Distal Femoral Replacement (DFR) for Tumor Prosthesis: A Comprehensive Medical SEO Guide
The field of orthopedic oncology has witnessed remarkable advancements, none more impactful in limb salvage than the evolution of endoprosthetic reconstruction. Among these, the Distal Femoral Replacement (DFR) using a tumor prosthesis stands as a cornerstone procedure, offering hope and functional restoration to patients facing aggressive bone tumors of the distal femur. This comprehensive guide delves into the intricate details of DFR tumor prostheses, from their sophisticated design and biomechanical principles to their critical role in clinical applications, maintenance, and the profound improvements they bring to patient outcomes.
1. Introduction & Overview: The Pinnacle of Limb Salvage
Distal Femoral Replacement (DFR) with a tumor prosthesis is a highly specialized surgical procedure involving the resection of a segment of the distal femur, typically affected by a malignant or aggressive benign tumor, and its reconstruction using a modular metallic implant. This procedure falls under the umbrella of limb-sparing surgery, a modern alternative to amputation that aims to remove the cancerous bone while preserving the limb's function and appearance.
Historically, limb-threatening tumors of the distal femur often necessitated above-knee amputation. However, breakthroughs in chemotherapy, diagnostic imaging, and implant technology have transformed the landscape. Today, DFR offers a viable and often superior option, allowing patients to retain their limb, achieve significant functional recovery, and maintain a better quality of life. The complexity of the knee joint, involving intricate ligamentous structures and articulation with the tibia and patella, makes distal femoral reconstruction particularly challenging and necessitates highly engineered prosthetic solutions.
2. Deep-Dive into Technical Specifications & Mechanisms
The success of a DFR tumor prosthesis hinges on its sophisticated design, choice of materials, and robust biomechanical properties. These devices are engineered to withstand significant loads, replicate knee joint kinematics, and integrate effectively with remaining bone and soft tissues.
2.1. Design Principles and Modularity
Modern DFR prostheses are predominantly modular systems. This modularity is crucial for several reasons:
* Customization: It allows surgeons to precisely match the resected bone length, accommodate varying bone diameters, and restore limb length, preventing significant limb length discrepancy.
* Intraoperative Flexibility: Different component sizes and types (e.g., varying hinge constraints, stem lengths) can be assembled during surgery to achieve optimal fit and stability.
* Revision Potential: Individual components can be replaced during revision surgery if one part fails, rather than the entire prosthesis.
Key Components of a Distal Femoral Tumor Prosthesis:
* Femoral Stem: Inserts into the remaining proximal femur, providing stable fixation. Can be cemented or uncemented (press-fit).
* Distal Femoral Segment: Replaces the resected distal femur, connecting the femoral stem to the knee articulation.
* Articulating Knee Component: Comprises a femoral condylar component (part of the distal femoral segment) and a tibial tray with a polyethylene insert, mimicking the knee joint.
* Hinge Mechanism: Often present in tumor prostheses due to extensive soft tissue and ligamentous resection. Provides inherent stability, controlling flexion-extension and preventing varus/valgus instability. Can be rotating hinge or fixed hinge.
* Augments/Spacers: Used to bridge larger bone defects or adjust limb length precisely.
* Patellar Resurfacing: Optional, depending on patellar involvement or wear.
2.2. Materials Science: Engineering for Longevity and Biocompatibility
The materials chosen for DFR prostheses are critical for their long-term performance, biocompatibility, and mechanical integrity.
| Component | Primary Materials | Key Properties |
|---|---|---|
| Femoral Stem, Distal Femur | Cobalt-Chromium (CoCr) Alloys, Titanium (Ti-6Al-4V) Alloys | High strength-to-weight ratio, corrosion resistance, biocompatibility |
| Articular Surface (Insert) | Ultra-High Molecular Weight Polyethylene (UHMWPE) | Low friction, wear resistance, shock absorption |
| Surface Coatings | Hydroxyapatite (HA), Porous Titanium/Cobalt-Chromium | Enhanced osteointegration (for uncemented fixation), improved bone ingrowth |
- Cobalt-Chromium (CoCr) alloys: Offer excellent wear resistance and high tensile strength, making them suitable for load-bearing components and articular surfaces in some designs.
- Titanium (Ti-6Al-4V) alloys: Known for their superior biocompatibility, lower elastic modulus (closer to bone, reducing stress shielding), and excellent fatigue strength. Often preferred for stems and porous coatings.
- UHMWPE: The gold standard for articular bearing surfaces due to its low coefficient of friction and good wear characteristics. Advanced forms, like highly cross-linked polyethylene, further enhance wear resistance.
- Surface Treatments: Porous coatings (e.g., plasma-sprayed titanium) promote biological fixation through bone ingrowth, while hydroxyapatite coatings can accelerate this process.
2.3. Biomechanics: Restoring Function and Stability
The biomechanical considerations for DFR are paramount to achieving functional outcomes.
* Load Transfer: The prosthesis must effectively transfer axial and rotational loads from the body to the remaining bone without causing stress shielding or implant loosening.
* Joint Kinematics: While a hinge mechanism provides stability, it can limit the natural rotational and translational movements of the knee. Design aims to optimize range of motion (ROM) while maintaining stability.
* Fatigue Resistance: Given the repetitive loading cycles, the implant must be highly resistant to fatigue failure over many years.
* Fixation Stability: Secure fixation of the femoral stem and tibial component is crucial to prevent aseptic loosening, a common long-term complication. Both cemented and uncemented techniques are employed, each with specific advantages and disadvantages regarding immediate stability and long-term biological fixation.
* Patellar Tracking: The design must ensure proper patellar tracking to prevent pain and improve knee extension strength.
3. Extensive Clinical Indications & Usage
DFR with a tumor prosthesis is a highly specialized procedure reserved for specific clinical scenarios following rigorous diagnostic workup and multidisciplinary team discussions.
3.1. Primary Indications
- Malignant Bone Tumors:
- Primary osteosarcoma of the distal femur.
- Ewing's sarcoma involving the distal femur.
- Chondrosarcoma (high-grade) of the distal femur.
- Metastatic carcinoma to the distal femur, especially with impending or pathological fracture, where long-term survival is expected.
- Aggressive Benign Tumors:
- Recurrent or aggressive giant cell tumor of the distal femur that has destroyed significant bone stock.
- Aneurysmal bone cysts with extensive bone destruction.
- Failed Limb-Sparing Procedures: Revision surgery for failed previous reconstructions (e.g., allograft failure, previous endoprosthesis failure).
- Severe Bone Loss: Trauma or infection leading to massive distal femoral bone loss, though less common for tumor prostheses.
3.2. Patient Selection Criteria
Ideal candidates are typically those with:
* Tumors amenable to wide surgical resection with adequate margins.
* Good overall health status, capable of tolerating major surgery and rehabilitation.
* No extensive neurovascular involvement precluding limb salvage.
* Realistic expectations regarding functional outcomes and potential limitations.
* Adequate soft tissue envelope for wound closure and coverage.
3.3. Pre-operative Planning: The Blueprint for Success
Metabolic and imaging studies are crucial:
* Imaging: MRI (for soft tissue extent and marrow involvement), CT scan (for cortical bone detail and prosthesis sizing), PET scan (for staging and metastatic workup), plain radiographs.
* Biopsy: Definitive tissue diagnosis is essential before planning definitive surgery.
* Surgical Margins: Meticulous planning to achieve wide oncologic margins while preserving critical neurovascular structures.
* Limb Length Discrepancy: Careful measurement and planning to prevent significant limb length differences post-reconstruction.
* Implant Sizing: Based on contralateral limb measurements and imaging, trial components are used to determine optimal prosthesis length and diameter.
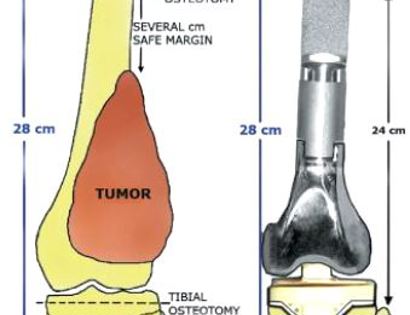
3.4. Detailed Surgical Procedure
The DFR procedure is complex and requires a highly skilled orthopedic oncology surgeon.
1. Patient Positioning and Anesthesia: Supine position, general anesthesia. Tourniquet application for bloodless field.
2. Surgical Approach: Typically an anterior or anteromedial approach to the distal femur and knee joint.
3. Tumor Resection: Meticulous dissection to identify and protect neurovascular bundles (femoral artery, vein, sciatic nerve, peroneal nerve). The distal femur containing the tumor is resected en bloc with adequate oncologic margins.
4. Bone Preparation: The remaining proximal femur is prepared for stem insertion (reaming the medullary canal). The tibia is prepared for the tibial tray component.
5. Trial Reduction: Trial components are assembled and inserted to assess fit, stability, limb length, and soft tissue tension.
6. Prosthesis Implantation: The definitive modular prosthesis components are assembled and implanted. The femoral stem is inserted (cemented or uncemented). The distal femoral component is attached, followed by the tibial tray and polyethylene insert. The hinge mechanism is secured.
7. Soft Tissue Reconstruction: Crucial for stability and function. Ligaments, muscles, and joint capsule are repaired or reconstructed around the prosthesis. Patellar tracking is assessed and adjusted.
8. Wound Closure: Meticulous closure of muscle layers, fascia, subcutaneous tissue, and skin. Drains are often placed.
3.5. Post-operative Care & Rehabilitation (Fitting/Usage Instructions)
- Pain Management: Aggressive multimodal pain control is essential.
- Deep Vein Thrombosis (DVT) Prophylaxis: Anticoagulants are typically administered.
- Weight-Bearing: Initial restrictions (e.g., partial weight-bearing) are common, gradually progressing to full weight-bearing based on surgeon protocol and fixation method.
- Physical Therapy:
- Early Mobilization: Gentle range of motion (ROM) exercises begin almost immediately to prevent stiffness.
- Strengthening: Progressive strengthening exercises for quadriceps, hamstrings, and gluteal muscles.
- Gait Training: Learning to walk with crutches or a walker, progressing to independent ambulation.
- Activity Modifications: Patients are guided on appropriate activities to protect the prosthesis and prevent injury.
- Regular Follow-ups: Essential for monitoring wound healing, infection, implant stability, and tumor recurrence. Imaging studies (X-rays) are performed periodically.
4. Risks, Side Effects, or Contraindications
While DFR offers significant benefits, it is a major surgical procedure with potential risks and complications.
4.1. General Surgical Risks
- Bleeding: Intraoperative and postoperative hemorrhage.
- Infection: Superficial or deep surgical site infection, including periprosthetic joint infection (PJI), which can be devastating.
- Nerve and Vascular Injury: Damage to the femoral artery/vein or sciatic/peroneal nerves during resection.
- Deep Vein Thrombosis (DVT) / Pulmonary Embolism (PE): Blood clot formation.
- Anesthetic Risks: Related to general anesthesia.
4.2. Prosthesis-Specific Risks & Side Effects
- Aseptic Loosening: The most common long-term complication, where the implant detaches from the bone without infection, often due to mechanical stress or bone resorption.
- Periprosthetic Fracture: Fracture of the bone around the implant, either during surgery or post-operatively.
- Mechanical Failure: Breakage or wear of implant components (e.g., hinge mechanism failure, polyethylene wear).
- Periprosthetic Joint Infection (PJI): A serious complication requiring prolonged antibiotic therapy and often revision surgery.
- Limb Length Discrepancy: Despite careful planning, minor differences can occur, potentially leading to gait abnormalities or back pain.
- Patellar Complications: Patellar fracture, patellar maltracking, or extensor mechanism disruption.
- Heterotopic Ossification: Formation of new bone in soft tissues around the joint, limiting ROM.
- Tumor Recurrence: Local recurrence at the surgical site or distant metastasis, requiring further oncologic treatment.
4.3. Contraindications
- Extensive Soft Tissue Involvement: When the tumor has invaded surrounding soft tissues or neurovascular structures to an extent that wide oncologic margins cannot be achieved without sacrificing the limb's viability or function.
- Poor General Health: Patients with severe comorbidities that make them unfit for major surgery and prolonged rehabilitation.
- Active Uncontrolled Infection: Pre-existing infection in the surgical area.
- Rapidly Progressing Metastatic Disease: Patients with very limited life expectancy due to widespread metastatic disease where the risks outweigh the benefits of limb salvage.
- Patient Non-Compliance: Unwillingness or inability to adhere to rigorous post-operative rehabilitation protocols.
5. Maintenance & Sterilization Protocols
Maintaining the integrity and sterility of DFR tumor prostheses and associated surgical instruments is paramount to patient safety and surgical success.
5.1. Manufacturing and Quality Control
- Design Validation: Rigorous testing of implant design for fatigue, wear, and mechanical strength.
- Material Certification: Use of medical-grade materials with certified specifications.
- Sterile Packaging: Most prostheses are supplied sterile by the manufacturer, typically sterilized by gamma irradiation or ethylene oxide, and double-pouched to maintain sterility until use.
- Traceability: Each implant has a unique serial number for tracking in case of recalls or adverse events.
5.2. Pre-operative Sterilization (for Reusable Instruments)
- Cleaning: Thorough cleaning of all reusable surgical instruments (e.g., reamers, guides, trial components) to remove bioburden.
- Disinfection: High-level disinfection followed by sterilization.
- Sterilization Methods:
- Steam Sterilization (Autoclave): Most common method for heat-stable instruments. Uses high-pressure saturated steam.
- Ethylene Oxide (EtO) Sterilization: Used for heat-sensitive instruments.
- Hydrogen Peroxide Gas Plasma: Another low-temperature sterilization method.
- Aseptic Technique: Strict adherence to sterile field protocols in the operating room to prevent contamination.
5.3. During Surgery
- Careful Handling: Implants must be handled with sterile gloves and instruments to avoid contamination or damage to polished surfaces or coatings.
- Inspection: Visual inspection of implant components for any defects or damage prior to implantation.
- Storage: Components not immediately used should remain in their sterile packaging until needed.
5.4. Long-term Patient Care and Monitoring
- Regular Follow-ups: Patients require lifelong follow-up with their orthopedic oncologist.
- Radiographic Surveillance: Periodic X-rays are crucial to monitor for signs of aseptic loosening, component wear, periprosthetic fracture, or tumor recurrence.
- Clinical Assessment: Evaluation of pain, function, range of motion, and any signs of infection.
- Revision Considerations: If complications arise (e.g., severe loosening, infection, mechanical failure), revision surgery may be necessary to replace part or all of the prosthesis.
6. Patient Outcome Improvements
DFR with a tumor prosthesis has revolutionized the management of distal femoral tumors, leading to significant improvements in patient outcomes.
- High Limb Salvage Rates: DFR has dramatically reduced the need for amputation, allowing a vast majority of eligible patients to retain their limb.
- Improved Functional Outcomes: Patients typically achieve good to excellent functional scores (e.g., Musculoskeletal Tumor Society - MSTS score, Knee Society Score). They can often ambulate independently, perform activities of daily living, and return to light recreational activities.
- Significant Pain Relief: Removal of the tumor and stabilization of the limb effectively alleviate tumor-related pain.
- Enhanced Quality of Life: Preserving the limb has profound psychological and social benefits, contributing to a better overall quality of life compared to amputation.
- Reduced Morbidity from Amputation: Avoids the complications associated with amputation, such as phantom limb pain, stump issues, and the need for prosthetic fitting.
- Oncologic Control: When combined with adjuvant therapies (chemotherapy, radiation), DFR provides excellent local tumor control, contributing to improved survival rates for certain tumor types.
7. Massive FAQ Section
Here are some frequently asked questions regarding Distal Femoral Replacement (DFR) with a tumor prosthesis:
Q1: What exactly is a Distal Femoral Replacement (DFR)?
A1: A Distal Femoral Replacement is a surgical procedure where a segment of the lower thigh bone (distal femur), usually affected by a tumor, is removed and replaced with a specialized modular metal implant (tumor prosthesis) that also reconstructs the knee joint.
Q2: Why is DFR used instead of amputation for bone tumors?
A2: DFR is a limb-sparing procedure designed to remove the tumor while preserving the patient's limb. It aims to restore function, reduce pain, and maintain a better quality of life, avoiding the significant physical and psychological impact of amputation.
Q3: What materials are used to make the tumor prosthesis?
A3: The prostheses are typically made from high-strength, biocompatible alloys such as Cobalt-Chromium (CoCr) and Titanium (Ti-6Al-4V). The joint surfaces are often made from Ultra-High Molecular Weight Polyethylene (UHMWPE) for low friction and wear resistance.
Q4: How long does the DFR surgery typically take?
A4: The surgery is complex and can take anywhere from 3 to 6 hours, depending on the tumor's extent, the complexity of resection, and the reconstruction required.
Q5: What is the typical recovery time after DFR?
A5: Initial hospital stay is usually 1-2 weeks. Full recovery and return to most activities can take 6-12 months, involving intensive physical therapy. Weight-bearing restrictions are common initially.
Q6: Will I be able to walk normally after a DFR?
A6: Most patients regain the ability to walk independently, often without assistive devices. However, some may have a slight limp or reduced range of motion compared to a natural knee. High-impact activities are usually restricted.
Q7: What are the most common complications associated with DFR?
A7: Common complications include infection (periprosthetic joint infection), aseptic loosening of the implant from the bone, mechanical failure of components, periprosthetic fracture, and tumor recurrence.
Q8: How long does a DFR tumor prosthesis typically last?
A8: The lifespan of a DFR prosthesis varies, but many can last 10-15 years or more. Factors like patient activity level, bone quality, and the type of prosthesis can influence its durability. Revision surgery may be needed if components wear out or loosen over time.
Q9: What activities can I participate in after receiving a DFR?
A9: Patients are generally encouraged to engage in low-impact activities such as walking, swimming, cycling, and golf. High-impact sports, running, and heavy lifting are usually discouraged to protect the prosthesis.
Q10: How important is physical therapy after DFR?
A10: Physical therapy is absolutely crucial. It starts almost immediately after surgery and is vital for regaining range of motion, strength, and proper gait. Adherence to the therapy program significantly impacts long-term functional outcomes.
Q11: Are there alternatives to DFR for distal femoral tumors?
A11: Yes, alternatives include traditional amputation, biological reconstructions (e.g., allograft-prosthesis composites, rotationplasty for pediatric patients), or in rare cases, limb salvage with custom implants or less extensive resections for very small, localized tumors. The choice depends on tumor type, stage, patient age, and functional goals.
Q12: How often will I need follow-up appointments after DFR?
A12: Regular follow-up appointments, often with X-rays, are typically scheduled every few months initially, then annually for many years. This is to monitor for tumor recurrence, implant stability, and any potential complications.