Functioning Free Neuromuscular Transfers: Principles and Surgical Techniques
Key Takeaway
Functioning free neuromuscular transfer (FFMT) involves the transplantation of a viable muscle with its neurovascular pedicle to restore voluntary motor function. First described by Tamai in 1970, this complex microsurgical procedure is indicated for severe volumetric muscle loss, ischemic contractures, or brachial plexus injuries. Success relies on meticulous preoperative planning, precise microvascular anastomosis, optimal muscle tensioning, and a dedicated postoperative rehabilitation protocol to ensure functional reinnervation and excursion.
INTRODUCTION TO FUNCTIONING NEUROMUSCULAR TRANSFERS
The advent of microsurgery revolutionized reconstructive orthopaedics, allowing for the restoration of severe functional deficits that were previously deemed irreparable. Tamai et al., in 1970, pioneered this frontier by demonstrating in a canine model that a functioning neuromuscular unit could be successfully transplanted using microvascular anastomoses and nerve repairs at a recipient site. Today, functioning free muscle transfers (FFMT) are a cornerstone in the armamentarium of the reconstructive microsurgeon.
Functioning free muscle transfers are primarily utilized for the replacement of the flexor and extensor compartments of the forearm, the muscles of facial expression, and the muscles in the extensor compartment of the leg. The fundamental advantage of this procedure is the restoration of a functional deficit by transferring a viable, vascularized muscle under voluntary control, resulting in minimal functional loss at the donor site.
However, the procedure is highly complex. The disadvantages include the sacrifice of a functioning donor muscle, a prolonged reinnervation time (often spanning several months to over a year), the absolute requirement for advanced microvascular skills, and the extended duration of the surgical procedure.
Surgical Pearl: If a simpler, reliable procedure—such as a regional tendon transfer—suffices to restore the desired function, it should always be utilized in preference to a free muscle transfer. FFMT is reserved for cases of massive volumetric muscle loss where local reconstructive options have been exhausted.
INDICATIONS AND PATIENT SELECTION
The success of a functioning neuromuscular transfer is heavily dependent on stringent patient selection. A high level of patient motivation is absolutely essential, as the postoperative rehabilitation protocol is arduous and requires sustained psychological resilience.
Primary Indications
- Volkmann’s Ischemic Contracture: Severe cases where the entire flexor or extensor compartment has undergone fibrotic replacement.
- Massive Trauma: Crush injuries or avulsions resulting in volumetric loss of musculotendinous units.
- Brachial Plexus Injuries: Late presentations or cases where primary nerve grafting has failed, requiring restoration of elbow flexion or hand prehension.
- Oncologic Resection: Wide local excisions of sarcomas that necessitate the removal of entire functional muscle compartments.
Contraindications
- Stiff Recipient Joints: The joints in the recipient extremity must be supple. A functional range of motion must be available in the elbow, wrist, and fingers prior to transfer.
- Lack of Donor Nerve: A single, undamaged motor nerve must be available in the recipient area to supply the muscle transplant.
- Poor Soft Tissue Bed: While a skin island can be transferred, an extensively irradiated or infected recipient bed precludes successful microvascular anastomosis.
- Unstable Proximal Joints: Stable proximal joints with balanced musculature must be present to allow the transferred muscle to act effectively on the distal segments.
BIOMECHANICS AND DONOR MUSCLE SELECTION
The selection of a given donor muscle is dictated by the specific biomechanical requirements of the recipient site, specifically the required strength and excursion.
- Strength: Proportional to the physiological cross-sectional area (PCSA) of the muscle.
- Excursion: Proportional to the resting length of the muscle fibers.
Muscles that have proven highly reliable for FFMT include the pectoralis major, latissimus dorsi, gracilis, rectus femoris, extensor digitorum brevis of the toes, and serratus anterior. Additionally, the semitendinosus, tensor fasciae latae, and brachioradialis can be utilized in specific scenarios.
Muscle Matching Guidelines
- Large Muscle Requirements: If a large muscle with high force generation is required (e.g., restoration of elbow flexion or massive forearm flexor loss), the latissimus dorsi or pectoralis major is preferred.
- Small Muscle Requirements: If a smaller muscle with specific excursion is needed (e.g., facial reanimation or isolated digit function), the gracilis, serratus anterior, or extensor digitorum brevis may suffice. The gracilis is often considered the "workhorse" for forearm flexor reconstruction due to its reliable neurovascular pedicle, excellent excursion, and minimal donor site morbidity.
Other critical factors in muscle selection include skin coverage requirements (the ability to harvest a chimeric or musculocutaneous flap), motor nerve availability and length, and the anatomical location of the ends of the recipient flexor tendons.
PREOPERATIVE EVALUATION AND PLANNING
Meticulous preoperative planning is the bedrock of a successful FFMT. The needs and the neurovascular anatomy of the recipient site must be assessed comprehensively.
Neurological Assessment
A single, undamaged motor nerve must be identified in the recipient area to innervate the transplant.
* Forearm Reconstruction: Median nerve branches to the flexor digitorum sublimis (FDS) or the anterior interosseous nerve (AIN) are most frequently utilized. On the extensor surface, branches of the radial nerve are targeted.
* Evaluation Modalities: The status of recipient nerves must be evaluated carefully through detailed history and physical examination, electromyography (EMG), and, if necessary, an exploratory surgical procedure prior to the definitive transfer.
Vascular Assessment
Arteriography (CT angiography or conventional angiography) is strictly indicated to assess the patency and caliber of recipient vessels, especially in extremities that have sustained severe crush injuries or ischemia. In some situations, imaging of the donor vessels is also warranted to rule out anatomical variants.
Soft Tissue and Joint Assessment
Good skin covering the recipient site is mandatory to allow frictionless gliding of the transferred muscle. If the local soft tissue envelope is deficient, a skin island may be carried with the transferred muscle (e.g., a musculocutaneous latissimus dorsi or gracilis flap). Furthermore, intensive preoperative physical therapy is often required to maximize passive joint suppleness.
SURGICAL PREPARATION AND SETUP
The complexity and duration of a functioning free muscle transfer necessitate a highly coordinated two-team approach. One team prepares the recipient site while the second team simultaneously harvests the donor muscle. This permits a more efficient and prompt completion of the procedure, minimizing total operative time and muscle ischemia.
Patient Positioning and Optimization
Before the procedure is begun, preparations must be made for:
* Appropriate monitoring of vital signs and core body temperature.
* Meticulous padding of all bony prominences to prevent positioning neuropathies during the prolonged surgery.
* Application of a forced-air heating blanket to maintain normothermia, which is critical for preventing peripheral vasospasm.
* Insertion of an indwelling urinary catheter.
SURGICAL TECHNIQUE: FOREARM PREPARATION
The following steps detail the preparation of the forearm recipient site (Surgical Technique 63-23), assuming the restoration of finger flexion.
1. Incision and Exposure
- Determine the general location of the recipient arteries and nerves based on previous surgical procedures on the forearm, preoperative clinical examination, electromyography, and angiography.
- Use a pneumatic tourniquet to allow rapid, bloodless initial dissection. Inflate and deflate the tourniquet as needed after the initial dissection to assess vessel pulsatility and control hemostasis.
- Usually, an extensive curved or a zigzag (Bruner-type) incision is required for adequate exposure of the entire volar forearm.
- If extensive scarring is present (e.g., post-Volkmann's contracture), carefully dissect from normal, uninjured areas proximally and distally into the scarred areas. This centripetal approach is vital to avoid iatrogenic injury to the delicate recipient vessels and nerves encased in scar tissue.
2. Neurovascular Identification and Template Planning
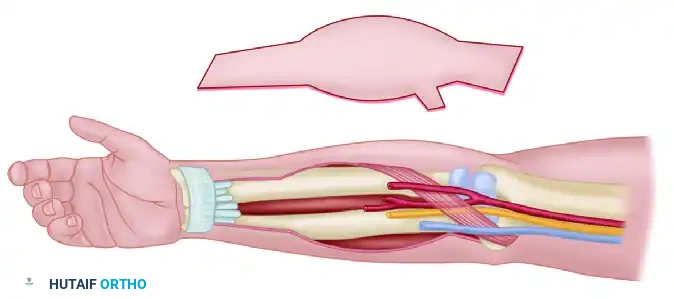
- Fashion a paper or sterile foil template to assist in locating the neurovascular pedicle and planning the inset. This template also helps to determine the exact area of needed skin coverage if a musculocutaneous flap is being utilized.

FIGURE 63-40: Free transfer, functioning muscle. A paper template assists in the preoperative and intraoperative planning of skin coverage requirements, musculotendinous attachments, and neurovascular repairs. NVB, neurovascular bundle.
- Arterial Selection: Plan to use the radial or ulnar artery, or a suitable large branch (e.g., common interosseous artery), as a recipient vessel. Ensure the vessel is cut back to healthy intima, free of post-traumatic fibrosis.
- Venous Selection: For venous drainage, use the venae comitantes of the selected arteries or robust superficial veins (e.g., cephalic or basilic veins) in the area.
- Neural Selection: On the flexor aspect, plan to expose the ulnar, median, and anterior interosseous nerves as needed. Exposure of the anterior interosseous nerve and artery may require sectioning of the pronator teres in a Z-configuration, allowing for later anatomical repair.
3. Tendon Preparation
- Expose the tendons of the flexor digitorum profundus (FDP), and mobilize them by dissecting them free from surrounding scar tissue to ensure satisfactory gliding.
- Identify the flexor tendons distally for flap attachment.
- Proximally, plan to expose the medial epicondyle and surrounding antebrachial fascia. This provides a robust, anatomical anchor point for the proximal attachment of the transferred muscle, mimicking the natural origin of the forearm flexors.
MUSCLE HARVEST, TRANSFER, AND INSET
(Note: The specific harvest of the donor muscle—e.g., Gracilis or Latissimus Dorsi—is performed simultaneously by the second surgical team according to standard microsurgical principles, ensuring a long neurovascular pedicle is obtained.)
Ischemia Time and Revascularization
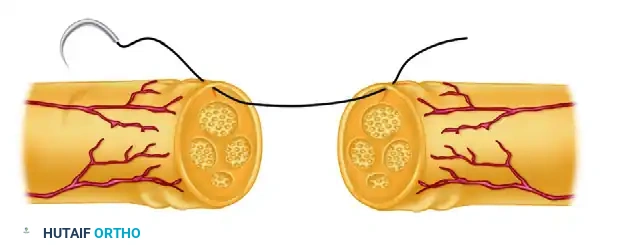
Once the muscle is detached from the donor site, the ischemia time begins. The muscle is transferred to the forearm, and the microvascular anastomoses are performed immediately to minimize warm ischemia.
* The arterial anastomosis is typically performed end-to-end or end-to-side to the radial or ulnar artery using 8-0 or 9-0 nylon under the operating microscope.
* At least one, preferably two, venous anastomoses are performed to ensure adequate outflow and prevent congestive failure of the flap.
Muscle Tensioning (The Blix Curve)
The biomechanical success of the transfer relies entirely on setting the muscle at its optimal resting length. According to the Blix curve (length-tension relationship), a muscle generates maximum active tension at its physiological resting length.
* Prior to harvest, placing marking sutures on the donor muscle at a known distance (e.g., exactly 5 cm apart) while the muscle is at resting length is a critical step.
* During inset at the recipient site, the proximal origin is secured to the medial epicondyle.
* The distal tendons are then interwoven (using a Pulvertaft weave) into the recipient FDP tendons. The tension is adjusted until the marking sutures are once again exactly 5 cm apart, ensuring the muscle is restored to its optimal physiological length.
Microneural Coaptation
The donor motor nerve is coapted to the recipient motor nerve (e.g., the AIN) using 9-0 or 10-0 epineurial sutures.
Surgical Warning: The nerve repair must be entirely tension-free and placed as close to the motor endplates of the transferred muscle as possible. This minimizes the distance regenerating axons must travel, thereby reducing the reinnervation time and preventing irreversible motor endplate degradation.
POSTOPERATIVE PROTOCOL AND REHABILITATION
The postoperative management of a functioning free muscle transfer is as critical as the surgical execution. A specialized, phased rehabilitation protocol is mandatory.
Phase 1: Immobilization and Vascular Protection (Weeks 0–3)
- The extremity is immobilized in a bulky, non-compressive splint. For flexor transfers, the wrist and fingers are placed in slight flexion to remove tension from the distal tendon repairs.
- Strict elevation and microvascular monitoring (e.g., implantable Doppler, surface temperature monitoring, or clinical flap checks) are instituted for the first 5 to 7 days.
- No active or passive motion of the involved joints is permitted to allow for tendon healing and to protect the microvascular anastomoses.
Phase 2: Early Passive Motion (Weeks 3–8)
- The splint is modified to allow controlled passive range of motion (PROM) to prevent joint stiffness and promote tendon gliding.
- Active contraction of the transferred muscle is strictly prohibited, as the tendon weaves are not yet fully mature, and the muscle is denervated.
Phase 3: Reinnervation and Active Motion (Months 3–12+)
- Clinical signs of reinnervation (Tinel's sign advancing along the nerve, visible muscle flicker, or EMG evidence of motor unit potentials) typically appear between 3 to 6 months, depending on the distance from the nerve repair to the motor endplates.
- Once reinnervation is confirmed, active motion exercises commence.
- Biofeedback and Cortical Plasticity: Because the transferred muscle is often innervated by a nerve that previously served a different function (e.g., using an intercostal nerve for arm flexion), the patient must undergo extensive neuromuscular re-education. Biofeedback and electrical muscle stimulation (EMS) are highly effective in helping the patient develop new cortical pathways to achieve voluntary control of the transferred muscle.
COMPLICATIONS AND PITFALLS
Despite meticulous technique, FFMT carries significant risks:
1. Microvascular Thrombosis: The most devastating complication, usually occurring within the first 72 hours. Immediate re-exploration is mandatory if vascular compromise is suspected.
2. Failure of Reinnervation: Can occur due to excessive tension on the nerve repair, a scarred recipient nerve bed, or prolonged denervation time leading to motor endplate fibrosis.
3. Inadequate Excursion or Strength: Often the result of improper intraoperative tensioning (failing to respect the resting length), severe postoperative adhesions, or selecting a donor muscle with insufficient PCSA for the required task.
4. Tendon Rupture: Premature active motion or aggressive passive stretching before the Pulvertaft weaves have achieved sufficient tensile strength can lead to catastrophic rupture at the musculotendinous junction.
You Might Also Like