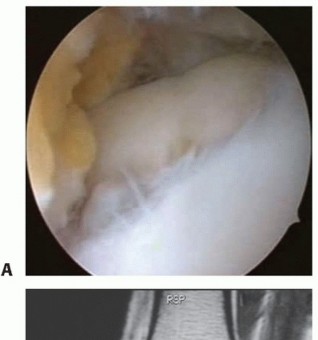
Articular Cartilage Injuries: Comprehensive Surgical Management
Key Takeaway
Articular cartilage injuries present a profound clinical challenge due to the tissue's avascular nature and limited intrinsic healing capacity. Management requires a meticulous, evidence-based approach, balancing patient demands with defect morphology. Surgical interventions range from palliative arthroscopic débridement to restorative techniques such as microfracture, osteochondral autograft transfer, and autologous chondrocyte implantation. Successful outcomes depend on precise patient selection, rigorous surgical technique, and strict adherence to postoperative rehabilitation protocols.
FUNDAMENTALS OF ARTICULAR CARTILAGE INJURIES
Articular cartilage is a highly specialized, complex connective tissue designed to withstand tremendous compressive and shear forces over millions of cycles throughout a human lifespan. Despite its remarkable biomechanical properties, it possesses a profound biological vulnerability: it is entirely avascular, aneural, and alymphatic. Consequently, it lacks the intrinsic ability to mount a meaningful healing response even after minor trauma. Over 250 years ago, the pioneering anatomist William Hunter astutely observed, “Ulcerated cartilage is a troublesome thing, once destroyed is not repaired.”
The overarching health, tribology, and kinematic function of any diarthrodial joint depend entirely on the viability of its articular cartilage. Without it, the inexorable progression toward osteoarthritis is guaranteed. In young, high-demand athletic populations, articular cartilage lesions are predominantly traumatic in etiology, often occurring in tandem with ligamentous instability. Conversely, in older populations, these changes represent the continuum of degenerative joint disease. Extensive knee arthroscopy registries demonstrate that articular cartilage lesions are incidentally or primarily identified in 60% to 66% of all patients undergoing knee arthroscopy.
Furthermore, acute articular cartilage lesions are frequently accompanied by significant hemarthrosis and occult subchondral trabecular microfractures (commonly referred to as bone bruises or bone blisters). Advanced imaging studies reveal that occult subchondral bone bruises are present in up to 80% of patients presenting with acute, complete ruptures of the anterior cruciate ligament (ACL).
Clinical Presentation and Evaluation
Patients harboring articular cartilage injuries typically present with a triad of symptoms: insidious or activity-related pain, recurrent effusion, and mechanical symptoms (catching, locking, or giving way).
- Femoral Condyle Lesions: Pain is generally localized to the medial or lateral tibiofemoral compartment. It is characteristically exacerbated by axial loading, weight-bearing, or high-impact activities.
- Patellofemoral Lesions: Patients report profound anterior knee pain aggravated by kneeling, stair climbing, descending stairs, and prolonged sitting with the knee flexed (the "theater sign").
Evaluation of a patient suspected of having a chondral or osteochondral lesion demands a rigorous, standardized radiographic protocol. This must include:
1. Weight-bearing anteroposterior (AP) views.
2. 45-degree flexion weight-bearing posteroanterior (PA) views (Rosenberg view) to assess the posterior functional weight-bearing zone.
3. True lateral views.
4. Patellar views (Merchant or sunrise) to evaluate the trochlear groove and patellar facets.
5. Bilateral standing hip-knee-ankle (long-leg) AP views to meticulously assess the mechanical axis.
Clinical Pearl: Any significant abnormality in the mechanical axis (varus or valgus malalignment) must be corrected (e.g., via high tibial osteotomy or distal femoral osteotomy) either before or concurrently with cartilage restoration procedures. Failure to address malalignment guarantees the mechanical failure of the cartilage repair.
Clinical suspicion should immediately prompt evaluation with high-resolution, cartilage-sensitive Magnetic Resonance Imaging (MRI) (e.g., T2 mapping, dGEMRIC) to accurately delineate lesion location, size, depth, and the integrity of the subchondral bone.
Classification Systems
Accurate classification is paramount for surgical decision-making. While MRI provides excellent preoperative mapping, definitive classification remains arthroscopic.
Classification of Articular Lesions by Configuration (Bauer and Jackson):
* Type I: Linear
* Type II: Stellate
* Type III: Flap
* Type IV: Crater
* Type V: Fibrillation
* Type VI: Degrading
Classification by Severity (ICRS / Modified Outerbridge):
* Grade 0: Normal, intact cartilage.
* Grade I: Softening and swelling (blistering) with an intact surface. Superficial fissures.
* Grade II: Fragmentation and fissures in an area less than 0.5 inches in diameter. Superficial ulceration less than 50% of the depth of the cartilage.
* Grade III: Deep ulceration, fibrillation, or chondral flaps extending more than 50% of the cartilage depth, but without exposed subchondral bone.
* Grade IV: Full-thickness wear with exposed subchondral bone.
NONOPERATIVE MANAGEMENT
Nonoperative management is the first line of treatment, particularly for low-demand patients or those with diffuse degenerative changes. The primary goal is to decrease the mechanical load on the joint. Modalities include:
* Load Modification: Weight loss and activity modification.
* Physical Therapy: Strengthening the dynamic stabilizers across the joint to absorb kinetic loads.
* Orthoses: Unloader braces to shift the mechanical axis away from the affected compartment.
* Pharmacotherapy: Analgesics, NSAIDs, and intra-articular injections (corticosteroids or hyaluronic acid).
OPERATIVE MANAGEMENT: SURGICAL TECHNIQUES
Operative intervention is indicated when nonoperative methods fail to relieve pain and mechanical symptoms. The choice of procedure is dictated by the lesion size, depth (chondral vs. osteochondral), patient age, and activity demands.
1. Arthroscopic Débridement and Lavage
Indications: Minimal symptoms, small lesions (< 2 cm) in low-demand patients.
Goal: Palliative relief by reducing joint inflammation and mechanical irritation.
The procedure involves the meticulous removal of loose, unstable chondral flaps that impinge within the joint during articulation. Débridement includes smoothing fibrillated surfaces to stable vertical margins, meniscal trimming, shaving impinging osteophytes, and resecting inflamed synovium. Copious joint lavage mechanically washes away cartilage fragments, inflammatory mediators, and calcium phosphate crystals. While this provides short-term palliative relief, it does not alter the natural history of the disease.
2. Marrow Stimulation (Microfracture)
Indications: Small lesions (< 2 cm), low-demand patients.
Goal: Reparative (Fibrocartilage formation).
Microfracture, pioneered by Steadman, relies on penetrating the avascular subchondral bone plate to access the highly vascularized cancellous bone marrow. This releases mesenchymal stem cells, growth factors, and a fibrin super-clot into the defect, stimulating an extrinsic reparative process.

Surgical Warning: The resulting repair tissue is Type I collagen-dominant fibrocartilage, which lacks the durability and resilience of native Type II hyaline cartilage. Clinical deterioration is often observed beginning 18 to 24 months postoperatively.
Five Critical Factors for Microfracture Success:
1. Preparation: The calcified cartilage layer must be meticulously removed using a ring curette to expose the subchondral bone plate, but aggressive abrasion that destroys the subchondral bone must be avoided.
2. Spacing: Using arthroscopic awls, penetrations should be made 3 to 4 mm deep, leaving a 1- to 2-mm bridge of intact bone between holes. This maintains the structural integrity of the subchondral plate while allowing marrow elements to fill the defect.
3. Rehabilitation: Joint function must be maintained postoperatively using early Continuous Passive Motion (CPM) to mold the regenerating tissue.
4. Weight-Bearing: Strict protected weight-bearing must be enforced for 6 to 8 weeks, depending on lesion location.
5. Alignment: Any mechanical axis abnormality must be corrected concurrently.
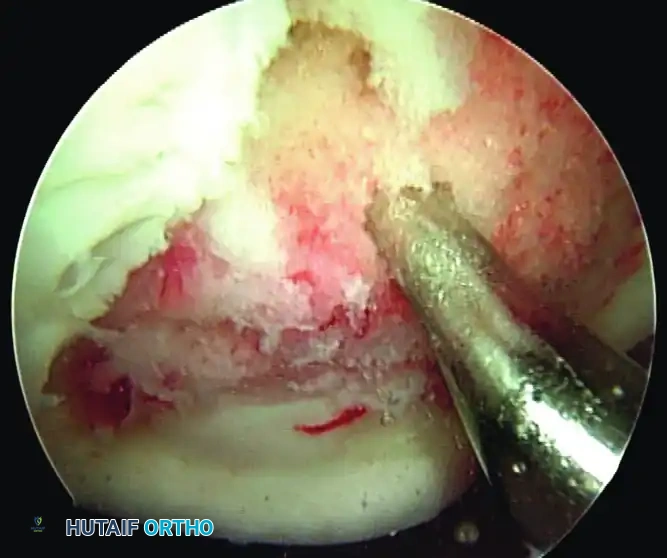
3. Osteochondral Autograft Transfer (OATS / Mosaicplasty)
Indications: Small to medium lesions (1.0 - 2.0 cm), high-demand patients.
Goal: Restorative (Hyaline cartilage replacement).
For focal, full-thickness defects, osteochondral autograft transfer provides immediate restoration of the articular surface with native, viable hyaline cartilage. Grafts are harvested from relatively non-weight-bearing regions of the knee (e.g., the periphery of the lateral femoral condyle or the intercondylar notch).
Surgical Technique:
The procedure can be performed arthroscopically or via a mini-arthrotomy. The recipient site is cored to a specific depth (typically 15 mm). A size-matched osteochondral plug is harvested perpendicularly from the donor site. The plug is then press-fit into the recipient socket. For larger defects, multiple smaller plugs can be used in a "mosaicplasty" configuration to recreate the complex contour of the condyle.
Pitfall: The graft must be inserted perfectly flush with the surrounding native cartilage. A proud graft will bear excessive load and fail, while a sunken graft will lead to poor integration and cyst formation.
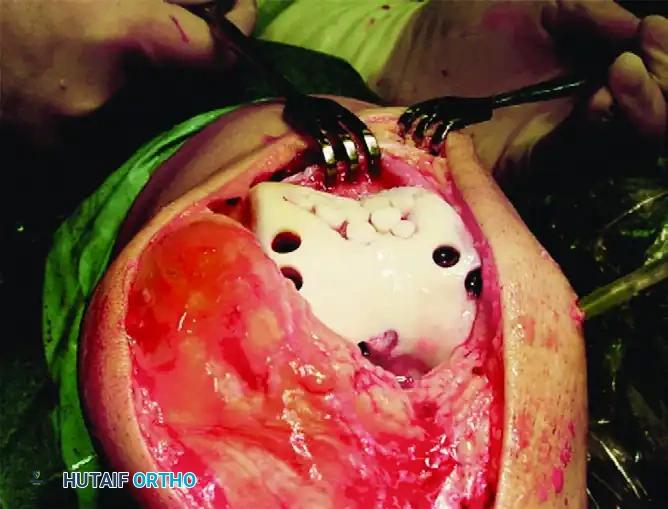
4. Osteochondral Allograft Transplantation
Indications: Large lesions (2.0 - 3.5+ cm), deep osteochondral defects, revision cases.
Goal: Restorative.
When lesions exceed 2 cm, or when there is significant subchondral bone loss, autograft harvesting causes unacceptable donor-site morbidity. In these scenarios, fresh osteoarticular size-matched allografts are indicated. Fresh allografts (used within 14 to 28 days of procurement) are critical, as they maintain chondrocyte viability of up to 80%.
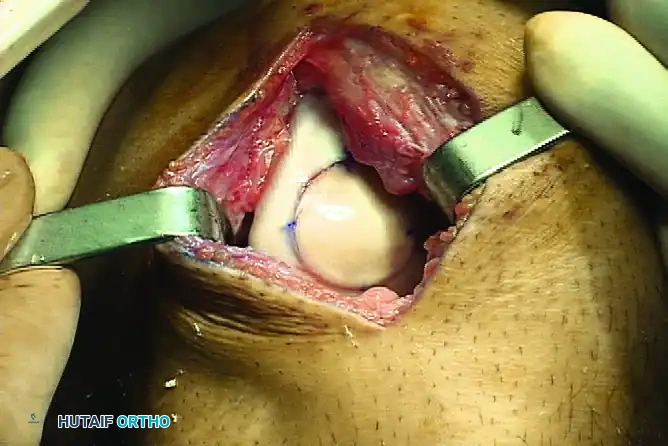
Surgical Technique:
1. Exposure and Preparation: A standard arthrotomy is performed to expose the defect. The lesion is sized, and a guide pin is placed centrally.
2. Recipient Coring: A cannulated reamer is used to core out the diseased cartilage and necrotic subchondral bone to a healthy, bleeding cancellous bed.
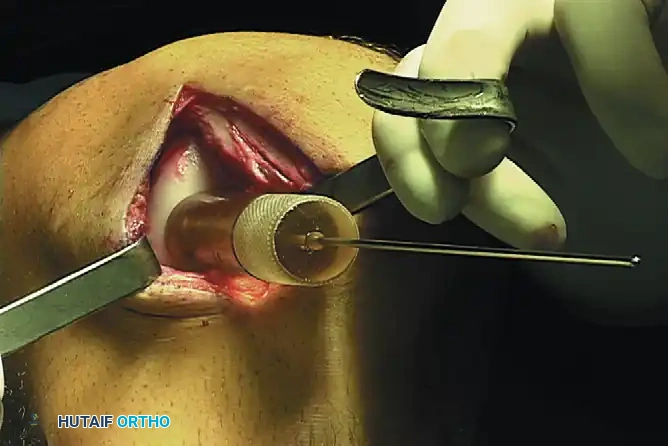
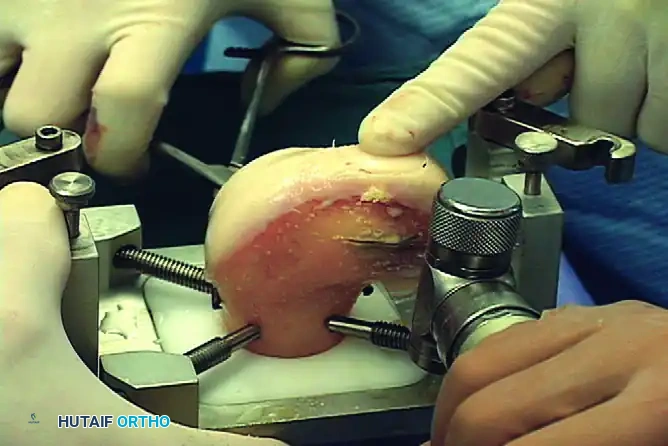
- Back-Table Graft Preparation: The fresh allograft hemicondyle is secured in a workstation. A matching donor plug is cored from the exact topographic location on the allograft to ensure matching radius of curvature.
- Implantation: The allograft plug is gently lavaged to remove marrow elements (reducing immunogenicity) and carefully press-fit into the recipient site. Supplemental fixation (e.g., bioabsorbable darts or headless compression screws) may be used if the press-fit is suboptimal.
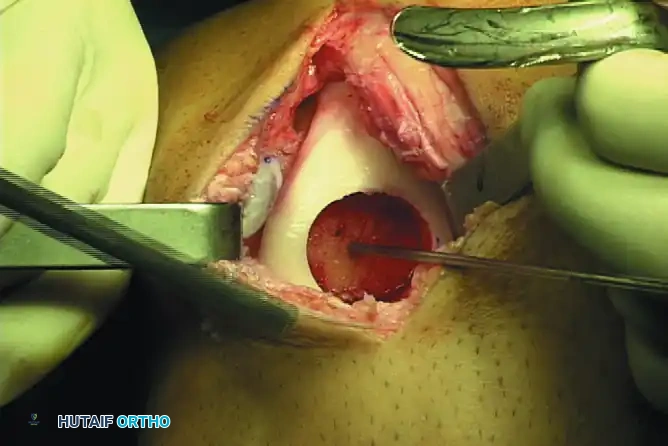
Clinical Pearl: The primary logistical disadvantage of fresh allografts is that patients must remain "on call" for immediate surgery once a size-matched, disease-cleared graft becomes available.
5. Autologous Chondrocyte Implantation (ACI) & MACI
Indications: Large lesions (2.0 - 10 cm), multiple lesions, high-demand patients.
Goal: Restorative.
ACI is a sophisticated, two-stage cell-based therapy designed to regenerate hyaline-like cartilage.
Stage 1: Biopsy
A minor arthroscopic procedure is performed to harvest 200 to 300 mg of healthy articular cartilage from a non-weight-bearing zone (e.g., the superior intercondylar notch). These cells are sent to a laboratory where they are enzymatically digested and expanded in vitro over 3 to 6 weeks, multiplying from a few hundred thousand to over 12 million cells.
Stage 2: Implantation
An open arthrotomy is performed. The defect is radically débrided to stable, vertical walls of healthy cartilage, taking care not to penetrate the subchondral bone (which would introduce bleeding and fibrocartilage-inducing stem cells).
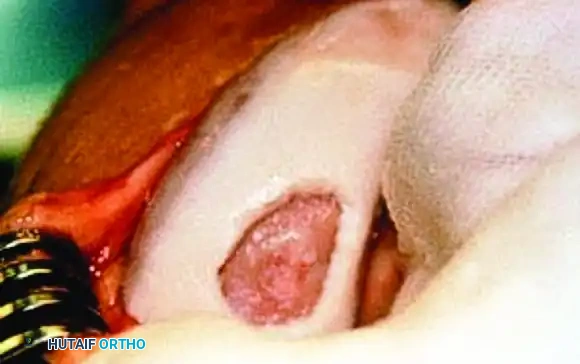
In first-generation ACI, a periosteal patch (harvested from the proximal medial tibia) is meticulously sutured over the defect using 6-0 Vicryl. The cambium layer of the periosteum must face the joint defect, as it contains chondrogenic precursors.
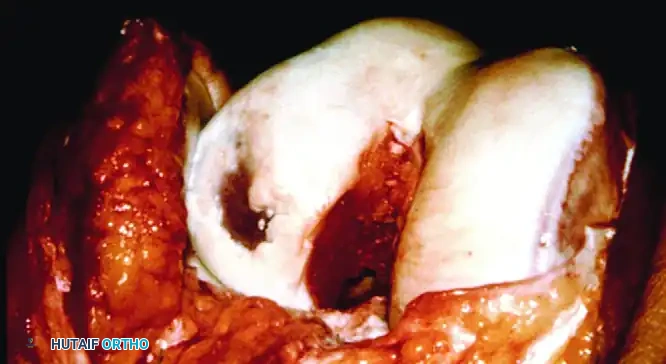
The suture line is sealed with fibrin glue to create a watertight compartment. The cultured chondrocytes are then injected beneath the patch, and the final opening is sealed.
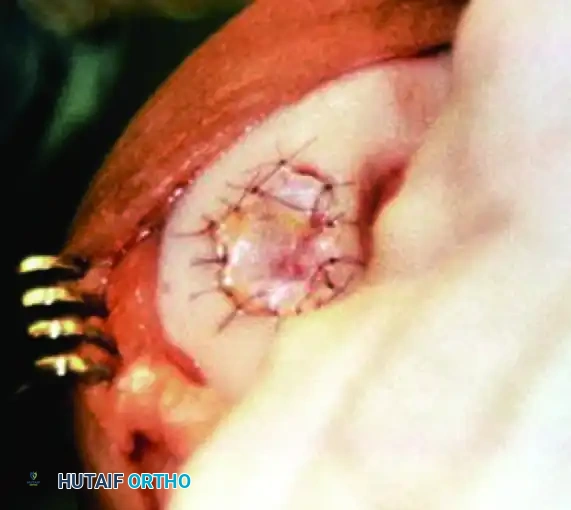
Advancements: MACI (Matrix-Induced ACI)
First-generation ACI faced complications such as periosteal hypertrophy (requiring secondary shaving) and uneven cell distribution. Second-generation techniques (MACI) seed the cultured chondrocytes directly onto a biodegradable Type I/III collagen or hyaluronan scaffold. Because these scaffolds are naturally adhesive, they can be cut to the exact template of the defect and secured with fibrin glue alone, eliminating the need for periosteal harvesting and complex suturing.
6. The "Sandwich" Technique for Deep Osteochondral Defects
Indications: Massive osteochondral defects with significant subchondral bone loss.
To avoid multiple staged bone-grafting procedures prior to ACI, the "sandwich" technique was developed for deep craters.
Surgical Technique:
1. The deep osseous void is packed with autologous cancellous bone graft.
2. A periosteal flap is sutured directly over the bone graft at the exact level of the native subchondral bone plate (cambium layer facing the joint).
3. A second periosteal membrane is sutured to the superficial rim of the articular cartilage defect (cambium layer facing down into the defect).
4. The cultured chondrocytes are injected into the space between the two periosteal layers.
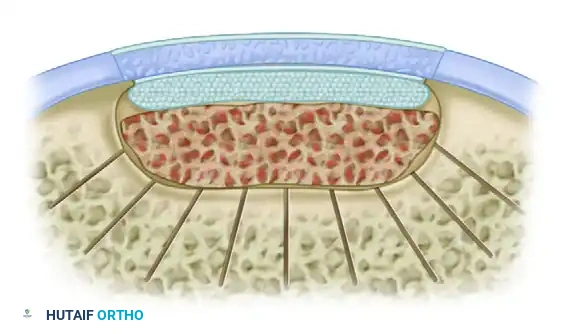
This highly complex, multi-layered reconstruction effectively reconstitutes both the structural osseous foundation and the biological articular surface in a single definitive procedure.
POSTOPERATIVE REHABILITATION PROTOCOLS
The success of any cartilage restoration procedure is inextricably linked to strict adherence to postoperative rehabilitation.
- Continuous Passive Motion (CPM): Initiated immediately in the recovery room. CPM stimulates chondrocyte metabolism, promotes the synthesis of Type II collagen, and prevents intra-articular adhesion formation. Patients typically use CPM for 6 to 8 hours daily for up to 6 weeks.
- Weight-Bearing Restrictions: For femoral condyle lesions, patients are restricted to toe-touch weight-bearing for 6 to 8 weeks to prevent mechanical shear forces from displacing the graft or disrupting the fragile regenerating tissue. Patellofemoral lesions may allow early weight-bearing in full extension (locked brace), as the patella does not engage the trochlea in extension.
- Return to Sport: Cartilage maturation is a slow biological process. Return to high-impact pivoting sports is generally prohibited for 12 to 18 months postoperatively, pending clinical evaluation and MRI confirmation of graft incorporation and tissue maturation.
You Might Also Like