Brachial Plexus Injuries: Comprehensive Diagnosis and Surgical Management
Key Takeaway
Brachial plexus injuries present complex reconstructive challenges, often resulting from high-energy trauma such as motorcycle accidents. Successful management requires precise anatomical localization, differentiating preganglionic from postganglionic lesions. This guide details the etiology, clinical evaluation, advanced electrodiagnostics, and step-by-step surgical interventions—including neurolysis, nerve grafting, and neurotization—aimed at restoring critical upper extremity function, prioritizing elbow flexion and shoulder abduction.
Etiology and Pathomechanics of Brachial Plexus Injuries
Brachial plexus injuries (BPI) represent one of the most devastating peripheral nerve injuries, often resulting in profound functional impairment of the upper extremity. The etiology of these injuries is broadly categorized into high-energy trauma, penetrating injuries, and iatrogenic or birth-related trauma.
In military combat, penetrating wounds from high-velocity missiles or shrapnel are the predominant cause. Conversely, in civilian populations, traction injuries secondary to high-energy blunt trauma are the most frequent mechanism. According to the epidemiological principles established by Narakas, approximately 70% of traumatic brachial plexus injuries occur secondary to motor vehicle accidents, and of these, 70% involve motorcycles or bicycles.
The pathomechanics of closed traction injuries dictate the pattern of neural damage. An impact that forcibly depresses the shoulder while laterally flexing the cervical spine to the contralateral side places immense tensile stress on the upper roots (C5, C6, and occasionally C7). Conversely, hyperabduction injuries, such as arresting a fall by grasping an object overhead, disproportionately tension the lower roots (C8, T1).
Associated Injuries
The sheer kinetic energy required to disrupt the brachial plexus frequently results in severe concomitant trauma.
* Skeletal Trauma: Fractures of the proximal humerus, scapula, clavicle, ribs, and cervical transverse processes are common. Scapulothoracic dissociation should be highly suspected in severe traction injuries.
* Vascular Injury: Rupture or intimal avulsion of the subclavian or axillary artery occurs in up to 20% of patients.
* Neurological Trauma: Concomitant spinal cord injury is reported in 2% to 5% of BPIs.
* Soft Tissue: Traumatic rotator cuff tears are frequently masked by the neurological deficit and must be considered during evaluation.
Clinical Pearl: In any high-energy shoulder girdle trauma presenting with a pulseless upper extremity and neurological deficit, vascular repair takes absolute precedence. The plexus should be tagged during the vascular intervention for delayed reconstruction.
Anatomical Classification of Brachial Plexus Injuries
Accurate classification is paramount for prognostication and surgical planning. The most critical distinction lies in determining whether the lesion is preganglionic (root avulsion) or postganglionic (rupture).
Preganglionic vs. Postganglionic Lesions
Preganglionic (Supraganglionic) Injuries: These occur proximal to the dorsal root ganglion (DRG), representing a true avulsion of the rootlets from the spinal cord. Because the central nervous system lacks robust regenerative capacity, these lesions are irrepairable by direct neurorrhaphy and necessitate nerve transfer (neurotization).

Postganglionic (Infraganglionic) Injuries: These occur distal to the neural foramen and DRG. The neurons remain connected to the spinal cord, allowing for Wallerian degeneration distally and potential regeneration if continuity is restored via neurolysis, primary repair, or interfascicular nerve grafting.
Topographical Classification
- Upper Plexus Injury (Erb-Duchenne Palsy): Involves C5 and C6 roots. Patients present with the classic "waiter's tip" posture. Deficits include paralysis of the deltoid, supraspinatus, infraspinatus, biceps, and brachialis. The primary disability is the inability to abduct/externally rotate the shoulder and flex the elbow.
- Lower Plexus Injury (Klumpke Palsy): Involves C8 and T1 roots. Characterized by paralysis of the intrinsic hand musculature and wrist/finger flexors, resulting in a "claw hand" deformity. Associated Horner syndrome (ptosis, miosis, anhidrosis) is a hallmark of T1 root avulsion.
- Cord-Level Injuries:
- Lateral Cord: Paralysis of the biceps and lateral pectoral muscles, with sensory loss over the anterolateral forearm.
- Posterior Cord: Paralysis of the deltoid, triceps, and wrist/finger extensors.
- Medial Cord: Combined ulnar and median nerve motor deficits (excluding pronator teres and flexor carpi radialis) with extensive medial sensory loss.
Diagnostic Modalities and Evaluation
A meticulous clinical examination forms the foundation of BPI diagnosis, but advanced imaging and electrodiagnostics are required to map the exact zone of injury.
Electrodiagnostic Studies (EMG/NCS)
Electromyography (EMG) and Nerve Conduction Studies (NCS) should be delayed until 3 to 4 weeks post-injury to allow for complete Wallerian degeneration.
* Sensory Nerve Action Potentials (SNAPs): The preservation of SNAPs in an anesthetic dermatome is the pathognomonic hallmark of a preganglionic injury. The sensory cell body in the DRG remains intact and viable, but its central connection to the spinal cord is severed.
* Motor Evoked Potentials: Fibrillation potentials indicate denervation. Serial EMGs are critical; the appearance of nascent motor units signals spontaneous reinnervation.
Advanced Imaging
Magnetic Resonance Imaging (MRI) and Computed Tomography (CT) Myelography are the gold standards for evaluating root integrity.
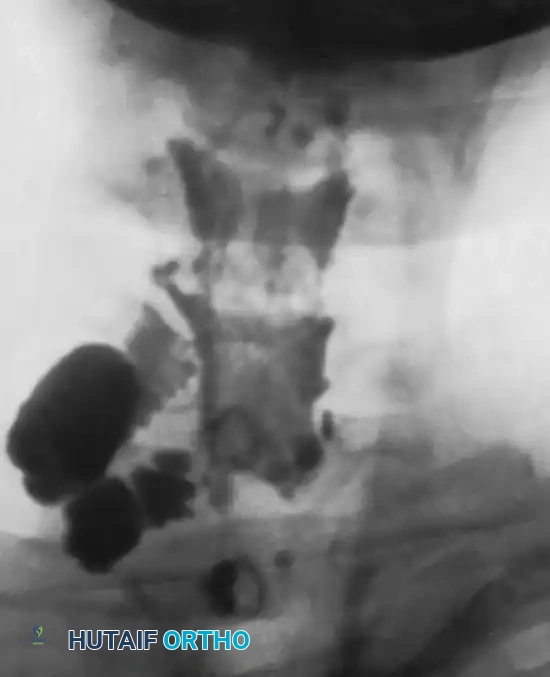
- CT Myelography: Highly sensitive for detecting pseudomeningoceles (extravasation of contrast through an empty dural root sleeve), which strongly correlates with root avulsion.
- MRI: Excellent for visualizing postganglionic neuromas, periplexus scarring, and the continuity of the trunks and cords.
The Histamine Axon Reflex Test
Though largely supplanted by modern electrodiagnostics, the cutaneous axon reflex remains a classic physiological test. A drop of histamine is scratched into the anesthetic skin. A normal response (vasodilation, wheal, and flare) in an anesthetic limb indicates an intact DRG and distal nerve (preganglionic lesion). An absent flare response indicates a postganglionic lesion.
Indications and Timing for Surgical Intervention
The management algorithm is dictated by the mechanism of injury (open vs. closed) and the progression of neurological recovery.
Open Injuries
- Sharp Lacerations: Immediate exploration and primary neurorrhaphy are indicated if the wound is clean and the patient is hemodynamically stable.
- High-Velocity Missile Wounds: Immediate exploration is contraindicated unless dictated by vascular injury. The concussive blast effect causes extensive neurapraxia and axonotmesis. A period of observation (3 to 4 weeks) followed by baseline EMG is recommended. If no recovery occurs by 3 to 6 months, exploration is warranted.
Surgical Warning: During emergency vascular repair in the setting of an open BPI, do not attempt exhaustive nerve repair. Tag the nerve ends with non-absorbable epineurial sutures or radiopaque clips, and interpose a Silastic sheet or viable muscle flap between the vascular graft and the neural elements to prevent tethering and facilitate delayed reconstruction.
Closed Traction Injuries
The majority of closed injuries are managed expectantly for the first 3 months. Physical therapy is instituted immediately to maintain passive range of motion and prevent contractures.
* Indications for Surgery at 3-6 Months:
1. Complete absence of clinical or EMG recovery.
2. Arrested recovery after initial improvement.
3. Severe, intractable neuropathic pain (often associated with root avulsions).
4. Confirmed preganglionic avulsions (early surgery at 3-4 weeks is advocated by some centers for isolated root avulsions to perform nerve transfers before motor endplate degradation).
Surgical Goals and Preoperative Planning
Reconstruction of the completely flail upper extremity requires a pragmatic approach. It is impossible to restore normal function. The internationally accepted hierarchy of surgical priorities is:
1. Restoration of Elbow Flexion: The absolute primary goal, allowing the hand to be brought to the mouth or workspace.
2. Restoration of Shoulder Abduction and External Rotation: Provides a stable platform for the hand and prevents glenohumeral subluxation.
3. Restoration of Protective Sensation: Specifically to the median nerve distribution (thumb and index finger).
4. Restoration of Wrist and Finger Flexion: Often requires free functioning muscle transfers (FFMT) in pan-plexus injuries.
Step-by-Step Surgical Approaches and Techniques
Patient Positioning and Preparation
The patient is positioned supine with a bump under the ipsilateral scapula. The head is turned to the contralateral side. The entire upper extremity, ipsilateral neck, chest, and both lower extremities (for sural nerve graft harvesting) are prepped and draped free. No paralytic agents should be used by anesthesia to allow for intraoperative nerve stimulation.
The Supraclavicular Approach
- Incision: A zigzag or L-shaped incision is made along the posterior border of the sternocleidomastoid (SCM), extending laterally parallel to the clavicle.
- Superficial Dissection: The platysma is divided. The external jugular vein and supraclavicular nerves are identified and preserved if possible.
- Deep Dissection: The omohyoid muscle is identified and divided. The transverse cervical and suprascapular vessels are ligated if they impede access.
- Identification of Landmarks: The anterior scalene muscle is the key landmark. The phrenic nerve is identified coursing from lateral to medial across its anterior surface and protected.
- Plexus Exposure: The roots (C5, C6, C7) are identified emerging between the anterior and middle scalene muscles. Dissection proceeds distally to the trunks.
The Infraclavicular Approach
- Incision: Extends from the supraclavicular incision, crossing the clavicle (osteotomy is rarely required but can be performed), and follows the deltopectoral groove.
- Dissection: The cephalic vein is retracted. The pectoralis major is retracted medially, and the pectoralis minor is tenotomized at the coracoid process to expose the cords and terminal branches surrounding the axillary artery.

Intraoperative Neurophysiology
Direct intraoperative nerve stimulation and recording of Nerve Action Potentials (NAPs) are mandatory.
* If a NAP is conducted across a neuroma-in-continuity, the internal fascicular architecture is intact. Simple neurolysis is indicated.
* If no NAP is recorded, the lesion represents a neurotmesis or severe axonotmesis with dense scarring. The neuroma must be resected back to healthy fascicles (evidenced by "mushrooming" of axoplasm) for grafting.
Interfascicular Nerve Grafting
When a postganglionic rupture is resected, the resulting gap cannot be closed under tension. Autologous nerve grafting is required.
* Donor Nerves: The sural nerve is the workhorse, providing up to 40 cm of graft material per leg. The medial antebrachial cutaneous nerve is an alternative.
* Technique: The grafts are reversed to minimize axonal loss through branching. They are interposed between the proximal and distal stumps and secured with 9-0 or 10-0 nylon epineurial sutures and fibrin glue under microscopic magnification.

Neurotization (Nerve Transfers)
In preganglionic root avulsions, no proximal stump is available. Expendable donor nerves are transferred directly to critical distal motor nerves close to the target muscle to minimize regeneration time.
* For Elbow Flexion:
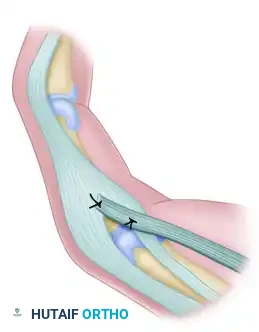
* Oberlin Transfer: Transfer of expendable motor fascicles from the ulnar nerve (usually supplying the FCU) directly to the biceps motor branch of the musculocutaneous nerve.
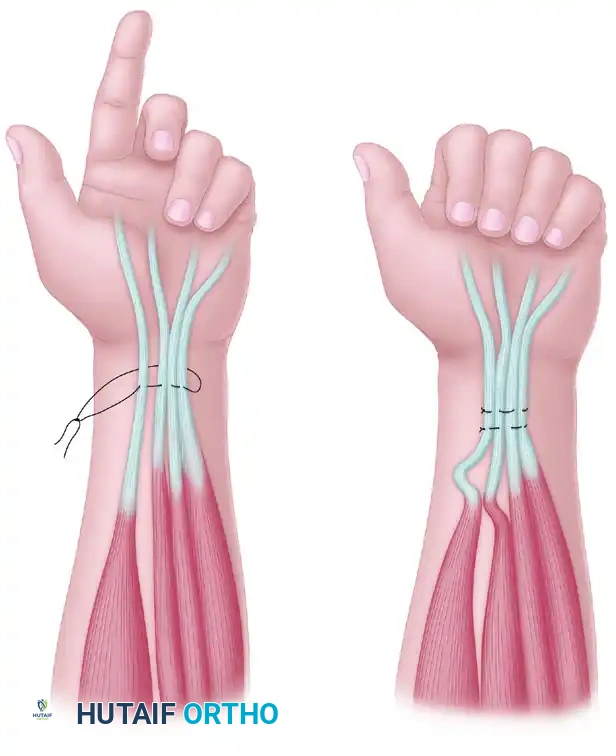
* Intercostal Nerve Transfer: T3-T5 intercostal nerves are harvested and coapted to the musculocutaneous nerve.
* For Shoulder Abduction:
* Spinal Accessory Nerve (CN XI) to Suprascapular Nerve: Restores supraspinatus and infraspinatus function. The distal CN XI is utilized, preserving the proximal branches to the upper trapezius.
* Somsak Transfer: Triceps motor branches (from the radial nerve) transferred to the axillary nerve to restore deltoid function.
Postoperative Protocol and Rehabilitation
Surgical reconstruction of the brachial plexus is only the beginning of a protracted recovery process. Axons regenerate at a rate of approximately 1 mm per day (1 inch per month).
- Immobilization: Postoperatively, the limb and neck are immobilized in a custom orthosis or sling for 3 to 4 weeks to prevent tension on the microvascular nerve coaptations.
- Passive Range of Motion (PROM): Initiated at 4 weeks to prevent joint contractures, particularly glenohumeral adhesive capsulitis and elbow flexion contractures.
- Biofeedback and Motor Re-education: As reinnervation occurs (often 12 to 18 months post-surgery), intensive cortical remapping is required. Patients who undergo an intercostal nerve transfer must learn to take deep breaths to initiate elbow flexion until cortical plasticity allows for independent movement.
- Secondary Procedures: If primary nerve reconstruction fails to yield adequate functional recovery by 18 to 24 months, secondary palliative procedures such as tendon transfers (e.g., Steindler flexorplasty), free functioning muscle transfers (Gracilis flap), or joint arthrodesis (shoulder or wrist) must be considered to maximize the patient's independence.
You Might Also Like