Operative Management of Bone Sarcomas and Multiple Myeloma
Key Takeaway
Malignant fibrous histiocytoma, fibrosarcoma, and multiple myeloma represent aggressive primary bone malignancies requiring multidisciplinary intervention. This guide details the evidence-based surgical management of these lesions, emphasizing wide resection principles for sarcomas and prophylactic stabilization techniques for myeloma-induced pathological fractures. Key insights include histopathological differentiation, neoadjuvant protocols, and step-by-step operative approaches to optimize patient survival and functional outcomes.
MALIGNANT FIBROUS HISTIOCYTOMA AND FIBROSARCOMA
Although malignant fibrous histiocytoma (MFH)—now frequently classified in modern pathology as Undifferentiated Pleomorphic Sarcoma (UPS) of bone—and fibrosarcoma are historically described in the literature as separate entities, their clinical distinction is often arbitrary. The presentation, prognosis, and treatment algorithms for these two entities are virtually identical, warranting a unified discussion regarding their operative and oncological management.
Malignant fibrous histiocytoma and fibrosarcoma of bone constitute approximately 3% to 5% of all primary malignant bone tumors. Excluding the first decade of life, they occur at any age with comparable frequency, and males and females are affected equally. While there is a slight predilection for the distal metaphysis of the femur or the proximal metaphysis of the tibia, any osseous structure may be involved.
Pathogenesis and Predisposing Conditions
A critical clinical feature of these sarcomas is their propensity to arise as secondary malignancies. Approximately 25% of these tumors are secondary to a preexisting bone abnormality.
Clinical Pearl: Always maintain a high index of suspicion for secondary sarcomatous transformation when a patient with a known benign osseous condition presents with new-onset, unremitting pain or rapid radiographic changes.
The most commonly reported predisposing conditions include:
* Paget Disease of Bone: Sarcomatous degeneration occurs in approximately 1% of Paget's patients, often presenting with a highly aggressive clinical course.
* Radiation Osteitis: Post-radiation sarcomas typically arise in the radiation field after a latency period of 5 to 15 years.
* Bone Infarction: Chronic medullary infarcts can undergo malignant transformation, typically at the periphery of the infarct.
* Giant Cell Tumor (GCT): Malignant transformation can occur, particularly following prior radiation therapy for the GCT.
* Dedifferentiated Chondrosarcoma: MFH or fibrosarcoma may represent the high-grade, non-cartilaginous component of a dedifferentiated chondrosarcoma.
Clinical Presentation and Advanced Imaging
As with other high-grade bone sarcomas, patients typically present with deep, aching pain that is worse at night and not relieved by rest. Due to the purely lytic and destructive nature of these tumors, patients have a significantly higher incidence (approximately 20%) of pathological fracture at presentation compared to osteosarcoma.
Radiographically, these tumors exhibit a highly aggressive appearance. They are typically purely lytic with indistinct, permeative borders (a wide zone of transition). They may appear as an area of aggressive bone destruction adjacent to an otherwise typical area of Paget disease or a calcified bone infarct. Periosteal reaction (such as Codman's triangle or a "sunburst" pattern) is notably absent unless a pathological fracture has already occurred.

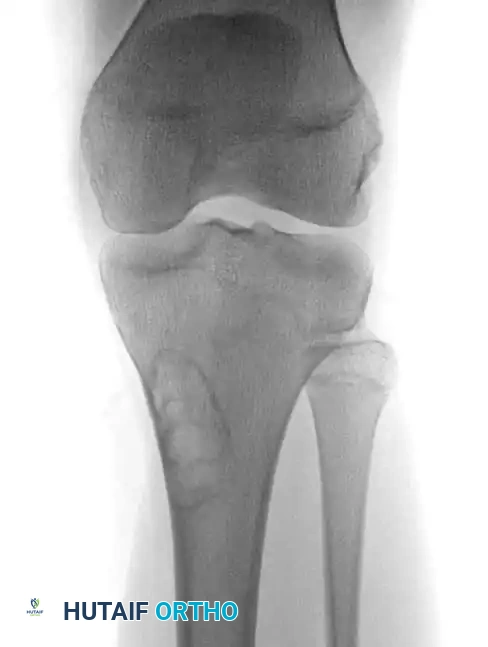
Anteroposterior and lateral radiographs of the knee of a 58-year-old man with malignant fibrous histiocytoma showing an aggressive area of bone destruction adjacent to multiple bone infarcts. Despite this appearance, the patient was treated for 6 months for degenerative joint disease before referral.
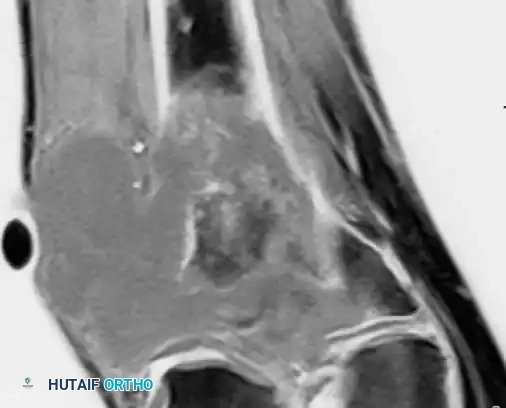
Magnetic Resonance Imaging (MRI) is the gold standard for local staging. It precisely delineates the intraosseous extent of the tumor, "skip" metastases within the same bone, and the degree of extraosseous soft-tissue extension.
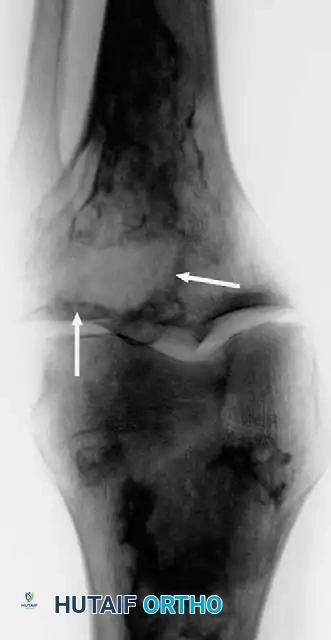
MRI demonstrating extensive bone destruction and a large soft-tissue mass adjacent to a bone infarct.
Histopathology and Diagnostic Criteria
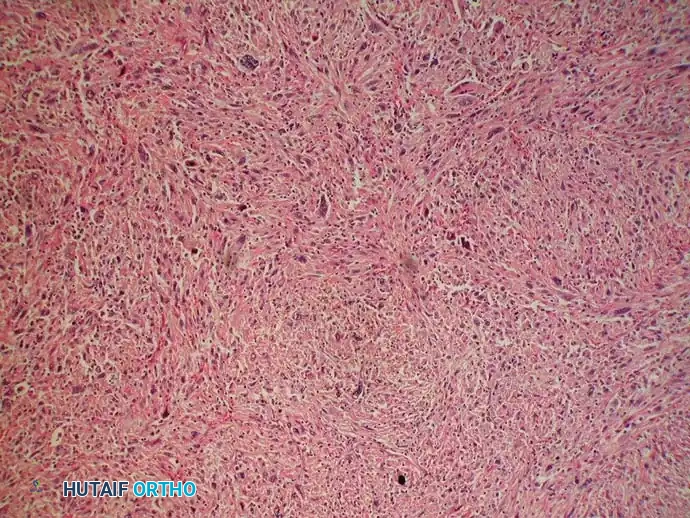
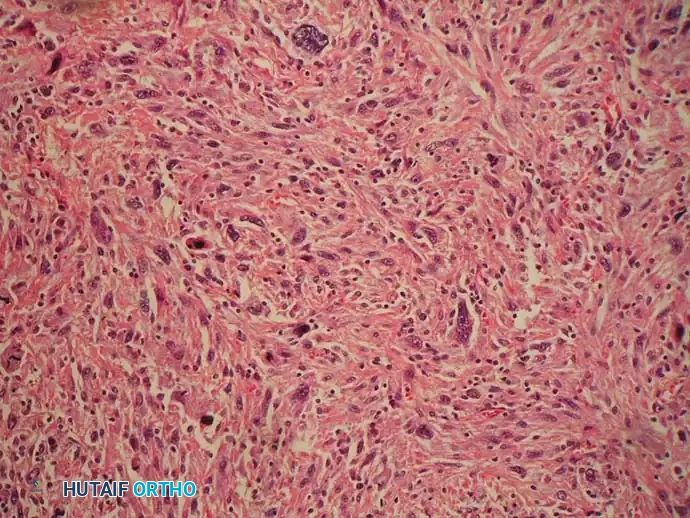
Histologically, the classic appearance of malignant fibrous histiocytoma is a high-grade spindle cell sarcoma arranged in a storiform or cartwheel pattern. However, the cellular appearance is highly pleomorphic. Tumors may exhibit benign and malignant multinucleated giant cells, cells with a histiocytic appearance (large, indented nuclei with abundant, well-defined cytoplasm), cells with foamy cytoplasm, inflammatory cells, and variable amounts of fibrosis.
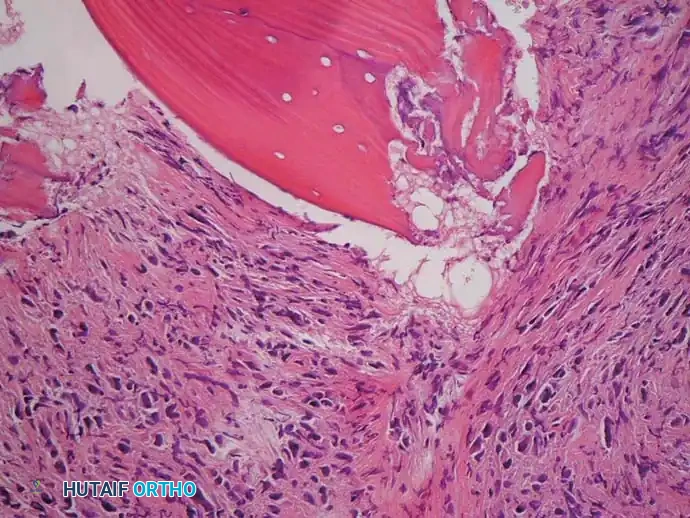
Low-power and high-power photomicrographs showing pleomorphic malignant cells arranged in a storiform pattern adjacent to a bone infarct.
The classic appearance of fibrosarcoma is that of a spindle cell neoplasm arranged in a herringbone pattern. Low-grade fibrosarcomas may exhibit abundant collagen production, whereas high-grade tumors are densely cellular with high mitotic rates.
Surgical Warning: The absolute defining characteristic of both MFH and fibrosarcoma is the lack of osteoid production by the malignant cells. Even a microscopic amount of malignant osteoid production mandates a change in diagnosis to osteosarcoma, which may alter the chemotherapy protocol.
Surgical Management and Operative Principles
At most tertiary orthopedic oncology centers, the treatment of MFH and fibrosarcoma of bone mirrors the treatment of osteosarcoma. Most patients with high-grade lesions are treated with neoadjuvant (preoperative) chemotherapy, followed by surgical resection, and concluding with adjuvant chemotherapy.
Compared with osteosarcoma, MFH may be more radiosensitive. Radiation therapy may be beneficial for patients with positive resection margins, intraoperative tumor contamination, or in surgically inaccessible locations (e.g., specific spinal or pelvic lesions).
Step-by-Step Surgical Approach: Wide Resection of the Distal Femur
When limb-salvage is feasible (intact neurovascular bundle, adequate soft-tissue coverage, and ability to achieve wide margins), a distal femoral resection with endoprosthetic reconstruction is performed.
- Positioning and Setup: The patient is placed supine. A sterile tourniquet is applied but not inflated unless catastrophic hemorrhage occurs, as tourniquet ischemia can obscure the assessment of viable margins.
- Incision and Exposure: A standard anterior or anteromedial utility incision is utilized, incorporating the previous biopsy tract (which must be excised en bloc with the tumor).
- Soft Tissue Dissection: The vastus medialis is elevated. The superficial femoral artery and vein are identified at the adductor hiatus and meticulously dissected free from the tumor pseudocapsule.
- Osteotomy: Based on preoperative MRI measurements, the femoral osteotomy site is marked (typically 3-5 cm proximal to the proximal-most extent of the marrow signal abnormality). The osteotomy is performed using an oscillating saw. A marrow sample from the remaining proximal femur is sent for frozen section to confirm a negative intramedullary margin.
- Joint Resection: The knee joint is entered. Depending on intra-articular involvement, an extra-articular resection (removing the entire knee joint en bloc) or an intra-articular resection (sacrificing the cruciates but sparing the proximal tibia) is performed.
- Reconstruction: A modular distal femoral replacement (megaprosthesis) is assembled. The femoral stem is cemented or press-fit into the proximal femur, and the tibial component is fixed to the proximal tibia.
- Closure: Meticulous hemostasis is achieved. The extensor mechanism is reconstructed, often requiring a medial gastrocnemius rotational flap if significant anterior soft tissue was resected.
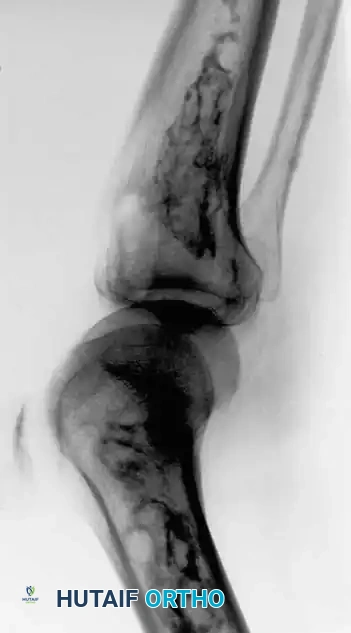
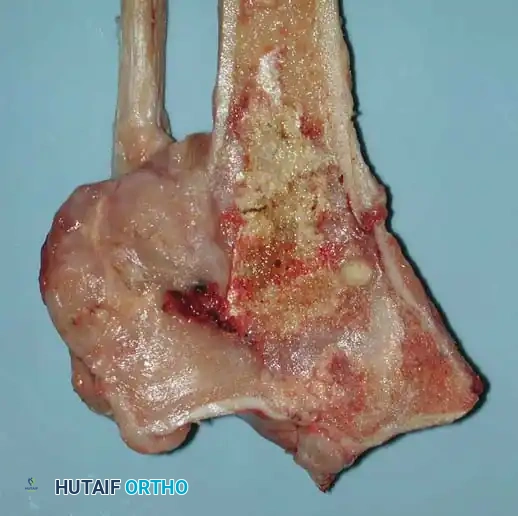
Gross photograph of the tumor after an above-knee amputation, demonstrating the extensive, destructive nature of the lesion when limb salvage is contraindicated.
Prognosis
The prognosis is heavily dependent on several factors:
* Presence or absence of distant metastases at presentation (most commonly pulmonary).
* Size and location of the tumor (axial vs. appendicular).
* Ability of the surgeon to achieve wide, negative margins.
* Histological grade of the tumor.
* Histological response to neoadjuvant chemotherapy (determined by the percentage of tumor necrosis in the resected specimen; >90% necrosis indicates a good response).
Overall, the 5-year survival rate for patients with high-grade tumors of the extremities without metastases at presentation is approximately 65%.
MULTIPLE MYELOMA AND PLASMACYTOMA
Multiple myeloma is the most common primary malignancy of bone, representing more than 40% of all primary bone cancers. It is a systemic hematological malignancy characterized by the monoclonal proliferation of plasma cells. Its peak incidence is in the fifth to seventh decades of life, with a 2:1 male predominance.
Systemic Manifestations and Clinical Presentation
Multiple myeloma and metastatic carcinoma must be the primary differential diagnoses for any patient older than age 40 presenting with a new, destructive bone lesion.
Bone pain is the most common presenting complaint. However, unlike primary bone sarcomas, multiple myeloma is a systemic disease. Patients frequently present with the classic "CRAB" criteria:
* Calcium elevation (Hypercalcemia)
* Renal failure (due to light chain cast nephropathy)
* Anemia (and thrombocytopenia due to marrow replacement)
* Bone lesions (lytic)
Peripheral neuropathy is also common, particularly with the osteosclerotic variant of multiple myeloma (POEMS syndrome). Symptoms are usually of short duration due to the aggressive nature of the disease, and pathological fractures are highly prevalent. The axial skeleton is most commonly involved, with the spine being the most frequent location, followed by the ribs, pelvis, and proximal appendicular skeleton.
Radiographic Evaluation and Staging
Radiographically, multiple myeloma classically appears as multiple, "punched-out," sharply demarcated, purely lytic lesions without any surrounding reactive sclerosis.
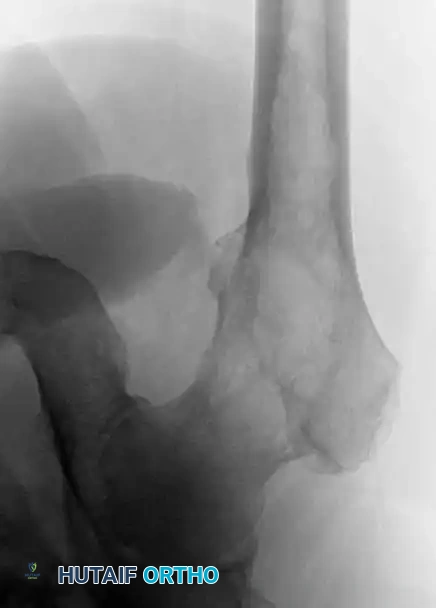
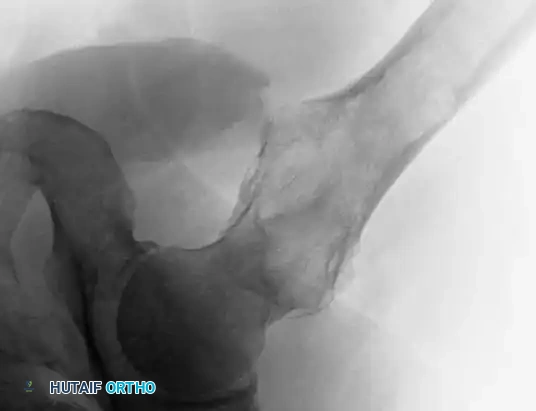
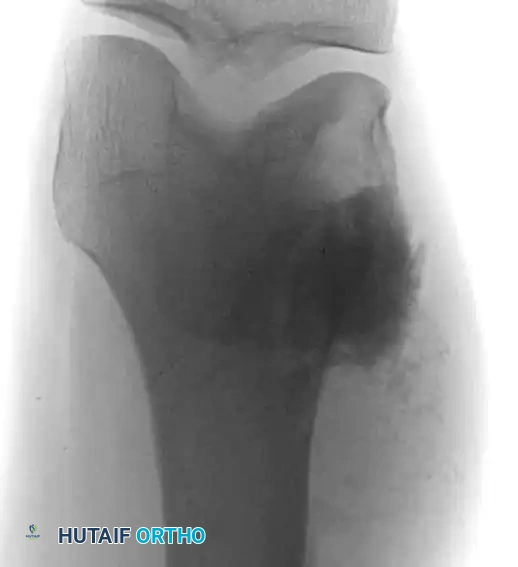
Anteroposterior and lateral radiographs of the proximal femur of a 61-year-old man with multiple myeloma showing multiple lytic lesions.
Because myeloma cells secrete osteoclast-activating factors (like RANKL) and suppress osteoblast activity, there is a profound lack of reactive bone formation. Consequently, most myeloma lesions are negative (cold) on a technetium-99m bone scan. A skeletal survey (plain radiographs of the entire skeleton) or whole-body low-dose CT is the preferred imaging modality for staging.
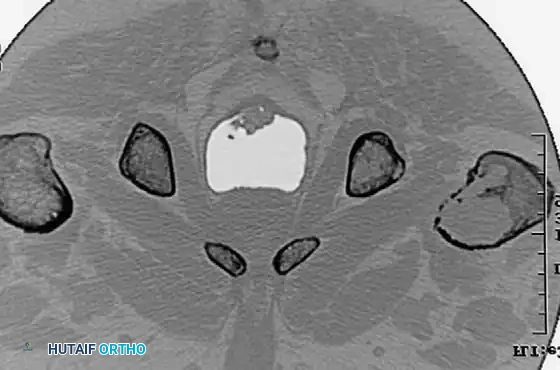
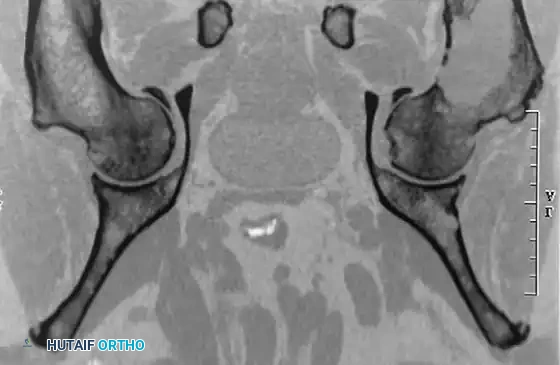
CT scans demonstrating the extensive cortical destruction of the lesions, highlighting the imminent biomechanical failure and the absolute need for prophylactic fixation.
Histopathology and Laboratory Diagnostics
The diagnosis is usually confirmed by serum and urine protein electrophoresis (SPEP/UPEP) and immunofixation, which demonstrate a monoclonal gammopathy (M-spike). A bone marrow biopsy is required for definitive staging.
Histologically, multiple myeloma appears as dense sheets of malignant plasma cells. These are small, round blue cells characterized by "clock face" (eccentric) nuclei and abundant cytoplasm with a prominent perinuclear clearing or "halo" (representing the Golgi apparatus). Amyloid production can be abundant.
Pathology Pearl: With the exception of patients on long-term hemodialysis, the presence of amyloid in bone tissue is almost pathognomonic for multiple myeloma.
Immunohistochemistry is vital for differentiating plasmacytoma from chronic osteomyelitis or lymphoma:
* Plasmacytoma/Myeloma: Exhibits monoclonal $\kappa$ or $\lambda$ light chains, stains positive for CD138 and the natural killer antigen CD56. Negative for CD45.
* Chronic Osteomyelitis: Plasma cells are polyclonal.
* Lymphoma: Cells usually stain positive for CD45 (leukocyte common antigen) and CD20 (a B-cell marker).
Orthopaedic Surgical Intervention: Pathological Fractures
The primary treatment for multiple myeloma is systemic chemotherapy (e.g., proteasome inhibitors, immunomodulatory drugs) and autologous stem cell transplantation. Symptomatic bone lesions usually respond rapidly to localized radiation therapy.
The orthopaedic surgeon is most commonly consulted to treat impending or actual pathological fractures, particularly of the spine, acetabulum, proximal femur, or proximal humerus.
Surgical Principle: Because patients with multiple myeloma often have a limited life expectancy and are systemically compromised, the surgical goal is not wide oncological resection. The goal is to perform a durable construct that allows for immediate, unrestricted weight-bearing and rapid return to function.
Step-by-Step: Prophylactic Cephalomedullary Nailing of the Proximal Femur
For extensive lytic lesions of the proximal femur (as seen in the provided CT scans), prophylactic stabilization is indicated to prevent catastrophic fracture.
- Positioning: The patient is placed supine on a fracture table. Gentle traction is applied.
- Approach: A minimally invasive lateral approach proximal to the greater trochanter is utilized.
- Tumor Debulking (Optional but Recommended): If the lesion is large, a small lateral cortical window can be made to curette the gross tumor volume. This reduces the tumor burden and provides a void for cement augmentation.
- Canal Preparation: A guide wire is passed down the femoral canal. The canal is reamed sequentially. Caution: Reaming in myeloma bone can easily cause iatrogenic fracture due to profound cortical thinning.
- Cement Augmentation: Polymethylmethacrylate (PMMA) bone cement is injected into the lytic defect in a doughy state.
- Nail Insertion: A long cephalomedullary nail is immediately passed through the curing cement to span the entire femur.
- Locking: Proximal reconstruction screws are placed into the femoral head, and distal interlocking screws are placed to control rotation.
If the articular surface or femoral neck is completely destroyed, a cemented bipolar hemiarthroplasty or total hip arthroplasty using a long-stem revision component is
Associated Surgical & Radiographic Imaging
You Might Also Like