Peripheral Nerve Injury: Pathophysiology, Degeneration, and Microsurgical Repair
Key Takeaway
Peripheral nerve injuries demand a profound understanding of neuronal degeneration and regeneration to optimize surgical outcomes. Following injury, the distal axon undergoes Wallerian degeneration, while the proximal segment experiences retrograde changes. Recovery depends on the injury severity, classified by Seddon and Sunderland. This guide details the pathophysiology, clinical evaluation, and advanced microsurgical techniques required for primary repair and nerve grafting, ensuring optimal functional restoration.
INTRODUCTION TO PERIPHERAL NERVE PATHOPHYSIOLOGY
The management of peripheral nerve injuries remains one of the most technically demanding and biologically complex challenges in operative orthopaedics. To achieve optimal functional restoration, the reconstructive microsurgeon must possess an exhaustive understanding of the cellular events that dictate neuronal degeneration and regeneration.
Any part of a neuron that becomes detached from its parent nucleus inevitably degenerates and is subsequently cleared by phagocytosis. This fundamental biological principle dictates the clinical course of all peripheral nerve injuries. The regenerative potential of a severed or crushed nerve is not merely a function of surgical coaptation, but rather a highly orchestrated cascade of molecular and cellular events that must traverse the hostile environment of the zone of injury.
THE BIOLOGY OF NEURONAL DEGENERATION
When a peripheral nerve is injured, a dual process of degeneration occurs, affecting both the distal and proximal segments of the severed axon. The timeline and severity of these degenerative processes vary significantly between sensory and motor segments, heavily influenced by the cross-sectional size and the degree of myelinization of the involved fibers.
Wallerian (Secondary) Degeneration
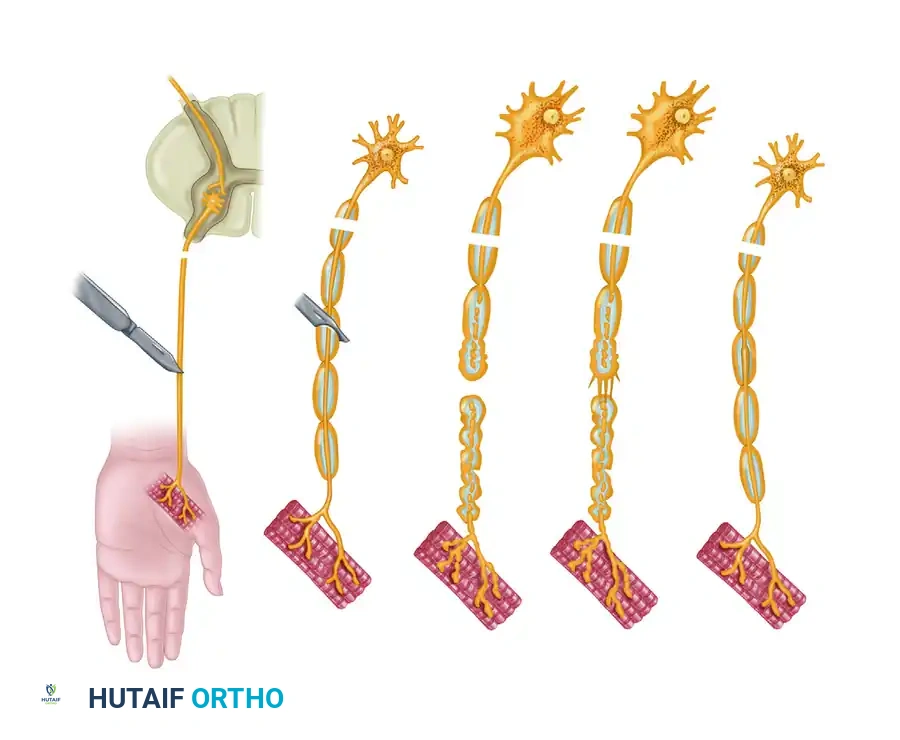
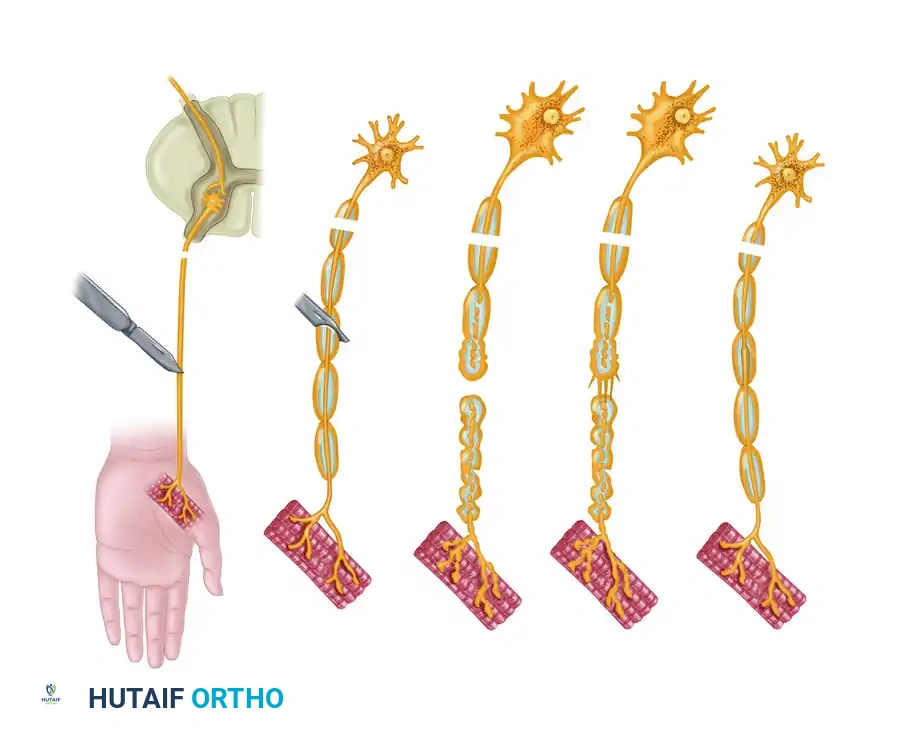
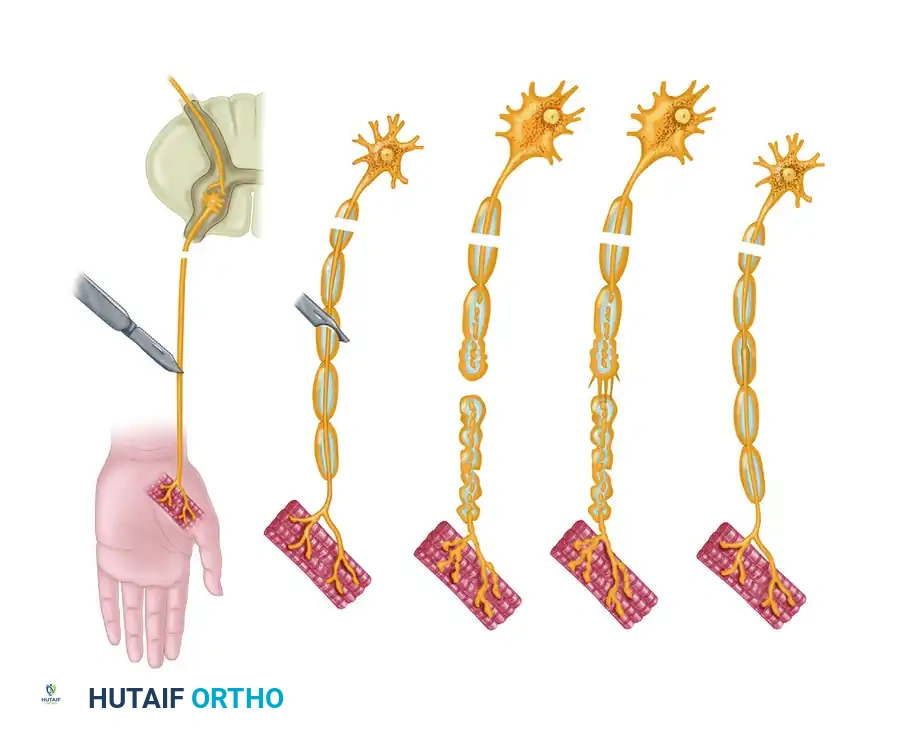
The process of degeneration distal to the point of injury is termed secondary, or Wallerian degeneration. This is an obligate process following axonal transection, preparing the distal endoneurial tubes for eventual regenerating axonal sprouts.

- Days 1 to 3: During the first 72 hours following injury, definite morphologic changes become apparent within the axoplasm. Crucially, response to faradic stimulation can still be obtained for periods ranging from 18 to 72 hours post-injury. This physiological window is critical when utilizing intraoperative nerve stimulation during early explorations.
- Days 2 to 3: The distal axonal segment becomes fragmented. Concomitant with subsequent fluid loss, these fragments begin to shrink, assuming a more oval or globular appearance. A parallel fragmentation and shrinkage of the myelin sheath mirrors the axonal degenerative changes.
- Day 7: Macrophages infiltrate the area in significantly greater numbers, initiating the phagocytosis of myelin and axonal debris. Simultaneously, Schwann cell division by mitosis becomes evident. These proliferating Schwann cells increase in number to fill the spatial void previously occupied by the axon and its myelin sheath, forming the critical "Bands of Büngner."
- Days 15 to 30: The clearing of axonal debris is virtually complete, leaving empty endoneurial tubes lined by hyperplastic Schwann cells, primed to receive regenerating axonal sprouts.
Clinical Pearl: The persistence of faradic excitability in the distal stump for up to 72 hours can be a diagnostic trap. Do not mistake this residual excitability for an intact nerve during early surgical exploration of acute trauma.
Primary (Retrograde) Degeneration
The reaction proximal to the point of detachment is termed primary, traumatic, or retrograde degeneration.
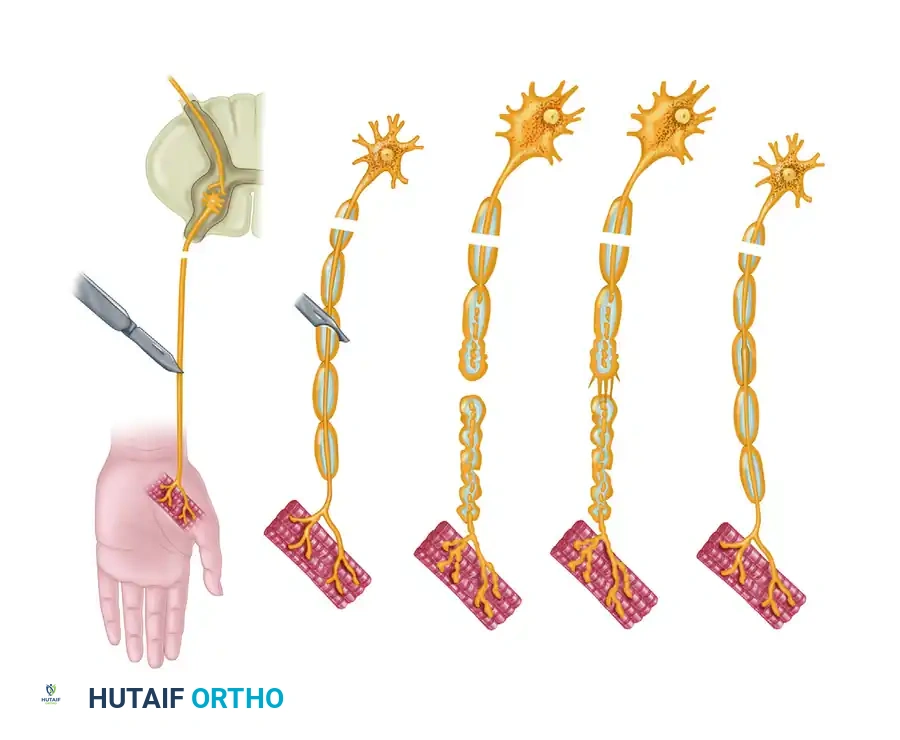
This retrograde process proceeds proximally for at least one internode or more, heavily dependent on the kinetic energy and degree of the proximal insult. Histologically, the axonal changes are identical to Wallerian degeneration. However, the parent cell body (soma) also undergoes profound metabolic shifts, a process known as chromatolysis.
The severity of these somatic changes varies with the type of cell and the proximity of the injury to the cell body. The more proximal the site of injury, the more severe the retrograde degeneration, increasing the risk of neuronal apoptosis. If the cell body survives, it undergoes hypertrophy, shifting its metabolic machinery from neurotransmitter production to the synthesis of structural proteins required for axonal regeneration.
CLASSIFICATION OF PERIPHERAL NERVE INJURIES
A rigorous understanding of nerve injury classification is paramount for determining prognosis and surgical indications. The classifications proposed by Seddon and expanded by Sunderland remain the gold standard in orthopaedic neurology.
First-Degree Injury (Neurapraxia)
In a first-degree injury, conduction along the axon is physiologically interrupted at the site of injury, but the axon itself is not structurally disrupted.
- Pathophysiology: No Wallerian degeneration occurs. The structural integrity of the axon, endoneurium, perineurium, and epineurium is preserved.
- Clinical Presentation: The loss of function varies, but motor function is typically more profoundly affected than sensory function. Sensory modalities are lost in a specific, predictable order of decreasing frequency: proprioception, touch, temperature, and pain. Sympathetic fibers are highly resistant to this type of injury.
- Recovery: Recovery is spontaneous and usually complete within a few days to weeks. A pathognomonic characteristic of neurapraxia is the simultaneous return of motor function in both proximal and distal musculature. There is no "motor march" and no advancing Tinel sign, as there is no regenerating axon tip to elicit it.
Second-Degree Injury (Axonotmesis)
In a second-degree injury, the axon is physically disrupted, leading to obligate Wallerian degeneration distal to the point of injury and retrograde degeneration proximally for one or more nodal segments.
- Pathophysiology: The critical distinguishing factor is that the integrity of the endoneurial tube (Schwann cell basal lamina) is maintained. This provides a perfect, uninterrupted anatomical conduit for regeneration.
- Clinical Presentation: Clinically, the neurological deficit is complete, presenting with total loss of motor, sensory, and sympathetic function in the distribution of the nerve.
- Recovery: Motor reinnervation occurs progressively from proximal to distal, strictly in the order in which nerve branches leave the parent trunk (the "motor march"). An advancing Tinel sign can be followed along the course of the nerve, typically advancing at a rate of 1 inch (2.5 cm) per month. Because the endoneurial tubes are intact, excellent functional return is usually achieved, provided the target end-organs have not undergone irreversible atrophy.
Third-Degree Injury
Third-degree injuries represent a significant escalation in severity, where both the axons and the endoneurial tubes are disrupted, but the perineurium remains preserved.
- Pathophysiology: The disruption of the endoneurial tubes leads to internal disorganization. As axons attempt to regenerate, scar tissue within the endoneurium can obstruct certain tubes and divert axonal sprouts into incorrect pathways (aberrant regeneration).
- Clinical Presentation: Neurological loss is complete.
- Recovery: Recovery is delayed and invariably incomplete. The regenerating axon tips require additional time to penetrate the intrafascicular fibrous barrier. The degree of functional return is highly variable and depends on the extent of intrafascicular scarring and axonal misdirection.
Fourth and Fifth-Degree Injuries (Neurotmesis)
- Fourth-Degree: The axon, endoneurium, and perineurium are disrupted, leaving only the epineurium intact. This results in a neuroma-in-continuity. Spontaneous functional recovery is impossible due to dense scar tissue blocking axonal progression.
- Fifth-Degree: Complete transection of the entire nerve trunk. Wide separation of the nerve ends occurs due to the inherent elasticity of the nerve.
Surgical Warning: Fourth and fifth-degree injuries will never recover spontaneously. They mandate meticulous microsurgical exploration, resection of the neuroma/scar, and tension-free coaptation or nerve grafting.
MECHANISMS OF NEURONAL REGENERATION
Following the initial phase of neuronal survival (1 to 2 weeks), the phase of neuronal regeneration commences (1 week to 3 months).
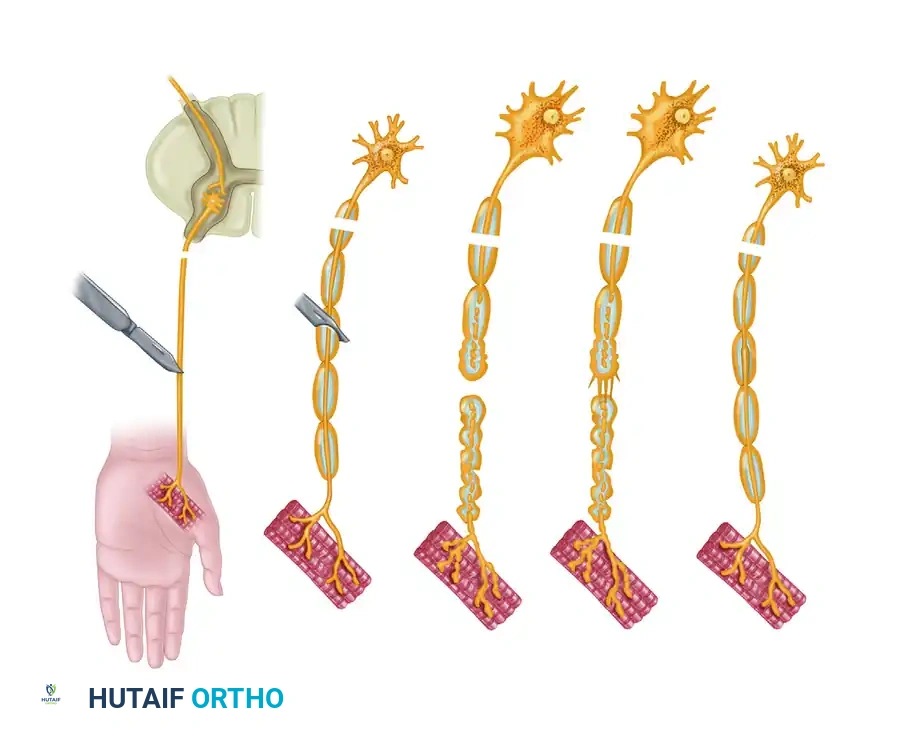
Axonal budding occurs at the proximal stump. A single proximal axon can produce up to 50 collateral sprouts. These sprouts navigate the injury site, guided by neurotrophic factors secreted by the distal Schwann cells. Experimental models utilizing silicone Y-chambers have demonstrated that regenerating axons exhibit a strong neurotrophic preference, growing toward nerve tissue rather than muscle or tendon.
However, this neurotrophic effect is distance-dependent; there is a critical gap (approximately 2 mm) beyond which this chemotactic guidance fails. If the regenerating fibers are obstructed by extensive scar tissue or wide separation, they form a disorganized, painful terminal neuroma, resulting in zero functional return.
SURGICAL MANAGEMENT AND OPERATIVE TECHNIQUES
The primary goal of peripheral nerve surgery is to provide an optimal anatomical and biological environment for regenerating axons to reach their target end-organs before irreversible motor end-plate degradation occurs (typically 12 to 18 months post-injury).
Indications and Timing
- Clean, Sharp Transections: (e.g., glass lacerations, knife wounds). Indicated for primary repair within 72 hours. Early repair prevents retraction of the nerve ends and allows for precise fascicular alignment before scar tissue distorts the anatomy.
- Blunt, Crush, or Avulsion Injuries: (e.g., high-velocity gunshot wounds, traction injuries). Primary repair is contraindicated due to the unknown extent of the zone of injury. These require delayed exploration (typically at 3 weeks) once the longitudinal extent of intraneural scarring has demarcated, allowing for accurate resection back to healthy fascicles.
Patient Positioning and Preparation
- Positioning: Position the patient to allow unhindered access to the entire course of the injured nerve. Ensure that adjacent joints can be manipulated intraoperatively to assess tension on the repair.
- Tourniquet: A pneumatic tourniquet is essential for a bloodless field. Exsanguinate the limb and inflate the tourniquet. Note: The tourniquet must be deflated prior to final epineurial closure to ensure absolute hemostasis, as a postoperative hematoma will cause devastating fibroplasia.
- Magnification: Operating loupes (minimum 3.5x to 4.5x) are required for the approach, transitioning to the operating microscope for the actual nerve preparation and coaptation.
Surgical Approach and Exposure
- Incision: Utilize extensile incisions. Never incise directly over the suspected neuroma or laceration.
- Proximal and Distal Control: Identify the nerve in virgin, uninjured tissue both proximally and distally to the zone of injury.
- Mobilization: Carefully mobilize the nerve toward the injury site. Preserve the segmental mesoneurial blood supply; excessive stripping will render the nerve ischemic and doom the repair.
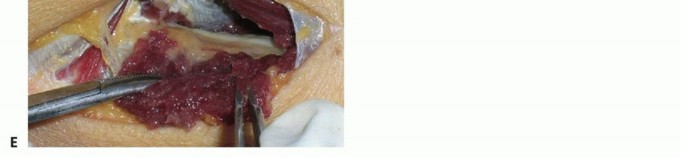
- Neuroma Resection: In delayed cases, a neuroma at the proximal stump and a glioma at the distal stump will be present. Using a fresh diamond knife or razor blade, serially section the nerve ends ("bread-loafing") until healthy, pouting fascicles are visualized. Healthy fascicles will exhibit a distinct "mushrooming" effect from the cut epineurial surface.
Microsurgical Repair Techniques
The choice between epineurial and grouped fascicular repair depends on the internal topography of the nerve at the level of injury.
- Epineurial Repair: Indicated for monofascicular or oligofascicular nerves, or when the internal topography is highly mixed.
- Place 8-0 or 9-0 nylon sutures in the epineurium at 180-degree intervals to align the nerve.
- Use surface landmarks (longitudinal epineurial vessels) to prevent rotational malalignment.
- Complete the repair with interrupted sutures, ensuring no fascicles are trapped within the suture line.
- Grouped Fascicular Repair: Indicated for nerves with distinct motor and sensory fascicular groups (e.g., the ulnar nerve at the wrist).
- Incise the epineurium longitudinally and dissect the major fascicular groups.
- Coapt corresponding groups using 10-0 nylon sutures placed through the perineurium.
Pitfall: Tension is the absolute enemy of nerve regeneration. If an 8-0 nylon suture cannot hold the nerve ends together without tearing, the repair is under excessive tension. A nerve graft must be utilized.
Management of Nerve Gaps
When tension-free primary repair is impossible, bridging the gap is mandatory.
- Autologous Nerve Grafting: The gold standard. The sural nerve is the most common donor, providing up to 40 cm of graft material. The graft acts as a biological scaffold, providing viable Schwann cells and intact endoneurial tubes. Grafts should be reversed to prevent axonal sprouts from escaping through transected branches.
- Nerve Conduits: Indicated only for small, non-critical sensory nerve gaps (less than 3 cm). Synthetic conduits (e.g., polyglycolic acid) or biological conduits (vein grafts) can guide regeneration but lack the robust cellular support of an autograft.
POSTOPERATIVE PROTOCOL AND REHABILITATION
The success of a flawless microsurgical repair can be entirely undone by improper postoperative management.
- Immobilization: The limb must be immobilized in a well-padded splint for 3 weeks to protect the coaptation site from tension during the critical early phases of healing.
- Progressive Mobilization: At 3 weeks, the splint is removed, and a supervised program of progressive, active-assisted range of motion is initiated. Extension blocks may be used to gradually stretch the nerve over the subsequent 3 to 4 weeks.
- Monitoring Regeneration: Clinical follow-up involves serial examinations for an advancing Tinel sign and the sequential return of motor function. Electromyography (EMG) can detect nascent motor unit potentials weeks before clinical contraction is visible, providing early evidence of successful reinnervation.
- Sensory Re-education: Once protective sensation begins to return, formal sensory re-education protocols are vital to help the cerebral cortex interpret the altered afferent signals generated by the misdirected axonal sprouts.
In conclusion, the operative management of peripheral nerve injuries requires a synthesis of profound biological knowledge and exacting microsurgical skill. By respecting the delicate pathophysiology of neuronal degeneration and adhering to strict principles of tension-free, anatomically aligned repair, the orthopaedic surgeon can maximize the potential for functional neuronal regeneration.
You Might Also Like