Orthopaedic Management of Hemophilic Arthropathy
Key Takeaway
The orthopaedic management of hemophilia requires a multidisciplinary approach, balancing meticulous surgical technique with precise hematological control. Elective procedures, including arthroscopic synovectomy and total joint arthroplasty, are now viable due to advanced factor concentrates. Success depends on strict preoperative factor assays, inhibitor screening, and specialized hemostatic protocols to minimize catastrophic bleeding, infection, and progressive joint destruction in hemophilic arthropathy.
Pathophysiology and Clinical Presentation of Hemophilic Arthropathy
Elective orthopaedic surgery for patients with classic hemophilia (factor VIII deficiency, Hemophilia A) and Christmas disease (factor IX deficiency, Hemophilia B) has evolved from a historically prohibitive risk to a highly successful, routine practice. This paradigm shift is entirely attributable to the modern availability of recombinant and plasma-derived factor VIII and factor IX concentrates. Prior to these advancements, surgical intervention was strictly reserved for lifesaving measures, carrying an unacceptably high mortality rate characterized by catastrophic wound hematomas, massive tissue sloughing, and overwhelming sepsis.
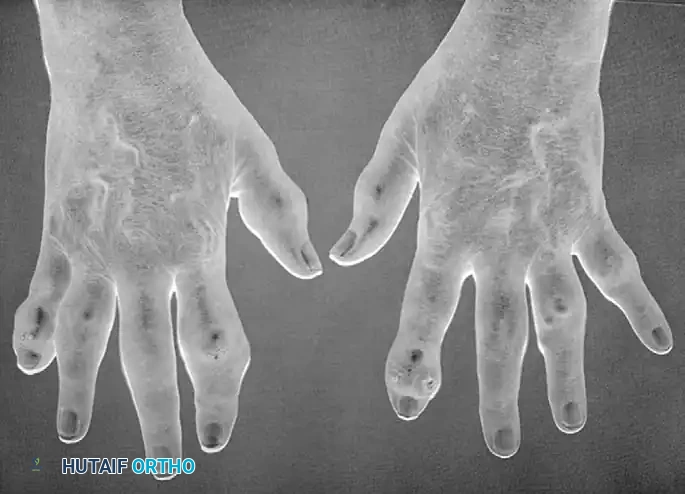
The hallmark of hemophilia in the musculoskeletal system is recurrent hemarthrosis, most frequently affecting the hinge joints: the knee, elbow, and ankle. The pathogenesis of hemophilic arthropathy begins with intra-articular bleeding, which triggers a profound inflammatory response. Hemosiderin-laden macrophages infiltrate the synovium, leading to chronic, progressive hypertrophic villous synovitis. This hypervascular synovial tissue becomes highly friable, creating a vicious cycle of recurrent bleeding and further synovial hypertrophy.
Over time, the hypertrophic synovium releases catabolic cytokines (such as IL-1 and TNF-alpha) and proteolytic enzymes that directly degrade articular cartilage. Concurrently, subchondral bone is compromised, leading to cyst formation, epiphyseal overgrowth in children, and ultimately, end-stage degenerative joint disease.
Nonoperative Management and Prophylaxis
Before considering surgical intervention, aggressive nonoperative management is mandatory. The primary goal is the prevention of hemarthrosis and the preservation of joint mechanics.
Hematological Prophylaxis
The current gold standard, as recommended by the National Hemophilia Foundation, is prophylactic factor replacement therapy. Nilsson et al. demonstrated that administering factor prophylactically from age 1 or 2 years through adolescence—maintaining factor VIII concentrations above 1% of normal—effectively prevents severe hemophilic arthropathy, limiting joint damage to minor defects.
Home therapy, allowing self-administration of factor VIII or IX at the earliest onset of periarticular stiffness or pain, has drastically reduced the incidence of end-stage degenerative arthritis. However, daily prophylactic intravenous administration often necessitates a central venous line, which carries a significant risk of contamination, thrombosis, and systemic infection.
Orthotic and Physical Therapy
Mechanical protection and rehabilitation are as critical as hematological control. Modalities include:
* Weight-relieving calipers and wedging casts: Utilized to offload compromised joints and correct early deformities.
* Spring-loaded braces (e.g., Dynasplint): Highly effective in applying low-load, prolonged stretch to soft-tissue contractures, particularly knee flexion contractures.
* Targeted Physical Therapy: Essential for maintaining muscle mass, proprioception, and joint range of motion (ROM).
Clinical Pearl: Conservative measures, combining orthotic stretching and aggressive physical therapy under factor coverage, may be as important as hematological management in avoiding the need for major reconstructive surgery.
Multidisciplinary Preoperative Optimization
Surgery in patients with hemophilia must never be undertaken casually. Catastrophic complications are minimized only through expert, multidisciplinary management and strict control of the clotting cascade.
The Role of the Hematologist
Successful surgery depends on an inseparable working relationship between the orthopaedic surgeon and an experienced hematologist. The hemorrhagic disorder must be accurately diagnosed and quantified before any surgical contemplation.
- Factor Assays and Inhibitor Screening: Correct replacement cannot occur without precise identification of the missing factor. Crucially, the surgical team must determine within days of the operation whether the patient has developed an alloantibody inhibitor against their deficient factor. The presence of a high-titer inhibitor severely complicates hematological therapy and often contraindicates elective or semi-elective surgery.
- Intraoperative and Postoperative Monitoring: A factor assay must be obtained at the time of surgery to ensure 100% correction. The hematocrit should be monitored for several days postoperatively, especially in patients with blood groups A, B, and AB, as a Coombs-positive hemolytic anemia may develop secondary to large volumes of factor concentrate.
- Infectious Disease Status: The patient’s HIV and Hepatitis C (HCV) status must be thoroughly investigated. In seropositive patients, T-lymphocyte (CD4) counts and viral loads must be optimized to assess the physiological ability to heal and the inherent risk of postoperative infection.
General Surgical Principles in Hemophilia
Post and Telfer outlined meticulous surgical techniques and preoperative evaluations that remain the bedrock of hemophilia surgery. When coagulation is strictly controlled, wound sloughing and infection are rare, resulting in profound pain relief and a cessation of recurrent bleeding.
The Post and Telfer Criteria:
- Consolidated Procedures: Perform as many procedures as the patient can tolerate in a single surgical session. This minimizes the number of times the patient is exposed to bleeding risks, reduces the astronomical cost of factor concentrates, and lowers the risk of inducing an inhibitor.
- Meticulous Asepsis and Tourniquet Use: Strict aseptic technique is non-negotiable. Pneumatic tourniquets should be used whenever anatomically possible to maintain a bloodless field.
- Avoidance of Electrocautery: Extensive use of electrocautery is contraindicated. Coagulated, charred tissues have a high tendency to slough postoperatively, creating a nidus for deep infection and secondary hemorrhage.
- Dead Space Management: Wound closure must be tight and meticulous to obliterate all dead space.
- Deep Wound Suction: Closed suction drains should be utilized in deep wounds for a minimum of 24 hours to prevent hematoma formation.
- Pharmacological Restrictions: Absolutely no aspirin or NSAIDs that inhibit platelet function should be administered postoperatively. Furthermore, intramuscular injections for pain relief are strictly forbidden due to the risk of muscle hematomas.
Indications for Surgical Intervention
The landscape of hemophilia surgery has shifted: there is a general decrease in the absolute need for surgery, an increase in the age of patients requiring intervention, and a transition toward joint-preserving and arthroscopic techniques. Current indications include:
- Chronic Hypertrophic Synovitis: Progressive synovial enlargement from repeated hemarthrosis that fails to respond to adequate factor replacement. Synovectomy is ideally performed before articular cartilage is significantly thinned.
- Severe Soft-Tissue Contractures: Contractures unresponsive to conservative measures (e.g., severe knee flexion contractures where serial casting causes posterior tibial subluxation).
- Severe Bony Deformity: Requiring corrective osteotomy.
- Expanding Pseudotumors: Encapsulated, expanding hematomas that destroy adjacent bone and soft tissue, continuing to enlarge despite factor replacement and radiation therapy.
- Useless or Chronically Infected Extremities: Necessitating amputation.
- End-Stage Hemophilic Arthropathy: Severe arthritic changes with incapacitating pain and hemorrhage, indicating total joint arthroplasty.
Management of Specific Pathologies
1. Hemophilic Pseudotumor and Osteochondritis Dissecans
Hemophilic pseudotumors are rare but limb-threatening complications caused by recurrent bleeding into muscles or subperiosteal spaces, leading to pressure necrosis of adjacent bone. In the pediatric hip, recurrent bleeding can compromise the vascular supply to the femoral head, leading to changes indistinguishable from Legg-Calvé-Perthes disease or severe osteochondritis dissecans.

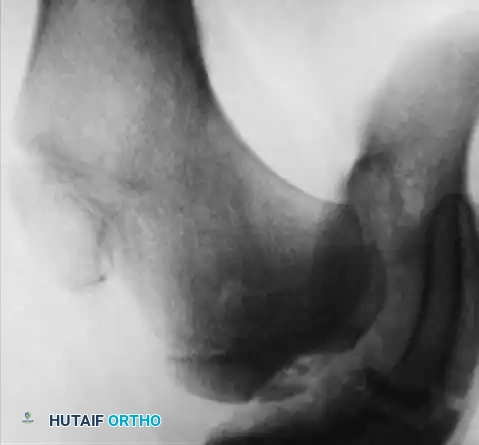
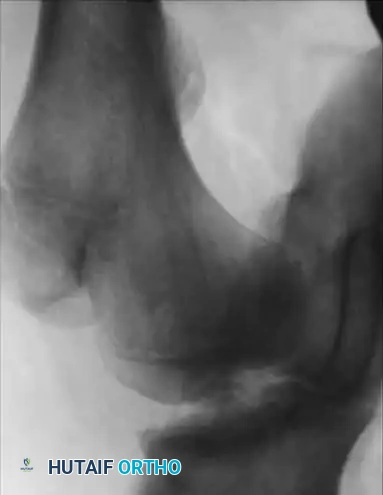
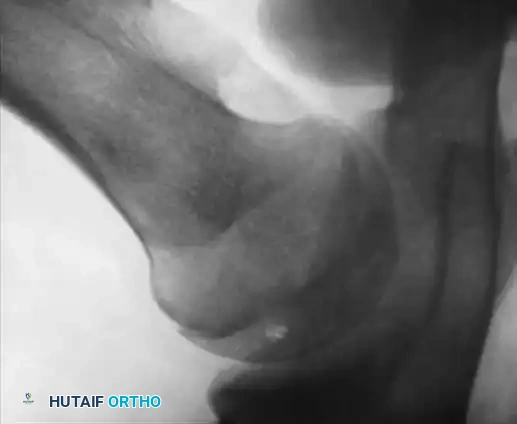
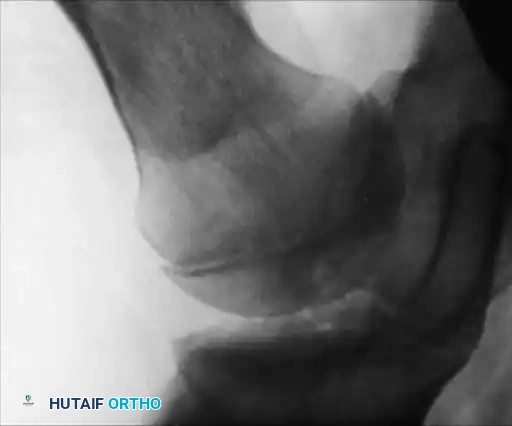
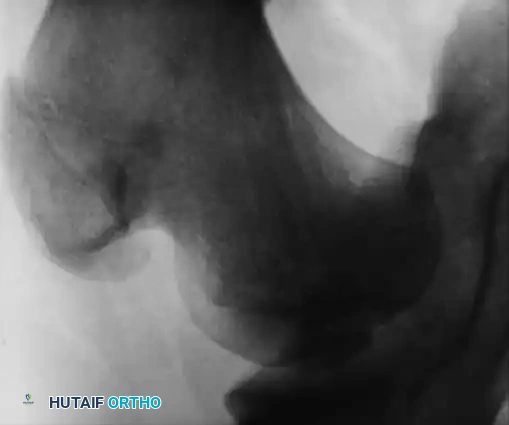
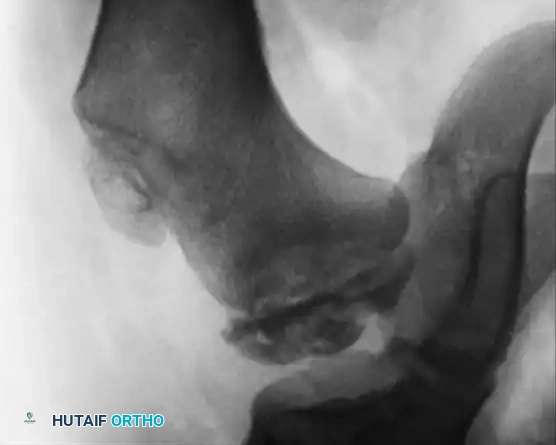
Fig. 29-34: Progression of osteochondritis dissecans of the hip in a hemophilic patient. Note the onset of Perthes-like disease, fragmentation, reossification, and eventual healing over an 8-year period, demonstrating the profound impact of intra-articular pressure and vascular compromise on the developing femoral head.
2. Soft-Tissue Contractures and Osteotomies
Knee flexion contractures are notoriously difficult to manage. If conservative measures fail, surgical release is indicated. For contractures less than 45 degrees, Rodriguez-Merchan et al. reported excellent results with hamstring release and posterior transverse capsulotomy.
If the contracture exceeds 50 to 60 degrees, soft-tissue release alone risks excessive traction on the popliteal neurovascular bundle. In these cases, a supracondylar extension osteotomy of the femur is beneficial, provided the patient retains 70 to 80 degrees of functional knee motion. Osteotomies for severe contractures should ideally be staged and performed after physeal closure to prevent growth arrest.
3. Synovectomy: Open, Arthroscopic, and Radionuclide
Synovectomy is highly effective at decreasing pain and the frequency of bleeding episodes; however, it does not halt the radiographic progression of joint destruction.
Arthroscopic vs. Open Synovectomy:
Triantafyllou et al. compared open and arthroscopic knee synovectomies. Both significantly reduced hemarthrosis. However, the open group experienced decreased or unchanged ROM in 75% of cases, with 62% requiring postoperative manipulation under anesthesia. Conversely, the arthroscopic group saw increased ROM in 80% of cases, required no manipulations, and experienced shorter hospitalizations and lower factor replacement costs.
Surgical Warning: While arthroscopic synovectomy is a low-risk procedure that effectively reduces recurrent hemarthrosis, Eickhoff and Wiedel noted that joint deterioration continues, albeit at a slower rate. Patients must be counseled that synovectomy is a palliative, joint-prolonging procedure, not a cure for arthropathy.
Joint-Specific Synovectomy:
* The Elbow: The second most frequent site of hemorrhage. Synovectomy combined with radial head excision provides excellent pain relief and frequently increases forearm rotation, though flexion/extension arcs rarely improve.
* The Ankle: Hypertrophic synovitis causes boggy swelling and impingement. Greene initially recommended open synovectomy due to the difficulty of accessing the posterior crypts of the malleoli. However, modern techniques by Patti, Mayo, and Dunn et al. have proven that arthroscopic ankle synovectomy is highly effective, yielding an 84% reduction in bleeding with stable or improved motion.
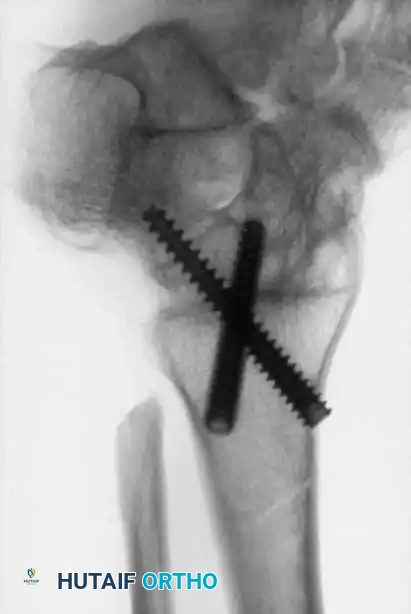
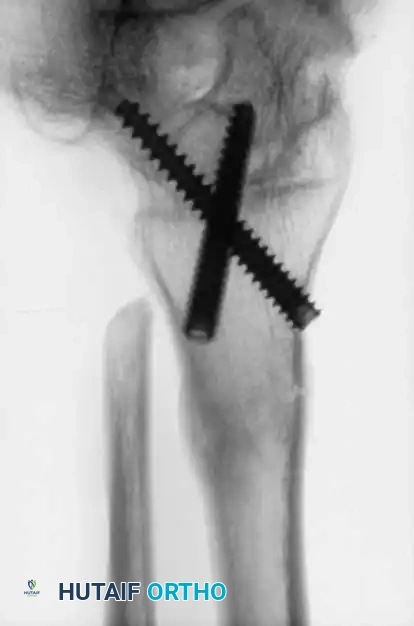
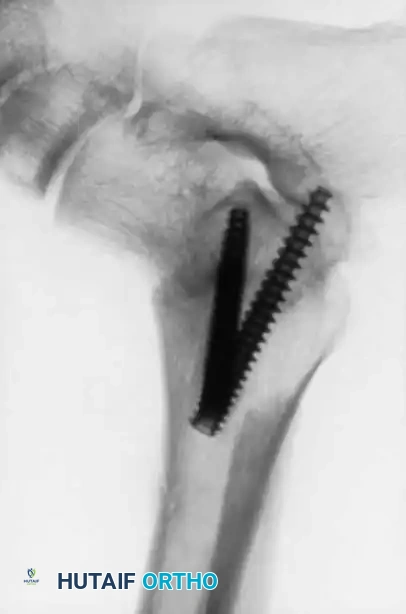
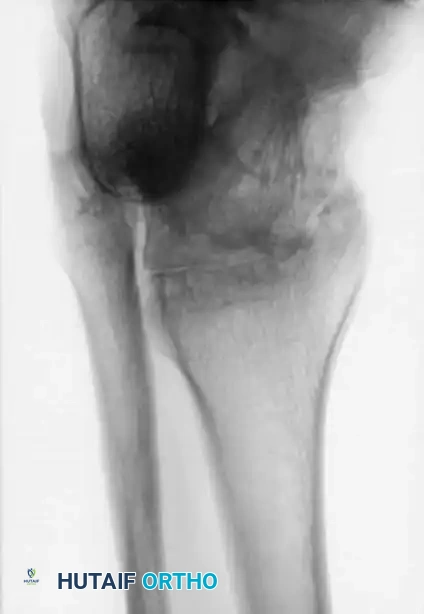
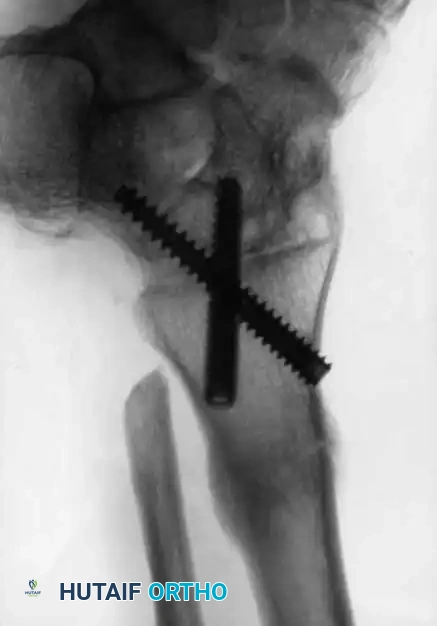
Advanced hemophilic arthropathy of the ankle often requires aggressive intervention. When arthroscopic synovectomy fails to control symptoms in end-stage disease, definitive stabilization via ankle arthrodesis utilizing rigid internal fixation becomes necessary to eliminate pain and prevent further hemorrhagic episodes.
Radionuclide Synovectomy (Synoviorthesis):
The intra-articular injection of a radioactive agent (e.g., Phosphorus-32 or Yttrium-90) to ablate synovial tissue has produced encouraging results, particularly in patients with high-titer inhibitors where surgical synovectomy is contraindicated.
4. Total Joint Arthroplasty (TJA)
When joint destruction reaches end-stage, total joint arthroplasty provides profound pain relief and functional restoration. McCollough et al. demonstrated that TJA is highly cost-effective; the postoperative reduction in required factor concentrate rapidly offsets the cost of the surgery.
Total Knee Arthroplasty (TKA):
TKA is indicated for advanced degenerative arthritis with incapacitating pain.
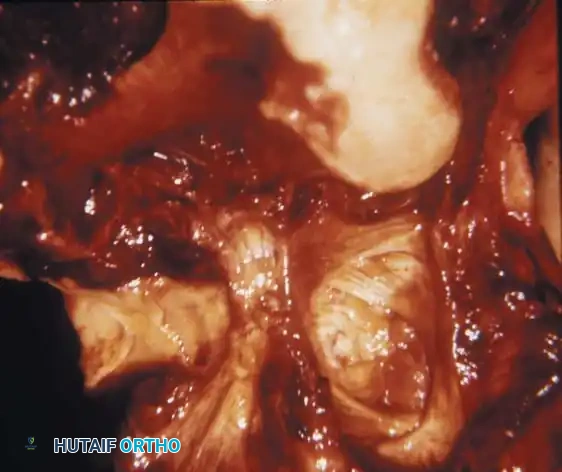
Fig. 29-35: Damaged knee joint with hemophilia (factor VIII deficiency). Note the marked destruction and erosion of the articular surface of the femoral condyle, gross erosion of the tibial plateaus, and complete destruction of the menisci by invasive hypertrophic synovium.
Preoperative planning is critical. TKA is unlikely to significantly increase the preoperative arc of motion; therefore, it should only be performed if the existing ROM is functional. Any flexion contracture greater than 30 degrees must be carefully evaluated and addressed during the procedure through aggressive posterior capsular releases and distal femoral resection. Because hemophilic patients are typically young, bilateral involvement is common. Bilateral arthroplasties, or arthrodesis of one knee and arthroplasty of the other, are viable strategies.
Total Hip Arthroplasty (THA):
THA is the procedure of choice for disabling hemophilic hip arthropathy. Nelson et al. reported successful outcomes in young patients, though they noted a higher rate of revision surgery over time. The longevity of the implants can be influenced by the patient's overall health, particularly in those with concomitant HIV or HCV infections.
Complications of Arthroplasty in Hemophilia:
Surgeons must be acutely aware of late complications. Disuse osteopenia is universally present in chronic hemophilic arthropathy, making the bone exceptionally brittle.
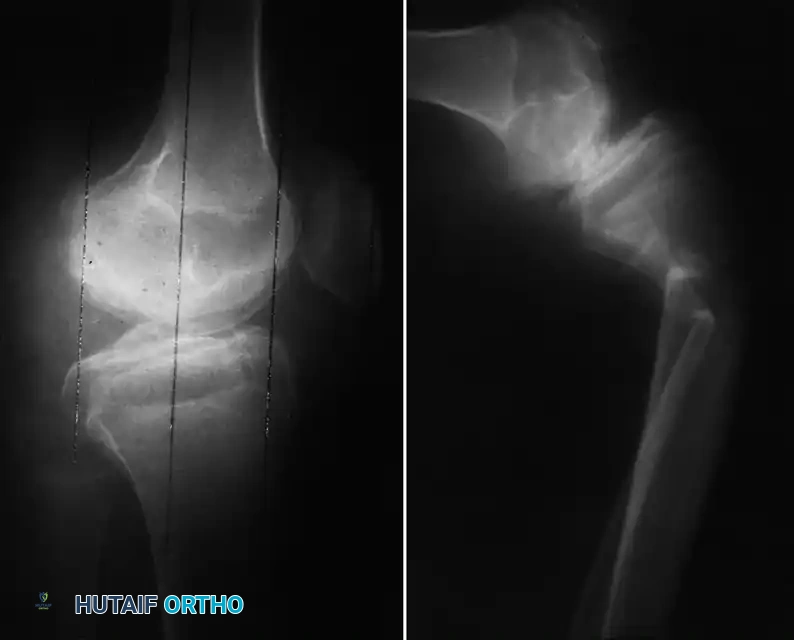
Fig. 29-36: Late complications of hemophilic arthropathy. Severe disuse osteopenia significantly increases the risk of periprosthetic fractures and iatrogenic fractures during postoperative manipulation or physical therapy.
Intraoperative fractures during implant seating, or postoperative fractures during physical therapy manipulation, are significant risks. Furthermore, the historical use of pooled plasma products left many older hemophilic patients HIV or HCV positive. This immunocompromised state drastically increases the risk of periprosthetic joint infection (PJI), requiring meticulous surgical technique and prolonged postoperative surveillance.
Postoperative Rehabilitation Protocol
Rehabilitation in the hemophilic patient requires a delicate balance between preventing stiffness and avoiding recurrent hemarthrosis.
1. Factor Coverage: Continuous or peak/trough factor coverage must be maintained at 100% for the first 7-10 days, gradually tapering to 50% during the intensive physical therapy phase (weeks 2-4).
2. Early Mobilization: Continuous Passive Motion (CPM) machines are frequently utilized immediately postoperatively for knee arthroplasties and synovectomies to prevent intra-articular adhesions.
3. Avoidance of Manipulation: Due to the severe osteopenia (as seen in Fig. 29-36), aggressive closed manipulation under anesthesia (MUA) should be avoided if possible, or performed with extreme caution to prevent supracondylar femur or proximal tibia fractures.
In conclusion, the operative management of hemophilia represents a triumph of modern multidisciplinary medicine. Through precise hematological control, meticulous surgical execution, and targeted rehabilitation, orthopaedic surgeons can successfully preserve joint function, eradicate pain, and dramatically improve the quality of life for patients suffering from hemophilic arthropathy.
📚 Medical References
- hemophilic arthropathy, Clin Orthop Relat Res 248:98, 1989.
- Forrest G, Fuchs M, Gutierrez A, et al: Factors affecting length of stay and need for rehabilitation after hip and knee arthroplasty, J Arthroplasty 13:186, 1998.
- Jackson M, Sarangi PP, Newman JH: Revision total knee arthroplasty: comparison of outcome following primary proximal tibial osteotomy or unicompartmental arthroplasty, J Arthroplasty 9:539, 1994.
- Katz MM, Hungerford DS, Krackow KA, et al: Results of knee arthroplasty after failed proximal tibial osteotomy for osteoarthritis, J Bone Joint Surg 69A:225, 1987.
- Larson KR, Cracchiolo A, Dorey FJ, et al: Total knee arthroplasty in patients after patellectomy, Clin Orthop Relat Res 264:243, 1991.
- Laskin R, Palleta G: Total knee replacement in the post patellectomy patient, J Arthroplasty 9:109, 1994.
- Lennox DW, Hungerford DS, Krackow KA: Total knee arthroplasty following patellectomy, Clin Orthop Relat Res 223:220, 1987.
- Meding JB, Keating EM, Ritter MA, et al: Total knee arthroplasty after high tibial osteotomy, Clin Orthop Relat Res 375:175, 2000.
- Meding JB, Reddleman K, Keating ME, et al: Total knee replacement in patients with diabetes mellitus, Clin Orthop Relat Res 416:208, 2003.
- Mont MA, Alexander N, Krackow KA, et al: Total knee arthroplasty after failed high tibial osteotomy, Orthop Clin North Am 25:515, 1994.
- Mont MA, Antonaides S, Krackow KA, et al: Total knee arthroplasty after failed high tibial osteotomy: a comparison with a matched group, Clin Orthop Relat Res 299:125, 1994.
- Paletta GA, Laskin RS: Total knee arthroplasty after a previous patellectomy, J Bone Joint Surg 77A:1708, 1995.
- Parvizi J, Hanssen AD, Spangehl MJ: Total knee arthroplasty following proximal tibial osteotomy: risk factors for failure, J Bone Joint Surg 86A:474, 2004.
- Parvizi J, Marrs J, Morrey BF: Total knee arthroplasty for neuropathic (Charcot) joints, Clin Orthop Relat Res 416:145, 2003.
- Saris DBF, van Rinsum AC, Dhert WJA, et al: Periarticular aneurysm formation in haemophilia, Lancet 349:766, 1997.
- Serna F, Mont MA, Krackow KA, et al: Total knee arthroplasty in diabetic patients: comparison to a matched control group, J Arthroplasty 9:375, 1994.
- Sledge C, Ewald F: Total knee arthroplasty experience at the Robert Brent Brigham Hospital, Clin Orthop Relat Res 145:78, 1979.
- Staeheli JW, Cass JR, Morrey B: Condylar total knee arthroplasty after failed proximal tibial osteotomy, J Bone Joint Surg 69A:28, 1987.
- Stern SH, Insall JN, Windsor RE, et al: Total knee arthroplasty in patients with psoriasis, Clin Orthop Relat Res 248:108, 1989.
- Windsor RE, Insall JN, Vince KG: Technical considerations of total knee arthroplasty after proximal tibial osteotomy, J Bone Joint Surg 70A:547, 1988.
- Complications Althausen PL, Lee MA, Finkemeier CG, et al: Operative stabilization of supracondylar femur fractures above total knee arthroplasty: a comparison of four treatment methods, J Arthroplasty 18:834, 2003.
- Asp JP, Rand JA: Peroneal nerve palsy after total knee arthroplasty, Clin Orthop Relat Res 261:233, 1990.
- Ayers DC: Supracondylar fracture of the distal femur proximal to a total knee replacement, Instr Course Lect 46:197, 1997.
- Barrack RL, Schrader T, Bertot AJ, et al: Component rotation and anterior knee pain after total knee arthroplasty, Clin Orthop Relat Res 392:46, 2001.
- Bayley JC, Scott RD, Ewald FC, et al: Failure of the metalbacked patellar component after total knee replacement, J Bone Joint Surg 70A:668, 1988.
- Berg M, Bergman BR, Hoborn J: Ultraviolet radiation compared to an ultra-clean air enclosure, J Bone Joint Surg 73B:811, 1991.
- Berger RA, Crossett LS, Jacobs JJ, et al: Malrotation causing patellofemoral complications after total knee arthroplasty, Clin Orthop Relat Res 356:144, 1998.
- Berry DJ, Rand JA: Isolated patellar component revision of total knee arthroplasty, Clin Orthop Relat Res 286:110, 1993.
- Booth RE, Lotke PA: The results of spacer block technique in revision of infected total knee arthroplasty, Clin Orthop Relat Res 248:57, 1989.
- Bose WJ, Gearen PF, Randall JC, et al: Long-term outcome of 42 knees with chronic infection after total knee arthroplasty, Clin Orthop Relat Res 319:285, 1995.
- Brause BD: Infected total knee replacement: diagnostic, therapeutic, and prophylactic considerations, Orthop Clin North Am 13:245, 1982.
- Brick GW, Scott RD: The patellofemoral component of total knee arthroplasty, Clin Orthop Relat Res 231:163, 1988.
- Burnett RS, Berger RA, Della Valle CJ, et al: Extensor mechanism allograft reconstruction after total knee arthroplasty, J Bone Joint Surg 87A (suppl 1, pt 2):175, 2005.
- Cadambi A, Engh GA: Use of a semitendinosus tendon autogenous graft for rupture of the patellar ligament after total knee arthroplasty, J Bone Joint Surg 74A:974, 1992.
- Calton TF, Fehring TK, Griffi n WL: Bone loss associated with the use of spacer blocks in infected total knee arthroplasty, Clin Orthop Relat Res 345:148, 1997.
- Crockarell JR Jr, Mihalko MJ:
You Might Also Like