Comprehensive Introduction and Patho-Epidemiology
Welcome, esteemed colleagues and surgical fellows, to this intraoperative masterclass. Today, we are undertaking a profoundly fascinating and technically demanding procedure: the posterior hemivertebra excision and spinal reconstruction. This intervention demands absolute meticulousness in planning, flawless precision in execution, and an intimate, three-dimensional understanding of spinal anatomy—particularly within the delicate and diminutive confines of the pediatric spine. Our overarching surgical objective is to achieve a complete, circumferential resection of the anomalous vertebral body utilizing exclusively a posterior approach. This resection must be immediately followed by robust segmental instrumentation and arthrodesis to definitively correct the progressive, unbalanced congenital scoliosis and kyphosis that invariably accompany this pathology.
Before we even consider making our initial incision, it is imperative that we thoroughly review the fundamental patho-epidemiology of the hemivertebra. At its core, a hemivertebra represents a distinct congenital anomaly of spinal formation. This embryologic failure typically occurs between the 8th and 12th weeks of gestation during the chondrification stage of vertebral development. Morphologically, it is characterized by the presence of only one lateral half of a vertebral body, a corresponding unilateral pedicle, and a single hemilamina. One must visualize a wedge-shaped osseous segment, which acts as a structural fulcrum, almost universally situated on the convex side of a developing and progressive scoliotic curve.
The clinical significance and natural history of a hemivertebra are intrinsically linked to its degree of segmentation from the adjacent normal vertebrae. A Fully Segmented Hemivertebra is our primary surgical indication today. It is anatomically separated from the normal vertebral bodies both cephalad and caudad by intact, albeit often dysplastic, intervertebral disc spaces. This complete segmentation signifies the presence of active, uninhibited superior and inferior endplate growth centers. Consequently, this leads to significant, asymmetrical, and unbalanced growth potential. These curves typically progress at an alarming rate of approximately 2 to 3 degrees per year and can easily exceed 45 to 50 degrees by skeletal maturity, rendering early surgical intervention not just elective, but mandatory to prevent severe truncal decompensation. Conversely, a Partially Segmented Hemivertebra is separated from one adjacent body by a disc but is osseously fused to the other. Its growth potential is substantially mitigated, typically progressing at less than 1 degree per year, and rarely exceeding 40 degrees, often allowing for conservative observation. Finally, an Unsegmented Hemivertebra is completely fused to both adjacent vertebral bodies, possesses virtually zero asymmetrical growth potential, and generally requires no surgical treatment.
Epidemiologically, congenital scoliosis occurs in approximately 1 in 1,000 live births, with hemivertebrae representing a significant proportion of these structural anomalies. The location of the hemivertebra is a critical determinant of clinical severity. For instance, hemivertebrae situated at the lumbosacral junction are particularly pernicious and almost always demand aggressive surgical treatment. Because the lumbar spine takes off at an oblique angle from the sacral foundation, the presence of a wedge at this transitional zone creates a long, sweeping compensatory curve in the otherwise normal cranial segments. This results in profound cosmetic deformity, severe coronal imbalance, and truncal shift. Furthermore, if the embryologic failure of formation is more pronounced anteriorly or posteriorly, the patient will present with a complex, multi-planar deformity, manifesting as a localized, rigid kyphoscoliosis or lordoscoliosis.
Detailed Surgical Anatomy and Biomechanics
To execute a safe and complete resection, the surgeon must possess a profound, three-dimensional mastery of the local surgical anatomy, which is invariably distorted by the congenital anomaly. The osseous anatomy of the hemivertebra itself is highly variable. The hemilamina is often thickened and malformed, and the unilateral pedicle may be foreshortened, sclerotic, or abnormally angulated compared to the adjacent normal segments. The wedge-shaped vertebral body projects laterally and often anteriorly, creating a localized apex of deformity. The adjacent normal vertebrae are also subject to secondary morphological changes due to the asymmetrical biomechanical forces applied over time, leading to wedging of the adjacent discs and remodeling of the adjacent endplates.
The neurovascular anatomy in the vicinity of a hemivertebra requires meticulous attention. The spinal cord and conus medullaris may be draped tightly over the convexity of the curve, particularly if a kyphotic component is present. The exiting nerve roots on the convex side (the side of the hemivertebra) are often stretched and vulnerable, while the roots on the concave side may be redundant or compressed within a narrowed neural foramen. Furthermore, one must always be acutely aware of the segmental vessels. The segmental artery and vein traversing the waist of the hemivertebra must be definitively identified, isolated, and ligated during the lateral and anterior dissection to prevent catastrophic hemorrhage. The proximity of the great vessels—the aorta on the left and the inferior vena cava on the right—dictates extreme caution during the anterior cortical breach and disc space preparation.
Biomechanically, a fully segmented hemivertebra acts as a continuous, asymmetrical growth engine. The intact growth plates on the convex side of the hemivertebra add longitudinal length to the convexity, while the contralateral concave side, lacking a corresponding growth center, acts as a tether. This creates a vicious cycle of progressive deformity, governed by the Hueter-Volkmann principle: increased compressive forces on the concave side further inhibit growth, while decreased pressure on the convex side accelerates it. This biomechanical reality dictates our surgical strategy: we must not only remove the asymmetrical growth center (the hemivertebra) but also close the resulting wedge-shaped defect to restore coronal and sagittal balance, thereby neutralizing the abnormal force vectors.
The posterior-only approach to hemivertebra excision relies heavily on the biomechanical principles of a closing wedge osteotomy. By resecting the posterior elements, the pedicle, and the vertebral body of the hemivertebra, we create a void. The adjacent normal segments are then instrumented with pedicle screws. By applying compression forces across the convex side of the instrumentation and carefully distracting or contouring the concave side, the spine is hinged upon the anterior longitudinal ligament (or the anterior cortical margin if the ALL is resected). This maneuver closes the osteotomy site, correcting the angular deformity and achieving bone-on-bone contact between the adjacent normal endplates, which is essential for a robust, long-term arthrodesis.
Exhaustive Indications and Contraindications
The decision to proceed with a posterior hemivertebra excision is highly nuanced and must be tailored to the individual patient's presentation, age, and associated comorbidities. The primary indication for surgical intervention is a documented, progressive congenital scoliosis or kyphoscoliosis driven by a fully segmented hemivertebra. The optimal window for this specific procedure is generally accepted to be between 18 months and 4 to 5 years of age. Intervening during this critical period allows the surgeon to capitalize on the relative flexibility of the adjacent compensatory curves, preventing the development of severe, rigid, and irrevocable secondary deformities. Furthermore, modern pediatric pedicle screw instrumentation systems are now suitably sized to achieve rigid fixation even in toddlers.
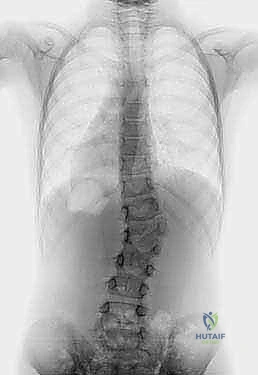
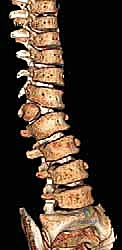
Absolute contraindications to the procedure include the presence of unaddressed, severe intraspinal anomalies. As previously noted, up to 40% of patients with congenital scoliosis harbor a concomitant spinal dysraphism, such as a tethered spinal cord, diastematomyelia (split cord malformation), or a significant syringomyelia. Attempting a major spinal deformity correction, particularly a closing wedge osteotomy that shortens the spinal column, in the presence of a tethered cord can result in catastrophic and irreversible neurologic injury. Therefore, neurosurgical intervention to untether the cord or resect a diastematomyelic spur must invariably precede the hemivertebra excision, often by several months.
Relative contraindications revolve primarily around the patient's age and overall medical fragility. In older children (typically over 8 to 10 years of age), the compensatory curves often become highly structured and rigid. A simple, single-level hemivertebra excision may be insufficient to restore global spinal balance in these older patients, necessitating more extensive, multi-level fusions or complex, multi-apical osteotomies. Additionally, severe congenital cardiac anomalies (present in roughly 20% of these patients) or profound pulmonary compromise may render the patient unfit to tolerate the physiological stress, potential blood loss, and prolonged anesthetic duration associated with a major spinal reconstruction.
Below is a comprehensive summary of the indications and contraindications for posterior hemivertebra excision:
| Category | Specific Criteria | Clinical Rationale / Considerations |
|---|---|---|
| Primary Indications | Documented progressive curve (>2-3 degrees/year) | Prevents severe, rigid secondary deformities. |
| Fully segmented hemivertebra | High asymmetrical growth potential mandates removal. | |
| Lumbosacral hemivertebra | High risk of severe truncal shift and pelvic obliquity. | |
| Optimal Age: 18 months to 5 years | Balances bone size for instrumentation with curve flexibility. | |
| Absolute Contraindications | Untreated spinal dysraphism (tethered cord) | Extremely high risk of iatrogenic neurologic injury during correction. |
| Active, untreated spinal infection | Risk of hardware seeding and systemic sepsis. | |
| Medically unstable for major surgery | Severe uncorrected VACTERL anomalies (e.g., severe cardiac defects). | |
| Relative Contraindications | Older age (> 8-10 years) | Structured compensatory curves may require multi-level fusions. |
| Unsegmented hemivertebra | Minimal growth potential; observation is usually preferred. | |
| Severe osteopenia/metabolic bone disease | High risk of instrumentation failure and pedicle screw pullout. |
Pre-Operative Planning, Templating, and Patient Positioning
The foundation of a successful hemivertebra excision is laid long before the patient enters the operating theater. A comprehensive, multi-disciplinary diagnostic workup is absolutely mandatory. Because the embryologic development of the spine occurs concurrently with other vital organ systems (between weeks 4 and 8 of gestation), congenital spinal anomalies are rarely isolated events. We must systematically screen for the VACTERL association (Vertebral, Anorectal, Cardiac, Tracheoesophageal, Renal, and Limb anomalies). A meticulous neurologic examination is crucial to identify subtle signs of dysraphism, such as asymmetric reflexes, cavus foot deformity, or cutaneous stigmata (hairy patches, hemangiomas, deep sacral dimples). Preoperative echocardiography and renal ultrasonography are non-negotiable screening tools.
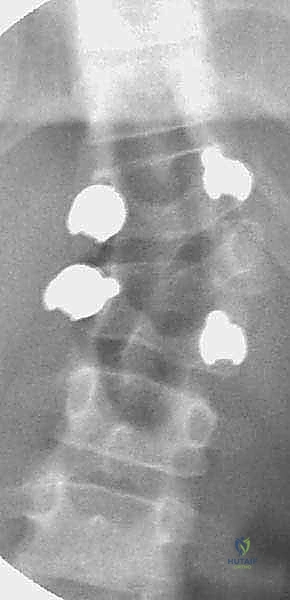
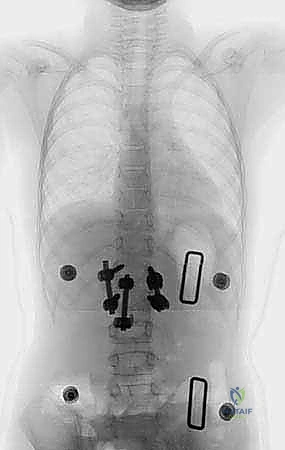
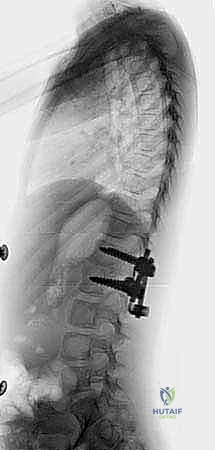
Imaging studies serve as our precise surgical roadmap. We initiate the workup with standing 36-inch posteroanterior (PA) and lateral radiographs to define the global deformity, measure the Cobb angles of the primary and compensatory curves, and assess overall coronal and sagittal balance. Supine bending radiographs are then obtained to evaluate the flexibility of the structural and compensatory curves, which directly dictates our selection of the upper and lower instrumented vertebrae (UIV and LIV). An MRI of the entire neuraxis (brainstem to sacrum) is absolutely mandatory to rule out the aforementioned intraspinal anomalies. Finally, a fine-cut CT scan with 3D reconstructions is critical. This allows us to map the distorted osseous anatomy, assess pedicle morphology (diameter, length, trajectory) for screw templating, and identify any unexpected posterior element deficiencies or synostoses that could complicate the exposure.
Templating is a rigorous exercise. Using the preoperative CT scan, we measure the pedicles of the vertebrae immediately adjacent to the hemivertebra. In a 3-year-old child, these pedicles can be exceptionally narrow, sometimes measuring less than 3.5 mm in diameter. We must pre-select our pedicle screw diameters and lengths accordingly, ensuring we have a wide array of pediatric-specific implants available. We also plan the trajectory of the osteotomy, calculating the exact angle of the wedge resection required to achieve orthogonal endplates upon closure of the defect.
Intraoperative patient positioning is a critical step that directly impacts surgical exposure and blood loss. The patient is carefully log-rolled into the prone position onto a radiolucent operating frame, typically a pediatric Jackson table or a specialized modular frame. The abdomen must hang completely free; any abdominal compression will engorge the epidural venous plexus, transforming a controlled dissection into a hemorrhagic nightmare. We frequently employ a technique called "airplaning" the table. By slightly tilting the table or bolstering the patient to elevate the convex side of the deformity, we significantly enhance our visualization down the shaft of the hemi-pedicle and across the anterior aspect of the vertebral body during the deep resection phase. Prior to prepping, a metallic marker is placed over the suspected level, and a spot radiograph confirms our location, preventing the disastrous complication of wrong-level surgery.
Step-by-Step Surgical Approach and Fixation Technique
Incision, Exposure, and Foundation Establishment
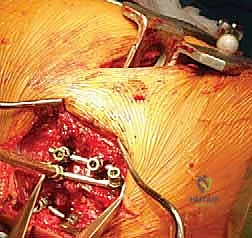
The procedure commences with a meticulously planned posterior midline incision. The length of the incision is dictated by our preoperative templating, ensuring adequate exposure of the hemivertebra and at least one normal vertebral level above and below for instrumentation. Following the incision of the skin and subcutaneous tissues, we divide the lumbodorsal fascia in the midline. Using a combination of electrocautery and Cobb elevators, a strict subperiosteal dissection of the paraspinal musculature is performed. It is paramount to stay precisely on the bone to minimize bleeding and preserve the vascular supply to the multifidus muscles. The exposure is carried out laterally to the tips of the transverse processes bilaterally. Extreme caution is exercised if the preoperative CT indicated any spina bifida occulta or dysplastic laminae, as the dura may lie immediately beneath the fascia.
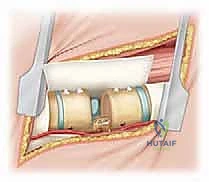
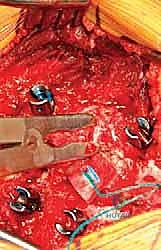
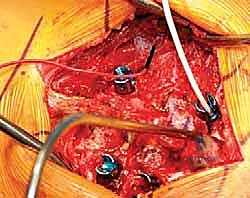
Once the posterior elements are fully exposed and hemostasis is achieved, we immediately establish our foundation by placing the pedicle screws in the adjacent normal vertebrae. We strongly advocate for bilateral pedicle screw fixation at the levels immediately cephalad and caudad to the hemivertebra to maximize the biomechanical stability of the construct. Given the altered anatomy and the small size of pediatric pedicles, we utilize a combination of anatomical landmarks, freehand probing techniques, and rigorous fluoroscopic or intraoperative navigation guidance. The pedicle is accessed using a high-speed burr or a pediatric awl, followed by a blunt, curved pedicle probe. The trajectory is carefully palpated with a ball-tipped feeler to ensure all five cortical walls (medial, lateral, superior, inferior, and anterior) are intact before inserting the appropriately sized screws.
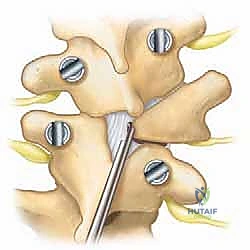
The Osteotomy: Posterior Element and Pedicle Resection
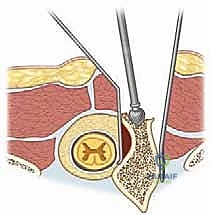
With the foundation secure, we proceed to the crux of the procedure: the hemivertebra excision. The first step is a complete laminectomy of the hemilamina. Using a high-speed burr and Kerrison rongeurs, the bone is carefully removed until the underlying dura and the exiting nerve roots are clearly visualized. The ligamentum flavum is excised cranially and caudally. We must identify the nerve root exiting above the hemi-pedicle and the root exiting below it; these roots must be protected throughout the remainder of the case.
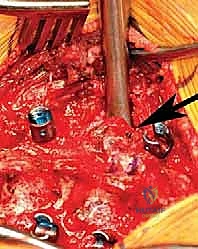
Next, we target the hemi-pedicle. This structure is often sclerotic and acts as a robust pillar connecting the posterior elements to the vertebral body. Using a high-speed burr, the pedicle is cored out from posterior to anterior, effectively "egg-shelling" it. The remaining thin cortical rim of the pedicle is then carefully micro-dissected away from the adjacent nerve roots and the dural sac using micro-curettes and fine Kerrison rongeurs. Removal of the pedicle provides direct, transpedicular access to the posterior aspect of the hemivertebral body.
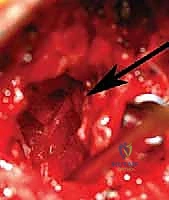
Vertebral Body Extirpation and Deformity Correction
The resection of the vertebral body is the most technically demanding and hazardous phase of the operation. Working through the void created by the resected pedicle, we use a combination of high-speed burrs, curettes, and pituitary rongeurs to decancellate the hemivertebral body. We work from the posterior margin towards the anterior cortex, taking great care not to breach the anterior longitudinal ligament prematurely, as this structure protects the great vessels. The lateral wall of the hemivertebra is carefully dissected subperiosteally. At this stage, the segmental vessels coursing over the waist of the vertebral body are identified, coagulated with bipolar electrocautery, and divided.
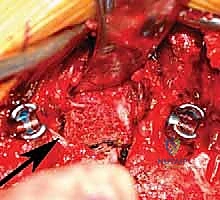
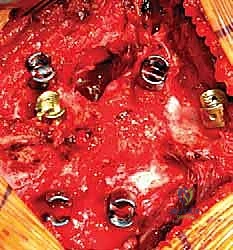
Once the cancellous core of the hemivertebra is removed, the superior and inferior intervertebral discs adjacent to the hemivertebra are excised completely, back to the posterior longitudinal ligament. The cartilaginous endplates of the adjacent normal vertebrae are meticulously removed using sharp curettes until bleeding subchondral bone is exposed; this is absolutely critical for achieving a solid interbody arthrodesis. The remaining cortical shell of the hemivertebral body is then collapsed inward and removed.
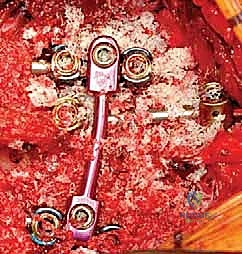
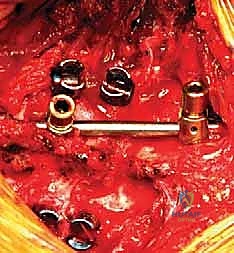
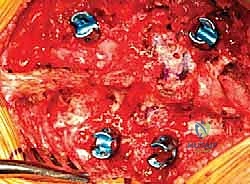
With the hemivertebra entirely extirpated, the deformity is ready for correction. A pre-contoured rod is secured to the pedicle screws on the convex side of the curve. By applying gradual, controlled compression across the pedicle screws on the convexity, the wedge-shaped defect is slowly closed. The spine hinges on the remaining anterior longitudinal ligament. Simultaneously, a rod is placed on the concave side, and gentle distraction or contouring is applied to assist in the correction and restore sagittal profile. Throughout this closure, intraoperative neuromonitoring (SSEPs and MEPs) is observed continuously. The exiting nerve roots are directly visualized to ensure they are not kinked or compressed as the bony defect closes. Once the endplates of the adjacent normal vertebrae are in firm, flush contact, the set screws are final-tightened. The resected bone from the hemivertebra is morselized and packed tightly over the decorticated posterior elements to facilitate a robust posterolateral fusion.
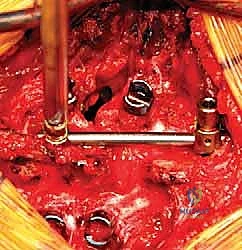
Complications, Incidence Rates, and Salvage Management
Despite meticulous technique, posterior hemivertebra excision is a high-stakes procedure fraught with potential complications. Neurologic injury is the most devastating, with reported incidence rates ranging from 1% to 3%. This can occur via direct mechanical trauma to the spinal cord or nerve roots during the osteotomy, ischemic injury due to excessive distraction or hypotension, or compression of the nerve root as the osteotomy is closed. If intraoperative Motor Evoked Potentials (MEPs) or Somatosensory Evoked Potentials (SSEPs) drop significantly during the correction phase, the surgeon must act immediately. The first step is to instantly release the corrective forces and return the spine to its pre-correction alignment. Mean arterial pressure (MAP) should be elevated by anesthesia to optimize spinal cord perfusion. If signals do not return, a Stagnara wake-up test may be indicated, and the procedure may need to be aborted or staged.
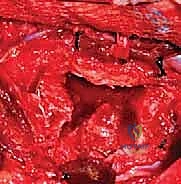
Dural tears are a relatively common intraoperative complication, occurring in approximately 5% to 8% of cases, particularly during the resection of the hemilamina or the medial wall of the hemi-pedicle where the dura may be adherent. In pediatric patients, the dura is exceptionally thin and friable. If a durotomy occurs, primary watertight repair using 5-0 or 6-0 non-absorbable suture is the gold standard. This should
Clinical & Radiographic Imaging Archive