Above-Elbow and Below-Elbow Amputations: Surgical Principles, Anatomy, and Biomechanics
Key Takeaway
Above-elbow (transhumeral) and below-elbow (transradial) amputations are definitive surgical interventions primarily for advanced upper extremity pathologies, including extensive sarcomas, severe trauma, or intractable infection. They require meticulous planning, focusing on maximizing residual limb length, preserving viable musculature for myodesis, and managing neurovascular structures to optimize prosthetic fitting and functional outcomes, thereby addressing complex patient needs.
Above-Elbow and Below-Elbow Amputations
Introduction & Epidemiology
Amputation of the upper extremity, specifically above-elbow (AEA) and below-elbow (BEA) levels, represents a definitive surgical intervention primarily necessitated by advanced pathologies where limb salvage is either infeasible or contraindicated. While the principles of upper extremity amputation remain consistent with those for the lower extremity, the functional imperative for preserving length and proprioceptive input is arguably more pronounced, given the intricate role of the hand and arm in daily activities and personal identity.
The primary indication for such major resections in the upper limb in an oncologic context involves extensive soft tissue and bone sarcomas that cause significant destruction, extend into critical neurovascular bundles, or present with inadequate margins for a reconstructive approach. As noted by Bickels et al., AEA may be required for advanced sarcomas of the forearm and elbow region, while BEA is indicated for tumors of the forearm and hand.
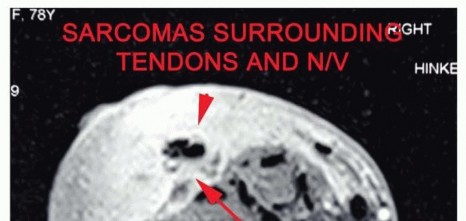
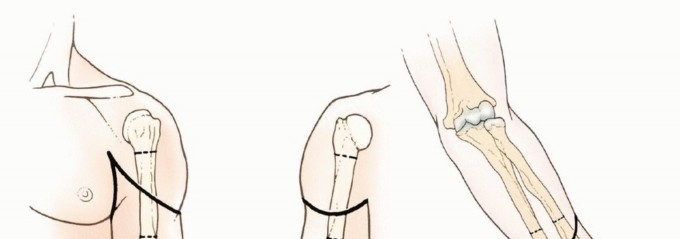
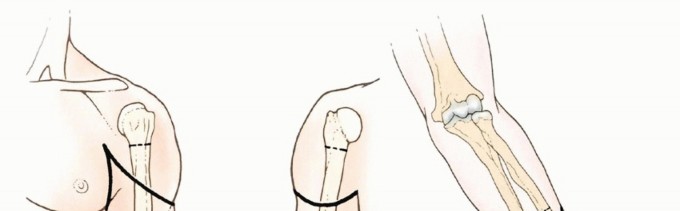
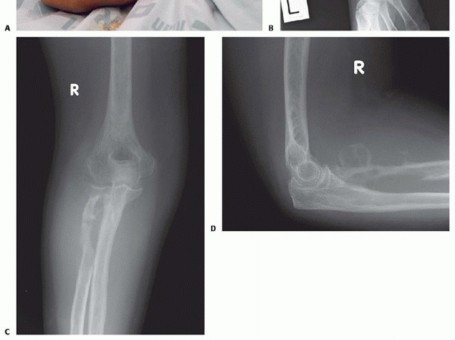
Figure 1A: Example of an advanced soft tissue sarcoma around the elbow requiring an above-elbow amputation.
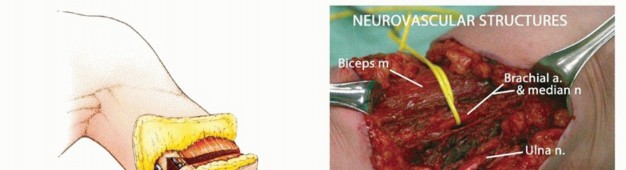
Figure 1B: Example of a tumor of the forearm necessitating a below-elbow amputation.
Amputations at these levels are comparatively rare. This infrequency is attributable to several factors:
* The upper arm, elbow, and forearm are less common sites for primary musculoskeletal tumors compared to the lower extremities.
* Tumors in these anatomically exposed regions are often noticed earlier, allowing for diagnosis and intervention at potentially more resectable stages.
* Advances in multimodal oncology, including highly effective preoperative chemotherapy, radiation therapy, and isolated limb perfusion techniques, have significantly expanded the scope of limb salvage surgeries, enabling local tumor control in a majority of patients who present with large or locally advanced lesions.
Nonetheless, AEA and BEA retain a critical, albeit niche, role in the management algorithm for aggressive soft tissue and bone tumors, especially in cases of extensive local recurrence, infection, intractable pain, or devastating trauma not amenable to reconstruction. The decision to proceed with amputation is invariably complex, requiring meticulous pre-operative planning, a multidisciplinary approach, and comprehensive patient counseling regarding functional outcomes and prosthetic options.
Surgical Anatomy & Biomechanics
A thorough understanding of the surgical anatomy and biomechanical implications is paramount for successful upper extremity amputation. The goal is to maximize functional length, preserve viable musculature for myodesis or myoplasty, manage neurovascular structures appropriately, and create a stump that is well-padded, conical, non-tender, and amenable to prosthetic fitting.
Above-Elbow Amputation (Transhumeral)
Anatomical Considerations
The humerus is surrounded by distinct muscular compartments separated by the medial and lateral intermuscular septa.
*
Anterior Compartment:
Biceps brachii, brachialis, coracobrachialis. Innervated by the musculocutaneous nerve. The biceps provides significant flexion power and contributes to shoulder stability.
*
Posterior Compartment:
Triceps brachii. Innervated by the radial nerve. Essential for elbow extension, and proximally, contributes to shoulder stability.
The main neurovascular bundle courses medially within the arm.
*
Brachial Artery:
The primary arterial supply to the forearm, typically accompanied by two brachial veins.
*
Median Nerve:
Travels alongside the brachial artery.
*
Ulnar Nerve:
Initially medial, it pierces the medial intermuscular septum to lie posterior to the medial epicondyle.
*
Radial Nerve:
Travels posteriorly in the spiral groove, then pierces the lateral intermuscular septum to enter the anterior compartment distal to the deltoid insertion.
The residual humeral length directly impacts the moment arm available for prosthetic control and the volume for socket fit. Preserving as much humerus as possible, while achieving oncologic clearance, is critical. High amputations may lose the ability to utilize deltoid or pectoralis major for myoelectric signals or direct muscular power.
Biomechanical Implications
A longer humeral remnant provides a greater lever arm, enhancing the control and stability of a prosthetic elbow joint. It also offers a larger surface area for socket suspension and reduces pressure points. Myodesis or myoplasty of the biceps and triceps over the end of the humerus provides padding, helps stabilize the soft tissue envelope, and can facilitate myoelectric control signals. Loss of the elbow joint significantly compromises complex multi-planar activities.
Figure 2: Illustrative anatomy of the upper arm musculature and neurovascular structures, critical for above-elbow amputation planning.
Below-Elbow Amputation (Transradial)
Anatomical Considerations
The forearm is characterized by a complex arrangement of muscles, nerves, and vessels, divided into anterior (flexor) and posterior (extensor) compartments.
- Anterior (Flexor) Compartment: Superficial (pronator teres, flexor carpi radialis, palmaris longus, flexor carpi ulnaris, flexor digitorum superficialis) and deep (flexor digitorum profundus, flexor pollicis longus, pronator quadratus) layers. Primarily innervated by the median nerve and ulnar nerve. Vascular supply mainly from radial and ulnar arteries.
- Posterior (Extensor) Compartment: Superficial (brachioradialis, extensor carpi radialis longus/brevis, extensor digitorum, extensor digiti minimi, extensor carpi ulnaris) and deep (supinator, abductor pollicis longus, extensor pollicis longus/brevis, extensor indicis) layers. Primarily innervated by the radial nerve (posterior interosseous nerve branch). Vascular supply mainly from the radial and posterior interosseous arteries.
The radius and ulna articulate to allow pronation and supination. Preserving the interosseous membrane and the relative lengths of these bones can influence stump stability and muscle attachment. Maximum length preservation is crucial, especially regarding the ability to retain pronation/supination, which is possible only with very distal amputations below the pronator quadratus. However, for most transradial amputations, this movement is lost.
Biomechanical Implications
The forearm stump length directly impacts prosthetic function. A longer transradial stump provides a greater lever arm for prosthetic control, allowing for better suspension, rotation, and potentially more nuanced myoelectric control. The preservation of specific muscles, such as the brachioradialis, pronator teres, and wrist flexors/extensors, allows for effective myodesis/myoplasty, providing robust soft tissue coverage, stump stability, and potential sites for targeted muscle reinnervation (TMR) for advanced prosthetics. The ideal transradial stump length is typically considered to be 5-7 inches distal to the olecranon to allow for prosthetic components without undue bulk.
Indications & Contraindications
The decision for upper extremity amputation is always made after careful consideration of all viable limb salvage options, anticipated functional outcomes, and patient-specific factors.
Indications
Oncologic Indications
- Unresectable Malignancy: Tumors extending beyond fascial planes into major neurovascular structures (brachial plexus, major vessels) where clean surgical margins cannot be achieved without sacrificing essential structures that cannot be reconstructed.
- Inability to Obtain Wide Margins: Despite extensive planning, a wide oncologic margin (typically >1 cm of uninvolved tissue) cannot be achieved with limb salvage, increasing the risk of local recurrence.
- Failed Limb Salvage: Persistence or recurrence of tumor after previous limb-sparing surgery, radiation, or chemotherapy.
- Intractable Pain or Ulceration: Advanced tumors causing severe, unmanageable pain or extensive skin/soft tissue breakdown that significantly impairs quality of life.
- Pathologic Fracture with Tumor Burden: Extensive bone destruction with tumor involvement that precludes stable reconstruction.
Traumatic Indications
- Devastating Crush Injuries: Irreversible damage to multiple tissue types (bone, muscle, nerve, vessel) rendering the limb non-viable or non-functional despite attempts at revascularization and reconstruction (e.g., mangled extremity severity score).
- Extensive Tissue Loss: Severe avulsion injuries or blast trauma leading to significant loss of skin, soft tissue, bone, and neurovascular structures.
- Ischemia: Prolonged, irreversible ischemia leading to necrosis not amenable to revascularization.
- Severe Infection: Uncontrolled, rapidly progressing necrotizing infection (e.g., necrotizing fasciitis, gas gangrene) that is limb-threatening and unresponsive to debridement and antibiotics, potentially systemic-threatening.
Other Indications
- Congenital Anomalies: Rare instances of severe malformations with absent or non-functional limb components where amputation and prosthetic fitting offer superior function.
- Severe Deformity/Contracture: End-stage conditions leading to a useless, painful, or non-functional limb that cannot be rehabilitated.
Contraindications
- Preservable Limb: Any situation where a reasonable attempt at limb salvage or reconstruction could yield a functional and durable outcome with acceptable morbidity.
- Patient Refusal: Even if medically indicated, amputation should not be performed without informed consent.
- Poor General Health/Prognosis: In patients with very limited life expectancy or severe comorbidities, the morbidity and functional recovery associated with amputation may not be justified, favoring palliative care.
- Psychological Instability: Significant psychiatric illness that could interfere with consent, post-operative care, or prosthetic rehabilitation.
The decision-making process is summarized in the table below:
| Indication Type | Operative Goal (Amputation) | Non-Operative Alternative / Consideration |
|---|---|---|
| Oncologic | Achieve wide oncologic margins, local disease control, pain relief, prevent systemic spread. | Limb salvage with wide resection, reconstructive surgery, neoadjuvant/adjuvant chemotherapy/radiation, palliative care. |
| Traumatic | Remove non-viable tissue, prevent infection, establish functional stump for prosthetics. | Revascularization, extensive debridement, complex reconstructive surgery (flaps, bone grafting, nerve repair). |
| Infectious | Remove septic focus, control systemic infection, prevent spread. | Aggressive debridement, targeted antibiotics, wound care, vacuum-assisted closure. |
| Vascular | Remove ischemic, necrotic tissue, prevent gangrene and systemic toxicity. | Revascularization (bypass, angioplasty), medical management of peripheral artery disease, local wound care for limited necrosis. |
| Congenital/Deformity | Establish functional stump for prosthetic fitting. | Orthotic management, corrective osteotomies, physiotherapy. |
Pre-Operative Planning & Patient Positioning
Meticulous pre-operative planning is crucial to optimize oncologic outcomes, functional results, and minimize complications.
Pre-Operative Planning
-
Multidisciplinary Tumor Board (Oncology Cases): Essential for reviewing imaging (MRI, CT, PET-CT), biopsy results, and discussing the full spectrum of treatment options (neoadjuvant/adjuvant therapies, limb salvage vs. amputation) with orthopedic oncologists, medical oncologists, radiation oncologists, pathologists, and radiologists.

Figure 3: Pre-operative imaging demonstrating tumor extent in the forearm, influencing amputation level.
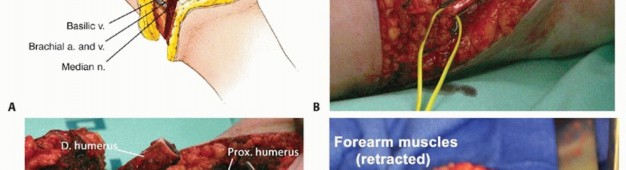
Figure 4: Detailed MRI of a soft tissue sarcoma, crucial for assessing neurovascular involvement and determining surgical margins. -
Imaging Review:
- MRI: Gold standard for soft tissue and bone tumor delineation, assessing neurovascular involvement, and planning safe surgical margins.
- CT Angiography: May be necessary to map vascular anatomy, especially in complex trauma or large tumors encroaching on major vessels.
- Plain Radiographs: To assess bone involvement, pathologic fractures, or pre-existing degenerative changes.
-
Prosthetic Consultation: Early involvement of a prosthetist is vital to discuss optimal stump length, desired prosthetic components, and anticipated functional capabilities. This informs surgical decisions regarding muscle handling and stump contour.
-
Psychological Evaluation & Support: Amputation is a life-altering event. Psychological counseling, peer support groups, and patient education about phantom limb phenomena are important components of holistic care.
-
Nutritional Status: Optimize nutrition, especially in oncologic or chronic infection cases, to enhance wound healing and reduce complication rates.
-
Pre-operative Optimization:
- Infection Control: Aggressive debridement, targeted antibiotics for septic cases.
- Smoking Cessation: Improves wound healing.
- Diabetic Control: Reduces infection and wound complications.
- Pre-operative Chemotherapy/Radiation: For oncologic cases, to downstage tumors, achieve better margins, and address micrometastasis.
-
Marking the Incision: Pre-operatively mark the desired skin flaps and anticipated bone cut level, considering both oncologic margins and optimal prosthetic fit.
Patient Positioning
- General Anesthesia: Typically required for these procedures.
- Supine Position: The patient is positioned supine on the operating table.
- Arm Abduction: The affected arm is abducted on an arm board or a specialized hand table, allowing full circumferential access to the limb.
- Padding: Meticulous padding of all pressure points (e.g., elbow, heel, sacrum) is critical to prevent iatrogenic nerve compression or skin breakdown.
- Tourniquet Application: A pneumatic tourniquet is routinely applied high on the arm or thigh (for AEA/BEA respectively) to ensure a bloodless field, which is crucial for precise neurovascular dissection and margin assessment. The tourniquet cuff should be well-padded and appropriately sized.
-
Surgical Prep:
The skin preparation extends proximally to the shoulder and ideally to the neck, and distally to the hand, encompassing the entire planned surgical field and allowing for potential extension if needed.
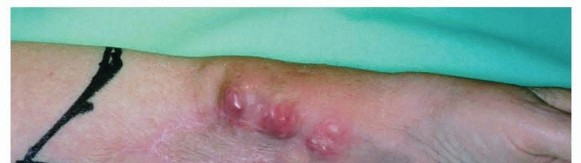
Figure 5: Patient positioning for upper extremity surgery, demonstrating arm abduction and sterile draping.

Figure 6: A wider view of the prepared surgical field, highlighting the extent of skin prep and draping for an amputation procedure.
Detailed Surgical Approach / Technique
The goal of both above-elbow and below-elbow amputations is to create a functional, well-padded, non-tender, and conical residual limb (stump) that can tolerate prosthetic fitting. Key principles include achieving adequate surgical margins (oncology), maximizing limb length, meticulous neurovascular management, and stabilizing musculature.
General Principles for Upper Extremity Amputations
-
Skin Flap Design: Typically, equal anterior and posterior flaps or a long posterior flap are utilized. The flaps should be broad-based, allowing for tension-free closure and good vascularity. The apex of the flap should extend approximately 1-2 cm distal to the planned bone transection level.
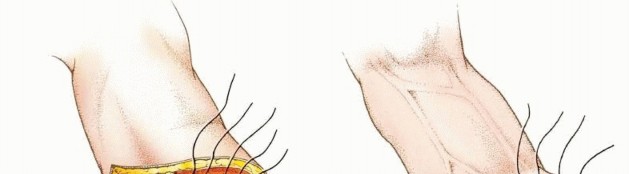
Figure 7: Example of initial skin incision lines for an upper extremity amputation. -
Muscle Management (Myodesis vs. Myoplasty):
- Myodesis: Direct attachment of muscle to bone. This provides robust stabilization of the muscle bellies, preventing retraction and atrophy, and creates a more stable, powerful stump. It is preferred when feasible.
-
Myoplasty:
Muscle-to-muscle closure. Used when myodesis to bone is not possible or practical, creating muscle slings to stabilize the stump.
Both techniques aim to provide padding over the bone end, facilitate prosthetic control, and reduce neuromas.
-
Neurovascular Management:
- Vessels: Major arteries and accompanying veins are ligated meticulously (triple ligation for arteries). Proximal ligation should be performed securely to prevent hematoma.
- Nerves: Nerves are identified, gently put on stretch, and sharply transected at a level that allows them to retract proximally into soft tissue. This reduces the risk of symptomatic neuroma formation by preventing the nerve end from being at a superficial, palpable, or weight-bearing site. Cauterization of nerve ends is generally avoided as it can induce neuroma formation.
-
Bone Transection: The bone is cut perpendicular to its long axis using an oscillating saw. Bone edges are meticulously smoothed and beveled (especially the anterior edge) to prevent sharp points that could erode through the soft tissue and skin or cause pressure points within a prosthesis.
-
Hemostasis and Drainage: Thorough hemostasis is achieved throughout the procedure. A suction drain is typically placed before closure to prevent hematoma formation, which can compromise wound healing and increase infection risk.
Above-Elbow Amputation (Transhumeral) Technique
The goal is to preserve maximum humeral length while achieving oncologic margins or removing non-viable tissue. An ideal length provides leverage for prosthetic fitting and muscle control.
-
Incision:
- Design equal anterior and posterior skin flaps. The length of the flaps should be approximately half the diameter of the arm at the level of planned bone resection.
- Incision extends through skin and subcutaneous tissue.
-
Muscle and Fascia Dissection:
- Incise the deep fascia circumferentially.
- Separate muscles of the anterior compartment (biceps, brachialis) from the posterior compartment (triceps) along the intermuscular septa.
-
Retract muscle groups proximally.
Figure 8: Dissection exposing major muscle groups in the upper arm prior to transection.
-
Neurovascular Dissection and Ligation:
- Identify the brachial artery and veins running medially with the median nerve . Ligate the brachial artery and veins securely.
- Identify the ulnar nerve (medial side) and the radial nerve (which spirals posteriorly, then anteriorly).
-
Gently pull each major nerve distally, sharply transect it, and allow it to retract into the muscle belly to prevent superficial neuromas.
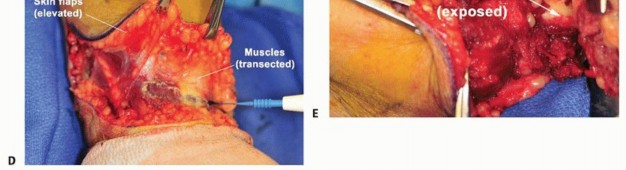
Figure 9: Identification and isolation of neurovascular bundles during a transhumeral amputation.
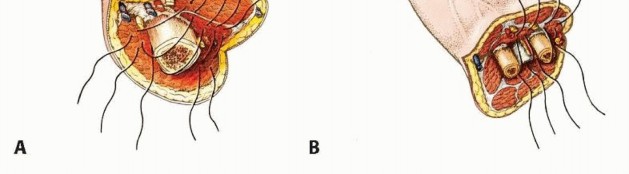
Figure 10: Ligation of major vessels, ensuring secure hemostasis.
-
Bone Transection:
- Retract all soft tissues proximally to expose the humerus at the planned level.
- Using an oscillating saw, transect the humerus perpendicular to its long axis.
-
Smooth and bevel the edges of the bone, particularly the anterior aspect, to create a rounded, non-irritating stump end.
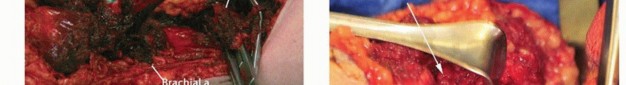
Figure 11: Transection of the humerus using an oscillating saw.
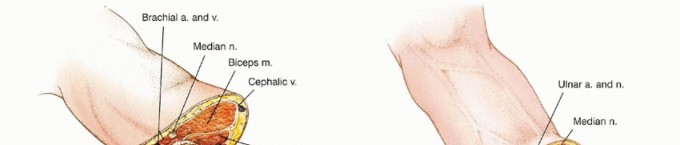
Figure 12: Smoothing and beveling of the bone edges after transection.
-
Muscle Stabilization (Myodesis/Myoplasty):
- The goal is to create a well-padded, stable, and conical stump.
- If possible, perform myodesis of the biceps and triceps over the end of the humerus. Drill small holes in the distal humerus and pass sutures through the tendons or muscle bellies for secure fixation. This maintains muscle tension and provides proprioceptive feedback for prosthetic control.
-
Alternatively,
myoplasty
involves suturing the anterior and posterior muscle groups to each other over the bone end.
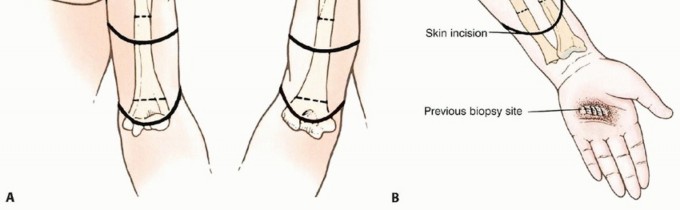
Figure 13: Suturing of muscle groups (myoplasty) over the bone end to create a padded stump.
-
Closure:
- Insert a closed suction drain (e.g., Jackson-Pratt).
- Close the deep fascia over the muscle mass.
- Close subcutaneous tissue, ensuring an even distribution of tissue.
-
Close the skin with interrupted or continuous non-absorbable sutures or staples, ensuring a tension-free closure.
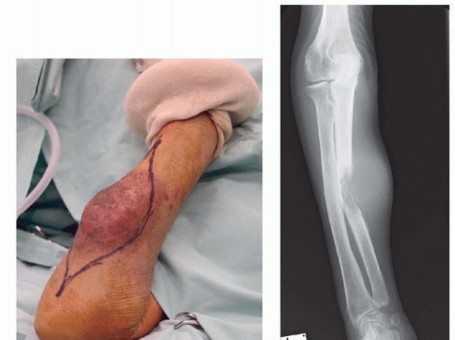
Figure 14: Final skin closure after a transhumeral amputation.
Below-Elbow Amputation (Transradial) Technique
Preserving maximal forearm length is critical for prosthetic function, as even short remnants can provide pronation/supination (if very distal) and crucial leverage.
-
Incision:
- Commonly, equal anterior and posterior skin flaps are used. The length of the flaps should allow for tension-free closure over the bone ends. A long posterior flap can also be considered if anterior tissue quality is compromised.
- The incision extends through skin and subcutaneous tissue.
-
Muscle and Fascia Dissection:
- Incise the deep fascia circumferentially.
- Separate the flexor compartment muscles from the extensor compartment muscles.
-
Retract muscle groups proximally.
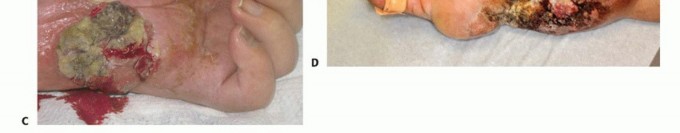
Figure 15: Dissection revealing the muscle compartments of the forearm.
-
Neurovascular Dissection and Ligation:
- Identify and ligate the radial artery and veins and the ulnar artery and veins .
- Identify the median nerve and its anterior interosseous branch.
- Identify the ulnar nerve .
- Identify the radial nerve and its superficial and posterior interosseous nerve (PIN) branches.
-
Gently pull each major nerve distally, sharply transect it, and allow it to retract into the muscle belly.
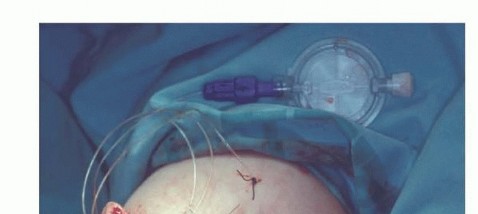
Figure 16: Isolation and management of the forearm neurovascular structures during transradial amputation.
-
Bone Transection:
- Retract soft tissues proximally to expose the radius and ulna at the planned level.
- Transect the radius and ulna using an oscillating saw. To prevent synostosis and create a better conical shape, the ulna is typically cut slightly shorter (e.g., 1-2 cm) than the radius, or vice-versa, depending on the desired stump contour and muscle attachments.
- Smooth and bevel all bone edges, particularly the anterior aspects, to prevent skin breakdown and provide a smooth surface for prosthetic fit.
-
Remove a small section of the interosseous membrane between the bone ends to further reduce the risk of synostosis.
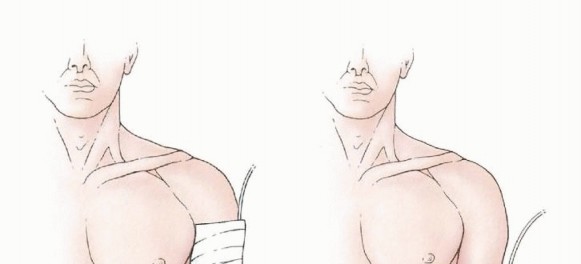
Figure 17: Transection of the radius and ulna, often at slightly different lengths to prevent synostosis and improve stump contour.
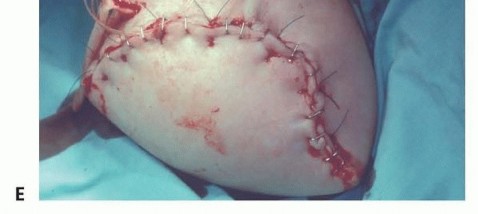
Figure 18: Smoothing and beveling of the radial and ulnar bone ends.
-
Muscle Stabilization (Myodesis/Myoplasty):
- The aim is to create a dynamic, well-padded, and conical stump.
- Myodesis is preferred. For example, the brachioradialis muscle can be attached to the ulna, and the common flexor group to the radius, or vice versa, to provide muscle balance and functional movement.
- Myoplasty involves suturing antagonist muscle groups (flexors to extensors) over the bone ends.
-
The pronator teres is often preserved and can be used for myodesis if the amputation is distal enough, offering valuable rotational input for advanced prosthetics.
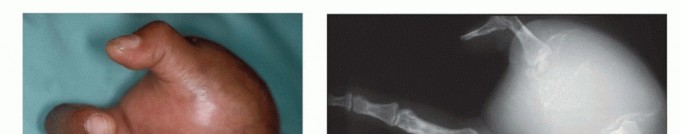
Figure 19: Myodesis technique, attaching remaining muscle groups to the distal radius and ulna.
-
Closure:
- Insert a closed suction drain.
- Close the deep fascia to cover the muscle mass and stabilize it.
- Close the subcutaneous tissue, ensuring an even distribution and minimal tension.
-
Close the skin with interrupted or continuous non-absorbable sutures or staples.
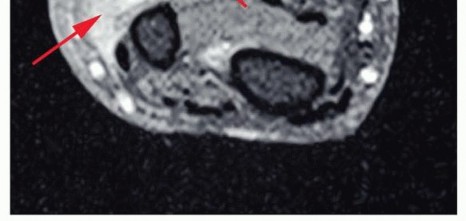
Figure 20: Completed skin closure following a transradial amputation.
Complications & Management
Amputations, despite being definitive surgical solutions, are associated with a range of potential complications, both acute and chronic. Proactive management and a multidisciplinary approach are essential for optimizing patient outcomes.
Common Complications
| Complication | Incidence / Likelihood | Management & Salvage Strategies |
|---|---|---|
| Wound Infection | 5-20% (higher in contaminated trauma/vascular cases) |
Prevention:
Meticulous surgical technique, appropriate perioperative antibiotics, sterile dressing changes, optimal nutritional status.
Management: Early recognition, wound exploration and debridement, targeted antibiotics based on culture, local wound care (e.g., VAC therapy), stump revision if severe or persistent. |
| Hematoma | ~5-10% |
Prevention:
Meticulous hemostasis intraoperatively, drain placement.
Management: Close monitoring, evacuation if large and symptomatic (may require return to OR), compression dressings. |
| Wound Dehiscence / Skin Necrosis | 5-15% (higher with poor vascularity, tension) |
Prevention:
Tension-free closure, well-vascularized flaps, avoid cautery to skin edges.
Management: Local wound care, serial debridement, secondary closure, skin grafting, or flap coverage for larger defects. Stump revision may be necessary in severe cases affecting prosthetic fit. |
| Phantom Limb Pain (PLP) | 50-80% of amputees experience some form |
Prevention:
Pre-emptive pain management, regional nerve blocks.
Management: Multimodal approach: pharmacotherapy (gabapentin, pregabalin, tricyclic antidepressants, opioids), nerve blocks, TENS, mirror therapy, psychological counseling, surgical neuroma resection, spinal cord stimulators, targeted muscle reinnervation (TMR). |
| Residual Limb Pain / Neuroma | 20-50% |
Prevention:
Tension-free nerve transection, burying nerve ends in soft tissue or bone.
Management: Conservative (NSAIDs, local injections, TENS), pharmacotherapy (neuropathic agents), surgical neuroma excision and repositioning, TMR (especially for high-risk neuromas). |
| Stump Edema | Common early post-op |
Prevention:
Compression dressings immediately post-op.
Management: Persistent compression (elastic bandages, shrinkers), elevation, physical therapy. |
| Stump Morphology Issues (Conical, Bulbous, Dog Ears) | Variable |
Prevention:
Careful flap design, muscle contouring (myodesis/myoplasty).
Management: Revision surgery (stump plasty) to optimize prosthetic fit and comfort. |
| Heterotopic Ossification (HO) | ~5-10% (especially in trauma/burns) |
Prevention:
Prophylactic radiation or NSAIDs (e.g., indomethacin) for high-risk patients (e.g., blast injuries, head injury).
Management: Conservative management for asymptomatic HO; surgical excision for symptomatic HO interfering with prosthetic fit or causing pain (usually after HO maturation). |
| Joint Contractures (Proximal) | Common if not actively prevented |
Prevention:
Early range of motion (ROM) exercises for proximal joints (shoulder for AEA, elbow for BEA), proper positioning.
Management: Aggressive physical therapy, stretching, splinting, botulinum toxin injections for spasticity, surgical release in refractory cases. |
| Psychological Distress / Body Image Issues | High incidence |
Prevention:
Pre-operative counseling, peer support, multidisciplinary team involvement.
Management: Psychological counseling, support groups, early prosthetic fitting and training, vocational rehabilitation. |
Specific Management Considerations
- Neuroma Management: Surgical treatment for symptomatic neuromas typically involves careful identification of the neuroma, sharp excision, and relocation of the nerve end into a less weight-bearing, well-vascularized soft tissue bed (e.g., muscle belly) or bone, ensuring it is under no tension. Advanced techniques like Targeted Muscle Reinnervation (TMR) can reroute transected nerves into denervated muscle targets to provide a physiological endpoint, reducing neuroma pain and creating new myoelectric control sites for advanced prosthetics.
- Stump Revision: This may be required for chronic wound breakdown, painful scars, poor stump shape preventing prosthetic fit, or refractory neuromas. The goal is always to improve the residual limb's function and comfort.
- Persistent Phantom Sensation/Pain: This can be debilitating. A comprehensive pain management strategy involving a pain specialist, physical therapist, and psychologist is often needed. Modalities include pharmacotherapy, TENS units, acupuncture, and psychological interventions like cognitive behavioral therapy.
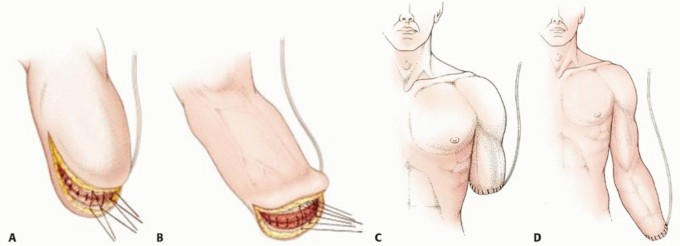
Figure 21: Post-operative wound healing with a well-formed stump.
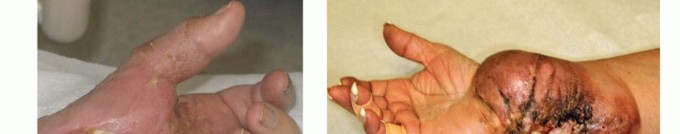
Figure 22: Image depicting a common site for neuroma formation post-amputation.
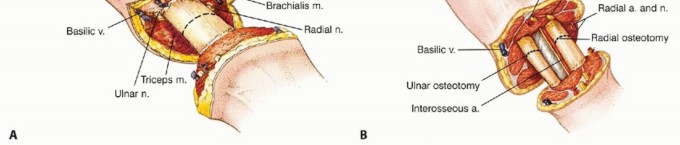
Figure 23: Example of heterotopic ossification observed in a residual limb.
Post-Operative Rehabilitation Protocols
Post-operative rehabilitation is a crucial, multi-stage process integral to maximizing functional recovery and facilitating successful prosthetic integration. It begins immediately post-surgery and extends through the patient's lifetime.
Phase 1: Immediate Post-Operative (Day 0 - Week 2)
- Pain Management: Aggressive, multimodal analgesia is essential to control incisional pain and manage early phantom limb sensations/pain. This includes opioids, NSAIDs, neuropathic agents (gabapentin, pregabalin), and regional nerve blocks.
- Wound Care: Daily inspection for signs of infection, hematoma, or dehiscence. Dressing changes per protocol.
- Stump Compression: Application of elastic bandages or a soft compression garment/shrinker immediately after drain removal. This helps reduce edema, shape the stump into a conical form, and prepare it for prosthetic fitting.
- Positioning: Proper positioning to prevent contractures (e.g., full elbow extension for BEA, neutral shoulder position for AEA).
- Early Mobilization: Gentle active and passive range of motion (ROM) exercises of the joints proximal to the amputation (shoulder for AEA, elbow for BEA) to prevent stiffness and maintain strength.
- Desensitization: Gentle tapping, rubbing, and pressure application to the stump to reduce hypersensitivity and prepare for prosthetic wear.
-
Patient Education:
Reinforce phantom limb phenomena, wound care, and early rehabilitation goals.

Figure 24: Immediate post-operative stump dressing and compression.
Phase 2: Pre-Prosthetic Training (Week 2 - 8)
- Stump Maturation: Continued use of compression therapy (shrinker socks or elastic bandages) to achieve optimal conical shape and reduce edema. The stump must be well-healed and non-tender.
- Strengthening: Progressive resistive exercises for the residual limb musculature and the proximal joints to improve strength and endurance.
- Range of Motion: Aggressive ROM exercises to ensure full, pain-free movement of the shoulder (AEA) or elbow (BEA). Avoidance of contractures is paramount.
- Stump Desensitization and Scar Mobilization: Continued desensitization techniques. Massage of scar tissue to prevent adherence and improve pliability.
- Proprioceptive Training: Exercises to enhance proprioception and neuromuscular control of the residual limb.
- One-Handed Task Training: For unilateral amputees, training in compensatory techniques for activities of daily living (ADLs) while awaiting prosthetic fitting.
-
Prosthetic Consultation and Casting:
If not initiated pre-operatively, this phase includes meeting with the prosthetist, discussing prosthetic options (body-powered, myoelectric, passive), and taking measurements or casts for the initial socket.

Figure 25: Application of a prosthetic shrinker to aid in stump maturation.
Phase 3: Prosthetic Fitting and Training (Week 8 onwards)
- Prosthetic Checkout: The initial test socket is fitted and evaluated for comfort, suspension, and alignment. Adjustments are made.
-
Basic Prosthetic Training:
- Donning and Doffing: Patients learn to apply and remove their prosthesis independently.
- Socket Tolerance: Gradually increasing wear time to build tolerance.
- Control Training: Learning to operate the terminal device (hook, hand) and, for AEA, the elbow joint. This involves either body power (cable control) or myoelectric signals (muscle contractions).
- Functional Use: Integrating the prosthesis into simple ADLs, starting with bilateral tasks and progressing to more complex unilateral tasks.
-
Advanced Prosthetic Training:
- Activity-Specific Training: Tailored training for vocational, recreational, and household tasks.
- Balance and Coordination: For both AEA and BEA, improving overall body balance and coordination, especially when using a heavier prosthesis.
- Problem-Solving: Learning to adapt to different environments and challenges with the prosthesis.
- Targeted Muscle Reinnervation (TMR) / Advanced Prosthetics: For patients who have undergone TMR, specific training protocols are implemented to maximize control of advanced myoelectric prostheses, allowing for more intuitive and multi-articulated movements.
-
Psychological Support:
Ongoing support to address body image, adaptation, and potential frustration with prosthetic use.
Figure 26: Patient undergoing prosthetic training, focusing on functional use of the terminal device.
Figure 27: Example of a fitted above-elbow prosthesis, demonstrating the aesthetic and functional outcome.
Long-Term Management
- Stump Health Monitoring: Regular skin checks, prosthetic adjustments, and addressing any signs of breakdown, irritation, or pain.
- Prosthetic Maintenance and Updates: Prostheses require regular maintenance and may be upgraded over time as technology advances or patient needs change.
- Lifelong Rehabilitation: Continuous engagement in physical activity, strengthening, and periodic re-evaluation by the rehabilitation team.
- Vocational Rehabilitation: Assisting patients in returning to work or finding new employment opportunities.
- Psychosocial Adjustment: Ongoing support to manage the psychological impact of limb loss and foster long-term adaptation.
Summary of Key Literature / Guidelines
The management of upper extremity amputations has evolved significantly, driven by advancements in surgical techniques, prosthetic technology, and rehabilitation science. Key principles are consistently emphasized across academic literature and clinical guidelines.
-
Multidisciplinary Team Approach: Consensus guidelines from organizations like the American Academy of Orthopaedic Surgeons (AAOS) and the Orthopaedic Trauma Association (OTA) underscore the necessity of a multidisciplinary team. This includes orthopedic surgeons (especially orthopedic oncologists for tumor cases), plastic surgeons, vascular surgeons, physiatrists, physical and occupational therapists, prosthetists, pain specialists, and psychologists. This collaborative model ensures comprehensive care from pre-operative planning through long-term rehabilitation.
-
Limb Salvage vs. Amputation Paradigm (Oncology): Modern orthopedic oncology prioritizes limb salvage whenever oncologically safe. The work by Enneking and others established the fundamental principles of wide surgical margins for sarcomas. However, for large, aggressive tumors with neurovascular involvement or failed previous treatments, amputation remains the gold standard for local disease control. Studies by Malawer et al. highlight the importance of careful patient selection and staging in this decision-making process. The use of neoadjuvant chemotherapy and radiation has significantly reduced amputation rates for many high-grade sarcomas.
-
Surgical Principles: The surgical execution of amputations is guided by established principles aimed at creating a functional, comfortable, and durable residual limb. The importance of myodesis and myoplasty for muscle stabilization, proprioception, and prosthetic control is emphasized in texts such as the AAOS Comprehensive Orthopaedic Review and surgical atlases. Nerve management to prevent painful neuromas, often involving tension-free proximal transection and burying in muscle, is a critical surgical detail consistently advocated in literature (e.g., specific reviews on neuroma prevention and treatment).
-
Prosthetic Advancement and Targeted Muscle Reinnervation (TMR): Recent decades have seen revolutionary advancements in prosthetic technology, particularly in the realm of myoelectric prostheses. TMR, pioneered by Dr. Todd Kuiken and colleagues, represents a significant surgical innovation. By transferring residual nerves to expendable motor points in the remaining muscle, TMR creates additional, more intuitive myoelectric control sites. Literature consistently demonstrates that TMR can improve functional outcomes, reduce phantom limb pain, and enhance the control of advanced multi-articulated prostheses for both above-elbow and shoulder disarticulation levels. This has led to the development of highly sophisticated bionic limbs.
-
Rehabilitation Guidelines: Rehabilitation protocols are well-established, emphasizing early intervention. The American Academy of Physical Medicine and Rehabilitation (AAPMR) and the American Physical Therapy Association (APTA) provide guidelines for progressive rehabilitation, including stump shaping, strengthening, ROM exercises, desensitization, and comprehensive prosthetic training. The shift towards early prosthetic fitting (within weeks of surgery) for stable, well-healed stumps is a recurring theme in modern rehabilitation literature, facilitating adaptation and improving functional integration.
-
Addressing Phantom Limb Pain: The high incidence of phantom limb pain has led to extensive research into its pathophysiology and management. Current guidelines suggest a multimodal approach, including pharmacotherapy, regional blocks, psychological interventions (CBT), mirror therapy, and in select cases, surgical options like TMR or neuromodulation.
In summary, above-elbow and below-elbow amputations, though less frequent than in the lower extremity, are complex procedures demanding a high level of surgical expertise and a comprehensive, multidisciplinary post-operative care pathway. The continuous evolution of oncologic management, surgical techniques, and prosthetic technology ensures that patients requiring these interventions can achieve optimal functional recovery and improved quality of life.
Clinical & Radiographic Imaging
You Might Also Like