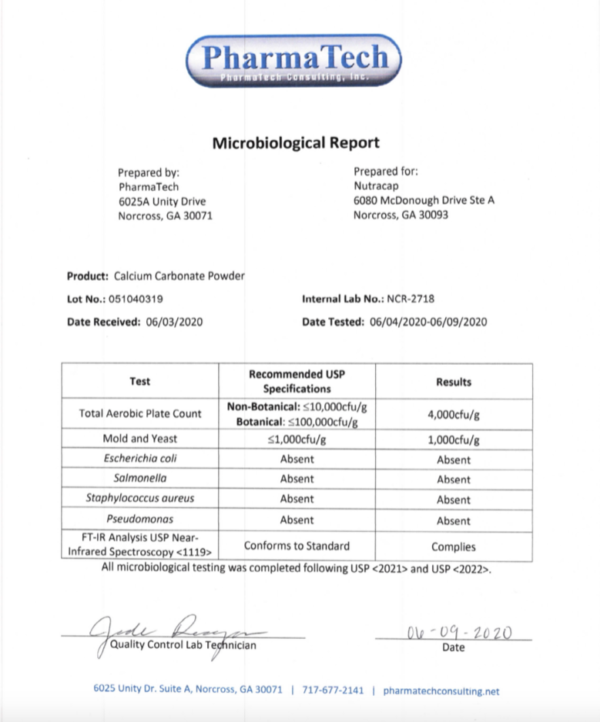
Calcium Carbonate: An Exhaustive Medical SEO Guide for Orthopedic Health and Beyond
Calcium carbonate is a widely recognized and essential compound, playing a pivotal role in human health. As an expert medical SEO copywriter and orthopedic specialist, this guide aims to provide a comprehensive, authoritative, and in-depth understanding of Calcium Carbonate, from its fundamental mechanisms to its diverse clinical applications and crucial safety considerations. Primarily known for its role in bone health, it also serves as a potent antacid and a critical phosphate binder in specific medical conditions.
1. Introduction & Overview of Calcium Carbonate
Calcium carbonate (CaCO₃) is a chemical compound commonly found in nature as minerals like calcite and aragonite, and as the primary component of shells of marine organisms, pearls, and eggshells. In medicine, it is a highly valued over-the-counter (OTC) supplement and medication. It is the most concentrated form of calcium available in supplements, containing approximately 40% elemental calcium by weight.
Its significance in the human body cannot be overstated. Calcium is the most abundant mineral in the body, vital not only for the structural integrity of bones and teeth but also for critical physiological processes including nerve transmission, muscle contraction, blood clotting, and hormonal secretion. When dietary intake of calcium is insufficient, Calcium Carbonate supplements become indispensable.
Beyond its role as a calcium source, Calcium Carbonate's alkaline properties make it an effective antacid, neutralizing stomach acid to relieve symptoms of heartburn and indigestion. Furthermore, in patients with chronic kidney disease, it is utilized as a phosphate binder to manage elevated serum phosphate levels.
2. Deep-Dive into Technical Specifications & Mechanisms
Understanding how Calcium Carbonate works is crucial for appreciating its therapeutic benefits and potential risks.
2.1. Mechanism of Action
Calcium Carbonate exerts its therapeutic effects through two primary mechanisms:
2.1.1. As a Calcium Supplement
When ingested, Calcium Carbonate dissociates in the acidic environment of the stomach into calcium ions (Ca²⁺) and carbonate ions (CO₃²⁻). The elemental calcium ions are then absorbed in the small intestine. This absorbed calcium is utilized by the body for various functions:
- Bone Formation and Remodeling: Calcium is the primary mineral component of hydroxyapatite crystals, which form the rigid matrix of bones. Adequate calcium intake is essential for bone density, strength, and continuous remodeling processes that repair microscopic damage.
- Nerve Impulse Transmission: Calcium ions play a critical role in the release of neurotransmitters at synapses, facilitating communication between nerve cells.
- Muscle Contraction: Calcium is essential for the interaction between actin and myosin filaments, enabling muscle cells to contract.
- Blood Coagulation: Calcium ions (Factor IV) are a vital cofactor in the cascade of reactions that lead to blood clot formation.
- Hormone Secretion: Calcium is involved in the release of various hormones and enzymes.
2.1.2. As an Antacid
In the stomach, Calcium Carbonate directly reacts with hydrochloric acid (HCl), the primary acid produced by the stomach, to form calcium chloride (CaCl₂), water (H₂O), and carbon dioxide (CO₂).
CaCO₃ (s) + 2HCl (aq) → CaCl₂ (aq) + H₂O (l) + CO₂ (g)
This neutralization reaction rapidly reduces stomach acidity, providing symptomatic relief from heartburn, indigestion, and sour stomach. The carbon dioxide gas produced can sometimes lead to belching or bloating.
2.1.3. As a Phosphate Binder
In patients with chronic kidney disease, impaired kidney function leads to an inability to excrete phosphate effectively, resulting in hyperphosphatemia. When taken with meals, Calcium Carbonate reacts with dietary phosphate in the gastrointestinal tract to form insoluble calcium phosphate compounds. These insoluble compounds are not absorbed and are subsequently excreted in the feces, thereby reducing the absorption of dietary phosphate and lowering serum phosphate levels.
2.2. Pharmacokinetics
The journey of Calcium Carbonate through the body involves several key stages:
- Absorption:
- Calcium Carbonate absorption primarily occurs in the small intestine.
- It requires an acidic environment for optimal dissolution and ionization into elemental calcium. Therefore, it is typically recommended to be taken with food, as stomach acid production is stimulated during meals.
- Absorption involves both active transport (saturable, vitamin D-dependent) and passive diffusion (non-saturable).
- Bioavailability is variable, generally ranging from 20-30%, but can be influenced by factors like age, vitamin D status, and concomitant food intake.
- Distribution:
- Once absorbed, calcium is transported in the blood, primarily bound to albumin (approximately 40-45%), with a smaller fraction complexed with anions (e.g., citrate, phosphate) and about 50% existing as free, ionized calcium (the biologically active form).
- The vast majority (99%) of total body calcium is stored in bones and teeth.
- Metabolism:
- Calcium is an elemental mineral and is not metabolized in the traditional sense by enzymes. Instead, it is actively involved in numerous metabolic pathways and physiological processes.
- Excretion:
- Excess calcium is primarily excreted by the kidneys via urine. Renal calcium excretion is tightly regulated by parathyroid hormone (PTH) and calcitonin.
- A smaller amount is excreted in feces (unabsorbed calcium and calcium secreted into the gut).
3. Extensive Clinical Indications & Usage
Calcium Carbonate's versatility makes it a cornerstone in managing various health conditions.
3.1. Calcium Supplementation
- Osteoporosis Prevention and Treatment: Crucial for maintaining bone mineral density, especially in post-menopausal women and the elderly, to prevent fractures. Often combined with Vitamin D.
- Osteopenia: Management of reduced bone density to prevent progression to osteoporosis.
- Hypocalcemia: Treatment of low blood calcium levels due due to various causes:
- Hypoparathyroidism (underactive parathyroid glands).
- Chronic renal failure (impaired vitamin D activation and phosphate retention).
- Vitamin D deficiency (leading to impaired calcium absorption).
- Pancreatitis.
- Rickets and Osteomalacia: In children and adults, respectively, often in conjunction with Vitamin D supplementation to support proper bone mineralization.
- Growth and Development: Essential for children and adolescents during periods of rapid bone growth.
- Pregnancy and Lactation: Increased calcium requirements during these periods to support fetal skeletal development and milk production.
- Post-menopausal Women: To counteract accelerated bone loss due to estrogen decline.
3.2. Antacid
- Dyspepsia: Relief of symptoms such as heartburn, indigestion, and sour stomach.
- Gastroesophageal Reflux Disease (GERD): Symptomatic relief of acid reflux. Not a primary treatment for erosive esophagitis but can manage episodic symptoms.
- Peptic Ulcer Disease: Adjunctive therapy for temporary symptom relief, though not a curative treatment.
3.3. Phosphate Binder
- Hyperphosphatemia in Chronic Kidney Disease (CKD): Used to reduce elevated serum phosphate levels in patients with CKD, particularly those on dialysis, by binding dietary phosphate in the gut.
3.4. Dosage Guidelines
Dosage varies significantly based on the indication, patient age, and individual needs. It is crucial to consider the elemental calcium content.
-
General Principles:
- Elemental Calcium: Calcium Carbonate is approximately 40% elemental calcium. For example, a 1250 mg tablet of calcium carbonate provides 500 mg of elemental calcium.
- Divided Doses: For optimal absorption and to minimize side effects, calcium supplements are best taken in divided doses, typically not exceeding 500-600 mg of elemental calcium per single dose.
- With Food: Always take Calcium Carbonate with food to enhance absorption (due to increased stomach acid production) and reduce gastrointestinal upset.
-
Typical Adult Dosages:
| Indication | Elemental Calcium (Daily) | Administration Notes |
|---|---|---|
| Calcium Supplementation | ||
| Osteoporosis Prevention | 1000-1200 mg | Usually in 2-3 divided doses with meals. |
| Hypocalcemia | Highly variable | Based on serum calcium levels, often higher doses. |
| Pregnancy/Lactation | 1000-1300 mg | Based on age, in divided doses. |
| Antacid | ||
| Heartburn/Indigestion | 500-1000 mg | As needed, up to 7500 mg (elemental) per day (short-term). |
| Phosphate Binder (CKD) | ||
| Hyperphosphatemia | 500-2000 mg | With each meal, dose titrated to phosphate levels. |
- Pediatric Doses: Should always be determined by a healthcare professional based on age, weight, and specific medical condition.
- Administration: Swallow tablets whole with water. Chewable forms should be thoroughly chewed. Avoid taking with high-fiber meals, as fiber can interfere with calcium absorption.
4. Risks, Side Effects, and Contraindications
While generally safe, Calcium Carbonate is not without risks.
4.1. Contraindications
- Hypercalcemia: Pre-existing high levels of calcium in the blood.
- Severe Hypercalciuria: Excessive calcium excretion in the urine.
- History of Renal Calculi (Kidney Stones): Especially calcium-oxalate stones, as calcium carbonate can exacerbate stone formation.
- Hypersensitivity: Known allergy to calcium carbonate or any of its components.
- Digitalis Toxicity: Calcium can potentiate the effects of cardiac glycosides like digoxin, increasing the risk of toxicity.
4.2. Side Effects
Most side effects are mild and gastrointestinal in nature.
-
Common Side Effects:
- Constipation
- Bloating
- Gas (due to CO₂ production)
- Stomach upset/discomfort
- Belching
-
Less Common/Serious Side Effects (often associated with high doses or prolonged use):
- Hypercalcemia: Symptoms include nausea, vomiting, loss of appetite, thirst, frequent urination, muscle weakness, fatigue, confusion, and in severe cases, cardiac arrhythmias or coma.
- Milk-Alkali Syndrome: A serious condition caused by excessive intake of calcium and absorbable alkali (like calcium carbonate), leading to hypercalcemia, metabolic alkalosis, and renal impairment.
- Kidney Stones: Increased risk, particularly in susceptible individuals.
- Renal Insufficiency: Worsening of kidney function in predisposed individuals.
4.3. Drug Interactions
Calcium Carbonate can significantly interact with various medications, affecting either its own absorption or the absorption/efficacy of other drugs. Careful spacing of doses is often required.
4.3.1. Drugs Whose Absorption/Efficacy is Decreased by Calcium Carbonate
- Bisphosphonates (e.g., Alendronate, Risedronate, Ibandronate): Calcium can chelate these drugs, significantly reducing their absorption. Separate by at least 30 minutes (risedronate) to 2 hours (alendronate).
- Thyroid Hormones (e.g., Levothyroxine): Calcium can bind to levothyroxine, reducing its absorption. Separate by at least 4 hours.
- Tetracycline Antibiotics (e.g., Doxycycline, Minocycline): Calcium forms insoluble chelates, reducing antibiotic absorption and efficacy. Separate by at least 2-4 hours.
- Fluoroquinolone Antibiotics (e.g., Ciprofloxacin, Levofloxacin): Similar to tetracyclines, calcium chelates these antibiotics. Separate by at least 2-6 hours.
- Iron Supplements: Calcium can inhibit iron absorption. Separate by at least 2 hours.
- Phenytoin: Calcium can reduce phenytoin levels. Monitor carefully.
- Gabapentin: Calcium can reduce gabapentin absorption. Separate by at least 2 hours.
- Mycophenolate Mofetil: Calcium can reduce its absorption.
4.3.2. Drugs That Affect Calcium Carbonate Absorption/Efficacy
- Proton Pump Inhibitors (PPIs) (e.g., Omeprazole, Pantoprazole) and H2 Blockers (e.g., Ranitidine, Famotidine): These medications reduce stomach acid, which is necessary for Calcium Carbonate dissolution and absorption. Consider Calcium Citrate (which doesn't require acid) if on long-term acid suppressants, or take Calcium Carbonate with food.
- Corticosteroids (e.g., Prednisone): Long-term use can decrease calcium absorption and increase calcium excretion, necessitating higher calcium intake.
4.3.3. Increased Risk of Side Effects with Concomitant Use
- Thiazide Diuretics (e.g., Hydrochlorothiazide): These diuretics reduce renal calcium excretion, increasing the risk of hypercalcemia when taken with calcium supplements.
- Digitalis Glycosides (e.g., Digoxin): Hypercalcemia can increase the risk of digitalis toxicity, leading to cardiac arrhythmias. Monitor calcium levels closely.
- Vitamin D Supplements: While often co-administered, excessive intake of both can lead to hypercalcemia and hypercalciuria.
4.3.4. Food Interactions
- Oxalates (e.g., spinach, rhubarb) and Phytates (e.g., whole grains, beans): These compounds can bind to calcium in the gut, reducing its absorption. While not typically a concern with balanced diets, very high intake can be problematic.
4.4. Pregnancy and Lactation Warnings
- Pregnancy: Calcium Carbonate is generally considered safe and often recommended during pregnancy to meet the increased calcium demands for fetal skeletal development. However, intake should not exceed the recommended daily allowances for pregnant women (typically 1000-1300 mg elemental calcium/day, depending on age), unless directed by a physician. Excessive calcium intake can be harmful.
- Lactation: Calcium requirements remain high during lactation to support milk production. Calcium Carbonate is safe and often recommended for breastfeeding mothers, again within recommended daily allowances. Small amounts of calcium are excreted in breast milk, but this is normal and beneficial for the infant.
4.5. Overdose Management
Acute or chronic overdose of Calcium Carbonate can lead to hypercalcemia.
-
Symptoms of Hypercalcemia: Mild hypercalcemia may be asymptomatic, but symptoms can include:
- Nausea, vomiting, constipation, abdominal pain, loss of appetite.
- Increased thirst (polydipsia) and frequent urination (polyuria).
- Fatigue, muscle weakness, lethargy.
- Confusion, disorientation, depression.
- In severe cases: Cardiac arrhythmias, kidney dysfunction, coma.
-
Treatment of Overdose:
- Discontinuation: Immediately stop Calcium Carbonate and any other calcium or vitamin D supplements.
- Hydration: Administer intravenous normal saline to promote renal calcium excretion and correct dehydration.
- Diuretics: Loop diuretics (e.g., Furosemide) may be used after adequate hydration to enhance calcium excretion, but thiazide diuretics should be avoided as they decrease calcium excretion.
- Pharmacological Interventions (for severe hypercalcemia):
- Calcitonin: Can rapidly but transiently lower serum calcium.
- Bisphosphonates: (e.g., Pamidronate, Zoledronic acid) can inhibit bone resorption and effectively lower calcium over several days.
- Corticosteroids: May be used if hypercalcemia is due to granulomatous diseases or vitamin D intoxication.
- Dialysis: In life-threatening hypercalcemia unresponsive to other treatments, hemodialysis may be necessary.
- Monitoring: Closely monitor serum calcium, phosphate, magnesium, potassium, renal function, and electrocardiogram (ECG) to detect arrhythmias.
5. Massive FAQ Section
Here are some frequently asked questions about Calcium Carbonate:
Q1: What is the best time to take Calcium Carbonate?
A1: Calcium Carbonate is best taken with meals. The stomach acid produced during digestion helps dissolve the calcium carbonate, making the elemental calcium more available for absorption.
Q2: Should I take Calcium Carbonate with food?
A2: Yes, absolutely. Taking Calcium Carbonate with food significantly enhances its absorption and helps minimize gastrointestinal side effects like gas and constipation.
Q3: What's the difference between Calcium Carbonate and Calcium Citrate?
A3: The main differences are elemental calcium content and absorption requirements. Calcium Carbonate contains about 40% elemental calcium and requires stomach acid for optimal absorption. Calcium Citrate contains about 21% elemental calcium but does not require stomach acid for absorption, making it a good option for people on acid-reducing medications or those with low stomach acid.
Q4: Can Calcium Carbonate cause constipation?
A4: Yes, constipation is one of the most common side effects of Calcium Carbonate. Ensuring adequate fluid intake and dietary fiber, or switching to a different form of calcium or a stool softener, may help.
Q5: Is Calcium Carbonate safe during pregnancy?
A5: Yes, Calcium Carbonate is generally considered safe and is often recommended during pregnancy to meet increased calcium needs for fetal development. However, always consult your doctor for appropriate dosing and to ensure it's within recommended daily allowances.
Q6: How much elemental calcium is in Calcium Carbonate?
A6: Calcium Carbonate contains approximately 40% elemental calcium by weight. So, a 1250 mg tablet of calcium carbonate provides 500 mg of elemental calcium.
Q7: Can Calcium Carbonate interact with other medications?
A7: Yes, Calcium Carbonate can interact with many medications, including certain antibiotics (tetracyclines, fluoroquinolones), bisphosphonates, thyroid hormones (levothyroxine), and iron supplements. It's crucial to separate the administration times of these medications by several hours. Always inform your doctor or pharmacist about all medications you are taking.
Q8: What are the signs of taking too much Calcium Carbonate (hypercalcemia)?
A8: Signs of hypercalcemia can include nausea, vomiting, constipation, increased thirst and urination, muscle weakness, fatigue, and confusion. If you experience these symptoms, stop taking calcium and seek medical attention immediately.
Q9: Does Calcium Carbonate help with acid reflux?
A9: Yes, Calcium Carbonate works as an antacid by neutralizing stomach acid, providing rapid, temporary relief from symptoms of heartburn and acid reflux.
Q10: Can children take Calcium Carbonate?
A10: Yes, children can take Calcium Carbonate for calcium supplementation, but the dosage must be carefully determined by a pediatrician based on the child's age, weight, and specific needs.
Q11: How long does it take for Calcium Carbonate to work as an antacid?
A11: As an antacid, Calcium Carbonate typically works very quickly, often within minutes, to neutralize stomach acid and relieve symptoms of heartburn or indigestion.
Q12: Is Calcium Carbonate good for osteoporosis?
A12: Yes, Calcium Carbonate is a primary and effective supplement for the prevention and treatment of osteoporosis, especially when combined with adequate Vitamin D, as it helps maintain and improve bone mineral density.
This exhaustive guide underscores the profound importance and multifaceted utility of Calcium Carbonate in maintaining overall health, particularly in orthopedic contexts. Always consult with a healthcare professional before starting or altering any medication or supplement regimen.