Peripheral Nerve Repair and Reconstruction: A Masterclass in Microsurgical Techniques
Key Takeaway
Peripheral nerve injuries in the upper extremity demand meticulous microsurgical techniques to restore motor and sensory function. This comprehensive guide details evidence-based protocols for primary neurorrhaphy, autologous nerve grafting, and the use of synthetic conduits. By mastering tension-free epineurial repair, fascicular alignment, and advanced neuroma management, orthopedic surgeons can optimize axonal regeneration and significantly improve patient outcomes following complex peripheral nerve trauma.
Introduction to Peripheral Nerve Reconstruction
The management of peripheral nerve injuries remains one of the most technically demanding and biologically complex challenges in orthopedic and hand surgery. The ultimate goal of peripheral nerve repair is to guide regenerating axons across a zone of injury to their appropriate distal end-organs, thereby restoring motor function, protective sensation, and proprioception.
Historically, the evolution of nerve repair has been driven by a deeper understanding of intraneural microcirculation, axonal regeneration, and the biomechanical consequences of tension at the coaptation site. Drawing upon the foundational work of pioneers such as Seddon, Sunderland, Millesi, and Lundborg, modern peripheral nerve surgery relies on exquisite microsurgical technique, precise fascicular alignment, and strict adherence to tension-free repair principles.
This comprehensive guide delineates the evidence-based principles of peripheral nerve repair, grafting, sensory restoration, and neuroma management, tailored for the practicing orthopedic consultant and advanced microsurgery fellow.
Applied Anatomy and Intraneural Microcirculation
A profound understanding of peripheral nerve architecture is non-negotiable for the operating surgeon. A peripheral nerve is not merely a conduit for electrical impulses; it is a highly vascularized, dynamic organ.
Structural Hierarchy
- Axon and Endoneurium: The fundamental functional unit. The endoneurium provides the immediate microenvironment for the axon and Schwann cells.
- Perineurium: A robust, metabolically active layer enclosing a group of axons to form a fascicle. It acts as the primary blood-nerve barrier and provides the nerve with its tensile strength.
- Epineurium: Divided into the internal epineurium (cushioning the fascicles) and the external epineurium (the outer sheath).
Intraneural Microcirculation
Lundborg and Rydevik’s seminal basic research demonstrated that the peripheral nerve possesses a dual vascular system: an extrinsic segmental system and an intrinsic longitudinal system.
Clinical Pearl: The intrinsic microcirculation is highly sensitive to mechanical compression and tension. Elongation of a nerve by merely 8% impairs venular flow, and 15% elongation causes complete ischemia. This physiologic absolute dictates the universal mandate for tension-free nerve repair.
Diagnostic Modalities and Preoperative Planning
Accurate preoperative assessment dictates the surgical timing and approach. The clinical examination remains the cornerstone of diagnosis, supplemented by judicious use of electrodiagnostic studies.
Clinical Evaluation
- Sensory Testing: As championed by Dellon, the use of vibratory stimuli (tuning forks at 30 Hz and 256 Hz) is highly sensitive for evaluating early peripheral nerve compression and regeneration. Two-point discrimination (static and moving) remains the gold standard for assessing functional tactile gnosis, though its reliability depends heavily on standardized testing protocols.
- Motor Testing: Precise isolation of individual muscle bellies is required to map the exact level of the lesion.
Electrodiagnostic Studies (EMG/NCS)
Electromyography (EMG) and Nerve Conduction Studies (NCS) are invaluable but must be timed correctly.
* Timing: Wallerian degeneration takes 10 to 21 days to manifest as fibrillation potentials and positive sharp waves on EMG. Ordering an EMG on day 2 post-injury is a common, yet futile, clinical error.
* Utility: EMG is most useful at 3 to 4 weeks to confirm the baseline extent of denervation, and at 3 to 6 months to detect subclinical nascent motor unit action potentials (MUAPs) indicating early reinnervation.
Surgical Warning: Do not rely solely on EMG for surgical decision-making in acute penetrating trauma. A clinically transected nerve requires exploration regardless of early electrodiagnostic ambiguity.
Principles of Primary Neurorrhaphy
Primary repair is indicated for sharp, clean transections where the nerve ends can be approximated without tension. The debate between epineurial and fascicular (perineurial) repair has been extensively studied by Sunderland, Tupper, and others.
Epineurial vs. Fascicular Repair
- Epineurial Repair: Involves placing sutures through the external epineurium. It is technically less demanding, minimizes intraneural scarring, and is the standard of care for oligofascicular nerves or mixed nerves where fascicular topography is not clearly segregated.
- Fascicular (Perineurial) Repair: Involves matching and suturing individual fascicles or groups of fascicles. While theoretically superior for aligning specific motor and sensory pathways, excessive intraneural dissection can devascularize the nerve and provoke robust scar formation.
Current consensus favors a group fascicular repair (or epineurial repair with fascicular alignment) for major mixed nerves (e.g., median nerve at the wrist), utilizing the surface epineurial vessels as landmarks for rotational alignment.
Step-by-Step Surgical Technique for Primary Repair
- Preparation and Exposure: Operate under tourniquet control and loupe magnification (or an operating microscope). Extend the incision proximally and distally to find normal nerve anatomy before tracing it into the zone of injury.
- Preparation of Nerve Ends: Resect the traumatized nerve ends back to healthy, pouting fascicles. The "mushrooming" of endoneurial tissue confirms viable nerve architecture.
- Alignment: Identify corresponding fascicular patterns and longitudinal epineurial blood vessels to ensure correct rotational alignment.
- Coaptation: Use 8-0 or 9-0 nylon for major nerves, and 10-0 nylon for digital nerves. Place the first two sutures 180 degrees apart to act as stay sutures.
- Suture Placement: Place just enough sutures to coapt the ends and prevent fascicular escape. Over-suturing causes ischemia and foreign-body reactions.
- Augmentation (Optional): Fibrin glue may be applied to reinforce the repair, potentially reducing the number of required sutures and minimizing trauma.
Pitfall: "Bridging the gap" by acutely flexing a joint (e.g., the wrist) to achieve primary repair is a historical error. Millesi demonstrated that the subsequent postoperative stretching of the nerve leads to profound intraneural fibrosis. If a tension-free repair cannot be achieved in a neutral joint position, a nerve graft or conduit is mandatory.
Management of Nerve Gaps: Grafts and Conduits
When primary repair is impossible due to segmental loss or retraction, bridging the gap is required. The choice of material depends on the gap length, nerve diameter, and the quality of the soft tissue bed.
Autologous Nerve Grafting
Autografting remains the gold standard for gaps exceeding 3 cm. The graft acts as a biological scaffold, providing viable Schwann cells and basal lamina tubes to guide regenerating axons.
- Sural Nerve: The workhorse donor nerve. It provides up to 30-40 cm of graft material. Anatomy dictates harvesting it posterior to the lateral malleolus, ascending alongside the lesser saphenous vein.
- Medial Antebrachial Cutaneous (MABC) Nerve: Excellent for digital nerve defects due to its size match and proximity to the primary surgical field in upper extremity trauma.
- Technique (Interfascicular Grafting): The graft must be reversed to prevent regenerating axons from escaping down branching points. The graft should be 10-15% longer than the defect to account for shrinkage and allow for a tension-free, undulating course.
Vascularized Nerve Grafts
Pioneered by Doi and Breidenbach, vascularized nerve grafts (e.g., vascularized ulnar nerve or sural nerve) are indicated for massive defects (>8-10 cm) or when the recipient bed is heavily scarred and avascular. By preserving the intrinsic blood supply, these grafts bypass the initial phase of ischemic necrosis seen in conventional grafts, theoretically accelerating regeneration.
Biological and Synthetic Conduits
For non-critical sensory nerves (e.g., digital nerves) with gaps less than 3 cm, conduits offer an alternative to autografting, sparing donor site morbidity.
* Vein Grafts: Autogenous vein grafts (often reversed to prevent valve obstruction) can be used for short gaps (<1 cm).
* Synthetic Conduits: Polyglycolic acid (PGA) tubes and biodegradable materials (e.g., Neurolac) have shown comparable results to autografts for digital nerve gaps up to 2.5 cm. The conduit allows for the accumulation of neurotrophic factors while preventing fibrous tissue ingrowth.
Regional Flaps for Sensory Restoration
In cases of irreparable nerve damage or severe soft tissue loss, restoring protective sensation to critical areas (e.g., the thumb pulp or the radial border of the index finger) is paramount to prevent pressure necrosis and restore hand function.
Neurovascular Island Pedicle Flaps
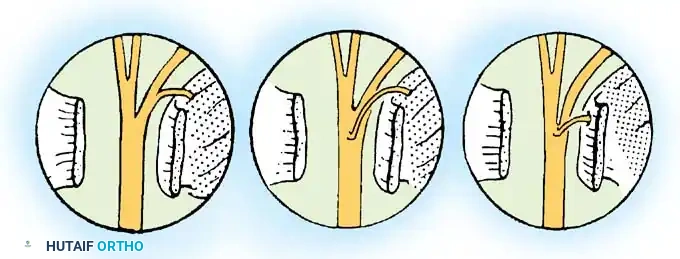
Described extensively by Omer, the neurovascular island flap transfers sensate skin from a less critical area (e.g., the ulnar border of the ring finger) to a critical area (e.g., the thumb pulp).
* Biomechanics: The flap is harvested on its neurovascular bundle (digital artery and nerve) and tunneled to the recipient site.
* Cortical Reintegration: A major challenge is cortical remapping; the patient initially perceives touch on the thumb as touch on the ring finger. Intensive sensory re-education is required.
Neurocutaneous Flaps
Advancements in flap surgery, such as those described by Bertelli, utilize neurocutaneous flaps based on the dorsal branches of the ulnar artery and nerve. These provide robust, sensate coverage for extensive hand reconstruction without sacrificing major digital vessels.
Management of Neuromas and Intractable Pain
A neuroma is a disorganized mass of regenerating axons and fibrous tissue that forms when a nerve fails to reach its distal target. While all transected nerves form a neuroma, only a subset becomes symptomatic (painful neuromas).
Surgical Interventions for Painful Neuromas
The management of painful neuromas, as outlined by Whipple and Wilson, is notoriously difficult. Simple excision frequently leads to recurrence.
1. Neurolysis: Indicated when the nerve is intact but compressed by surrounding scar tissue. External neurolysis frees the nerve from the scar bed, while internal neurolysis (separating fascicles) is rarely indicated due to the risk of devascularization.
2. Excision and Transposition: The most reliable treatment for a terminal neuroma is wide excision of the neuroma followed by transposition of the proximal nerve stump into a healthy, well-vascularized, and mechanically protected environment.
* Muscle Implantation: Burying the nerve stump deep into a muscle belly (e.g., pronator quadratus or brachioradialis) prevents mechanical irritation.
* Bone Implantation: Drilling a hole into the medullary canal of a bone and inserting the nerve stump.
3. Centrocentral Anastomosis: Coapting two adjacent neuroma stumps together to create a closed loop, theoretically downregulating the regenerative drive.
Clinical Pearl: The key to successful neuroma management is altering the mechanical environment of the nerve ending. No technique is universally successful, and managing patient expectations preoperatively is as critical as the surgical execution.
Postoperative Rehabilitation Protocols
The success of a flawless microsurgical repair can be entirely undone by poor postoperative rehabilitation. Rehabilitation, as structured by Frykman and Waylett, is divided into three distinct phases.
Phase 1: Immobilization and Protection (Weeks 0-3)
- The extremity is splinted in a position that minimizes tension on the repair.
- Strict elevation and edema control are enforced.
- Recent studies suggest that limited, highly protected early motion may not be detrimental to digital nerve repairs, but conservative immobilization remains the standard for major mixed nerves.
Phase 2: Motor Re-education and Mobilization (Weeks 3-8)
- Gradual mobilization of the joints is initiated to prevent contractures.
- As motor function begins to return (often months later), biofeedback and targeted muscle strengthening are employed.
- Galvanic stimulation of denervated muscle remains controversial; while it may delay muscle atrophy, it does not alter the ultimate functional outcome of nerve regeneration.
Phase 3: Sensory Re-education and Desensitization (Months 3+)
- Desensitization: As the nerve regenerates, patients often experience hypersensitivity (dysesthesia). Desensitization techniques using varying textures (silk, cotton, Velcro) and immersion baths (rice, sand) help normalize sensory input.
- Sensory Re-education: Once moving and static touch return, the brain must be "retrained" to interpret the new, often distorted, neural signals. This involves visually guided tactile exercises to improve spatial recognition and two-point discrimination.
Conclusion
Peripheral nerve repair and reconstruction require a synthesis of profound anatomical knowledge, meticulous microsurgical skill, and immense patience. From the initial decision to perform a tension-free primary epineurial repair, to the complex utilization of vascularized nerve grafts and synthetic conduits, every surgical step must be optimized to support axonal regeneration. By adhering to these evidence-based principles and integrating rigorous postoperative rehabilitation, the orthopedic surgeon can maximize functional recovery and restore quality of life to patients suffering from devastating peripheral nerve injuries.
You Might Also Like