Complications of Total Knee Arthroplasty: A Comprehensive Surgical Guide
Key Takeaway
Total knee arthroplasty (TKA) is a highly successful procedure, yet complications such as thromboembolism, periprosthetic joint infection, and extensor mechanism failure demand rigorous clinical management. This guide details evidence-based protocols for prophylaxis, diagnostic algorithms, and step-by-step surgical interventions—including debridement, resection arthroplasty, and arthrodesis—equipping orthopedic surgeons with the advanced techniques necessary to optimize patient outcomes and salvage complex arthroplasty failures.
INTRODUCTION TO TKA COMPLICATIONS
Total knee arthroplasty (TKA) is one of the most successful and frequently performed orthopedic procedures worldwide, offering profound pain relief and functional restoration for end-stage knee arthropathy. However, the management of its complications remains a formidable challenge for the arthroplasty surgeon. Complications can be broadly categorized into systemic issues, such as thromboembolic events, and local complications, including periprosthetic joint infection (PJI), aseptic loosening, patellofemoral maltracking, extensor mechanism disruption, and periprosthetic fractures.
This comprehensive guide delineates the pathophysiology, diagnostic algorithms, and step-by-step surgical management of these complex scenarios, providing evidence-based protocols for postgraduate orthopedic residents, fellows, and practicing consultants.
THROMBOEMBOLIC COMPLICATIONS
One of the most significant and potentially devastating systemic complications following TKA is the development of deep vein thrombosis (DVT), which can precipitate a life-threatening pulmonary embolism (PE).
Risk Factors and Prevalence
The etiology of thromboembolism in TKA is multifactorial, classically aligning with Virchow’s triad: endothelial injury (surgical trauma), venous stasis (tourniquet use and postoperative immobility), and hypercoagulability (systemic inflammatory response to surgery).
Factors correlated with an increased risk of DVT include:
* Age older than 40 years
* Exogenous estrogen use
* History of stroke, congestive heart failure, or myocardial infarction
* Malignancy or nephrotic syndrome
* Prolonged immobility or obesity
* Previous history of thromboembolism
Without any form of mechanical or pharmaceutical prophylaxis, the overall prevalence of DVT after TKA ranges from 40% to 84%. Proximal thrombi (popliteal vein and above) occur in 9% to 20% of patients and pose a significantly greater risk of PE than distal calf vein thrombi (40% to 60%). However, calf thrombi possess a propensity to propagate proximally in 6% to 23% of cases. The risk of asymptomatic PE is estimated at 10% to 20%, while symptomatic PE occurs in 0.5% to 3% of patients, carrying a mortality rate of up to 2%.
Diagnostic Modalities
Clinical examination is notoriously unreliable for detecting DVT, as the majority of clots remain asymptomatic.
* Venography: Historically the "gold standard" for research, venography carries risks of anaphylaxis to contrast media and a paradoxical risk of inducing DVT.
* Duplex Ultrasound: Currently the preferred screening and diagnostic modality due to its non-invasive nature, low cost, and repeatability. While sensitivities range from 52% to 86%, its accuracy is highly operator-dependent.
Prophylaxis Protocols
Modern DVT prophylaxis employs a multimodal approach combining mechanical devices (pneumatic compression boots, foot pumps) and pharmacological agents.
Clinical Pearl: Mechanical compression is highly advantageous as it carries no bleeding risk, but its efficacy is strictly limited by patient compliance and the duration of hospitalization. It should be used as an adjunct, not a standalone therapy, in high-risk patients.
Pharmacological options include:
* Aspirin: While historically debated, modern protocols utilizing aspirin (e.g., 81 mg BID) have shown excellent efficacy in low-risk patients when combined with early mobilization.
* Warfarin: Adjusted to maintain an International Normalized Ratio (INR) between 2.0 and 3.0. Disadvantages include delayed onset, required monitoring, and drug interactions.
* Low-Molecular-Weight Heparin (LMWH) & Fondaparinux: Highly effective with standard dosing and no routine monitoring. However, they carry a higher cost and increased bleeding incidence.
Surgical Warning: LMWH must be used with extreme caution in patients receiving epidural or spinal anesthesia due to the catastrophic risk of epidural hematoma. The highest risk occurs on postoperative day 3 upon catheter removal.
Current Institutional Protocol: We advocate for chemical prophylaxis (LMWH, warfarin, or aspirin based on risk stratification) combined with pneumatic compression for at least 14 days in standard patients, extended to 6 weeks for those with a prior history of thromboembolism.
PERIPROSTHETIC JOINT INFECTION (PJI)
Infection is arguably the most dreaded complication in arthroplasty, with an incidence ranging from 1.6% to 2.6%. The presence of a foreign body lowers the threshold for infection, as bacteria (commonly Staphylococcus aureus and Staphylococcus epidermidis) form a protective glycocalyx biofilm on the implant surface, rendering them highly resistant to host defenses and systemic antibiotics.
Prevention Strategies
Prevention begins in the operating room with strict adherence to aseptic technique.
* Operating Room Environment: Minimizing personnel traffic, utilizing filtered vertical laminar flow, and employing body exhaust suits significantly reduce bacterial contamination.
* Prophylactic Antibiotics: A first-generation cephalosporin (e.g., cefazolin) is standard. Vancomycin is reserved for patients with severe penicillin allergies or in institutions with high MRSA prevalence.
Diagnosis of PJI
A high index of suspicion must be maintained for any patient presenting with a consistently painful TKA or acute onset of pain in a previously well-functioning joint.
- Serology: Erythrocyte sedimentation rate (ESR) and C-reactive protein (CRP) are first-line markers. CRP typically peaks 48-72 hours postoperatively and normalizes within 3 weeks. Serum Interleukin-6 (IL-6) is highly accurate, boasting 100% sensitivity and 95% specificity.
- Radiography: Advanced infections may show bone resorption at the cement-bone interface or periosteal reaction.
- Joint Aspiration: The definitive diagnostic step. A synovial fluid white blood cell (WBC) count > 2,500 cells/mm³ with > 60% polymorphonuclear (PMN) cells is highly indicative of PJI.
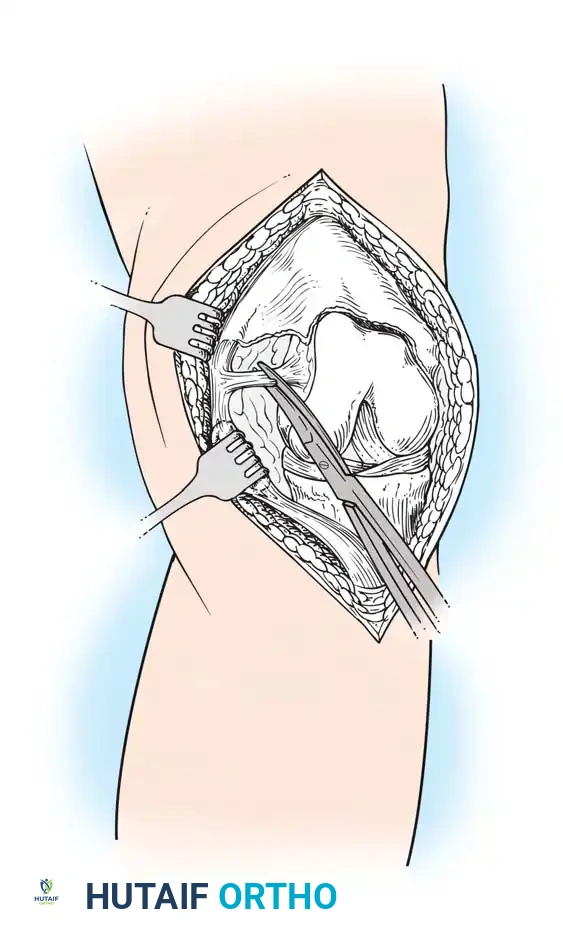
SURGICAL MANAGEMENT OF PJI
Treatment selection depends on the chronicity of the infection, host immune status, and implant stability.
Debridement, Antibiotics, and Implant Retention (DAIR)
Indicated strictly for early postoperative infections (< 4 weeks) or acute hematogenous infections with a well-fixed prosthesis.
Surgical Steps:
1. Approach: Re-open the previous incision. Perform a radical synovectomy and thorough debridement of all necrotic tissue.
2. Polyethylene Exchange: The modular polyethylene insert must be removed to access the posterior capsule and thoroughly irrigate the joint.
3. Irrigation: Copious pulsatile lavage (minimum 9 liters) with sterile saline.
4. Closure: Change gowns, gloves, and instruments before inserting the new polyethylene liner and closing the wound. Follow with 6 weeks of targeted intravenous antibiotics.
Resection Arthroplasty
Resection arthroplasty involves the complete removal of the infected prosthesis and cement, followed by radical debridement.

Fig. 6-57 Resection arthroplasty of knee. The components are removed, and the joint space is left to form a fibrous pseudoarthrosis.
Indications: Ideal for patients with severe polyarticular rheumatoid arthritis and limited ambulatory demands, where maintaining some knee flexion for sitting is preferable to a stiff, fused knee.
Outcomes: While infection resolution is high (83%), functional outcomes are poor. Most patients require extensive bracing and assistive devices to ambulate due to profound instability.
Knee Arthrodesis
Arthrodesis provides a stable, painless limb at the cost of joint mobility.
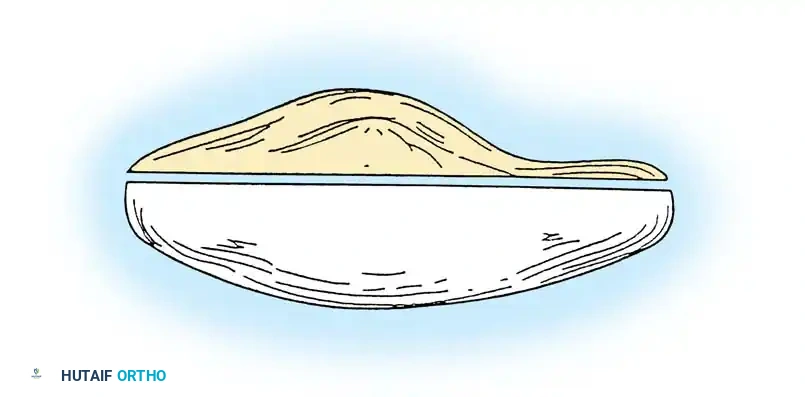
Radiographic appearance of a knee arthrodesis utilizing a long intramedullary nail following failed infected TKA.
Indications: High functional demands in a young patient, deficient extensor mechanism, poor soft-tissue envelope, or infection with highly virulent organisms precluding reimplantation.
Surgical Technique (IM Nailing):
1. Positioning: Supine on a radiolucent table.
2. Preparation: Remove all implants and cement. Prepare the femoral and tibial canals using flexible reamers.
3. Bone Apposition: Ensure flat, bleeding cancellous bone surfaces on both the distal femur and proximal tibia.
4. Implantation: Pass a long, custom or modular intramedullary nail antegrade through the femur into the tibia, applying compression across the arthrodesis site.
Two-Stage Revision Arthroplasty
The gold standard for chronic PJI.
* Stage 1: Explantation of components, radical debridement, and placement of an antibiotic-impregnated cement spacer.
* Stage 2: After 6-8 weeks of IV antibiotics and normalization of inflammatory markers, the spacer is removed, and revision components are implanted.
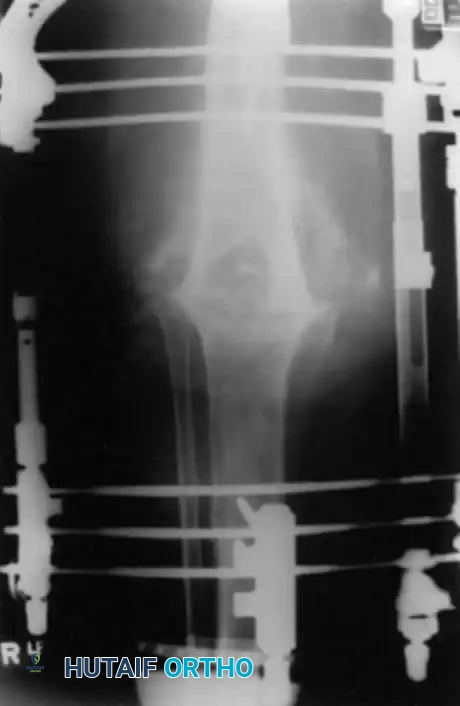
Complex two-stage revision TKA utilizing diaphyseal engaging stems and metaphyseal augments to manage severe bone loss.
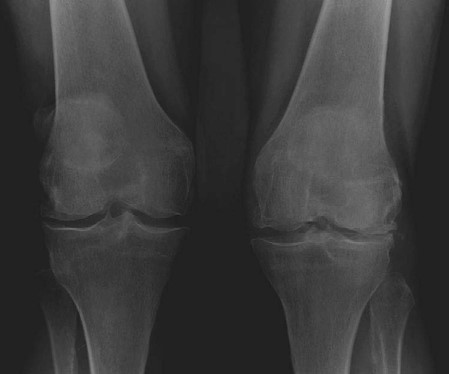
ASEPTIC LOOSENING AND OSTEOLYSIS
Aseptic loosening remains a leading cause of late TKA failure. It is primarily driven by particulate wear debris (usually polyethylene), which is phagocytosed by macrophages. This triggers a cytokine cascade (TNF-α, IL-1, IL-6) leading to osteoclast activation and subsequent periprosthetic osteolysis.
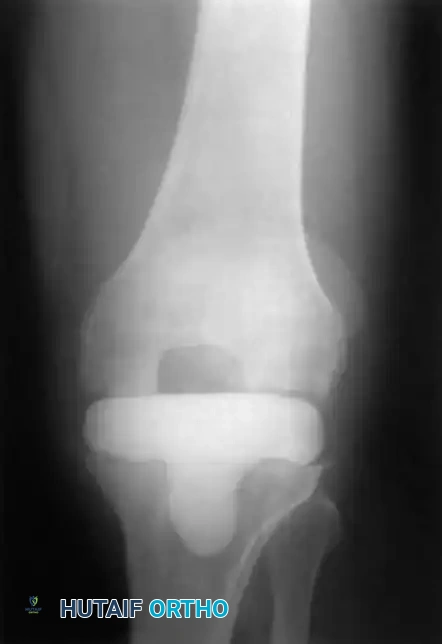
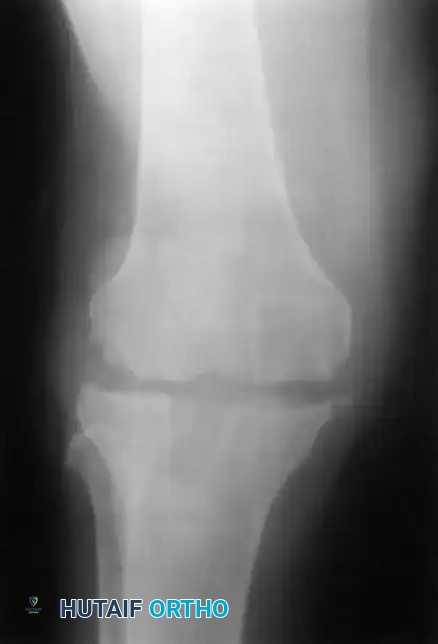
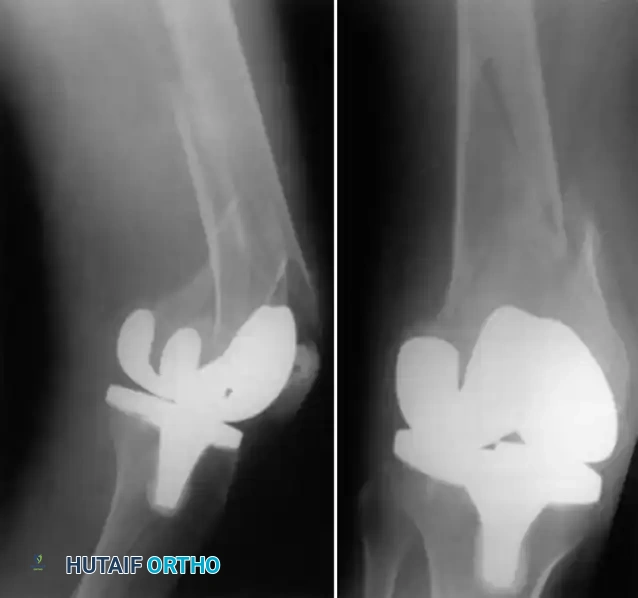
Standard AP radiograph of a well-fixed TKA. Baseline imaging is critical for future comparison.
Radiograph demonstrating significant radiolucent lines and osteolysis beneath the tibial tray, indicative of aseptic loosening.
Diagnosis: Serial radiographs demonstrating progressive radiolucent lines > 2mm, component migration, or subsidence.
Management: Revision arthroplasty utilizing stems to bypass metaphyseal defects and engage diaphyseal bone, supplemented with metal augments or structural allografts.
PATELLOFEMORAL COMPLICATIONS
The patellofemoral joint is historically the most common source of complications following TKA, encompassing anterior knee pain, maltracking, instability, and patellar clunk syndrome.
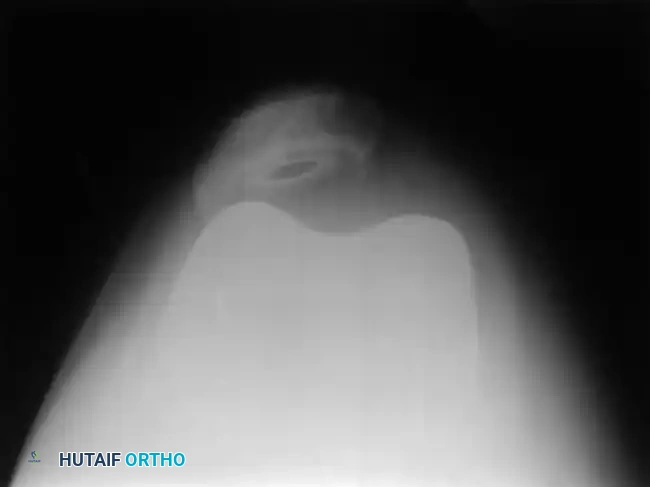
Patellar Resurfacing and Maltracking
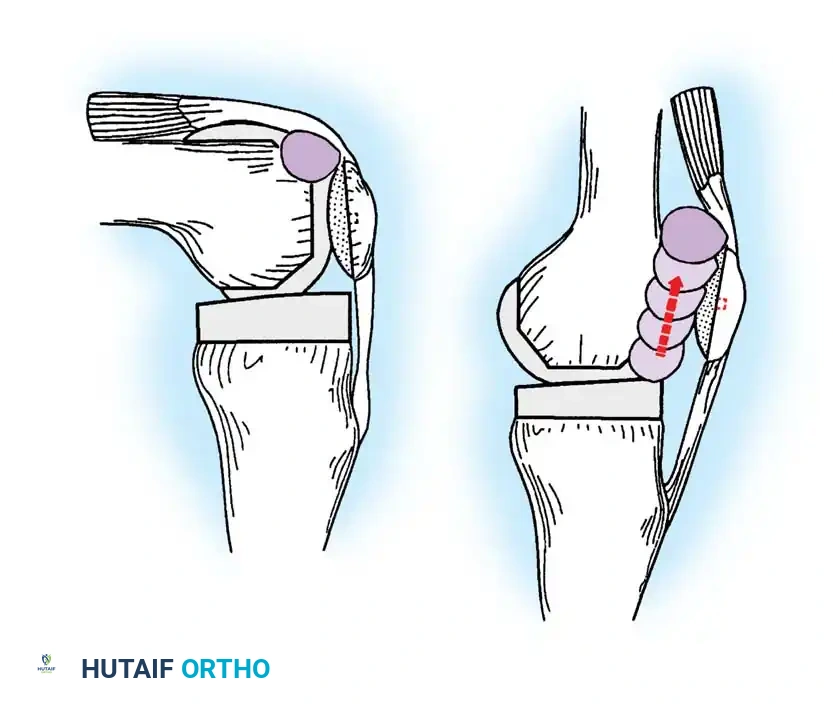
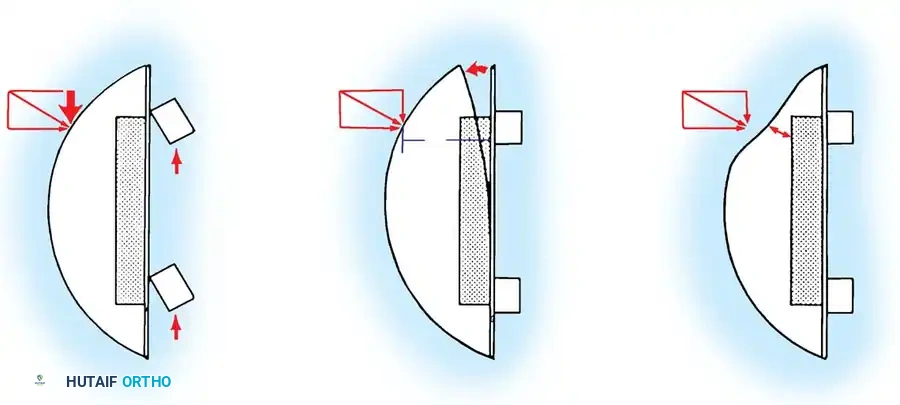
Proper patellar tracking relies on the delicate balance of the extensor mechanism and precise component positioning. Internal rotation of the femoral or tibial components is a primary driver of patellar maltracking and subluxation.
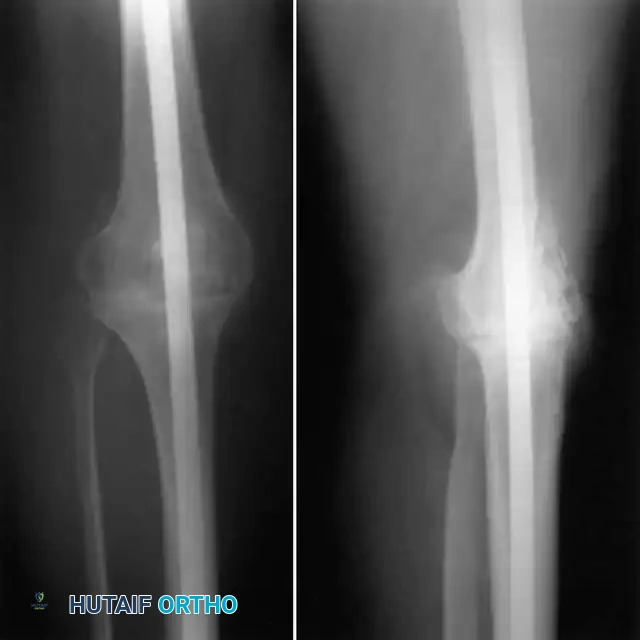
Diagram illustrating the biomechanics of patellar component positioning. Medialization of the patellar dome improves tracking by increasing the Q-angle mechanically.
When resurfacing the patella, restoring the native composite thickness is paramount. Overstuffing the joint leads to decreased flexion and increased retinacular strain, while excessive resection risks patellar fracture.
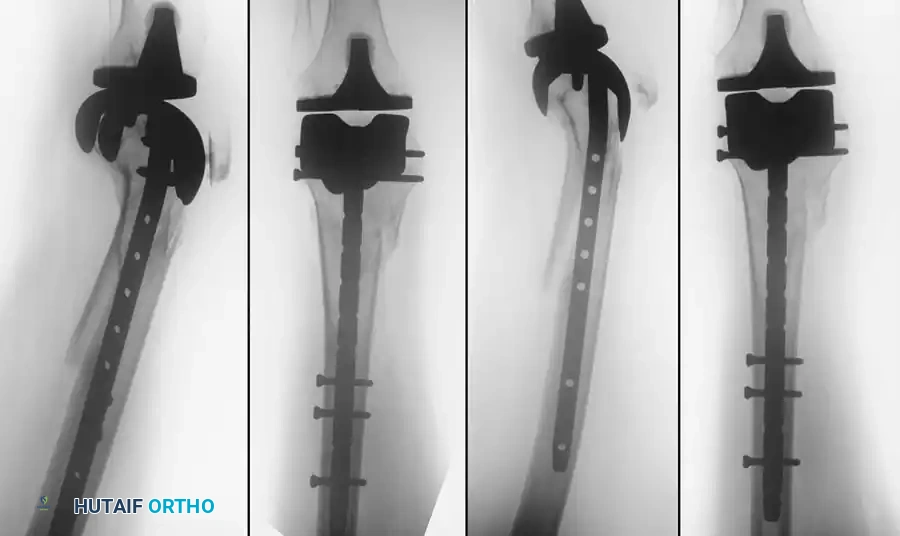
Diagram showing proper patellar resection. The composite thickness of the remnant bone and the polyethylene button should equal the original native patellar thickness.
Skyline (Merchant) view radiograph demonstrating proper patellar tracking within the trochlear groove of the femoral component.
Patellar Clunk Syndrome
Patellar clunk syndrome occurs when a fibrous nodule forms at the superior pole of the patella or the distal quadriceps tendon. During knee flexion, this nodule drops into the intercondylar box of a posterior-stabilized (PS) femoral component. Upon active extension, the nodule catches on the superior margin of the box, then abruptly pops out with a painful, audible "clunk" at approximately 30 to 45 degrees of flexion.
Diagrammatic representation of Patellar Clunk Syndrome. Note the fibrous nodule engaging the intercondylar box of the posterior-stabilized femoral component.
Management: Arthroscopic or open excision of the fibrous nodule is highly curative. Modern femoral component designs with modified, smoother intercondylar boxes have significantly reduced the incidence of this complication.
EXTENSOR MECHANISM DISRUPTION
Rupture of the extensor mechanism (quadriceps tendon, patella fracture, or patellar tendon) is a catastrophic complication that severely compromises knee function. Patellar tendon ruptures are particularly challenging due to the poor healing potential of the tendon to the proximal tibia.
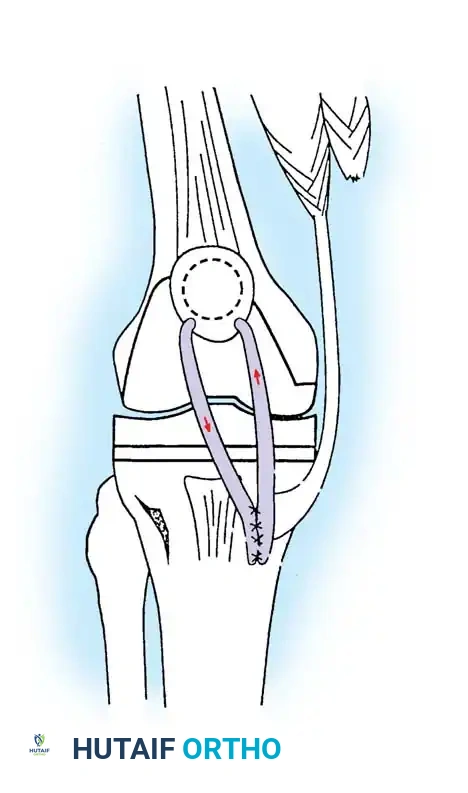
Surgical technique for extensor mechanism repair. Heavy non-absorbable sutures are passed through the patellar tendon and secured through transosseous drill holes in the tibial tubercle.
Surgical Management:
1. Direct Repair: For acute avulsions, direct repair using heavy non-absorbable sutures (e.g., #5 FiberWire) passed through transosseous tunnels in the tibial tubercle is performed. The repair is often augmented with a cerclage wire or heavy suture passed through the quadriceps tendon and tibial tubercle to protect the primary repair during early healing.
2. Allograft Reconstruction: For chronic ruptures or failed primary repairs, reconstruction using a complete extensor mechanism allograft (tibial tubercle, patellar tendon, patella, and quadriceps tendon) or synthetic mesh is required. The host bone is prepared, and the allograft tubercle is press-fit and cabled into a trough on the host tibia.
Postoperative Protocol: Following extensor mechanism repair, the knee is immobilized in full extension in a cylinder cast or locked brace for 6 weeks. Flexion is then gradually introduced in a controlled, hinged brace, avoiding active terminal extension against resistance for at least 3 months.
PERIPROSTHETIC FRACTURES
Periprosthetic fractures, particularly supracondylar femur fractures, are increasing in incidence due to an aging population and the rising number of TKAs performed. Risk factors include advanced age, osteoporosis, chronic steroid use, and anterior femoral notching during the index arthroplasty.
Radiographs demonstrating a periprosthetic supracondylar femur fracture managed with a lateral locking plate.
Classification and Management:
The Lewis and Rorabeck classification is widely used for supracondylar fractures:
* Type I: Undisplaced fracture, prosthesis intact. Managed non-operatively with a cast or brace.
* Type II: Displaced fracture, prosthesis intact. Managed with open reduction and internal fixation (ORIF) using lateral locking plates or retrograde intramedullary nails.
* Type III: Fracture with a loose prosthesis. Requires revision arthroplasty using a long-stemmed femoral component, often with distal femoral replacement (megaprosthesis) if bone loss is severe.
Surgical Technique (ORIF with Lateral Locking Plate):
1. Positioning: Supine on a radiolucent table with a bump under the ipsilateral hip.
2. Approach: A lateral subvastus or standard lateral approach to the distal femur.
3. Reduction: Indirect reduction techniques are preferred to preserve the periosteal blood supply.
4. Fixation: A pre-contoured distal femoral locking plate is applied. Distal fixation requires multiple locking screws around the femoral component box. Proximal fixation utilizes bicortical locking or non-locking screws.
CONCLUSION
The management of complications following total knee arthroplasty requires a profound understanding of joint biomechanics, meticulous surgical technique, and a multidisciplinary approach to patient care. Whether addressing the insidious onset of periprosthetic joint infection, the mechanical failure of aseptic loosening, or the catastrophic disruption of the extensor mechanism, the orthopedic surgeon must be equipped with a versatile armamentarium of diagnostic and surgical strategies. By adhering to strict evidence-based protocols, surgeons can effectively salvage failed arthroplasties, restore function, and significantly improve the quality of life for their patients.
You Might Also Like