Methods and Indications for Skin Closure in Hand Surgery
Key Takeaway
The reconstructive ladder in hand surgery dictates the optimal method for skin closure, ranging from secondary intention and direct suturing to complex free tissue transfer. Primary closure is preferred for uncontaminated wounds, while exposed deep structures—such as tendons devoid of paratenon, nerves, or cortical bone—mandate vascularized flap coverage. Meticulous handling of soft tissues, precise hemostasis, and appropriate donor site management are paramount for restoring both form and function in the injured hand.
PRINCIPLES OF SOFT TISSUE RECONSTRUCTION IN THE HAND
The fundamental goal of soft tissue reconstruction in the hand is to achieve rapid, stable wound closure that preserves maximal function, minimizes scar contracture, and restores protective sensation. The decision-making process is governed by the "reconstructive ladder," a hierarchical approach that begins with the simplest, least morbid techniques (healing by secondary intention, direct suture) and progresses to highly complex interventions (local flaps, regional flaps, and free tissue transfer) only when dictated by the nature of the defect.
In hand surgery, the unique biomechanical properties of the skin must be respected. The dorsal skin is thin, highly mobile, and accommodates the extreme flexion of the digits and wrist. Conversely, the volar (palmar) skin is thick, glabrous, and firmly tethered to the underlying palmar aponeurosis by robust fascial ligaments (Cleland’s and Grayson’s ligaments) to withstand high shear forces during grip. Understanding these biomechanical constraints is paramount when selecting the appropriate method for skin closure.
DIRECT SUTURE: INDICATIONS AND BIOMECHANICS
Unless a wound is severely contaminated, crushed, or exhibits significant tissue loss, primary closure by direct suture is the gold standard for hand lacerations. Healing by primary intention minimizes fibrotic scarring, reduces the risk of infection, and accelerates functional recovery.
Evidence-Based Non-Operative Management
While primary suturing is the standard, selected lacerations may be managed conservatively. A landmark randomized controlled trial by Quinn et al. demonstrated that full-thickness hand lacerations smaller than 2 cm—provided they are devoid of underlying tendon, joint, fracture, or nerve complications—can heal by secondary intention. When treated without suturing, these specific wounds healed with a cosmetic appearance equivalent to sutured lacerations and were associated with significantly less patient discomfort.
Surgical Technique for Direct Closure
Most incised wounds can be closed via simple direct suturing. The subcutaneous tissue in the hand is typically not sutured separately to avoid introducing foreign body reactions near superficial gliding structures.
Surgical Pearl: Care must be taken to avoid inversion of the skin edges. Eversion is critical for optimal epidermal healing. Careful, pinpoint hemostasis using bipolar electrocautery is necessary to prevent postoperative hematoma, which serves as a nidus for infection and subsequent fibrosis.
Closure is markedly facilitated when all viable skin edges are meticulously preserved during the initial debridement and pulsatile lavage.
Managing Dorsal Defects:
A unique biomechanical advantage of the dorsal hand is its skin mobility. A small, irregular skin defect on the dorsal surface can often be converted into a transverse elliptical defect. By extending the wrist, the dorsal skin is placed on slack, allowing the ellipse to be closed in a transverse line without excessive tension. This technique prevents longitudinal scar contracture, which could otherwise restrict metacarpophalangeal (MCP) joint flexion.
SKIN GRAFTS: AUTOGRAFTS, ALLOGRAFTS, AND XENOGRAFTS
When primary closure cannot be achieved without excessive tension, but a well-vascularized wound bed is present, skin grafting is indicated. Skin grafts obtained from the patient (autografts) are categorized as either split-thickness skin grafts (STSG) or full-thickness skin grafts (FTSG).
Graft Survival and Vascularity Requirements
Skin grafts rely entirely on the vascularity of the recipient bed. They survive through three distinct phases:
1. Plasmatic Imbibition (Days 1-2): The graft absorbs nutrients directly from the wound bed exudate.
2. Inosculation (Days 3-4): Capillary networks from the recipient bed align and connect with the graft's vessels.
3. Angiogenesis (Days 5+): True neovascularization occurs, permanently securing the graft.
Grafts will not survive over exposed avascular structures, including:
* Tendons denuded of their paratenon.
* Cortical bone stripped of its periosteum.
* Exposed nerves or joint capsules.
Flap Viability and Venous Congestion
Wounds with distally attached, random-pattern flaps may appear to have sufficient skin for primary closure, but they frequently lack adequate venous drainage. This deficient drainage initiates a catastrophic cascade: venous engorgement leads to distention, subsequent microvascular thrombosis, and ultimate tissue necrosis.
Clinically, the color of a failing flap transitions predictably: from a deep, congested blue, to purple, and finally to black (eschar). Retrograde flaps resulting from crushing or avulsion injuries are at an exceptionally high risk of failure due to the initial microvascular trauma.
Surgical Warning: A distally based random flap on the dorsal surface of the hand or forearm is significantly less likely to survive than a similar flap on the palm, due to the differing venous architectures and subdermal plexuses.

Fig. 62-2 Flap attached distally on dorsum of hand (A) is less likely to survive than is similar one on palm (B).
If doubt exists regarding the viability of a traumatized flap, the ischemic skin should be sharply excised and replaced immediately with a split-thickness graft.
Biological Dressings
In cases of severe contamination or when the wound bed requires time to granulate, temporary coverage is necessary. Allografts (from human cadaveric donors) and xenografts (e.g., porcine skin) serve as excellent biological dressings. They provide temporary coverage, reduce fluid loss, and stimulate the development of a healthy capillary bed. Although an allograft may temporarily revascularize, inevitable immunologic rejection occurs, necessitating eventual replacement with an autograft.
SKIN FLAPS: CLASSIFICATION AND PRINCIPLES
When a skin defect leaves deep, avascular structures exposed (nerves, tendons without paratenon, cortical bone), a skin graft is insufficient. A skin flap—bringing its own subcutaneous tissue and intrinsic blood supply—is mandatory to provide durable coverage and support the survival of these critical structures.
Nomenclature and Classification
Flaps are classified based on three primary criteria: location, blood supply, and technique of transfer.
1. By Location:
* Local Flaps: Harvested from tissue immediately adjacent to the defect (e.g., advancement, rotation, or transposition flaps).
* Regional/Distant Flaps: Harvested from a separate anatomical region (e.g., groin flap, cross-arm flap).
2. By Blood Supply:
* Random Pattern Flaps: Receive their circulation through the unnamed subdermal or subcutaneous vascular plexus. Because they lack a dominant arterial axis, their survival depends strictly on their geometry. To ensure survival, the base of a random flap must be as wide as its length (a 1:1 length-to-width ratio).
* Axial Pattern Flaps: Receive their circulation from a specific, named artery running along the longitudinal axis of the flap (e.g., the radial forearm flap based on the radial artery). Because of this robust vascularity, the length-to-width ratio can safely exceed 3:1. Axial flaps are further subdivided by their tissue composition into cutaneous, musculocutaneous, and fasciocutaneous flaps.
3. By Technique of Transfer:
* Pedicled Flaps: Remain attached to their donor site blood supply while the distal end is inset into the defect. They require a second surgical stage (division and inset) weeks later.
* Free Flaps: Completely detached from the donor site and transferred in a single step, requiring microvascular anastomosis of the artery and veins to recipient vessels at the defect site.
COVERAGE OF SPECIFIC ANATOMICAL AREAS
The selection of a flap is highly dependent on the anatomical zone of the injury, the size of the defect, and the functional requirements of the affected digit or hand segment.
Fingertip and Distal Phalanx Injuries
Fingertip injuries with isolated skin loss of less than 1 square centimeter can be treated highly satisfactorily with healing by secondary intention, often resulting in excellent contour and sensation.
If the defect is larger but lacks exposed bone, a full-thickness skin graft (often harvested from the hypothenar eminence or volar wrist crease) provides durable coverage and the potential for the return of protective sensation. For amputations with exposed bone, local advancement flaps (e.g., V-Y advancement, Atasoy, or Kutler flaps) or neurovascular island flaps are indicated to provide sensate, padded coverage.
Dorsal Digital Defects
A large skin defect on the dorsum of a finger that exposes extensor tendons devoid of paratenon requires flap coverage.
* Double Local Flap: Frequently, a double local flap can be constructed by rotating a proximally based local flap on one side of the defect and a distally based local flap on the contralateral side. The donor defects created by these rotations are then covered with split-thickness skin grafts.
* Reverse Cross-Finger Flap: For more proximal dorsal injuries where local rotational flaps are insufficient, a reverse cross-finger flap is utilized. This involves elevating the subcutaneous tissue from the dorsum of an adjacent finger (leaving the skin intact as a "book page") and turning it over the defect, followed by skin grafting over the transposed tissue.
Volar Digital Defects
Skin defects on the volar (palmar) surface of a finger that expose flexor tendons are classically managed with a standard Cross-Finger Flap.
* Technique: The flap is raised from the dorsal surface of an adjacent, uninjured finger. It extends from the mid-lateral line of one side to the mid-lateral line of the other, designed slightly wider than the recipient defect to avoid tension.
* Important Caveat: While a flap from the dorsal surface of one finger can safely cover a volar defect on another, the reverse is never indicated. Volar skin is too specialized, tethered, and critical for grip to be utilized as a donor site for a dorsal defect.
Proximal Palmar and Dorsal Digital Defects
For defects at the base of the digits or the web spaces, a Flag Flap (an axial pattern flap based on the dorsal metacarpal arteries) is highly effective. If multiple fingers are involved, or if a massive area requires coverage, a distant pedicled flap, such as a subpectoral flap, may be appropriate.
Pitfall: Thick subcutaneous fat, such as that found on the lower abdomen, is highly undesirable for digital reconstruction. It creates a bulky, insensate appendage that severely limits joint mobility and hand function. If an abdominal flap is used, aggressive secondary defatting procedures are inevitably required.
Extensive Hand and Palmar Defects
Skin defects on the palm or dorsum of the hand exposing vital structures offer multiple reconstructive options depending on the defect's magnitude:
1. Filleted Finger Flap: If an adjacent finger is unsalvageable due to severe trauma (e.g., devastating crush or devascularization), it can be "filleted." The bone and tendons are excised, and the remaining vascularized soft tissue envelope is unfolded to create an excellent, sensate pedicle graft for large palmar or dorsal defects.
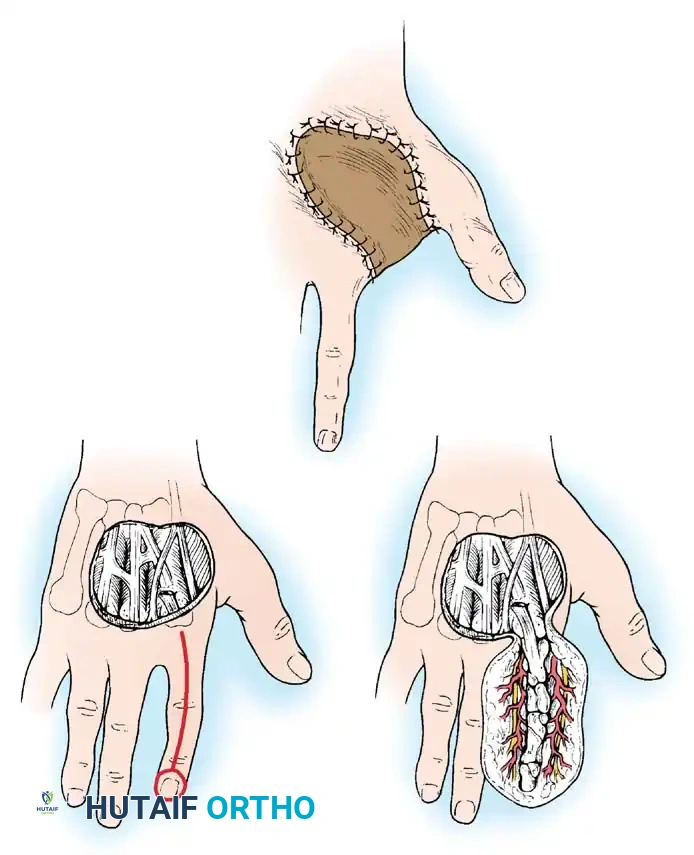
Fig. 62-3 A-C, Filleted graft fashioned from injured finger.
- Axial Forearm Flaps: An arterialized axial flap from the ipsilateral forearm (e.g., Radial Forearm or Posterior Interosseous Artery flap) provides robust coverage and allows for comfortable, independent positioning of the upper limb postoperatively.
- Distant Pedicled Flaps: Cross-arm and cross-forearm flaps provide excellent skin quality, but they carry the significant disadvantage of immobilizing both upper extremities together for weeks, leading to profound stiffness. An ipsilateral groin or abdominal pedicle flap permits more comfortable positioning.
- Free Tissue Transfer: In modern practice, for massive defects or when composite tissue (bone, tendon, skin) is required, free tissue transfer via microvascular technique (e.g., Anterolateral Thigh flap, Lateral Arm flap) often provides the most definitive and functional coverage in a single stage.
MANAGEMENT OF THE DONOR AREA
The morbidity of the donor site must never be overlooked. Proper management is essential to prevent delayed healing, severe pain, and unsightly scarring. The donor area (and any raw, exposed undersurface of a pedicled flap that does not make contact with the recipient defect) should be meticulously covered with split-thickness skin grafts.
Dressing Techniques for Donor Sites
Several acceptable, evidence-based methods are utilized for treating the donor area:
- Traditional Non-Adherent Dressings: The donor area is dressed with a single layer of finely woven nylon or silk gauze (e.g., Xeroform or Adaptic).
- Warning: If the secondary bulky dressing prevents adequate drying, the donor area tends to become macerated. Maceration invites secondary bacterial colonization, infection, and full-thickness necrosis, potentially converting a partial-thickness donor site into a full-thickness defect requiring its own skin graft. To prevent this, the outer dressings are often removed early, the part is left uncovered, and rapid desiccation (drying) of the area is actively encouraged.
- Synthetic Adhesive Films: A modern alternative involves placing a semi-permeable synthetic adhesive film (e.g., Tegaderm or Opsite) directly over the donor site. Serum and blood will accumulate beneath the film daily for the first 1 to 2 days; the film may be carefully aspirated or changed if leakage occurs. After approximately 7 to 10 days, the film is removed, revealing a re-epithelialized, satisfactorily healed surface.
Regardless of the dressing choice, mechanical shear must be avoided. Bed sheets and blankets should be kept strictly off the donor site using a rigid bed cradle support to prevent traumatic avulsion of the fragile, newly forming epithelium.
POSTOPERATIVE PROTOCOLS AND REHABILITATION
The success of any skin closure, graft, or flap relies heavily on the postoperative regimen.
Immobilization and Positioning
Strict immobilization of the affected joints is required to prevent shear forces that disrupt the delicate microvascular anastomoses or the inosculation of skin grafts. The hand should be elevated continuously above the level of the heart to promote venous and lymphatic drainage, thereby reducing edema and preventing flap congestion.
Flap Monitoring
For the first 48 to 72 hours, pedicled and free flaps require rigorous monitoring. Clinical assessment includes:
* Color: Should match the donor site. Pallor indicates arterial insufficiency; a blue/purple hue indicates venous congestion.
* Capillary Refill: Normal is 1-3 seconds. Brisk refill (<1 sec) suggests venous congestion; sluggish refill (>3 sec) suggests arterial inflow compromise.
* Turgor and Temperature: The flap should feel warm and soft. A swollen, tense flap is a surgical emergency requiring immediate evaluation of the pedicle.
Mobilization
Once graft take is confirmed (typically post-operative day 5-7) or flap inset is secure, a carefully guided rehabilitation program initiated by a certified hand therapist is crucial. Early, protected active range of motion prevents tendon adhesions and joint contractures, ensuring that the sophisticated soft tissue reconstruction translates into a functionally restored hand.
You Might Also Like