Masterclass in Functioning Neuromuscular Transfers and Vascularized Bone Grafts
Key Takeaway
Functioning free neuromuscular transfers restore critical motor deficits by transplanting a viable muscle unit using microvascular and microneural anastomoses. Success relies on meticulous preoperative planning, selecting a donor muscle with matching excursion, and precise intraoperative tensioning. This procedure is primarily indicated for replacing the flexor or extensor compartments of the forearm, facial expression muscles, and lower extremity defects, requiring advanced microsurgical proficiency and dedicated postoperative rehabilitation.
FUNCTIONING NEUROMUSCULAR TRANSFERS: PRINCIPLES AND EVOLUTION
The advent of functioning free neuromuscular transfers represents a pinnacle of reconstructive microsurgery, allowing surgeons to restore voluntary motor function to severely traumatized or anatomically deficient limbs. Tamai et al. first demonstrated the viability of this concept in a 1970 canine model, proving that a functioning neuromuscular unit could be transplanted using microvascular anastomoses and microneural repairs. Subsequent experimental and clinical validation by Kubo, Ikuta, Tsuge, Harii, and surgeons at the Sixth People’s Hospital in Shanghai cemented its clinical utility.
Pioneers such as Manktelow, Buncke, Gordon, and Schenck further refined the technique, demonstrating that transferred muscle not only survives ischemic transfer via microvascular revascularization but also regains voluntary contractility following microneural repair and subsequent reinnervation.
Today, functioning free muscle transfers (FFMT) are primarily utilized for the replacement of the flexor and extensor compartments of the forearm, facial reanimation (muscles of facial expression), and the reconstruction of the extensor compartment of the leg.
Indications and Donor Muscle Selection
The selection of a donor muscle is dictated by the biomechanical requirements of the recipient site, specifically the required strength, excursion, skin coverage needs, and the availability of a suitable motor nerve.
Commonly utilized donor muscles include:
* Gracilis: The workhorse for forearm flexor/extensor reconstruction and facial reanimation. It offers excellent excursion, reliable neurovascular anatomy, and minimal donor-site morbidity.
* Latissimus Dorsi: Preferred when massive power and a large skin paddle are required for extensive soft-tissue defects.
* Pectoralis Major: Offers substantial bulk and power, though donor-site cosmesis can be a limiting factor.
* Other Options: Rectus femoris, extensor digitorum brevis (for facial reanimation), serratus anterior, semitendinosus, tensor fasciae latae, and brachioradialis.
Surgical Warning: Manktelow’s Principle dictates that if a simpler, reliable procedure—such as a regional tendon transfer—can sufficiently restore the desired function, it must be utilized in preference to a free muscle transfer. FFMT is a salvage procedure reserved for cases where local reconstructive options are exhausted.
Advantages and Disadvantages
Advantages:
* Restoration of a profound functional deficit by transferring a viable, vascularized muscle under voluntary control.
* Minimal functional loss at the donor site when synergistic muscles remain intact.
* Ability to bring vascularized soft-tissue coverage (chimeric or myocutaneous flaps) to a scarred recipient bed.
Disadvantages:
* Sacrifice of a functioning native muscle.
* Prolonged reinnervation time (often 6 to 12 months before functional recovery is noted).
* Requires advanced microvascular and microneural proficiency.
* Extended operative times, necessitating two synchronized surgical teams.
PREOPERATIVE EVALUATION AND PLANNING
A high level of patient motivation and compliance is absolute; without dedicated postoperative rehabilitation, the transfer will fail to achieve functional excursion. Manktelow, Ikuta, Egloff, and Buncke emphasize a rigorous preoperative assessment.
Recipient Site Assessment
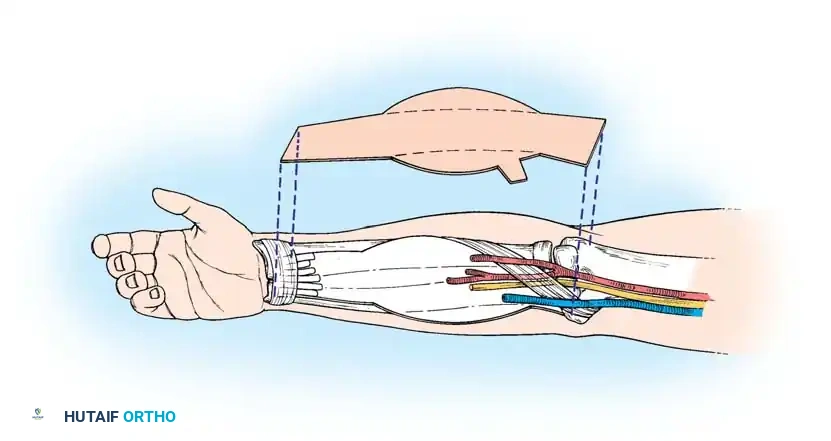
- Motor Nerve Availability: A single, undamaged, expendable motor nerve must be available at the recipient site. In forearm flexor reconstruction, branches of the median nerve (specifically those to the flexor digitorum superficialis) or the anterior interosseous nerve (AIN) are most frequently utilized.
- Nerve Viability: The status of the recipient nerve must be confirmed via clinical history, physical examination, electromyography (EMG), and, if necessary, preliminary surgical exploration.
- Vascular Assessment: Arteriography or high-resolution CT angiography is indicated to assess the patency and caliber of recipient vessels, particularly in limbs with a history of severe crush injury or ischemia.
- Joint Suppleness: The joints of the recipient extremity (elbow, wrist, and fingers) must possess a full, functional passive range of motion. A transferred muscle cannot overcome fixed joint contractures.
- Soft Tissue Envelope: Adequate skin coverage is mandatory. If the recipient bed is heavily scarred, a skin island must be harvested with the donor muscle to ensure tension-free closure over the neurovascular pedicle.
SURGICAL TECHNIQUE: FREE TRANSFER OF FUNCTIONING MUSCLE
The procedure demands meticulous coordination. Two surgical teams are essential to minimize total operative time and muscle ischemia. Prior to incision, ensure the patient is optimized with core temperature monitoring, a heating blanket, adequate padding of bony prominences, and an indwelling urinary catheter.
Preparation of the Forearm (Recipient Site)
- Incision and Exposure: Utilize an extensive curved or zigzag incision to provide wide exposure of the flexor or extensor compartments. If extensive scarring is present, dissection must proceed from normal, uninjured proximal/distal tissues into the zone of injury to protect the recipient neurovascular structures.
- Template Utilization: Fashion a sterile paper template based on preoperative planning to determine the exact location of the neurovascular pedicle and the dimensions of the required skin paddle.

- Vascular Preparation: Isolate the radial or ulnar artery (or a suitable large branch) to serve as the recipient artery. Ensure robust, pulsatile flow before committing to the transfer. For venous drainage, isolate the venae comitantes of the selected artery or robust superficial veins (e.g., cephalic or basilic systems).
- Neural Preparation: On the flexor aspect, isolate the anterior interosseous nerve (AIN) or median nerve branches. Exposure of the AIN may require a Z-lengthening of the pronator teres. On the extensor surface, isolate robust branches of the radial nerve.
- Tendon Preparation: Expose the recipient tendons (e.g., flexor digitorum profundus). Mobilize them extensively, freeing them from surrounding scar tissue to ensure frictionless gliding. Prepare the medial epicondyle (for flexor replacement) or lateral epicondyle (for extensor replacement) to serve as the proximal bony attachment for the transferred muscle.
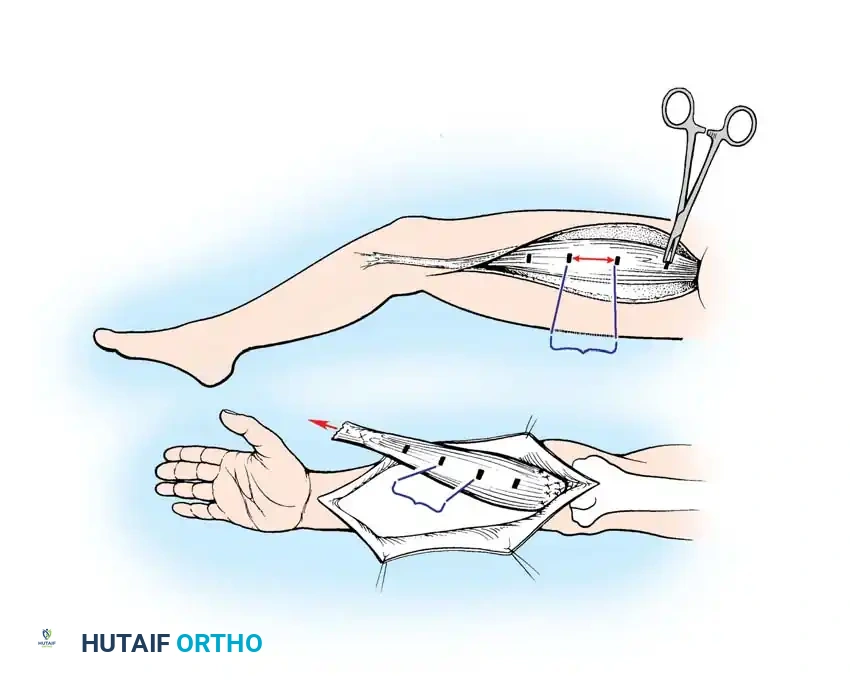
Harvesting and Tensioning the Donor Muscle (Manktelow Technique)
The donor muscle is dissected but left perfused on its native vascular pedicle until the recipient site is entirely prepared. This minimizes the critical ischemia time.
Clinical Pearl: The 5-cm Rule for Muscle Tensioning
Establishing the correct resting tension of the transferred muscle is the most critical biomechanical step. A muscle sutured too loosely will expend its excursion taking up slack; a muscle sutured too tightly will suffer ischemic contracture or fail to achieve full excursion.
- Determine Maximal Physiological Length: Position the donor extremity to stretch the muscle to its maximal physiological length. For the gracilis, this is achieved with the knee fully extended and the hip abducted. For the latissimus dorsi or pectoralis major, this requires maximal humeral abduction.
- Place Suture Markers: With the muscle at this maximal length, place permanent suture markers (or metal clips) on the surface of the muscle belly at exactly 5-cm intervals.
- Harvest: Once the markers are placed and the recipient site is ready, ligate the neurovascular pedicle, detach the origin and insertion, and transfer the muscle to the recipient limb.
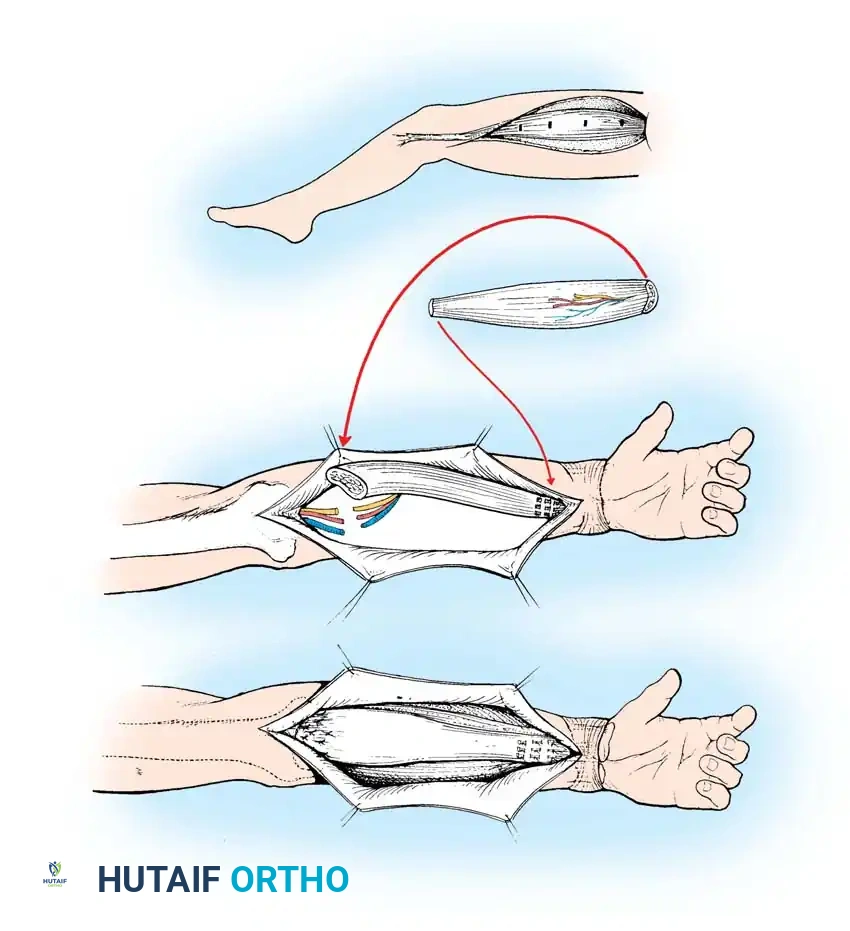
Inset and Microvascular Anastomosis
- Positioning: Transfer the muscle to the arm. It is often necessary to reverse the orientation of the muscle (origin becomes insertion) to optimally align the donor neurovascular pedicle with the recipient vessels and nerves.
- Proximal Attachment: Secure the origin of the muscle to the medial epicondyle and surrounding antebrachial fascia (for flexor reconstruction) or the lateral epicondyle (for extensor reconstruction). Suture it loosely to prevent displacement during microsurgery.
- Microvascular Repair: Position the muscle to allow tension-free arterial and venous anastomoses. While the order of repair (artery vs. vein) is surgeon-dependent, minimizing total ischemia time is paramount.
- Microneural Repair: The nerve repair must be positioned as close to the motor endplates of the muscle as possible (ideally within 2 to 3 cm) to minimize the distance regenerating axons must travel, thereby shortening the denervation period. Perform a tension-free, epineurial or grouped fascicular repair using 10-0 or 11-0 nylon under the operating microscope.
Distal Attachment and Tendon Weaving
- Restoring Tension: Apply distal traction to the transferred muscle until the previously placed suture markers are once again exactly 5 cm apart. This perfectly restores the muscle's maximal physiological length in its new bed.
- Tendon Weaving: Mark the intersection of the donor tendon and the recipient FDP tendons. Secure the recipient tendons to each other with side-to-side sutures to ensure a unified cascade. Weave the recipient tendons into the donor tendon (e.g., Pulvertaft weave) under the established tension.
- Closure: Cover the distal musculotendinous junction with the transferred skin paddle or a local flap. The proximal muscle belly can be covered with a split-thickness skin graft if necessary. Close all wounds loosely to prevent compartment syndrome or pedicle compression.
POSTOPERATIVE PROTOCOL AND REHABILITATION
The success of a functioning neuromuscular transfer is heavily reliant on postoperative care.
- Immediate Postoperative Phase (0-3 Weeks): Maintain systemic hydration and peripheral perfusion. The limb is immobilized in a plaster splint with the wrist and fingers moderately flexed to remove all tension from the musculotendinous repair. Anticoagulation protocols (e.g., aspirin, low-molecular-weight heparin, or dextran) are utilized based on institutional preference.
- Early Mobilization (3-8 Weeks): Gentle, controlled passive stretching exercises are initiated at 3 weeks to ensure tendon gliding and prevent adhesions. This continues until full passive range of motion is achieved.
- Reinnervation Phase (2-4 Months): As regenerating axons reach the motor endplates (typically at 2 to 4 months), early active muscle contraction becomes palpable. Gravity-eliminated active flexion is initiated.
- Strengthening Phase (6-12 Months): Once active motion is confirmed, progressive resistive exercises are incorporated. Biofeedback and electrical stimulation may be used as adjuncts.
- Maturation: Manktelow notes that maximum muscle strength and excursion typically stabilize 2 to 3 years post-transplantation.
FREE VASCULARIZED BONE GRAFTS
While functioning muscle transfers restore dynamic motion, massive segmental skeletal defects require structural reconstruction. Conventional non-vascularized autografts (e.g., iliac crest) are limited by creeping substitution, making them prone to resorption, stress fractures, and failure in defects exceeding 6 cm, particularly in poorly vascularized or irradiated beds.
Free vascularized bone grafts (FVBG), pioneered by Taylor, revolutionize the treatment of large skeletal defects, congenital pseudarthrosis, and tumor resections by transferring living bone with its intrinsic endosteal and periosteal blood supply intact.
Advantages of Vascularized Bone Grafts (Taylor's Principles)
- Primary Bone Healing: The graft heals to the recipient bed via primary osteoblastic activity rather than creeping substitution, allowing for rapid incorporation.
- Mechanical Superiority: A vascularized tubular bone (like the fibula) maintains its structural integrity and is biomechanically stronger than an onlay cortical graft, which undergoes an initial phase of necrotic weakening.
- One-Stage Reconstruction: Allows for immediate structural bridging of massive defects.
- Infection Resistance: The transfer of well-vascularized tissue significantly improves the eradication of chronic osteomyelitis in the recipient bed.
- Fail-Safe Mechanism: If the microvascular anastomosis thromboses, the transferred bone simply acts as a traditional, non-vascularized cortical bone graft.
Disadvantages and Limitations
- Complexity: Requires prolonged operative times and advanced microsurgical expertise.
- Donor Site Morbidity: Harvesting the fibula can lead to ankle instability, flexor hallucis longus weakness, or peroneal nerve neuropraxia.
- Monitoring: The patency of the buried intraosseous anastomosis is difficult to monitor postoperatively unless a cutaneous skin paddle is included.
- Vessel Sacrifice: Requires the sacrifice of a major vascular axis in both the donor and recipient limbs.
Donor Site Selection
The fibula and the iliac crest are the premier donor sites for vascularized bone grafts, each offering distinct anatomical advantages.
The Vascularized Fibula
For the majority of orthopedic long-bone reconstructions, the fibula is the gold standard.
* Anatomy: It provides a straight, dense cortical strut. Up to 26 cm of bone can be harvested from an adult.
* Vascular Pedicle: Supplied by the peroneal artery (1.5 to 2.5 mm in diameter) and its two venae comitantes (2 to 3 mm in diameter). The pedicle length can range from 1 to 5 cm.
* Versatility: It can be harvested with a skin paddle (osteocutaneous flap) based on septocutaneous perforators, or with a portion of the soleus or flexor hallucis longus muscle. In pediatric patients, the proximal fibular physis can be transferred based on the anterior tibial recurrent artery to allow for continued longitudinal growth.
* Surgical Considerations: The dissection is relatively straightforward, though meticulous care must be taken to protect the common peroneal nerve proximally and to preserve the distal 6 to 8 cm of the fibula to maintain ankle mortise stability.
The Vascularized Iliac Crest
- Anatomy: Provides a curved, corticocancellous block of bone, ideal for metaphyseal defects, pelvic reconstruction, or maxillofacial surgery.
- Dimensions: A length of 8 to 10 cm can be reliably harvested.
- Vascular Pedicle: Can be based on either the superficial circumflex iliac artery (SCIA, 0.5 to 3 mm diameter) or the deep circumflex iliac artery (DCIA), paired with the superficial inferior epigastric veins or deep venae comitantes.
- Advantages: The rich cancellous bone promotes rapid union at the osteotomy sites, and it can be easily contoured. It is frequently harvested as a massive osteocutaneous flap for complex composite defects.
You Might Also Like