General Considerations and Operative Management of Peripheral Nerve Injuries
Key Takeaway
The management of peripheral nerve injuries requires meticulous microsurgical technique and strict adherence to tension-free repair principles. Critical factors influencing outcomes include the mechanism of injury, the length of the nerve gap, and the timing of neurorrhaphy. While clean lacerations warrant immediate repair, high-energy or crush injuries benefit from a delayed approach at three to six weeks to allow for accurate delineation of the zone of injury and optimal metabolic conditions for axonal regeneration.
GENERAL CONSIDERATIONS IN THE TREATMENT OF NERVE INJURIES
The operative management of peripheral nerve injuries represents one of the most technically demanding and biologically complex challenges in orthopedic surgery. As in any major trauma, the initial management of a patient with peripheral nerve damage must be strictly governed by Advanced Trauma Life Support (ATLS) principles. Careful assessment of vital functions is paramount; appropriate actions to prevent cardiopulmonary failure, hemorrhagic shock, and life-threatening systemic complications must inevitably precede the definitive management of focal neurologic deficits. Once the patient is stabilized and limb viability is assured, the surgeon must systematically evaluate the peripheral nerve injury, taking into account the mechanism of trauma, the zone of injury, and the anticipated biological behavior of the regenerating axons.
The mechanism of injury dictates the macroscopic and microscopic pathology of the nerve. In sharp, clean lacerations (e.g., glass or scalpel wounds), the nerve ends typically retract, but the resulting gap can usually be overcome with minimal mobilization. Conversely, when a high-velocity missile or severe crush injury severs a nerve, the proximal and distal nerve damage is extensive due to cavitation, thermal necrosis, and longitudinal traction. In these high-energy scenarios, both the proximal and distal stumps must ultimately be widely resected to expose healthy, normal funiculi (fascicles), inevitably producing a substantial gap. This gap is further exacerbated if a segment of the nerve is physically carried away by the missile or shrapnel.
Surgical Warning: Failure to resect a nerve stump back to healthy, unscarred fascicles (characterized by a "mushrooming" of axoplasm from the cut end) is the most common cause of neurorrhaphy failure. Suturing fibrotic or contused nerve ends guarantees a neuroma-in-continuity and blocks axonal regeneration.
MANAGEMENT OF NERVE GAPS AND THE TENSION-FREE PRINCIPLE
The fundamental tenet of peripheral nerve surgery is that a neurorrhaphy must be entirely tension-free. Widespread consensus and extensive biomechanical studies confirm that excessive tension on a primary repair severely compromises intraneural microvascular perfusion, leading to ischemia, profound scarring, and ultimately, failure of nerve regeneration.
When faced with a nerve gap, the surgeon has a hierarchical armamentarium of techniques to achieve closure. These methods include:
1. Nerve Mobilization: Freeing the nerve proximally and distally.
2. Nerve Transposition: Rerouting the nerve to shorten its anatomical path (e.g., anterior transposition of the ulnar nerve).
3. Joint Flexion: Positioning the adjacent joint to relax the nerve bed.
4. Nerve Grafting: Bridging the defect with autologous nerve tissue.
5. Bone Shortening: Resecting bone to shorten the limb (typically reserved for major replantations).
The Challenge of Funicular Mismatch
The greater the nerve defect, the more dissimilar the funicular (fascicular) pattern between the proximal and distal ends. Peripheral nerves are not static cables; their internal topography consists of a constantly changing, complex plexus of fibers as the nerve progresses distally. This intraneural plexification is particularly pronounced in the more proximal portions of peripheral nerves. Therefore, forcing a primary repair over a large gap not only induces ischemic tension but also guarantees severe fascicular mismatch, directing regenerating motor axons into sensory endoneurial tubes and vice versa.
Limits of Primary Repair
The observed upper limit of a nerve gap beyond which primary repair results deteriorate precipitously is approximately 2.5 cm. The classic observations of Kirklin, Murphey, and Berkson in 1949—that functional recovery is significantly better when the gap is relatively small—remain entirely valid in contemporary microsurgery.
Clinical Pearl: Nerve grafting is strictly advised if, after adequate mobilization, the nerve gap cannot be closed by flexing the main joint of the limb 90 degrees. If a repair requires more than 90 degrees of flexion to coapt, the tension upon subsequent joint extension will inevitably rupture the repair or induce ischemic fibrosis.
TIMING OF SURGICAL INTERVENTION: DELAY BETWEEN INJURY AND REPAIR
The timing of neurorrhaphy is a critical determinant of functional outcome and must be tailored to the mechanism of injury, the condition of the soft tissue envelope, and the specific nerve involved.
Impact on Motor vs. Sensory Recovery
Delay in neurorrhaphy affects motor recovery far more profoundly than sensory recovery. This discrepancy is primarily dictated by the finite survival time of denervated striated muscle. Following denervation, motor endplates undergo progressive fibrosis and irreversible degradation.
For every delay of 6 days between injury and repair, there is a variable loss of potential motor recovery that averages approximately 1% of maximal performance. After 3 months of delay, this loss increases exponentially. Furthermore, the return of function in distal musculature (e.g., intrinsic muscles of the hand or foot) is notoriously poor when suturing is delayed, as the regenerating axons (growing at roughly 1 mm per day) cannot reach the target motor endplates before irreversible muscle atrophy occurs.
The influence of delay on sensory return is less rigid. Historical data, including landmark Veterans Administration studies, demonstrated that delay has surprisingly little negative influence on sensory recovery. Useful protective sensation has been documented to return in a subset of patients even when neurorrhaphy was performed up to 2 years after the initial injury. The absolute critical limit of delay beyond which sensation cannot return remains unknown, though early repair is universally preferred.
Primary vs. Secondary Repair Strategies
Early suture is paramount in reducing the incidence of painful paresthesias, preventing debilitating neuromas, and regaining a highly useful degree of sensation. Interestingly, some studies indicate that if neurorrhaphy is performed within 3 months after injury, sensory return may actually be superior after secondary repair compared to immediate primary repair, particularly in digital nerve injuries.
This phenomenon is explained by the consistent timetable of intracellular metabolic events following nerve transection. Between 3 and 6 weeks post-injury, the degenerative (Wallerian degeneration) and reparative changes within the neuronal cell body (chromatolysis) and the proximal/distal nerve trunks are well established. At this stage, the Schwann cells are highly active, and the neuron is metabolically primed for maximal axonal sprouting.
Evidence-Based Treatment Algorithm:
* Clean, Sharp Wounds: In cases of sharp lacerations (e.g., knife wounds) with no crush component, our practice is to perform primary neurorrhaphy immediately or during the first 3 to 7 days.
* High-Energy, Crush, or Contaminated Wounds: In the presence of extensive soft tissue contusion, laceration, crushing, or gross contamination—where the proximal and distal extent of intraneural damage is impossible to delineate acutely—a delayed secondary repair at 3 to 6 weeks is strongly preferred. This delay allows the zone of injury to declare itself, ensuring that the subsequent resection margins are composed of healthy, viable fascicles.
INFLUENCE OF THE LEVEL OF INJURY
The anatomical level of the nerve injury is a profound prognostic indicator. The general rule of peripheral nerve surgery is: the more proximal the injury, the more incomplete the overall return of motor and sensory function, especially in the most distal structures.
Conditions are more favorable for the recovery of proximal muscles because the distance regenerating axons must travel is shorter. A proximal injury (e.g., brachial plexus or proximal sciatic nerve) requires axons to regenerate over vast distances. Even at an optimal regeneration rate of 1 to 1.5 mm per day, it may take years for axons to reach distal targets like the intrinsic muscles of the hand or foot. By the time the axons arrive, the motor endplates have often undergone irreversible fibrosis. Consequently, proximal nerve injuries often require supplementary procedures, such as tendon transfers or nerve transfers (neurotization), to restore distal function.
OPERATIVE TECHNIQUE: PRINCIPLES OF NEURORRHAPHY
When performing a neurorrhaphy, meticulous microsurgical technique is mandatory. The procedure should be performed under loop magnification or an operating microscope, utilizing microsurgical instruments.
Preparation and Debridement
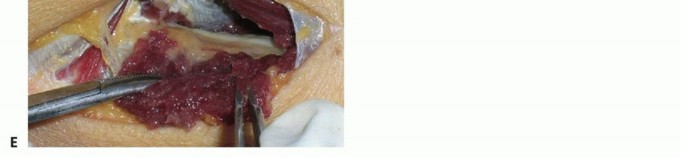
- Exposure: The nerve must be exposed in healthy tissue proximal and distal to the zone of injury before approaching the scarred or traumatized segment.
- Neurolysis and Resection: The injured nerve ends are mobilized. In delayed repairs, the terminal neuroma on the proximal stump and the glioma on the distal stump must be serially sectioned using a fresh diamond knife or razor blade.
- Assessment of Fascicles: Sectioning continues until healthy fascicles pout from the epineurium. The proximal stump must show healthy axoplasm, and the distal stump must show open, unscarred endoneurial tubes.
Pitfall: Inadequate resection of the proximal neuroma is a catastrophic error. If the surgeon encounters dense, fibrotic tissue without distinct fascicular architecture, further resection is mandatory, even if it necessitates a nerve graft.
Epineurial vs. Fascicular Repair
For the majority of peripheral nerve trunks, a meticulous group fascicular repair or an epineurial repair is performed.
* Epineurial Repair: Involves placing sutures (typically 8-0 or 9-0 non-absorbable monofilament) through the external epineurium. This is standard for nerves with a polyfascicular pattern where distinct group matching is impossible.
* Group Fascicular Repair: Involves suturing the interfascicular epineurium of matching fascicle groups. This is indicated when distinct motor and sensory fascicle groups can be anatomically identified (e.g., the ulnar nerve at the wrist).
Alignment is critical. The surgeon must use surface landmarks, such as longitudinal epineurial blood vessels, to ensure correct rotational alignment of the proximal and distal stumps, thereby minimizing funicular mismatch.
Nerve Grafting Technique
If a tension-free primary repair cannot be achieved, an autologous nerve graft is the gold standard. The sural nerve is the most commonly utilized donor due to its length (up to 30-40 cm can be harvested), predictable anatomy, and acceptable donor site morbidity (loss of sensation over the lateral border of the foot).
* The graft is reversed to prevent regenerating axons from escaping through severed branches.
* Multiple cables of the sural nerve are used to match the cross-sectional area of the injured nerve.
* The grafts are sutured in place using 9-0 or 10-0 nylon under zero tension.
POSTOPERATIVE PROTOCOL AND REHABILITATION
The success of a meticulously performed neurorrhaphy can be easily undone by improper postoperative care.
- Immobilization: The limb is immobilized in a well-padded splint or cast to protect the repair. If joint flexion was utilized to achieve a tension-free repair, the joint is immobilized in that exact position.
- Duration: Absolute immobilization is maintained for 3 to 4 weeks to allow the epineurial repair site to gain sufficient tensile strength.
- Progressive Mobilization: After 3 to 4 weeks, the splint is modified to allow gradual, progressive extension of the joint (typically 10 to 15 degrees per week). Aggressive or sudden stretching must be strictly avoided to prevent traction injury to the regenerating axons.
- Rehabilitation: Once full range of motion is achieved, the patient begins a rigorous program of physical therapy. This includes maintaining passive range of motion of denervated joints to prevent contractures, electrical stimulation of denervated muscles (though controversial, often used to delay atrophy), and eventually, sensory re-education and motor strengthening as reinnervation occurs.
In conclusion, the operative management of peripheral nerve injuries demands a profound understanding of nerve biomechanics, Wallerian degeneration, and microsurgical principles. By respecting the tension-free principle, appropriately timing the intervention based on the mechanism of injury, and executing precise surgical technique, the orthopedic surgeon can optimize the biological environment for axonal regeneration and maximize functional recovery for the patient.
You Might Also Like