nerve) carry a significantly worse prognosis for distal motor and sensory recovery compared to distal injuries (e.g., wrist-level median nerve). This is due to three primary physiological factors:
- Retrograde Neuronal Changes: Neurons that innervate the distal portions of the limb are more severely affected by retrograde changes (chromatolysis) and cell death after a proximal injury. The metabolic burden on the cell body to regenerate an axon over a massive distance is immense.
- Cross-Sectional Topography: In proximal nerve trunks, a greater proportion of the cross-sectional area is occupied by fibers destined for proximal muscles. The sheer volume of axons that must navigate the coaptation site increases the statistical likelihood of misdirection.
- Axonal Disorientation: The potential for disorientation of regrowing axons and for axonal loss during regeneration is exponentially greater for distal muscles than for proximally situated muscles following a high injury. Intraneural plexus formation causes fascicles to branch and merge repeatedly down the limb, making accurate topographical matching at a proximal level nearly impossible.
Despite these challenges, useful function can at times return regardless of the level of injury—except for certain severe preganglionic brachial plexus avulsions—provided the critical limit of delay has not passed and the end organs remain viable.
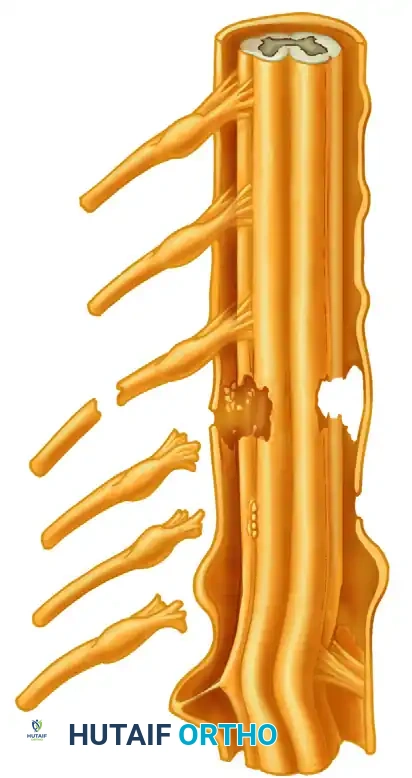
CONDITION AND PREPARATION OF NERVE ENDS
The condition of the nerve ends at the time of neurorrhaphy is the most critical local factor determining the success of axonal crossing. A neurorrhaphy with a pristine external epineurial appearance is absolutely no guarantee of optimal internal fascicular alignment.
Morphological Changes in the Distal Stump
Following transection, the distal stump undergoes Wallerian degeneration. Concurrently, the endoneurial tubes shrink, and the surrounding connective tissue contracts.
* Distal Shrinkage: Distal stump shrinkage has been found to be maximal at about 4 months post-injury.
* Size Mismatch: This shrinkage leaves the distal fascicular cross-sectional area diminished to 30% to 40% of its normal size. This creates a significant caliber mismatch between the swollen proximal neuroma and the atrophic distal stump, making accurate fascicular alignment and appropriate axonal regeneration exceedingly difficult.
Surgical Preparation: The "Bread-Loafing" Technique
It is universally agreed that the nerve ends must be prepared in such a way that a satisfactory, healthy fascicular pattern is apparent in both the proximal and distal stumps.
- Resection: Using a diamond knife or a fresh scalpel blade on a sterile wooden tongue depressor, the surgeon must sequentially slice the nerve ends (bread-loafing) until healthy tissue is reached.
- Visual Cues: Healthy proximal fascicles will "pout" or "mushroom" out of the epineurium, indicating normal axoplasmic flow. The tissue should be soft, and there should be punctate bleeding from the vasa nervorum.
- Eradication of Scar: Absolutely no scar, foreign material, or necrotic tissue should be allowed to remain around the ends. Fibrous tissue acts as an impenetrable physical barrier to the delicate regenerating growth cones.
Pitfall: Inadequate resection of the proximal neuroma is the most common cause of failure in secondary nerve repair. Surgeons are often hesitant to resect too much nerve for fear of creating an unbridgeable gap. However, coapting scarred nerve guarantees failure. It is always better to resect back to healthy tissue and bridge the resulting gap with a graft.
BIOMECHANICS OF NEURORRHAPHY: THE DETRIMENT OF TENSION
Meticulous handling of the nerve ends, strict asepsis, careful nerve mobilization, and preservation of the neural blood supply are foundational principles. However, the avoidance of tension is arguably the most critical biomechanical mandate in peripheral nerve surgery.
The Ischemic Threshold
Clinical and experimental evidence unequivocally indicates that excessive tension on the neurorrhaphy causes catastrophic intraneural ischemia and subsequent fibrosis.
* Tension exceeding 8% of the nerve's resting length significantly diminishes intraneural blood flow.
* Tension exceeding 15% completely occludes the vasa nervorum, leading to necrosis of the coaptation site and dense scar formation.
The Fallacy of Extreme Joint Flexion
Historically, surgeons would acutely flex adjacent joints (e.g., hyperflexing the wrist and elbow for a median nerve repair) to overcome large nerve gaps and achieve an end-to-end repair. While this allows for coaptation on the operating table, it is biologically disastrous. When the acutely flexed limb is mobilized weeks later, the regenerating nerve is subjected to massive traction forces, causing micro-tears, chronic ischemia, and excessive intraneural fibrosis.
Interfascicular Nerve Grafting
Sometimes, resection of the nerve ends to expose satisfactory fasciculi leaves a gap that simply cannot be closed by end-to-end repair without violating the principles of tension. In these scenarios, the interfascicular nerve grafting technique, pioneered and advocated by Millesi, Meissl, and Berger, is the absolute standard of care.
- The Millesi Principle: It is vastly preferable to bridge a gap with autologous nerve grafts under zero tension than to perform a primary repair under even mild tension.
- Graft Selection: The sural nerve is the workhorse donor for cable grafting, providing up to 30-40 cm of graft material with minimal donor site morbidity (lateral foot numbness). Other options include the medial antebrachial cutaneous nerve or the terminal branch of the posterior interosseous nerve.
- Technique: The epineurium of the proximal and distal stumps is resected. The fascicles are grouped according to their topographical alignment. Multiple strands (cables) of the donor nerve are then interposed between the matching fascicular groups and secured with 9-0 or 10-0 microsutures and/or fibrin glue.
STEP-BY-STEP SURGICAL PROTOCOL FOR NERVE REPAIR
To achieve the textbook standard of care, the following operative sequence should be strictly adhered to:
1. Preoperative Setup and Exposure
- Tourniquet: Utilize a pneumatic tourniquet to ensure a bloodless field during the initial exposure, but deflate it prior to final coaptation to assess the vasa nervorum and ensure hemostasis.
- Incision: Design extensile incisions. Never dissect directly through the zone of injury initially. Identify the normal nerve proximally and distally in virgin tissue, then trace it toward the scarred epicenter.
2. Mobilization and Bed Preparation
- Mobilize the nerve gently using vessel loops. Avoid stripping the mesoneurium over long distances, as this devascularizes the nerve trunk.
- Prepare a healthy, well-vascularized soft-tissue bed for the repaired nerve. Avoid placing the coaptation site directly over bare bone, metallic fracture implants, or beneath tight fascial bands.
3. Fascicular Alignment and Coaptation
- Utilize an operating microscope (magnification 10x to 40x).
- Align the superficial epineurial vessels.
- Match the fascicular topography based on size, shape, and grouping. In mixed nerves (e.g., median or ulnar), intraoperative nerve stimulation (in awake or lightly anesthetized patients) or histochemical staining (e.g., Karnovsky-Roots stain for motor vs. sensory fascicles) can aid in differentiation, though these are highly specialized techniques.
- Place epineurial or group-fascicular sutures using 8-0 to 10-0 non-absorbable monofilament. Use the minimum number of sutures required to coapt the ends without gaping; excessive suture material provokes a foreign body inflammatory response.
4. Postoperative Immobilization and Rehabilitation
- Immobilization: The limb is immobilized in a bulky, non-compressive splint in a neutral, tension-free position for 3 to 4 weeks. This allows the initial fibrin clot at the coaptation site to organize into mature connective tissue.
- Mobilization: After 3 to 4 weeks, a highly supervised, graduated active and passive range-of-motion protocol is initiated to prevent joint contractures and promote nerve gliding.
- Sensory Re-education: As axonal regeneration progresses (at a rate of approximately 1 mm per day), formal sensory re-education and motor biofeedback programs are instituted by specialized hand or neuro-physiotherapists to maximize cortical neuroplasticity and functional integration.
CONCLUSION
The operative management of peripheral nerve injuries is a delicate synthesis of timing, biology, and microsurgical execution. While primary repair remains the ideal for sharp, clean lacerations, the surgeon must possess the clinical acumen to recognize when delayed or secondary repair is mandated by the zone of injury. Above all, the absolute eradication of tension, the meticulous preparation of nerve ends to healthy fascicles, and the judicious use of interfascicular nerve grafting are the cornerstones of successful peripheral nerve reconstruction. Mastery of these principles ensures that the regenerating axons are provided the optimal biological conduit to reach their distal targets, thereby restoring form and function to the denervated extremity.
You Might Also Like