Legg-Calvé-Perthes Disease: Pathogenesis, Classification, and Surgical Management
Key Takeaway
Legg-Calvé-Perthes disease is an idiopathic avascular necrosis of the pediatric capital femoral epiphysis. Successful management relies on early diagnosis, accurate classification using the Herring Lateral Pillar system, and achieving spherical femoral head remodeling through biological plasticity. Treatment ranges from observation to surgical containment via femoral varus derotational osteotomy or pelvic osteotomies, dictated by the patient's age at onset and the extent of epiphyseal necrosis.
LEGG-CALVÉ-PERTHES DISEASE
Legg-Calvé-Perthes disease (LCPD) represents a complex, idiopathic osteonecrosis (avascular necrosis) of the capital femoral epiphysis in the pediatric population. The condition is characterized by a temporary disruption of blood supply to the femoral head, leading to an ischemic cascade of bone death, subsequent fragmentation, and eventual revascularization and remodeling. The overarching goal of the orthopedic surgeon is to prevent femoral head deformation during the fragile fragmentation phase, ensuring a congruent, spherical joint at skeletal maturity.
Etiology and Pathogenesis
The precise etiology of Legg-Calvé-Perthes disease remains elusive and has provoked considerable academic controversy. Historically, the prevailing hypothesis suggested that an inherited thrombophilia promoted thrombotic venous occlusion within the femoral vein, precipitating bone death in the femoral head. However, rigorous contemporary investigations have largely debunked this theory.
Numerous recent studies, notably those by Hresko et al., have failed to identify a statistically significant correlation between inherited hypercoagulability or protein C deficiency and the onset of LCPD. Current evidence strongly indicates that inherited thrombophilia is not associated with the osteonecrosis seen in this disease. While research into the microvascular anatomy of the medial circumflex femoral artery continues, it is now widely accepted that systemic coagulation disorders are not primary etiologic factors.

Clinical Presentation and Differential Diagnosis
Differentiating the acute symptoms of Legg-Calvé-Perthes disease from an irritable hip caused by transient synovitis is a common and critical clinical challenge. Both conditions present with a limp, hip or knee pain, and restricted range of motion (particularly internal rotation and abduction).
Erken and Katz delineated several distinguishing epidemiological and clinical characteristics between the two entities:
* Gender Predilection: Irritable hip syndrome occurs twice as frequently in boys as in girls. In contrast, LCPD exhibits a stronger male predominance, occurring three times more frequently in boys.
* Age of Onset: The average age for transient synovitis is 3 years, whereas the classic presentation of LCPD peaks at an average age of 7 years.
* Symptom Duration: Children with transient synovitis typically experience symptoms for an average of 6 days. Conversely, children presenting with LCPD report an average symptom duration of 6 weeks prior to diagnosis.
Further diagnostic nuances have been identified in the literature. Eggl et al. observed that ultrasonographic evidence of capsular distention persisting longer than 6 weeks is highly predictive of the onset of LCPD. Keenan and Clegg demonstrated that a delay in bone age of more than 2 years is a reliable indicator of the early phases of LCPD. Consequently, a plain radiograph of the left wrist serves as a highly effective screening tool to stratify patients requiring advanced imaging (MRI) or serial radiographic follow-up.
Clinical Pearl: Meyer dysplasia is a critical differential diagnosis that can easily be mistaken for LCPD, potentially leading to unnecessary surgical interventions. Rowe et al. noted that Meyer dysplasia predominantly affects boys younger than 4 years old and is bilateral in 59% of cases. Radiographically, it presents with delayed, small, or "cracked" ossification centers without the condensation, subchondral fractures, fragmentation, or subluxation pathognomonic of LCPD.
Additionally, children with perinatal human immunodeficiency virus (HIV) possess an elevated risk for osteonecrosis of the hip. LCPD must be highly suspected in an HIV-positive child presenting with a limp. The exact mechanism—whether attributable to the virus itself, associated opportunistic complications, or antiretroviral therapies—remains under investigation.
Imaging Evaluation
Historically, plain radiographic changes lagged behind clinical symptoms by 6 weeks or more. Today, advanced imaging allows for rapid, definitive diagnosis and precise preoperative planning.
Plain Radiography:
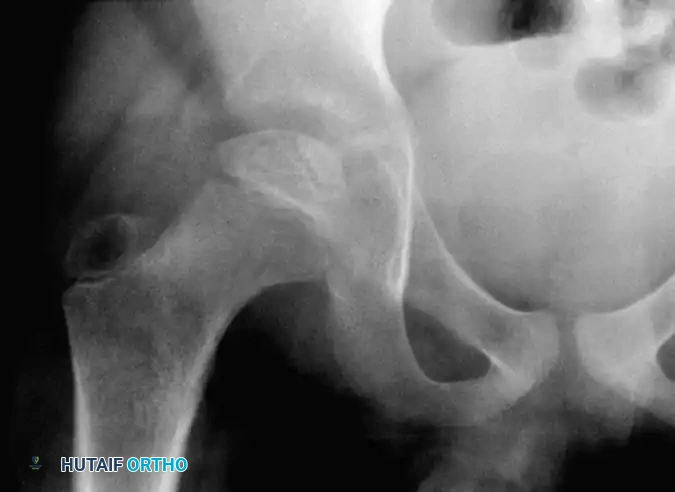
Standard anteroposterior (AP) and frog-leg lateral radiographs of the pelvis remain the baseline. Early signs include a smaller ossification center, increased radiodensity (sclerosis), and widening of the medial joint space (Waldenström sign).
Magnetic Resonance Imaging (MRI):
MRI has become the gold standard at our institution for determining the extent of epiphyseal involvement, classifying the disease, and formulating treatment plans. Henderson et al. demonstrated that MRI is vastly superior to plain radiographs and bone scintigraphy in delineating the exact location and extent of necrosis during the evolutionary stages of LCPD.
Song et al. utilized MRI to elucidate the etiology of medial joint space widening seen on plain films. They found the space is filled with:
1. Overgrown cartilage in the initial stage.
2. Overgrown cartilage combined with a true widened medial joint space during the fragmentation stage.
3. A widened true medial joint space during the healing stage.
During healing, the apparent widening decreases as the overgrown cartilage ossifies, despite the presence of lateral subluxation secondary to coxa magna. Thus, true lateral subluxation of the original femoral head is primarily indicated by medial joint space widening during the fragmentation stage.
Classification Systems
Accurate classification is paramount for determining prognosis and guiding surgical intervention. The primary aim of treatment is the containment of the femoral head within the acetabulum. As described by Salter, containment leverages "biological plasticity," allowing the necrotic femoral head to be molded into a concentric, spherical shape by the spherical acetabulum during the revascularization phase.
1. Catterall Classification
Catterall classified LCPD based on the radiographic volume of epiphyseal involvement:
* Group I: Partial head involvement (less than 50%).
* Group II & III: More than 50% involvement with sequestrum formation.
* Group IV: Total epiphyseal involvement.
Catterall also identified "Head-at-Risk" signs, which correlate with poor outcomes and indicate impending deformation:
1. Lateral subluxation of the femoral head.
2. Speckled calcification lateral to the capital epiphysis.
3. Diffuse metaphyseal reaction (metaphyseal cysts).
4. A horizontal physis.
5. Gage Sign: A radiolucent V-shaped defect in the lateral epiphysis and adjacent metaphysis.
2. Salter-Thompson Classification
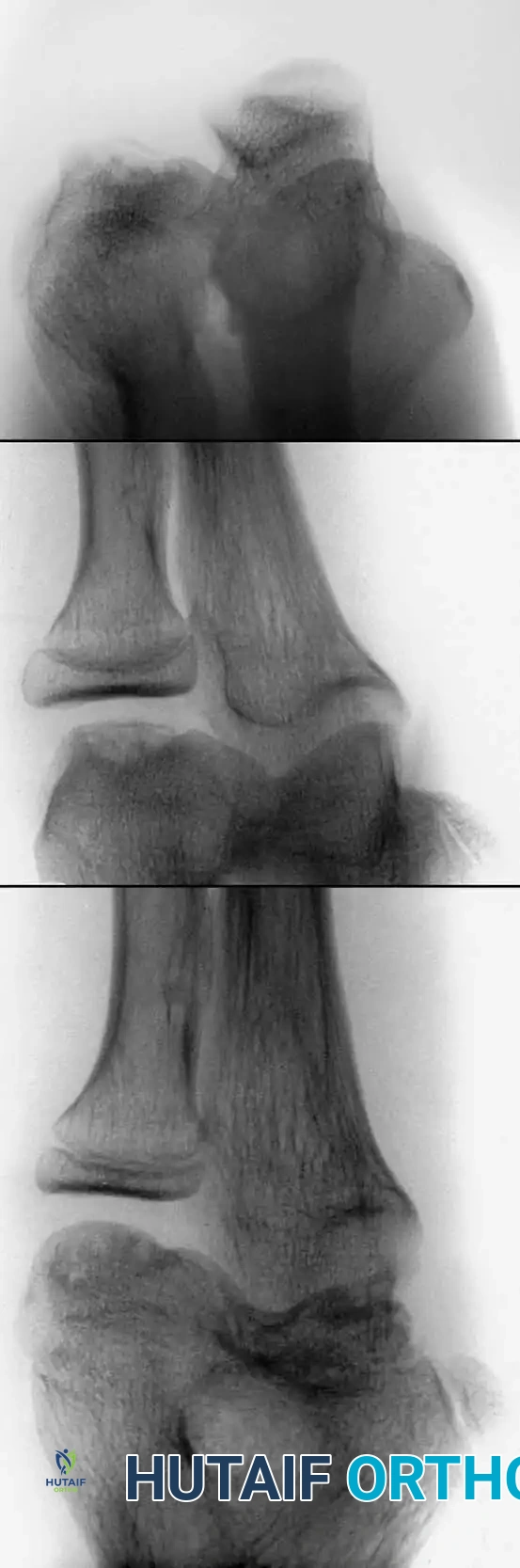
Salter and Thompson simplified early classification by measuring the extent of the subchondral fracture (crescent sign) in the superolateral femoral head on frog-leg lateral radiographs.
* Type A: Fracture line involves less than 50% of the superior dome (Good prognosis).
* Type B: Fracture line involves more than 50% of the superior dome (Fair/Poor prognosis, often requiring innominate osteotomy).
While Mukherjee and Fabry validated its accuracy, our institutional experience notes that subchondral fractures are visible early in only one-third of patients, limiting its universal application.
3. Herring Lateral Pillar Classification
The Herring classification is currently the most widely utilized and prognostically accurate system. It evaluates the height of the lateral pillar of the capital femoral epiphysis on an AP radiograph during the early fragmentation phase.
* Group A: No involvement of the lateral pillar (100% height maintained). Uniformly good outcomes.
* Group B: Greater than 50% of lateral pillar height maintained. Good outcomes if onset is < 9 years of age; less favorable if > 9 years.
* Group C: Less than 50% of lateral pillar height maintained. Worst outcomes, typically resulting in aspherical femoral heads regardless of age.
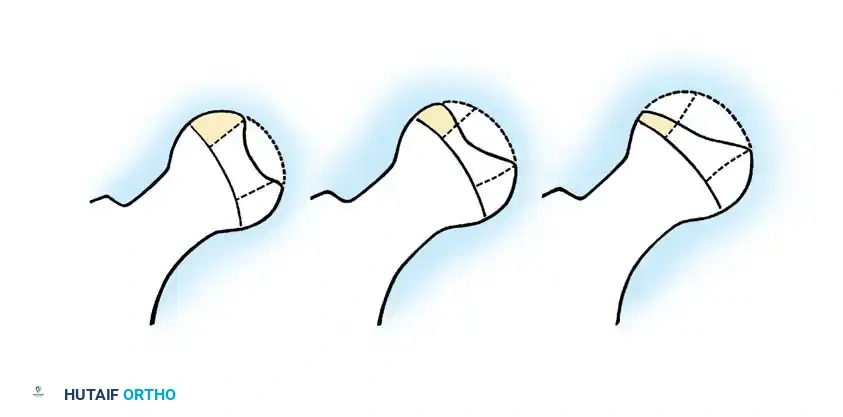
Ritterbusch et al. and Ippolito et al. confirmed that the Lateral Pillar classification boasts superior interobserver reliability and is a more accurate predictor of the final Stulberg outcome than the Catterall system.
Bilateral Involvement
Bilateral LCPD occurs in 10-15% of cases and must be carefully distinguished from Multiple Epiphyseal Dysplasia (MED). Skeletal surveys and bone age assessments are critical here.
While early literature suggested bilateral cases were inherently more severe, Guille et al. evaluated 83 patients and found that the development of bilateral LCPD represents two independent ischemic events. The second hip affected is generally less severe (78% of cases), and the Stulberg outcome is independent of bilaterality. Unilateral disease does not predispose the contralateral hip to a more severe disease course.
Principles of Treatment and Biomechanics
A 22-year longitudinal follow-up of 80 children at our institution yielded critical insights:
1. 84% of patients achieve good results with noncontainment methods.
2. Satisfactory clinical function is often maintained despite imperfect radiographic appearance.
3. Poor results are almost exclusively seen in patients with Catterall III/IV (Herring B/C) involvement who are older than 6 years at onset.
The fundamental biomechanical principle of treatment is containment. As the child ages, the remodeling capability of the acetabulum decreases (Madan et al.). In younger children, the acetabulum can remodel to fit a deformed femoral head. In older children, a deformed head will subluxate laterally, impinging on the lateral acetabular margin.
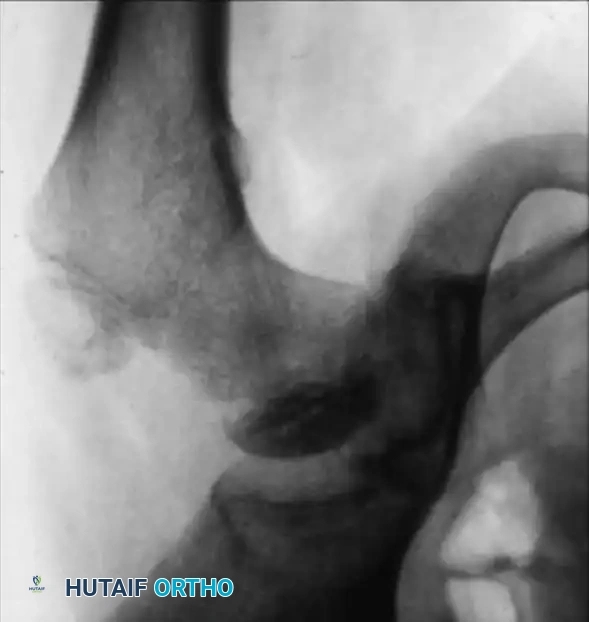
Surgical Warning: Hinged Abduction
Reinker described "hinged abduction," a severe complication where the extruded, enlarged anterolateral portion of the femoral head impinges against the lateral acetabular rim during abduction. Instead of gliding concentrically, the hip hinges open medially. Failure of the lateral epiphysis to slide under the acetabulum on an internally rotated/abducted radiograph is diagnostic. This requires immediate intervention, often a valgus osteotomy, to prevent catastrophic joint destruction.
Non-Operative Management
For children under 6 years of age, or those with Herring Group A disease, non-operative management is the standard of care. This includes observation, activity modification, and physical therapy to maintain range of motion.
Historically, abduction bracing and Petrie casts were utilized extensively to enforce containment.
While Petrie casts (bilateral long-leg casts with an abduction bar) are still occasionally used to regain motion in an irritable hip prior to surgery, prolonged bracing has largely fallen out of favor due to poor patient compliance and lack of definitive evidence proving superiority over natural history in severe cases.
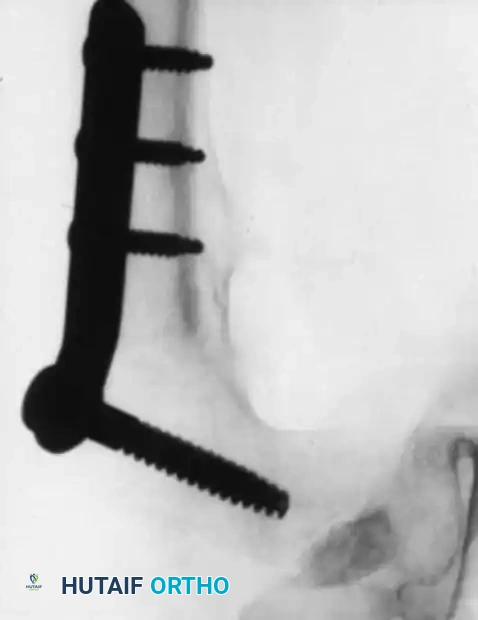
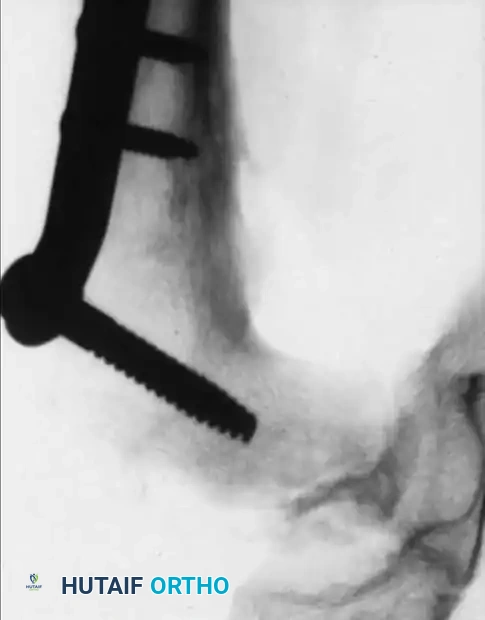
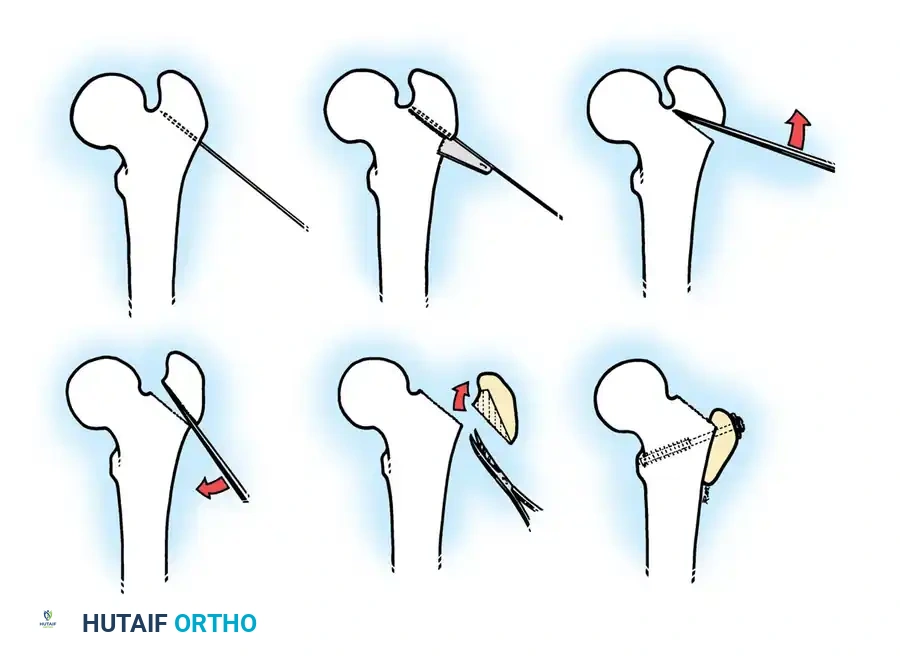
Surgical Management: Femoral Varus Derotational Osteotomy (VDRO)
Surgical containment is indicated for children over the age of 8 with Herring Group B or B/C disease, or any child exhibiting progressive loss of containment and "head-at-risk" signs.
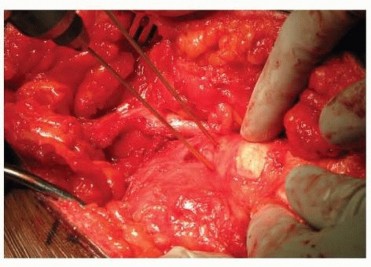
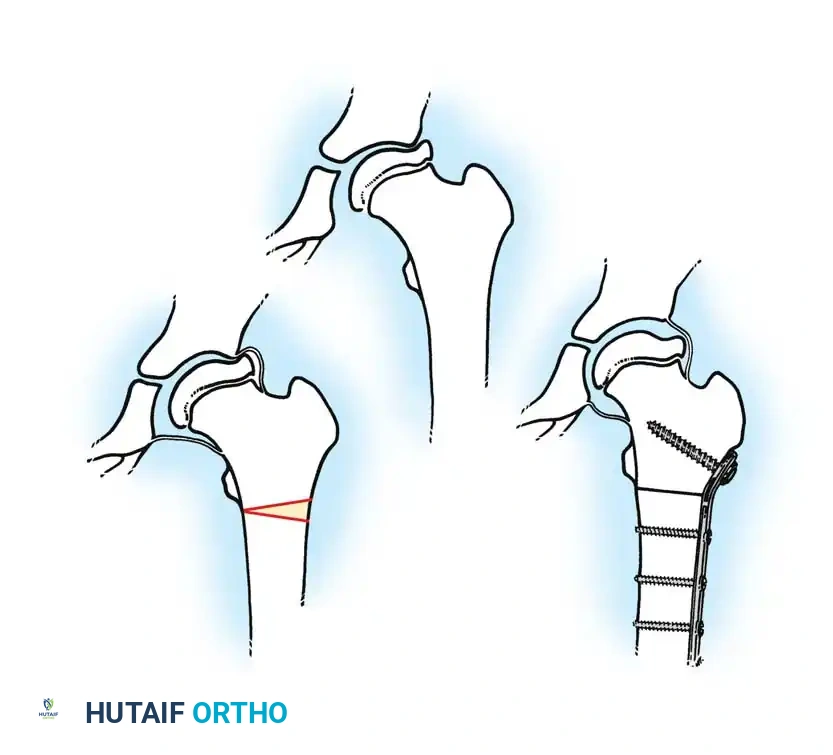
The Femoral Varus Derotational Osteotomy (VDRO) is a powerful technique that redirects the vulnerable anterolateral portion of the femoral head deeply into the acetabulum, relieving it from the deforming forces of the acetabular rim.

Step-by-Step Surgical Technique:
1. Positioning: The patient is placed supine on a radiolucent Jackson table. A bump is placed under the ipsilateral hip.
2. Approach: A standard lateral approach to the proximal femur is utilized. The fascia lata is incised, and the vastus lateralis is elevated off the intermuscular septum and reflected anteriorly to expose the proximal femoral shaft.
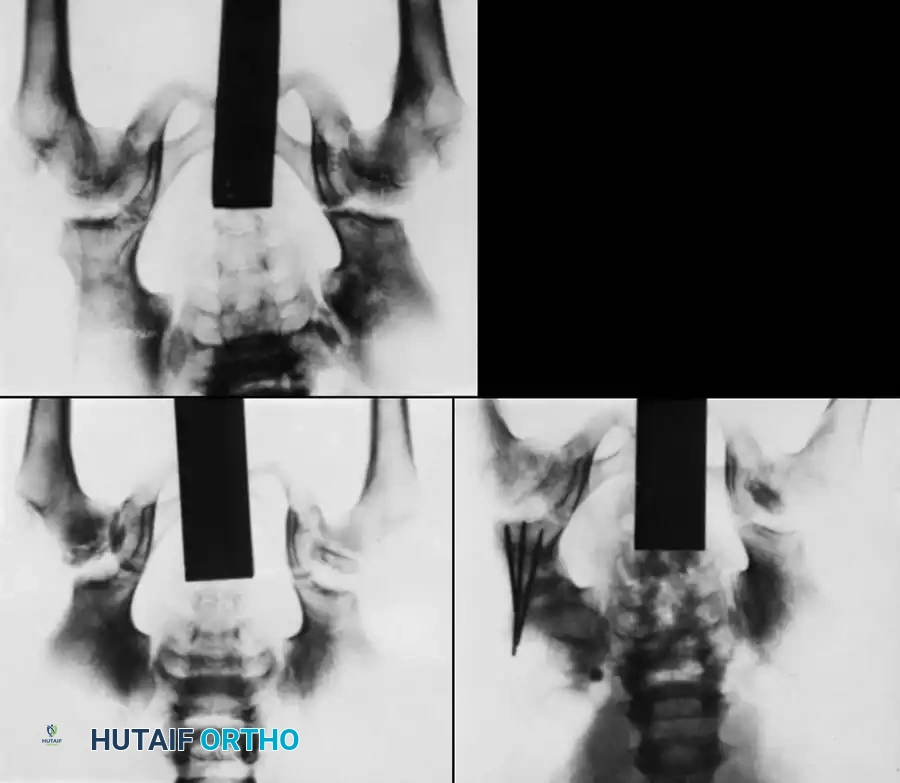
3. Guidewire Placement: Under strict fluoroscopic guidance, a guidewire is advanced into the femoral neck and head, parallel to the planned angle of the blade plate. The goal is typically to achieve a neck-shaft angle of 110 to 115 degrees.
4. Osteotomy: A transverse or closing-wedge osteotomy is performed at the intertrochanteric level using an oscillating saw.
5. Derotation and Varus: The distal fragment is externally rotated (typically 15-20 degrees) to correct the anteversion, and the proximal fragment is allowed to fall into varus.
6. Fixation: A pediatric locking plate or a fixed-angle blade plate is impacted into the proximal fragment and secured to the distal shaft with cortical screws. Compression is applied across the osteotomy site.
Postoperatively, the varus angulation results in a temporary limb length discrepancy and a Trendelenburg gait, which typically resolves as the child grows and the neck-shaft angle remodels.
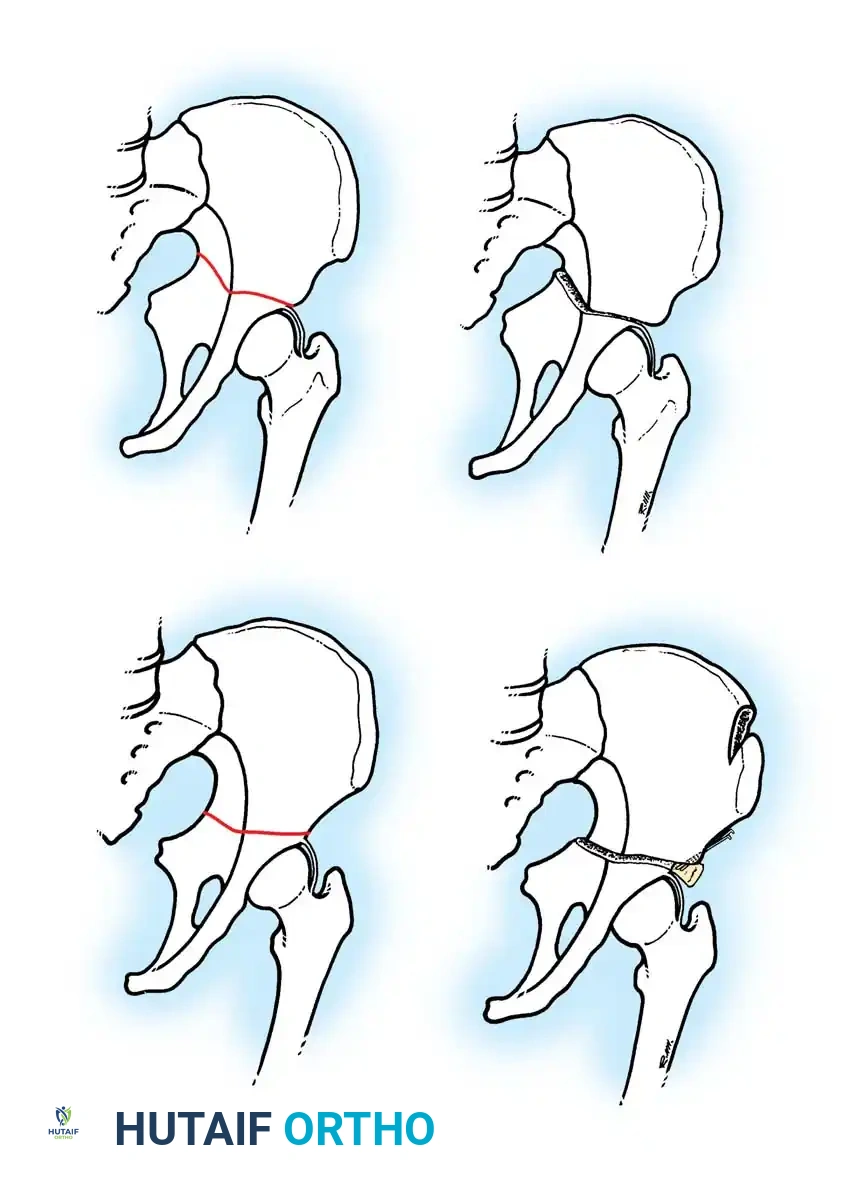
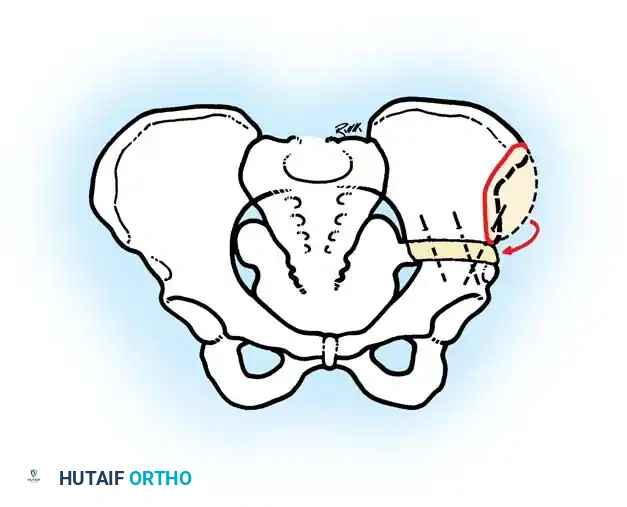
Surgical Management: Pelvic Osteotomies
Innominate osteotomies, such as the Salter osteotomy, provide containment by redirecting the acetabulum to cover the anterolateral aspect of the femoral head without altering the proximal femoral anatomy. This is particularly advantageous in older children where remodeling of a varus femur is less predictable, or in cases where limb shortening is undesirable.

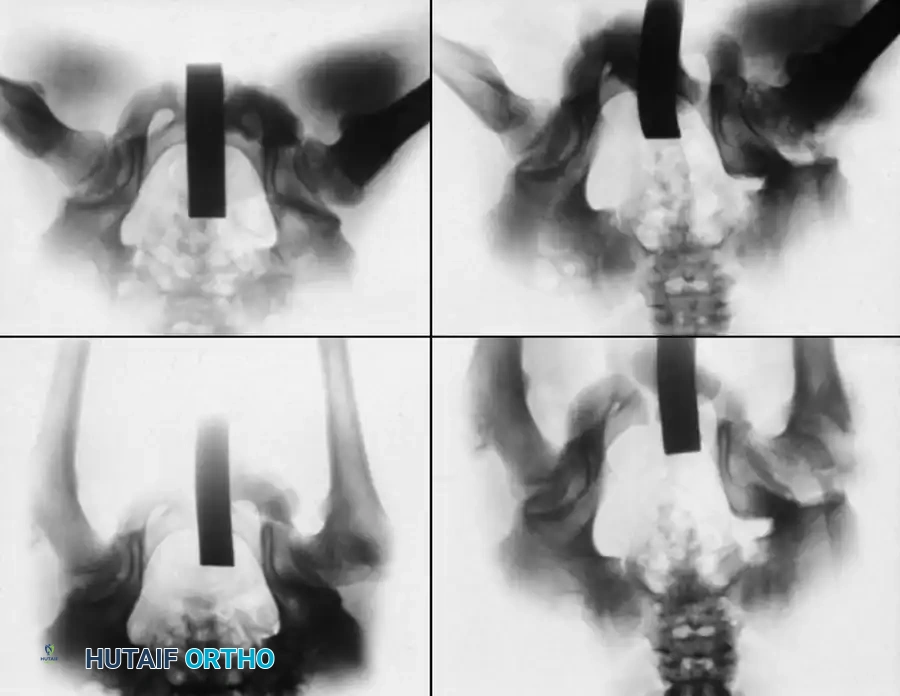
Salter Innominate Osteotomy Technique:
1. Approach: A Smith-Petersen (anterior) approach is utilized. The interval between the tensor fasciae latae and the sartorius is developed.
2. Exposure: The iliac apophysis is split, and the inner and outer tables of the ilium are stripped subperiosteally down to the greater sciatic notch.
3. Osteotomy: A Gigli saw is passed through the sciatic notch and directed anteriorly to exit at the anterior inferior iliac spine (AIIS).
4. Redirection: The distal acetabular fragment is hinged on the pubic symphysis and rotated anterolaterally, pivoting on the triradiate cartilage.
5. Grafting and Fixation: A triangular wedge of bone (harvested from the anterior iliac crest) is impacted into the osteotomy gap. The construct is stabilized with two or three threaded Kirschner wires driven from the proximal ilium, through the graft, and into the distal fragment.
Salvage Procedures for Late Disease
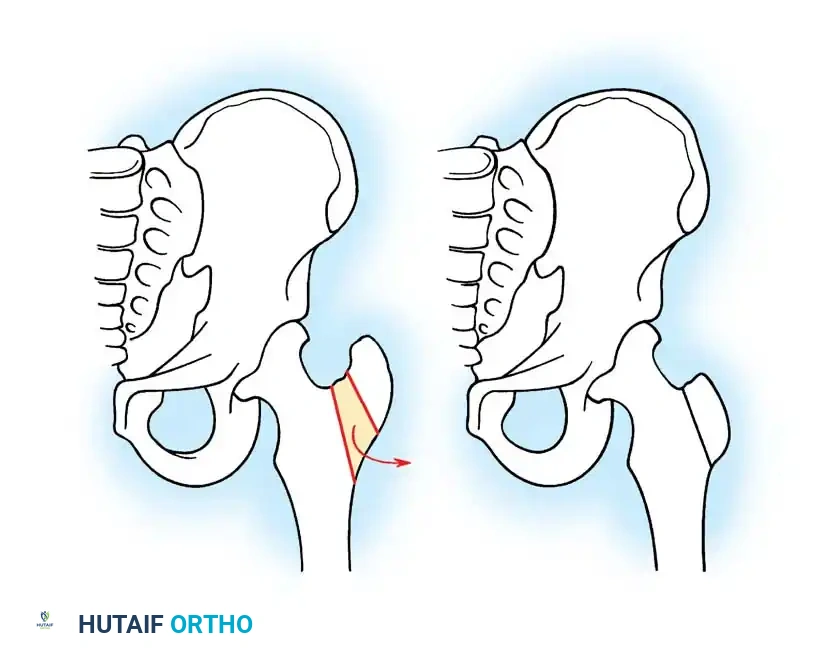
In late-stage LCPD presenting with established hinged abduction, severe coxa magna, and loss of sphericity, traditional containment procedures are contraindicated as they will exacerbate impingement.
Valgus Extension Osteotomy:
For patients with hinged abduction, a proximal femoral valgus osteotomy is the procedure of choice. By placing the femur into valgus, the extruded anterolateral bump is rotated away from the lateral acetabular rim, allowing the medial, more spherical portion of the femoral head to articulate with the acetabulum. This dramatically relieves pain and improves abductor mechanics.
Postoperative Protocols and Long-Term Outcomes
Postoperative management depends heavily on the stability of the fixation and the compliance of the patient. Following a VDRO or Salter osteotomy, patients are typically restricted to toe-touch weight-bearing for 6 to 8 weeks. Spica casting is rarely required with modern rigid internal fixation but may be utilized in non-compliant or very young patients.
Radiographic union is usually evident by 8 weeks, at which point progressive weight-bearing and aggressive abductor strengthening are initiated. Hardware removal is generally planned for 6 to 12 months postoperatively to prevent physeal tethering or stress shielding.
The ultimate prognosis of Legg-Calvé-Perthes disease is best evaluated at skeletal maturity using the **Stulberg
📚 Medical References
- Legg-Calvé-Perthes Disease Agus H, Kalenderer O, Eryanilmaz G, et al: Intraoberver and interobserver reliability of Catterall, Herring, Salter-Thompson and Stulberg classifi cation systems in Perthes disease, J Pediatr Orthop 13B:166, 2004.
- Akgun R, Yazici M, Aksoy MC, et al: The accuracy and reliability of estimation of lateral pillar height in determining the Herring grade in , J Pediatr Orthop 24;651, 2004.
- Aksoy MC, Caglar O, Yazici M, et al: Comparison between braced and non-braced Legg-Calvé-Perthes disease patients: a radiological outcome study, J Pediatr Orthop 13B:153, 2004.
- Arruda VR, Belangero WD, Ozelo MC, et al: Inherited risk factors for thrombophilia among children with LeggCalvé-Perthes disease, J Pediatr Orthop 19:84, 1999.
- Atsumi T, Yoshiwara S: Rotational open wedge osteotomy in a patient aged older than 7 years with Perthes’ disease—a preliminary report, Arch Orthop Trauma Surg 122:346, 2002.
- Axer A: Subtrochanteric osteotomy in the treatment of Perthes’ disease, J Bone Joint Surg 47B:489, 1965.
- Balasa VV, Gruppo RA, Glueck CA, et al: Legg-Calvé-Perthes disease and thrombophilia, J Bone Joint Surg 86A:2642, 2004.
- Bellyei A, Mike G: Acetabular development in Legg-CalvéPerthes disease, Orthopedics 11:407, 1988.
- Bennett JT, Mazurek RG, Cash JD: Chiari’s osteotomy in the treatment of Perthes’ disease, J Bone Joint Surg 73B: 225, 1991.
- Bowen JR, Kumar VP, Joyce JJ, et al: Osteochondritis dissecans following Perthes disease: arthroscopic-operative treatment, Clin Orthop Relat Res 209:49, 1986.
- Bowen JR, Schreiber FC, Foster BK, et al: Premature femoral neck physeal closure in Perthes’ disease, Clin Orthop Relat Res 171:24, 1982.
- Bursali, A, Erkula G: Lateral shelf acetabuloplasty in the treatment of Legg-Calvé-Perthes disease, J Pediatr Orthop 13B:150, 2004.
- Cahuzac JPH, Onimus M, Trottmann F, et al: Chiari pelvic osteotomy in Perthes disease, J Pediatr Orthop 10:163, 1990.
- Canale ST, d’Anca AF, Cotler JM, et al: Use of innominate osteotomy in Legg-Calvé-Perthes disease, J Bone Joint Surg 54A:25, 1972.
- Canario AT, Williams L, Weintroub S, et al: A controlled study of the results of
You Might Also Like