Peripheral Nerve Decompression: Lateral Femoral Cutaneous and Common Peroneal Nerve Masterclass
Key Takeaway
This masterclass guides fellows through the intricate surgical decompression of the Lateral Femoral Cutaneous Nerve (LFCN) and Common Peroneal Nerve (CPN). We meticulously detail preoperative planning, precise patient positioning, comprehensive surgical anatomy, and granular intraoperative execution with real-time directives. Essential pearls, critical pitfalls, and robust postoperative management strategies are emphasized to optimize outcomes and prevent complications.
Introduction and Epidemiology
The management of peripheral nerve pathology represents a complex and technically demanding subdiscipline within orthopedic surgery. Care of peripheral nerve problems requires a profound knowledge and understanding of nerve pathology, anatomic nerve variations, patterns of nerve damage, and entrapment syndromes that follow both high-energy trauma and common operative procedures. Unlike other surgical disciplines where primary intervention is the norm, a large proportion of peripheral nerve surgery in the lower extremity attempts to correct neuropathy in the postoperative patient and, therefore, is reoperative and reconstructive in nature.

Lateral femoral cutaneous nerve (LFCN) neuropathy, clinically recognized as meralgia paresthetica, is frequently encountered in the orthopedic patient following injuries or procedures in proximity to the anterior superior iliac spine (ASIS), the inguinal region, or the anterior thigh. Iatrogenic injury to the LFCN is a known complication of the anterior approach to the hip, pelvic osteotomies, and iliac crest bone grafting. Because this nerve carries only sensory signals, the symptoms are strictly limited to pain, dysesthesias, or paresthesias in the anterolateral thigh.

Conversely, the common peroneal nerve (CPN) is highly susceptible to injury secondary to surgical procedures and trauma involving the lateral knee. The CPN can become entrapped in postoperative scar tissue, subjected to severe traction during multiligamentous knee injuries or knee dislocations, or inadvertently directly damaged during proximal fibular osteotomies, lateral meniscus repairs, or total knee arthroplasty. The resulting neuropathy typically presents as foot drop, a devastating complication that severely impairs patient mobility and quality of life.
Surgical Anatomy and Biomechanics
Lateral Femoral Cutaneous Nerve Anatomy
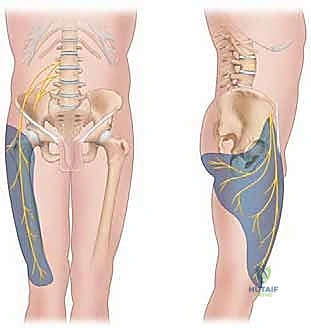
The LFCN arises from the lumbar plexus through contributions from the dorsal divisions of the L2 and L3 spinal roots. It emerges from the lateral border of the psoas major muscle, crosses the iliacus muscle obliquely toward the ASIS, and provides strictly sensory innervation to the anterolateral thigh.

In the majority of the population, the nerve courses medial to the ASIS and traverses the groin crease under the inguinal ligament as it descends to innervate the thigh. However, the nerve is highly prone to iatrogenic injury when its anatomy is aberrant. Surgeons must maintain a high index of suspicion for anatomic variations. The LFCN can run directly through the tendinous fibers of the inguinal ligament, rest directly against the ASIS, or course over the most medial portion of the ASIS rather than maintaining its usual medial trajectory.

Common Peroneal Nerve Anatomy
The CPN is the smaller terminal branch of the sciatic nerve, formed from contributions from the posterior divisions of the sacral plexus (L4 to S2). It diverges from the tibial nerve in the distal third of the posterior thigh, coursing laterally along the medial border of the biceps femoris.

Pathology of the CPN is classically observed as it wraps around the neck of the fibula. At this level, the nerve is superficial, tethered, and directly overlies bone, making it exceptionally vulnerable to direct compression and traction. It passes deep to the fibrous edge of the peroneus longus muscle—a common site of entrapment—just before it bifurcates into its deep and superficial branches.

The CPN provides crucial motor innervation: the deep branch innervates the anterior compartment of the leg (tibialis anterior, extensor hallucis longus, extensor digitorum longus, peroneus tertius) for foot dorsiflexion, while the superficial branch innervates the lateral compartment (peroneus longus and brevis) for foot eversion. Sensory innervation is supplied to the anterolateral lower leg and the majority of the dorsum of the foot and toes.
General Peripheral Nerve Vascularity and Biomechanics
The peripheral nerve possesses a highly specialized and significant intrinsic blood supply that permits the surgeon to safely lift the nerve from its anatomic bed, open the epineurium, and operate between the individual fascicles.

The endoneurial and perineurial microvessels maintain excellent vascularity to the peripheral nerve. Segmental blood vessels enter the peripheral nerve through the mesoneurium, analogous to the mesentery of the bowel. In addition, an extensive network of longitudinal vessels within the epineurium, perineurium, and endoneurium supplies the nerve tissue.
Classic biomechanical and microvascular studies by Maki et al. have demonstrated that the safe length a nerve can be elevated from its bed—relying solely on its internal longitudinal vascular supply rather than segmental vessels—is a distance of approximately 60 times the diameter of the nerve. Therefore, during neurolysis or transposition, the surgeon must balance the need to mobilize the nerve into a healthy, unscarred tissue bed against the risk of devascularization. The primary goal is to remove external compressive forces while acknowledging and preserving internal microvascular perfusion.
Indications and Contraindications
The decision to proceed with operative intervention for LFCN or CPN neuropathy depends on the mechanism of injury, the duration of symptoms, the progression of neurologic deficits, and the response to conservative management.

Open injuries with suspected nerve transection (neurotmesis) warrant immediate or early surgical exploration. Closed injuries, such as traction neurapraxia or axonotmesis following a knee dislocation, are typically observed for 3 to 6 months to allow for spontaneous regeneration. Failure to demonstrate clinical or electrodiagnostic improvement within this window is a strong indication for surgical exploration, neurolysis, or nerve reconstruction.
Operative vs Non Operative Management
| Clinical Scenario | Management Strategy | Rationale and Timing |
|---|---|---|
| Closed Traction Injury (CPN) | Non-Operative initially | Observe for 3-6 months. Serial EMG/NCS at 6 and 12 weeks to assess for early reinnervation potentials. |
| Open Laceration with Deficit | Operative (Immediate) | High probability of neurotmesis. Requires primary end-to-end repair or interposition grafting. |
| Progressive Motor Deficit | Operative (Urgent) | Suggests expanding hematoma, compartment syndrome, or progressive entrapment requiring immediate decompression. |
| Meralgia Paresthetica | Non-Operative initially | NSAIDs, weight loss, avoidance of tight clothing, corticosteroid/local anesthetic injections. |
| Refractory LFCN Neuropathy | Operative (Elective) | Failure of >6 months of conservative care. Options include neurolysis or neurectomy depending on nerve viability. |
| Neuroma in Continuity | Operative (Elective) | Resection and grafting if no distal conduction is present; neurolysis if partial conduction remains. |
Pre Operative Planning and Patient Positioning
Thorough preoperative planning is paramount for successful outcomes in peripheral nerve surgery. A detailed clinical examination must map the exact sensory deficit and grade motor function using the Medical Research Council (MRC) scale. The presence and progression of a Tinel's sign can help track the advancing front of regenerating axons, which typically grow at a rate of 1 mm per day.

Electrodiagnostic and Imaging Modalities
Electromyography (EMG) and Nerve Conduction Studies (NCS) are the gold standard for evaluating nerve integrity. However, timing is critical. Wallerian degeneration takes approximately 3 weeks to manifest as fibrillation potentials and positive sharp waves on EMG. Therefore, early EMG may yield false-negative results regarding the severity of axonal loss. Follow-up studies at 3 months are crucial to detect nascent motor unit action potentials (MUAPs), which indicate early reinnervation before clinical motor recovery is apparent.
High-resolution ultrasound (HRUS) and Magnetic Resonance Neurography (MRN) have revolutionized preoperative planning. These modalities allow the surgeon to visualize neuromas, partial transections, scar tethering, and anatomic variations prior to incision. Ultrasound is particularly useful for dynamically assessing the LFCN as it crosses the inguinal ligament.

Patient Positioning and Preparation
For LFCN exploration, the patient is positioned supine with a bump under the ipsilateral hip to elevate the pelvis. The entire anterior thigh and groin are prepped and draped to allow for proximal tracing of the nerve into the retroperitoneum if necessary.
For CPN exploration, patient positioning depends on the planned procedure. A supine position with a bump under the ipsilateral hip allows access to the lateral knee, while a lateral decubitus position provides excellent exposure of the popliteal fossa and the proximal course of the CPN. A sterile tourniquet is applied to the proximal thigh to provide a bloodless surgical field during the initial dissection, though it should be deflated prior to nerve stimulation and final hemostasis.
Detailed Surgical Approach and Technique
Lateral Femoral Cutaneous Nerve Decompression and Neurectomy
The surface anatomy and the most common site of impingement for the LFCN are crucial landmarks for the surgical approach.

An oblique incision is made approximately 2 cm medial and inferior to the ASIS, following the Langer lines of the groin crease. The subcutaneous tissue is carefully dissected. The LFCN is typically found deep to the superficial fascia but superficial to the fascia lata, just distal to the inguinal ligament.
Once identified, the nerve is traced proximally to its intersection with the inguinal ligament. If the nerve is compressed but appears structurally intact without significant neuroma formation, a neurolysis is performed. This involves dividing the overlying fascia and a portion of the inguinal ligament to ensure the nerve is completely free of tethering throughout its course.
If the nerve is severely damaged, encased in dense scar, or presents with a painful neuroma, a neurectomy is often preferred over neurolysis. The nerve is sharply transected well proximal to the zone of injury, ideally allowing the proximal stump to retract deep into the retroperitoneal space, away from the surgical scar and joint motion, thereby minimizing the risk of recurrent neuroma pain.
Common Peroneal Nerve Decompression and Reconstruction
Surgical exposure of the CPN requires a meticulous understanding of the lateral knee anatomy.

A curvilinear incision is made starting posterior to the biceps femoris tendon, extending distally across the fibular neck, and curving anteriorly over the anterior compartment of the leg. The deep fascia is incised, and the CPN is identified proximal to the fibular head, where it lies adjacent to the biceps femoris.
The nerve is then traced distally. The most critical step in CPN decompression is the release of the sharp fascial edge of the peroneus longus muscle. The muscle fibers are elevated off the fibula, and the fibrous arch is divided completely. The surgeon must follow the nerve until it bifurcates into the deep and superficial branches, ensuring both divisions are entirely free from compression.

If a neuroma-in-continuity is encountered, intraoperative nerve stimulation is utilized. If stimulation across the neuroma yields distal muscle contraction, an external neurolysis (and potentially internal epineurotomy) is performed, preserving the intact fascicles. If there is no conduction, the neuroma must be resected back to healthy, bleeding fascicular tissue.
For segmental defects following neuroma resection or trauma, tension-free repair is mandatory. Given the poor outcomes of primary repair under tension, interposition nerve grafting is frequently required. Autologous sural nerve grafts remain the gold standard. The grafts are reversed to prevent axonal escape down side branches and are sutured into place using 8-0 or 9-0 non-absorbable monofilament sutures under microscopic magnification. Fibrin glue may be used to augment the repair.
Complications and Management
Peripheral nerve surgery carries specific risks, and complications can profoundly impact patient outcomes. The reoperative nature of many of these procedures inherently increases the risk of complications such as infection, hematoma, and failure to improve.

The most challenging complication is the formation of a painful terminal neuroma following nerve transection or failed repair. Neuromas occur when regenerating axons escape the epineurial boundary and form a disorganized mass of nerve tissue and scar. Management requires surgical excision of the neuroma and relocation of the nerve stump into a protected environment, such as burying it deep within an adjacent muscle belly or utilizing advanced techniques like Targeted Muscle Reinnervation (TMR) or Regenerative Peripheral Nerve Interfaces (RPNI).
Another significant complication is the failure of motor recovery following CPN repair. Due to the distance regenerating axons must travel from the knee to the anterior compartment musculature, target muscle atrophy and motor endplate degradation may occur before reinnervation is achieved.
Common Complications and Salvage Strategies
| Complication | Incidence | Prevention and Salvage Strategies |
|---|---|---|
| Recurrent Neuroma (LFCN) | 10-15% | Prevention: Proximal transection and retroperitoneal burying. Salvage: Revision neurectomy, TMR, or RPNI. |
| Failure of Motor Recovery (CPN) | 30-50% (Grafts >6cm) | Prevention: Early intervention before endplate loss; tension-free grafting. Salvage: Tendon transfer (e.g., Posterior Tibial Tendon to dorsiflexors) or static AFO. |
| Postoperative Hematoma | 2-5% | Prevention: Meticulous hemostasis, deflation of tourniquet prior to closure, use of closed suction drains. Salvage: Immediate surgical evacuation to prevent compressive neuropathy. |
| Infection | 1-3% | Prevention: Strict sterile technique, prophylactic antibiotics. Salvage: Operative debridement, targeted antimicrobial therapy. |
| Complex Regional Pain Syndrome | <5% | Prevention: Gentle handling of nerve tissue, early postoperative mobilization. Salvage: Multidisciplinary pain management, sympathetic blocks, gabapentinoids. |
Post Operative Rehabilitation Protocols
The postoperative rehabilitation phase is as critical as the surgical intervention itself. The protocol must be tailored to the specific procedure performed—whether a simple decompression, a primary repair, or a complex nerve graft.

For isolated nerve decompressions (e.g., LFCN neurolysis or CPN release), early mobilization is encouraged to prevent the nerve from adhering to the newly formed surgical scar. Patients are typically allowed weight-bearing as tolerated, and physical therapy focuses on maintaining joint range of motion and nerve gliding exercises.
In cases of nerve repair or grafting, the surgical site must be protected from tension. The affected joint (e.g., the knee for CPN repair) is often immobilized in a slightly flexed position for 3 to 4 weeks to allow the microvascular anastomosis and epineurial healing to consolidate. Following this period of immobilization, a graduated range of motion protocol is initiated.
For patients with persistent foot drop during the prolonged recovery phase of a CPN injury, a custom dynamic Ankle-Foot Orthosis (AFO) is essential. The AFO prevents equinus contracture of the Achilles tendon, facilitates a normal heel-strike during the gait cycle, and prevents tripping hazards.
Sensory re-education and desensitization therapies are integral components of the rehabilitation process, particularly for LFCN injuries or as sensory function begins to return to the anterolateral leg following CPN repair. Therapists utilize varying textures, temperatures, and pressures to help the central nervous system recalibrate to the altered afferent input, thereby reducing neuropathic pain and hypersensitivity.
Summary of Key Literature and Guidelines
The foundational principles of peripheral nerve surgery are heavily reliant on the classic classifications of nerve injury described by Seddon and Sunderland. Seddon's classification (Neurapraxia, Axonotmesis, Neurotmesis) and Sunderland's five-degree system provide the framework for predicting spontaneous recovery and determining the necessity of surgical intervention.
The microvascular anatomy detailed by Maki et al. remains a cornerstone of modern neurolysis techniques, establishing the biomechanical limits of nerve mobilization based on the 60-times-diameter rule for preserving intrinsic blood supply.
Recent systematic reviews and clinical guidelines emphasize the critical nature of timing in CPN injuries. Literature demonstrates that primary repair or grafting performed within 3 to 6 months of injury yields significantly superior motor recovery compared to delayed interventions. Furthermore, studies evaluating interposition grafting for CPN defects indicate that outcomes precipitously decline when graft lengths exceed 6 centimeters, largely due to the prolonged time required for axonal regeneration and the subsequent irreversible atrophy of distal motor endplates. In such cases, early consideration for concurrent tendon transfer procedures is increasingly advocated in modern orthopedic literature to ensure functional restoration of foot dorsiflexion.
Clinical & Radiographic Imaging















You Might Also Like

