Early Management and Microsurgical Repair of Peripheral Nerve Injuries

Key Takeaway
Early management of peripheral nerve injuries demands meticulous assessment, optimal surgical timing, and precise microsurgical technique. Primary neurorrhaphy is indicated for clean, sharp lacerations, whereas delayed repair suits crush or blast injuries, allowing tissue demarcation. Successful nerve regeneration relies on patient age, tension-free coaptation, accurate fascicular alignment, and minimizing surgical delay to preserve motor endplate viability and maximize functional recovery.
GENERAL CONSIDERATIONS IN PERIPHERAL NERVE TRAUMA
The initial management of a patient presenting with peripheral nerve damage must strictly adhere to Advanced Trauma Life Support (ATLS) principles. As with any high-energy or penetrating trauma, the preservation of life and limb takes absolute precedence over the restoration of neurologic function. Cardiopulmonary stabilization, hemorrhage control, and the prevention of shock are the primary objectives. Once major visceral injuries have been addressed and resuscitative measures are underway, the orthopedic surgeon must systematically evaluate the peripheral nerve deficit.
A meticulous clinical examination is paramount. The specific nerve deficit must be documented with discrete motor grading and sensory mapping before any anesthetic is administered. In the setting of an open wound, systemic antibiotics and tetanus prophylaxis are administered immediately.
Wound Management and Surgical Timing
The nature of the wound dictates the timing of surgical intervention.
Immediate Primary Repair:
If the wound is clean and sharply incised (e.g., a knife or glass laceration), the patient is hemodynamically stable, and the surgical environment is optimal (availability of an operating microscope, microsurgical instruments, and experienced personnel), immediate primary repair of the nerve is the gold standard.
Delayed Primary Repair (3 to 7 Days):
If the patient's general medical condition precludes immediate lengthy microsurgery, or if the surgical team lacks the necessary equipment at the time of presentation, delayed primary repair within the first 3 to 7 days is preferred. The wound is thoroughly irrigated, débrided of foreign material and necrotic tissue, and closed or covered with a sterile dressing. The patient is closely observed for signs of sepsis.
Secondary Repair (3 to 6 Weeks):
When open wounds are the result of high-velocity missiles, severe crush injuries, or heavy contamination (e.g., agricultural accidents), immediate repair is contraindicated. The zone of injury in these cases is often indistinguishable acutely.
1. The wound undergoes aggressive, serial débridement.
2. If the severed nerve ends are identified during débridement, they should be tagged with non-absorbable, radio-opaque sutures (such as stainless steel or prolene) to facilitate future identification.
3. In the absence of a significant gap, loose end-to-end apposition with a single epineurial suture prevents retraction. If a segmental gap exists, the nerve ends should be tacked to adjacent stable soft tissues.
4. Definitive neurorrhaphy is delayed for 3 to 6 weeks. By this time, soft tissues have healed, the extent of intraneural scarring and neuroma formation is clearly demarcated, and the risk of infection is minimized.
Surgical Warning: Never attempt definitive primary neurorrhaphy in a heavily contaminated wound or a blast injury. The microscopic zone of trauma extends far beyond the macroscopic laceration, guaranteeing failure and severe neuroma-in-continuity formation.
Management of Closed Nerve Injuries
Closed peripheral nerve injuries (e.g., traction injuries, blunt trauma) require a conservative initial approach. Careful documentation of residual function is essential.
* Rehabilitation: Once initial pain subsides, early active motion of all uninvolved joints must commence. Gentle passive range of motion (ROM) exercises are instituted to prevent soft-tissue contractures and keep joints supple.
* Splinting: Dynamic and static splinting should be used intermittently to support paralyzed joints and prevent fixed deformities.
* Monitoring: The progress of reinnervation is tracked clinically and via serial Electromyography (EMG) and Nerve Conduction Velocity (NCV) studies, typically beginning 3 to 4 weeks post-injury.
* Surgical Exploration: If a closed fracture is complicated by a nerve deficit, early exploration is generally avoided, awaiting spontaneous recovery (neurapraxia or axonotmesis). Conversely, if a nerve deficit acutely follows the closed reduction or casting of a fracture, immediate surgical exploration is mandated to rule out nerve entrapment or iatrogenic laceration.
FACTORS INFLUENCING REGENERATION AFTER NEURORRHAPHY
The success of peripheral nerve repair is multifactorial. Extensive data from military conflicts and civilian trauma centers have elucidated several critical variables that dictate functional outcomes. Associated vascular injuries, which cause tissue ischemia, severely compromise nerve regeneration and must be addressed concurrently.
1. Age of the Patient
Age is arguably the most significant independent prognostic factor for nerve regeneration. Neurorrhaphies are vastly more successful in children and adolescents than in adults.
* Neuroplasticity: The superior results in youth are largely attributed to the immense potential for central nervous system adaptation to altered peripheral sensory input.
* Clinical Outcomes: Studies by Omer and Onne demonstrate that patients under 20 years of age achieve the most robust recovery. For median and ulnar nerve repairs, patients under 10 years old frequently achieve 2-point discrimination of less than 10 mm. In contrast, adults between 20 and 40 years typically plateau at 30 mm.
2. Gap Between Nerve Ends and Tension
The mechanism of injury dictates the size of the nerve defect. Sharp lacerations result in minimal tissue loss, allowing for primary end-to-end repair. High-velocity missile injuries cause extensive proximal and distal necrosis, necessitating wide resection of the damaged nerve ends until healthy, pouting fascicles are visualized.

The image above illustrates the resection of a neuroma and the subsequent attempt at end-to-end repair. Note that excessive tension or extreme joint flexion used to close a large gap leads to ischemia, intraneural fibrosis, and ultimate failure of regeneration.
Methods to overcome nerve gaps include:
* Extensive proximal and distal nerve mobilization.
* Anatomic transposition (e.g., anterior submuscular transposition of the ulnar nerve).
* Mild joint flexion (must be used with extreme caution to avoid tension upon extension).
* Interfascicular nerve grafting.
* Bone shortening (rarely indicated, typically reserved for severe replantation scenarios).
Clinical Pearl: Tension is the enemy of nerve regeneration. Millesi's foundational research proved that tension at the coaptation site induces ischemia and dense intraneural scarring. If a gap exceeds 2.5 cm after mobilization, or if an 8-0 nylon suture cannot hold the nerve ends together without tearing, an interfascicular nerve graft is strictly indicated.
3. Delay Between Injury and Repair
The timing of repair profoundly affects motor recovery due to the progressive, irreversible atrophy of denervated striated muscle and the degradation of motor endplates.
* Motor Decline: For every 6 days of delay between injury and repair, there is an approximate 1% loss of maximal potential motor recovery. After 3 months, this loss accelerates exponentially.
* Sensory Return: Sensory receptors are more resilient to prolonged denervation. Useful protective sensation has been documented even when repair is delayed up to 2 years, though early repair significantly reduces painful paresthesias and improves tactile gnosis.
* Metabolic Timetable: Ducker's experimental work established that between 3 and 6 weeks, the cell body (chromatolysis) and the nerve trunks (Wallerian degeneration) reach peak metabolic readiness for axonal regeneration, supporting the rationale for delayed primary repair in complex wounds.
4. Level of Injury
The anatomical level of the injury inversely correlates with the quality of recovery. Proximal lesions (e.g., brachial plexus, proximal sciatic nerve) yield poorer outcomes in distal structures compared to distal lesions (e.g., wrist-level median nerve).
* Retrograde Degeneration: Neurons innervating distal targets suffer more severe retrograde cell death following a proximal injury.
* Axonal Misdirection: The distance regenerating axons must travel increases the probability of axonal misrouting into incorrect endoneurial tubes.
* Time to Target: Because axons regenerate at approximately 1 mm per day, proximal injuries may result in target muscle denervation times that exceed the viability of the motor endplates (typically 12 to 18 months).
5. Condition of the Nerve Ends and Fascicular Alignment
Meticulous handling of the nerve ends is critical. The surgeon must ensure asepsis, preserve the delicate segmental vasa nervorum, and provide a well-vascularized soft-tissue bed free of restrictive scar tissue.
Distal stump shrinkage occurs over time, reducing the cross-sectional area by up to 40% at 4 months. Furthermore, the internal topography of peripheral nerves is highly complex, with fascicles constantly dividing and merging (intraneural plexus formation).
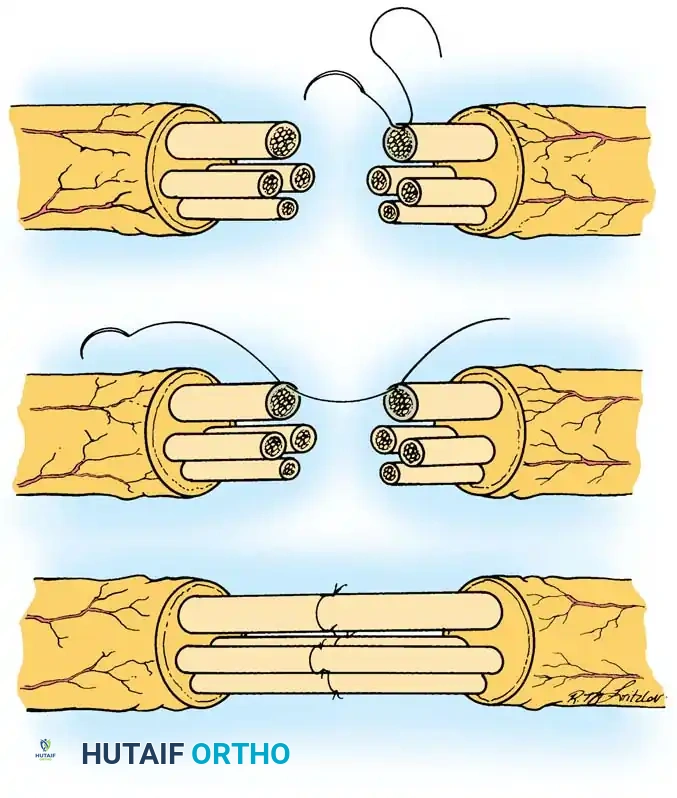
The image above demonstrates the technique of group fascicular repair. Precise alignment of corresponding motor and sensory fascicular groups is paramount to prevent axonal cross-innervation and ensure functional recovery.
Edshage demonstrated that a visually perfect external epineurial repair does not guarantee internal fascicular alignment. Therefore, the nerve ends must be sharply trimmed using a diamond knife or specialized nerve scissors until healthy, herniating fascicles are visible. No fibrotic tissue or foreign material can remain at the coaptation site.
SURGICAL TECHNIQUES IN NERVE REPAIR
Preparation and Positioning
- Anesthesia: General anesthesia or a robust regional block is required to prevent patient movement during microsurgery.
- Tourniquet: A pneumatic tourniquet is utilized for a bloodless field during exposure and preparation but should be deflated prior to final coaptation to ensure absolute hemostasis. Hematoma at the repair site promotes fibrosis.
- Magnification: Operating loupes (minimum 3.5x to 4.5x) are used for exposure. An operating microscope is mandatory for fascicular dissection and coaptation.
Epineurial vs. Fascicular Repair
- Epineurial Repair: Indicated for pure motor or pure sensory nerves, or sharply lacerated mixed nerves where the fascicular topography perfectly matches. Interrupted 8-0 or 9-0 non-absorbable monofilament sutures are placed through the epineurium.
- Group Fascicular Repair: Indicated for mixed nerves (e.g., median or ulnar nerves at the wrist) where motor and sensory fascicle groups can be distinctly identified based on topography or intraoperative awake stimulation. Sutures (10-0 nylon) are placed through the perineurium of matching fascicular groups.
Nerve Grafting Technique
When a tension-free primary repair is impossible, autologous nerve grafting is the gold standard. The sural nerve is the most commonly utilized donor due to its length (up to 40 cm), favorable fascicular architecture, and minimal donor-site morbidity (lateral foot numbness).
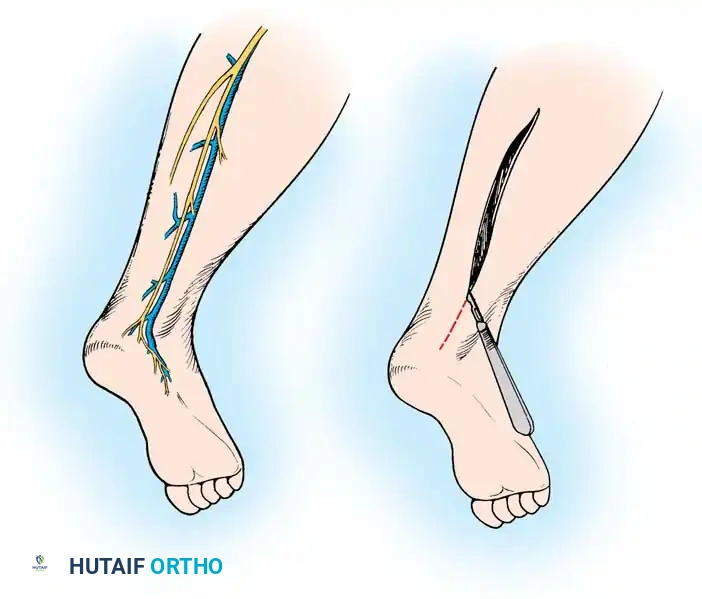
The image above depicts the surgical approach for harvesting the sural nerve. A longitudinal or step-ladder incision is made along the posterolateral calf, tracing the nerve alongside the lesser saphenous vein.
Sural Nerve Harvest Steps:
1. Position the patient prone or lateral decubitus.
2. Identify the sural nerve posterior to the lateral malleolus.
3. Make a longitudinal incision (or multiple step-ladder incisions) tracking proximally toward the popliteal fossa.
4. Carefully dissect the nerve from the lesser saphenous vein, ligating any crossing venous branches.
5. Transect the nerve proximally and distally, yielding the required length.
6. Reverse the graft during interposition. Because nerves branch distally, reversing the graft prevents regenerating axons from escaping through severed side branches.
7. Perform interfascicular coaptation using 10-0 nylon under the microscope, ensuring zero tension. Fibrin glue may be used to augment the repair.
POSTOPERATIVE PROTOCOL AND REHABILITATION
The success of a flawless microsurgical repair can be entirely undone by improper postoperative care.
- Immobilization: The limb is immobilized in a well-padded plaster splint for 3 weeks to protect the coaptation site from tension. The joints are positioned in a neutral, relaxed posture. Extreme flexion to protect the repair is avoided, as it leads to severe joint contractures and subsequent nerve traction during rehabilitation.
- Progressive Mobilization: At 3 weeks, the splint is removed, and a supervised, progressive active ROM program is initiated. A hinged brace may be used to gradually increase extension by 10 degrees per week.
- Sensory Re-education: As Tinel's sign advances and protective sensation returns, formal sensory re-education programs are vital to maximize cortical integration of the new sensory input.
- Motor Rehabilitation: Electrical stimulation of denervated muscle remains controversial, but active engagement of intact synergistic muscles is mandatory. Dynamic splinting (e.g., a radial nerve palsy extension splint) is utilized to prevent overstretching of denervated muscles and to maintain functional hand use while awaiting reinnervation.
- Long-Term Follow-Up: Serial clinical examinations and EMG studies are conducted every 3 to 6 months. If regenerating axons fail to cross the repair site (arrested Tinel's sign) or if functional recovery plateaus prematurely, secondary procedures such as tendon transfers or nerve neurolysis must be considered.
You Might Also Like