Masterclass in Elbow Arthroplasty: Biomechanics, Surgical Techniques, and Clinical Outcomes
Key Takeaway
Elbow arthroplasty aims to restore a painless, functional arc of motion in patients with inflammatory or degenerative arthritis. Success relies on meticulous patient selection, understanding of the complex ginglymoarthrodial biomechanics, and precise surgical execution. This guide covers the historical evolution, anatomical considerations, and step-by-step surgical techniques for modern semiconstrained and unconstrained elbow prostheses, providing orthopedic surgeons with evidence-based protocols for optimal patient outcomes.
Introduction to Elbow Arthroplasty
Although extremity arthroplasty has been performed for over 150 to 200 years, the elbow remains one of the most unforgiving joints to reconstruct. To date, no single arthroplasty design has been developed that universally meets the needs of every patient with a disabled elbow joint. The primary goal—restoration of a painless, functional arc of elbow motion in active individuals with inflammatory or degenerative arthritis—is often incomplete.
In lower-demand individuals, such as elderly patients with rheumatoid arthritis or complex distal humerus fractures, total elbow arthroplasty (TEA) provides excellent pain relief and functional restoration. However, careful patient selection is paramount. The complication rates for elbow implant arthroplasty remain higher than those for hip or knee arthroplasty, and managing these complications (e.g., aseptic loosening, infection, triceps insufficiency) requires advanced reconstructive expertise.
Historical Evolution of Elbow Reconstruction
The historical development of elbow arthroplasty can be viewed through four distinct eras, as originally categorized by Coonrad. Understanding these eras provides critical insight into the biomechanical rationale behind modern implant designs.
Era 1: Resection and Anatomical Arthroplasty (1885–1947)
Resection arthroplasty, pioneered by Verneuil and Ollier in the 1800s, involved the radical removal of the entire elbow joint via distal humeral and proximal radioulnar resection.

While its use in posttuberculous ankylosis eradicated infection, it rendered the elbow grossly unstable. Nevertheless, in the 1930s, Buzby recommended it strictly for pain control. A variation known as "functional arthroplasty" involved a wedge-shaped distal humeral resection left as a fulcrum for the proximal ulna. Advocated by Hass, this technique is now primarily reserved as a salvage procedure following catastrophic infection or failed arthroplasty.
In anatomical arthroplasty, only the articular surfaces were removed, exposing the underlying cancellous bone. To minimize ankylosis, biological interposition materials were popularized in Europe by Payr and Putti. In the United States, Murphy introduced arthroplasty in the 1900s, and Lexer advocated for fascia and fat interposition. In 1918, Baer experimented with materials ranging from zinc and silicone to chromicized pig bladder (Baer membrane). Ultimately, fascia lata became the most popular interpositional substance due to its ease of harvest and minimal donor-site morbidity, with successful series reported by Campbell, Henderson, and MacAusland.
Era 2: Constrained Hinge Arthroplasty (1947–1970)
Despite successes with interpositional arthroplasty, efforts shifted toward replacing the joint entirely. Early designs included metal for the olecranon, acrylic for the lower humerus, and Vitallium for the radial head. Metal-to-metal hinged prostheses emerged but frequently failed. Because these rigid hinges lacked an anatomical center of rotation, they severely limited flexion and transmitted massive stresses to the bone-implant interface, leading to rapid aseptic loosening, skin breakdown, and triceps tendon ruptures.

Era 3: Polymethyl Methacrylate Fixation (1970–1975)
Dee initiated the third major era by utilizing polymethyl methacrylate (PMMA) bone cement to fix metal-to-metal hinged prostheses. While short-term results were encouraging, the rigid constraint combined with rigid fixation simply accelerated catastrophic implant loosening within 2 to 3 years. These designs were largely abandoned.
Era 4: Modern Prosthetic Design (1975–Present)
Since 1975, two dominant prosthetic philosophies have evolved to address the biomechanical failures of rigid hinges:
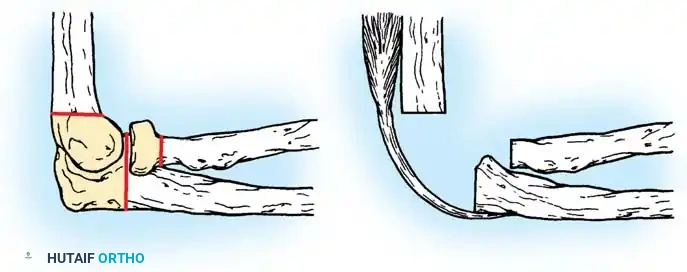
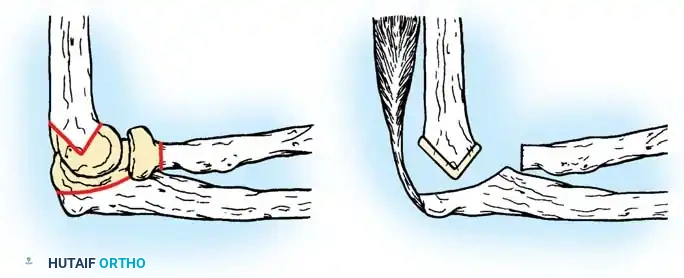
- Semiconstrained Metal-to-Polyethylene Hinges: Implants such as the Mayo, Coonrad-Morrey, GSB III, and Discovery designs incorporate a "sloppy hinge." This design allows 7 to 10 degrees of varus-valgus and rotational laxity, dissipating stresses away from the cement-bone interface and into the surrounding soft tissues.
- Unconstrained Resurfacing Arthroplasties: Implants like the Ewald capitellocondylar, Kudo, and Souter-Strathclyde rely entirely on intact capsuloligamentous structures and balanced musculature for stability.
Modern semiconstrained implant design.
Modern unconstrained implant design.
Anatomy and Biomechanics of the Elbow
The elbow is a highly congruent ginglymoarthrodial (hinge-pivot) joint composed of two independent uniaxial articulations: the humeroulnar joint (ginglymoid) and the radiocapitellar/proximal radioulnar joint (trochoid). This allows two degrees of freedom: flexion-extension and pronation-supination.
Capsuloligamentous Stabilizers
Stability is maintained by a delicate interplay of joint geometry, capsuloligamentous integrity, and dynamic muscle forces (biceps, brachialis, anconeus, triceps).
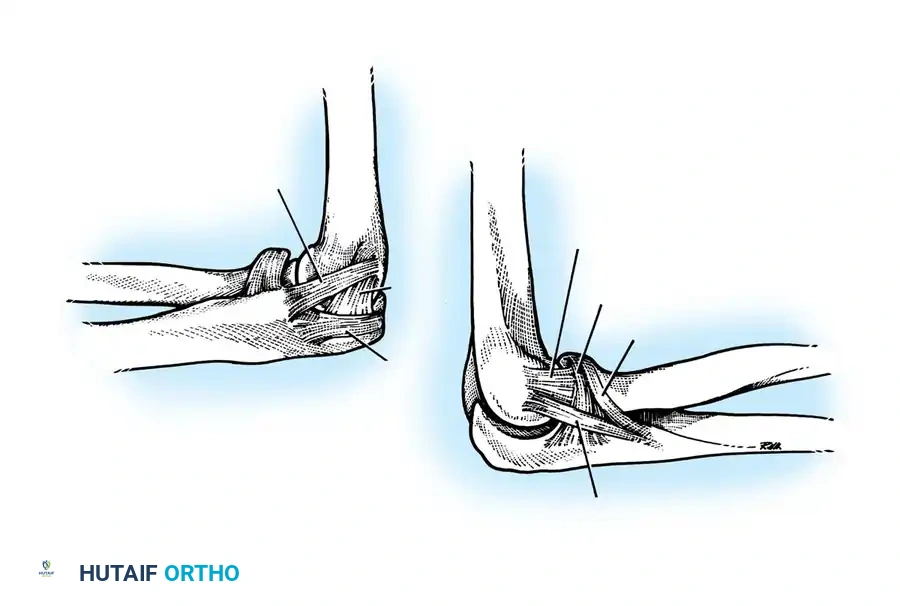
The Medial Collateral Ligament (MCL) complex consists of anterior, posterior, and transverse bundles. The anterior bundle is the primary restraint to valgus stress and remains taut throughout the flexion-extension arc.
The Lateral Collateral Ligament (LCL) complex, as described by Morrey, includes the radial collateral ligament, the lateral ulnar collateral ligament (LUCL), the accessory lateral collateral ligament, and the annular ligament. The LUCL is the critical structure preventing posterolateral rotatory instability (PLRI), extending superficial to the annular ligament to insert on the crista supinatorius of the ulna.
Kinematics and Joint Forces
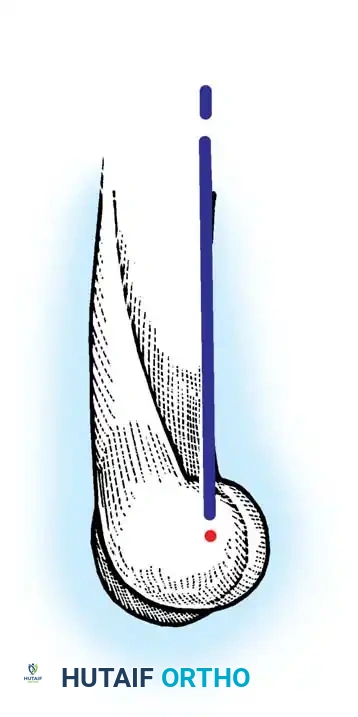
The instant center of rotation for elbow flexion and extension is remarkably constant. It lies at the center of concentric circles formed by the lateral projection of the capitellum and trochlea, passing colinearly with the anterior cortex of the distal humerus.
The carrying angle varies dynamically, moving from approximately 11 degrees of valgus in full extension to 6 degrees of varus in full flexion.
Clinical Pearl: When placing an unconstrained prosthesis, recreating this exact anatomical axis of rotation is mandatory. Failure to do so results in eccentric loading, accelerated polyethylene wear, and early clinical failure.
Joint reaction forces at the elbow are surprisingly high. Static analyses reveal that forces are greatest in extension and pronation. During strenuous lifting, compressive forces across the joint can reach three times body weight.
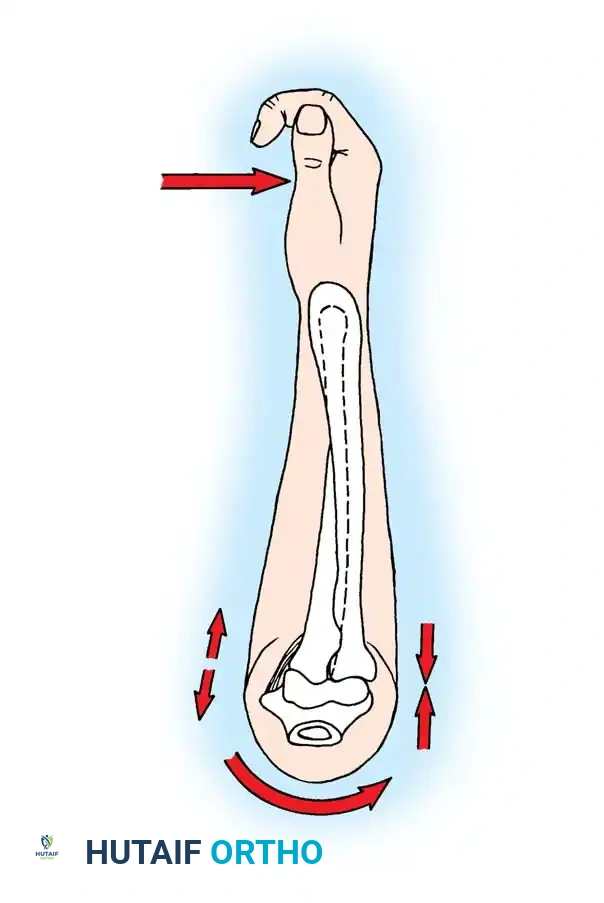
With the elbow extended and axially loaded, 40% of the stress is transmitted through the ulnohumeral joint, and 60% through the radiocapitellar joint. If the radial head is excised, the humeroradial force is entirely shifted to the ulna. This drastically increases tension on the MCL and concentrates massive loads on the lateral edge of the coronoid process, potentially applying forces up to nine times body weight to the medial structures.
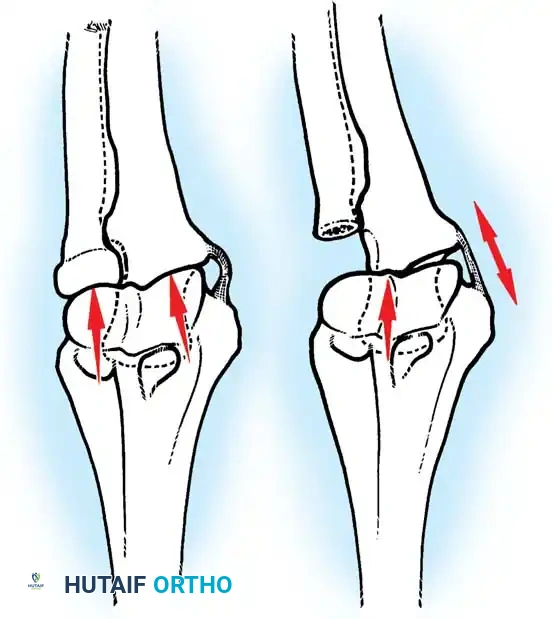
Principles of Soft-Tissue Balancing
Soft-tissue balancing is as critical in the elbow as it is in any major joint reconstruction. While historical texts (such as Wiater and Levine's work on shoulder arthroplasty) note that a 1 cm gain in anterior capsular length increases external rotation by 20 degrees, the analogous principle applies to the elbow: meticulous release of the anterior capsule is required to restore extension, while posterior capsular release is necessary to restore flexion. Intracapsular and extracapsular releases must be balanced perfectly to prevent iatrogenic instability, particularly when utilizing unconstrained implants.
Preoperative Planning and Positioning
Thorough preoperative templating is essential to assess bone stock, canal diameter, and the presence of deformity. Standard AP and lateral radiographs are mandatory; CT scans are highly recommended for post-traumatic cases or revision scenarios to evaluate bone loss.
Positioning:
The patient is typically placed in the lateral decubitus position with the operative arm draped over a padded post, allowing 120 degrees of flexion. Alternatively, a supine position with the arm brought across the chest can be utilized. A sterile tourniquet is applied high on the brachium.

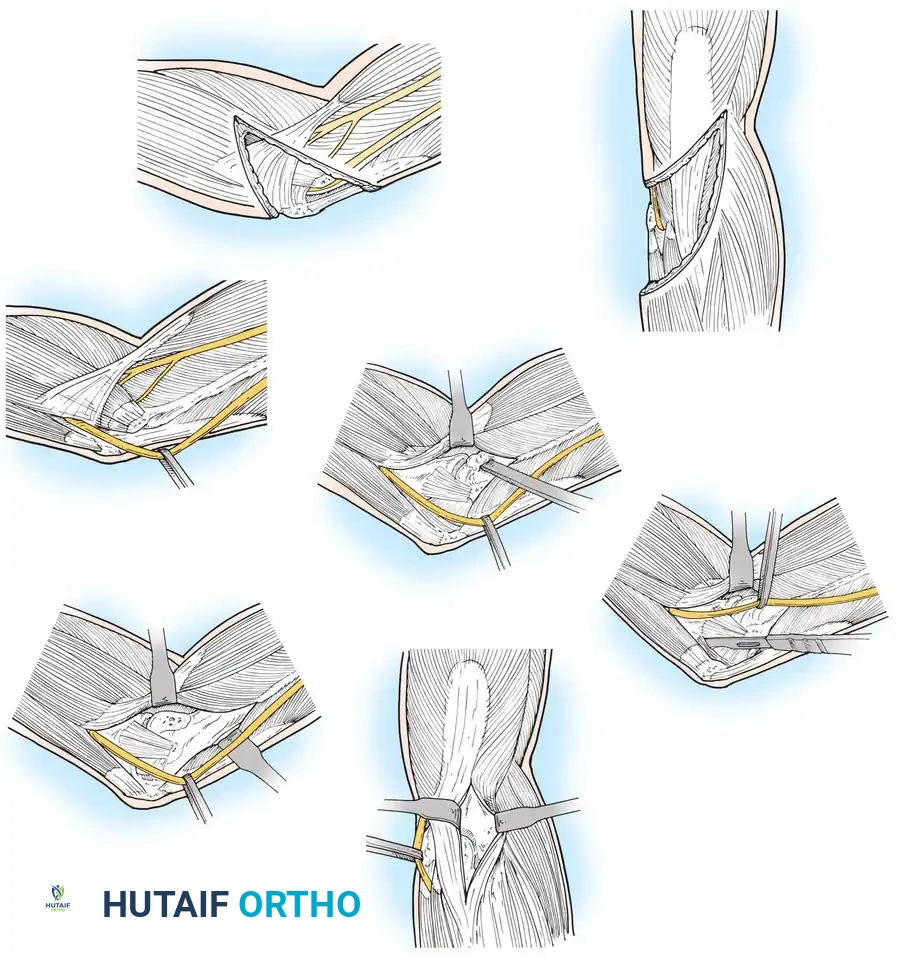
Surgical Approaches and Step-by-Step Technique
1. Incision and Ulnar Nerve Management
A straight posterior incision is made, curving slightly lateral to the tip of the olecranon to avoid placing the scar directly over the bony prominence.

The ulnar nerve must be identified proximally at the medial intermuscular septum, mobilized distally through the cubital tunnel, and protected. Routine anterior transposition of the ulnar nerve is performed by most surgeons to prevent postoperative neuropathy.
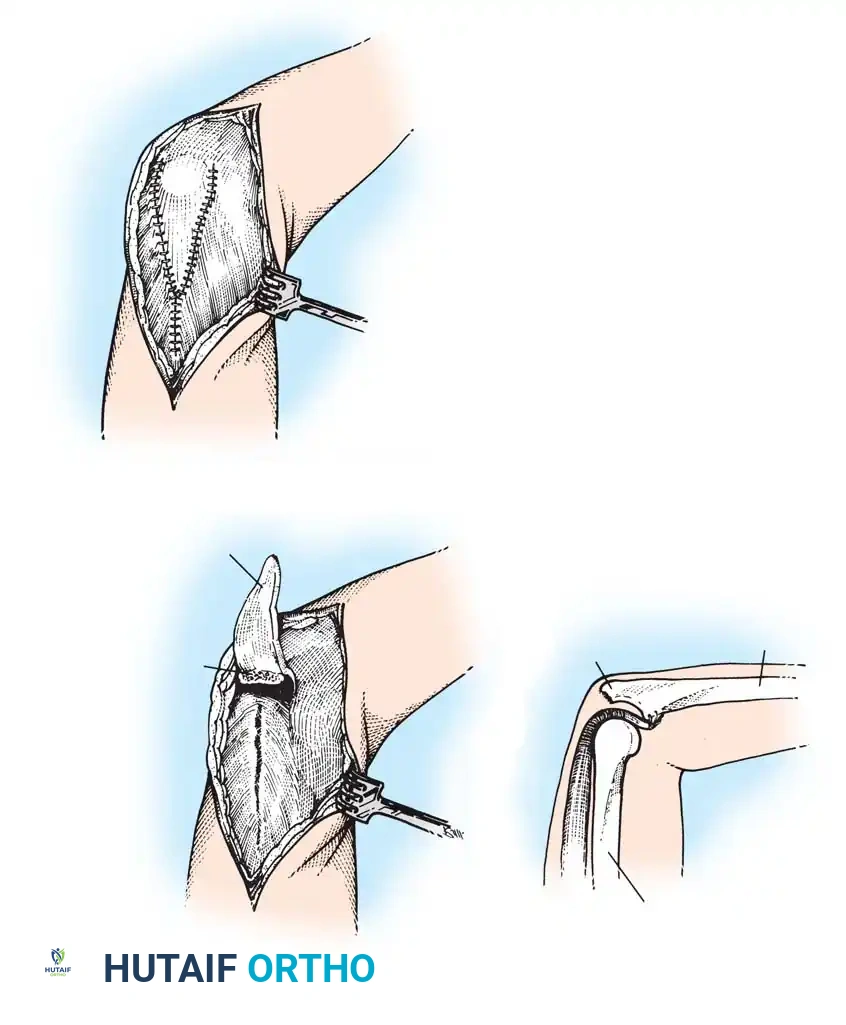
2. Triceps Management
Several options exist for managing the extensor mechanism:
* Triceps-Reflecting (Bryan-Morrey): The triceps is reflected from medial to lateral in continuity with the anconeus and forearm fascia.
* Triceps-Splitting: The triceps is split longitudinally.
* Triceps-Sparing: Used primarily for unconstrained resurfacing, preserving the extensor attachment entirely.
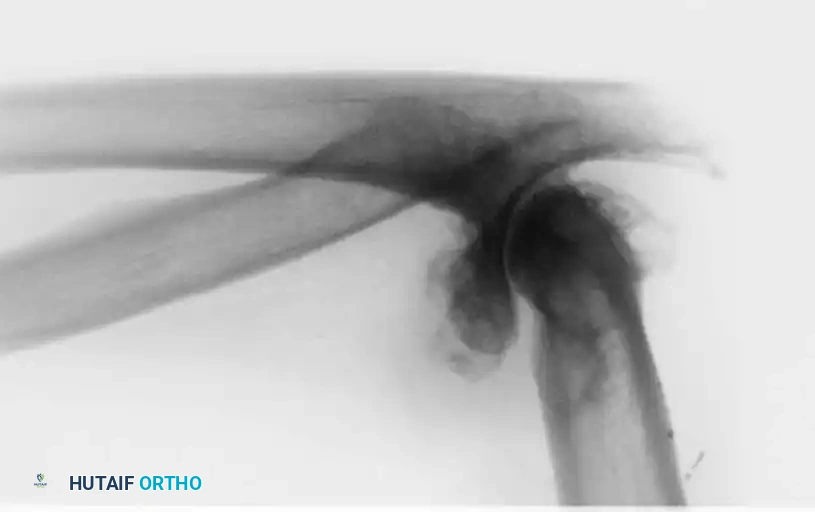
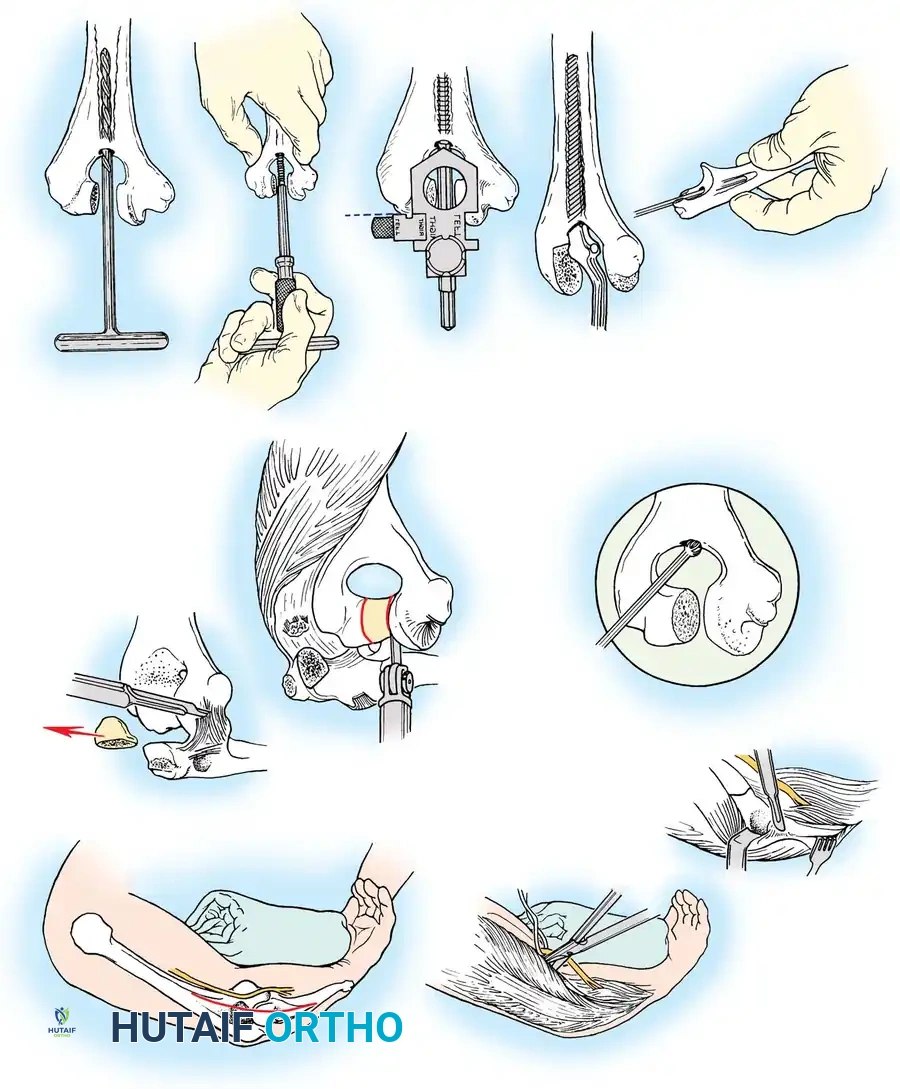
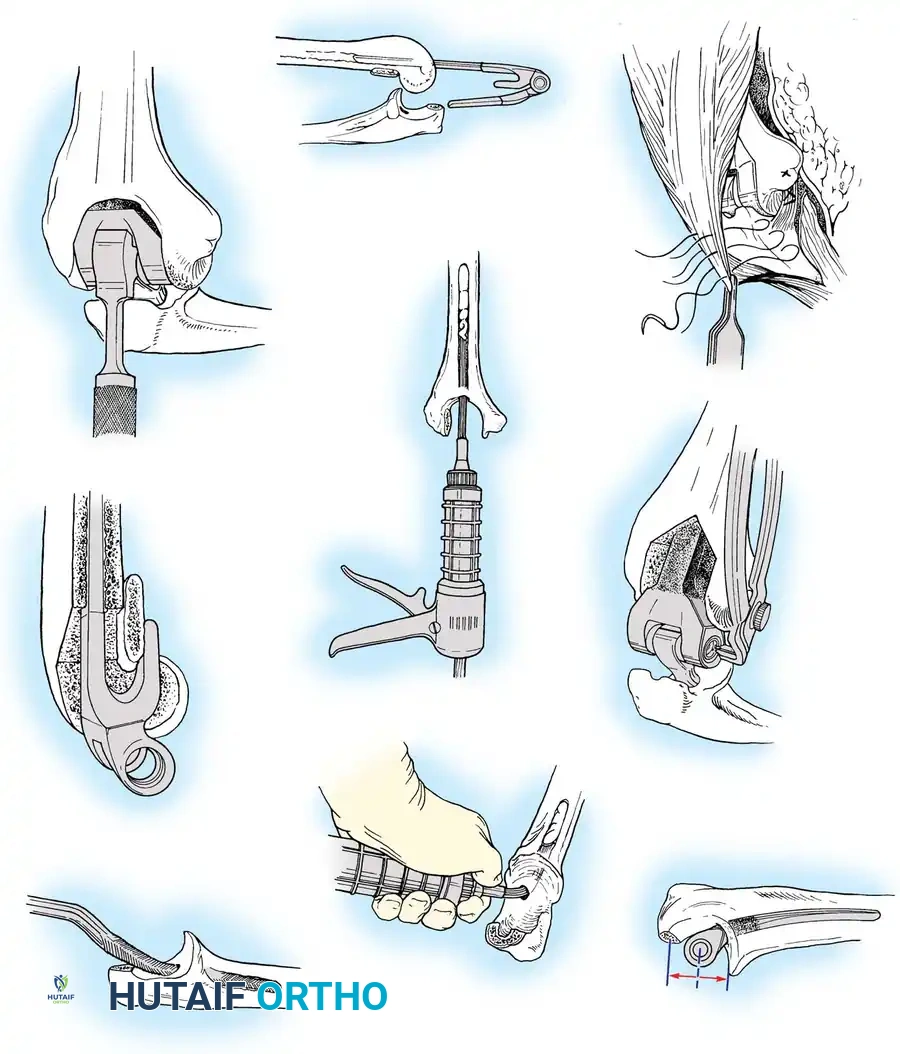
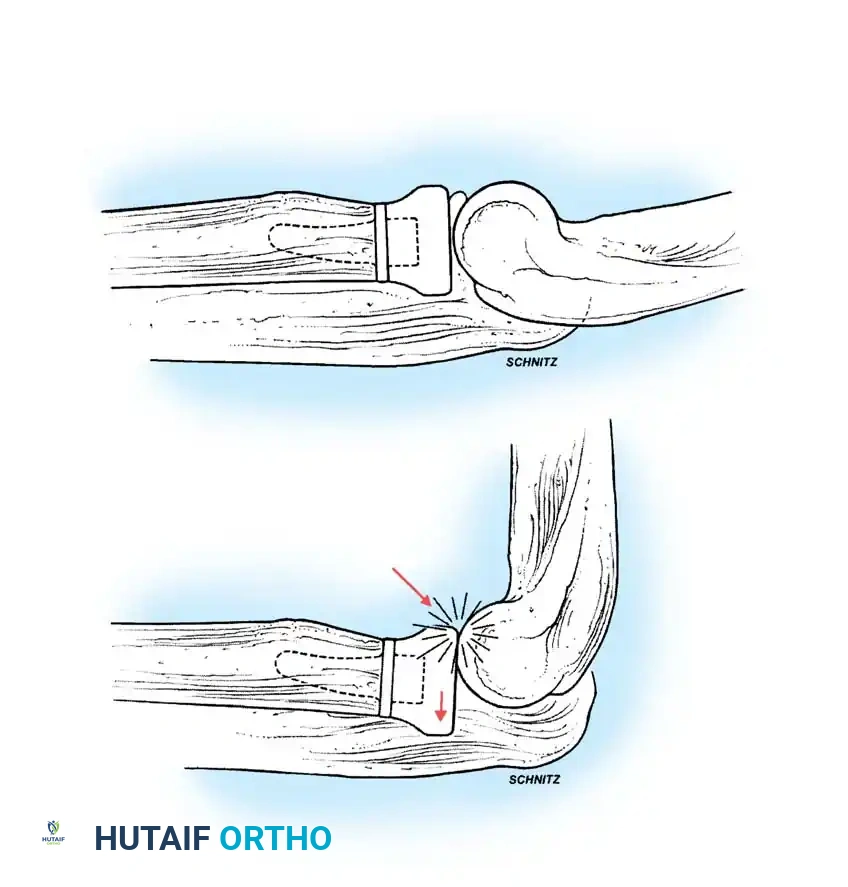
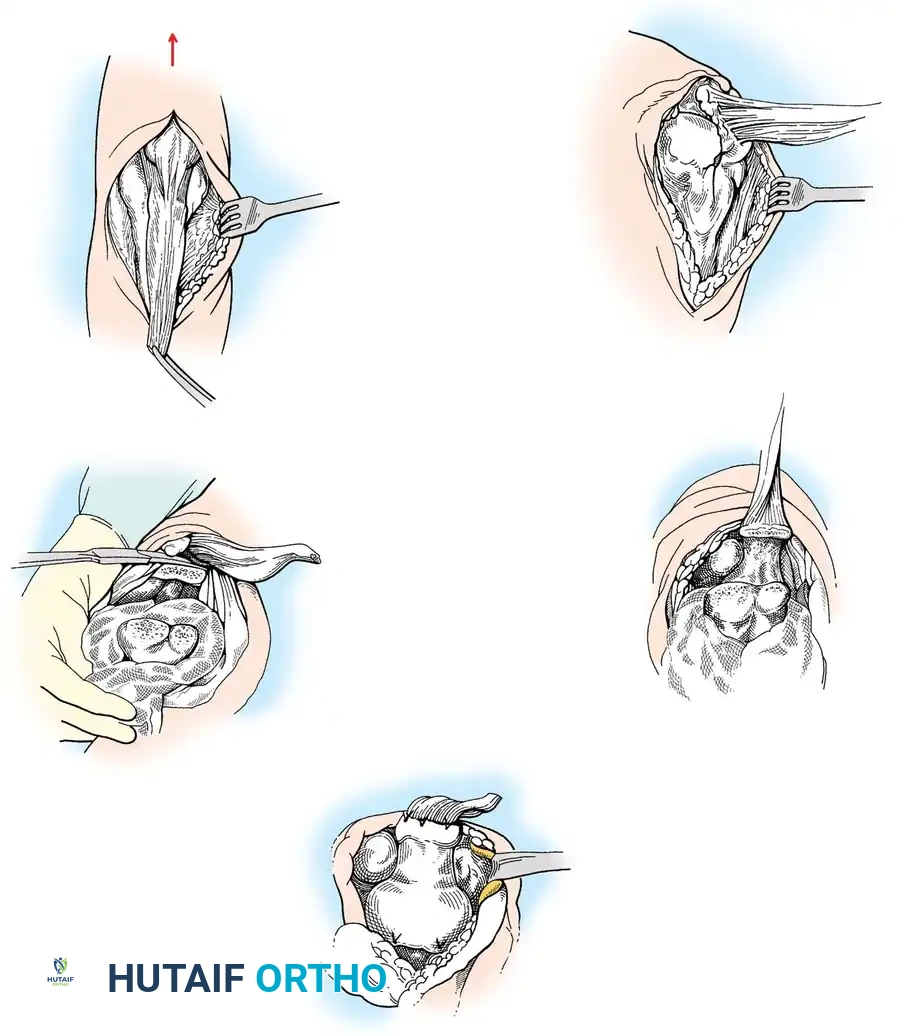
3. Joint Exposure and Bone Preparation
The collateral ligaments are released (or preserved, depending on implant constraint), and the joint is dislocated. The tip of the olecranon may be excised to improve exposure.
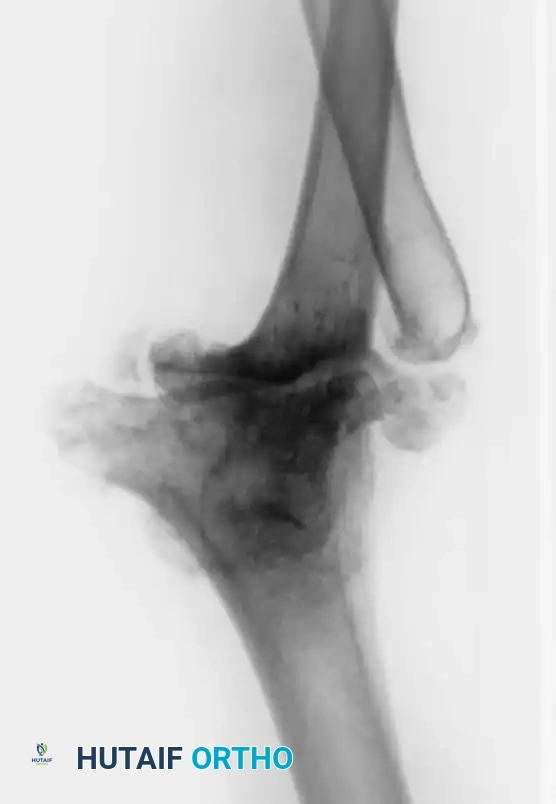
Humeral Preparation:
The humeral canal is accessed via the
Associated Surgical & Radiographic Imaging
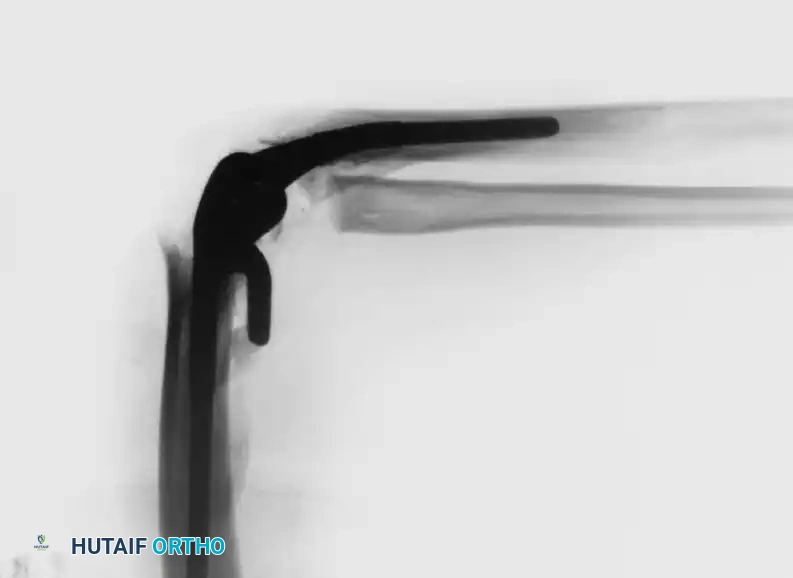
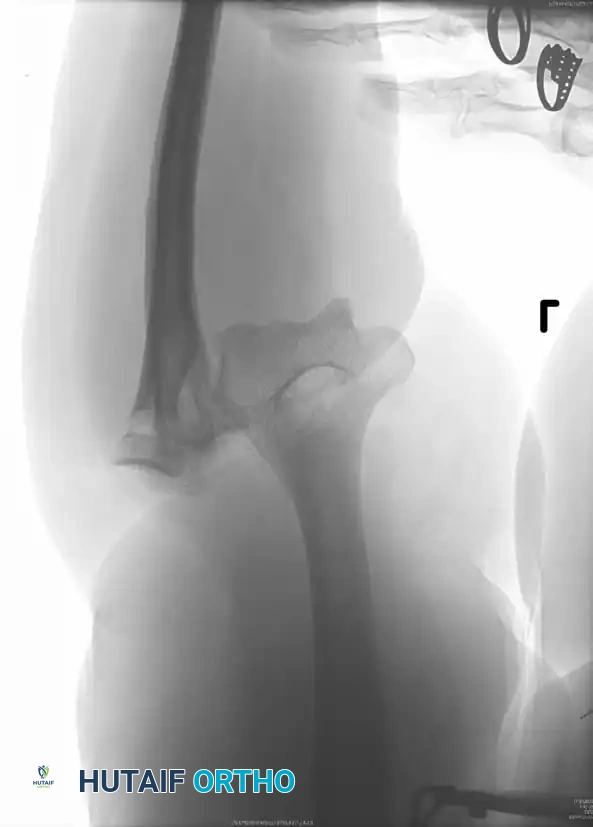
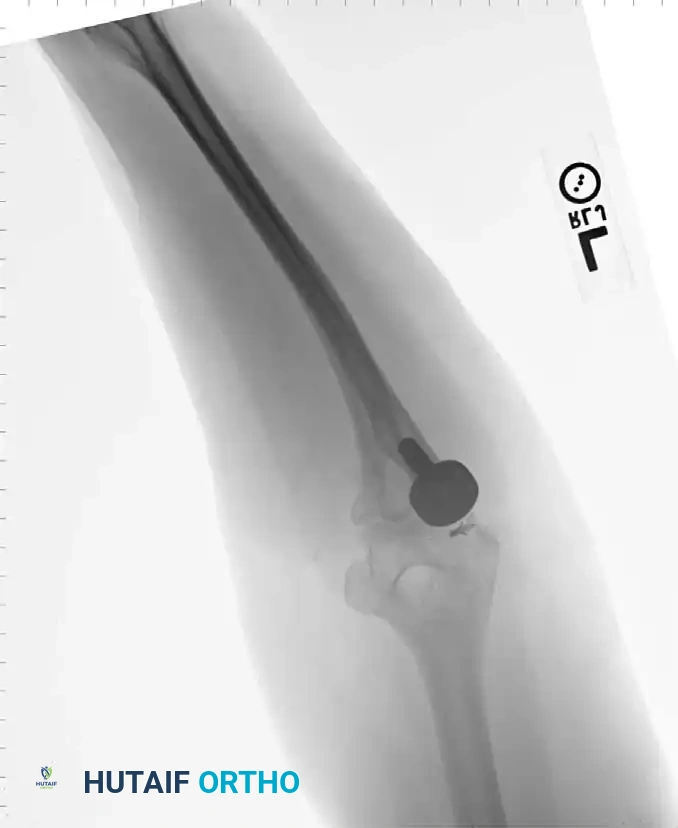
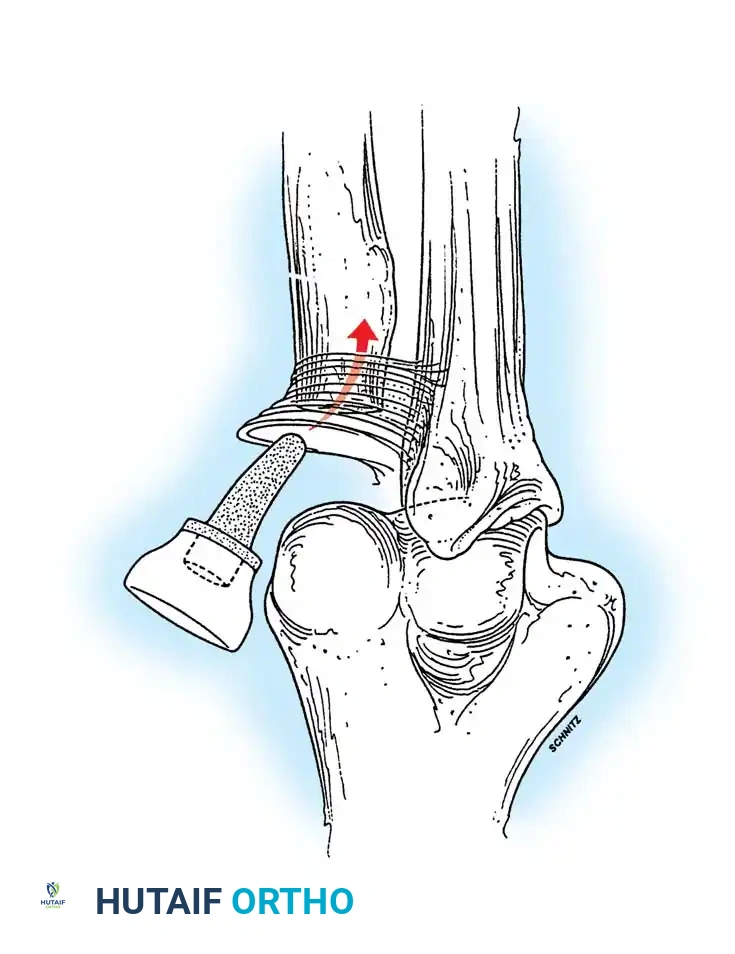
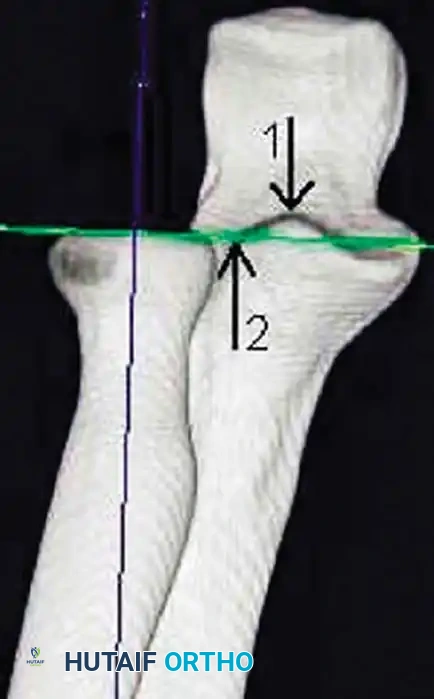
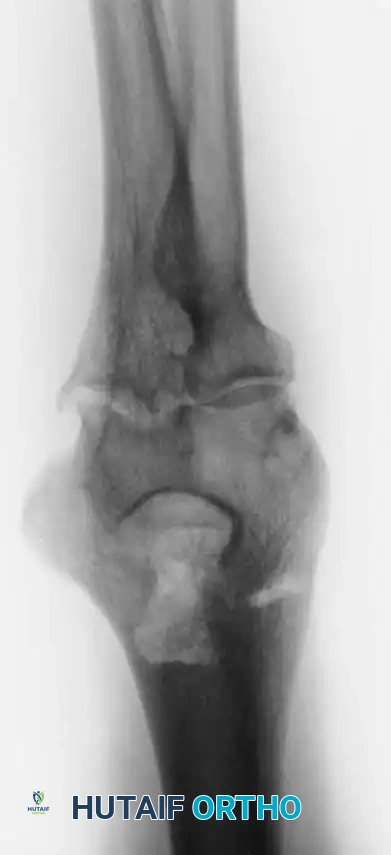
📚 Medical References
- elbow arthroplasty: two to fi ve year followup in rheumatoid arthritis, J Bone Joint Surg 62A: 1239, 1980.
- Hotchkiss R: Compass universal hinge: surgical technique , Memphis, Tenn, Smith and Nephew, 1998.
- Kaplan EB: Surgical approach to the proximal end of the radius and its use in fractures of the head and neck of the radius, J Bone Joint Surg 23:86, 1941.
- Key JA, Conwell HE: The management of fractures, dislocations, and sprains , 2nd ed. St Louis, Mosby, 1937.
- Kocher T: Textbook of operative surgery , 3rd ed, London, 1911, Adam & Charles Black (Translated by HJ Stiles, CB Paul). MacAusland WR: Ankylosis of the elbow: with report of four cases treated by arthroplasty. JAMA 64:312, 1915.
- Mehta JA, Bain GI: Surgical approaches to the elbow, Hand Clin 20;375, 2004.
- Molesworth WHL: Operation for complete exposure of the elbow joint, Br J Surg 18:303, 1930.
- Morrey BF, Bryan RS, Dobyns JH, et al: Total elbow arthroplasty: a fi ve-year experience at the Mayo Clinic, J Bone Joint Surg 63A:1050, 1981.
- Müller ME, Allgöwer M, Schneider R, et al: Manual of internal fi xation: techniques recommended by the AO-ASIF group, 3rd ed, Berlin, Springer-Verlag, 1991.
- Patterson SO, Bain GI, Mehta JA: Surgical approaches to the elbow, Clin Orthop Relat Res 370:19, 2000.
- Wadsworth TG: A modified posterolateral approach to the elbow and proximal radioulnar joints, Clin Orthop Relat Res 144:151, 1979.
- Wilkinson JM, Stanley D: Posterior surgical approaches to the elbow: a comparative anatomic study, J Shoulder Elbow Surg 10:380, 2001.
- Radius Henry AK: Exposures of long bones and other surgical methods, Bristol, England, 1927, John Wright & Sons. Strachan JCH, Ellis BW: Vulnerability of the posterior interosseous nerve during radial head resection, J Bone Joint Surg 53B:320, 1971.
- Thompson JE: Anatomical methods of approach in operations on the long bones of the extremities, Ann Surg 68:309, 1918.
- Ulna Boyd HB: Surgical exposure of the ulna and proximal third of the radius through one incision, Surg Gynecol Obstet 71:86, 1940.
You Might Also Like