Articular Cartilage Matrix Scaffold (MACI): The Cutting-Edge in Cartilage Repair
Articular cartilage defects, often resulting from trauma, repetitive stress, or degenerative conditions, present a significant challenge in orthopedic medicine. Unlike many other tissues, articular cartilage has a limited capacity for self-repair, often leading to progressive joint pain, dysfunction, and ultimately, osteoarthritis. The Articular Cartilage Matrix Scaffold, commonly known as MACI (Matrix-Applied Chondrocyte Implantation), represents a pioneering advancement in biological cartilage restoration. This comprehensive guide delves into the intricacies of MACI, offering an authoritative overview for medical professionals and patients seeking in-depth understanding.
1. Comprehensive Introduction & Overview
MACI is an innovative, two-stage surgical procedure designed to repair symptomatic, full-thickness articular cartilage defects. It utilizes a patient's own (autologous) chondrocytes, grown and expanded in a laboratory, which are then seeded onto a resorbable porcine collagen membrane. This cellularized scaffold is subsequently implanted into the cartilage defect, promoting the regeneration of hyaline-like cartilage.
Key Principles of MACI:
- Autologous: Uses the patient's own cells, minimizing immune rejection risk.
- Biological: Aims to regenerate tissue that closely mimics native hyaline cartilage.
- Matrix-Supported: The collagen membrane provides a scaffold for chondrocyte adherence, proliferation, and differentiation, facilitating easier surgical handling compared to earlier ACI techniques.
- Targeted Repair: Specifically addresses focal chondral defects rather than widespread arthritic conditions.
Evolution of Cartilage Repair:
MACI builds upon the foundation of Autologous Chondrocyte Implantation (ACI), which emerged in the 1990s. Early ACI involved injecting a suspension of chondrocytes under a periosteal flap. MACI refines this by providing a pre-seeded, ready-to-implant matrix, simplifying the surgical procedure and potentially improving cell retention and distribution within the defect.
2. Deep-Dive into Technical Specifications / Mechanisms
2.1. Design and Materials
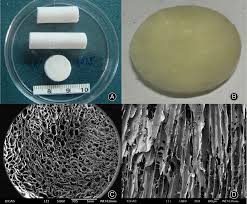
The MACI product consists of two primary components:
- Autologous Chondrocytes: These are healthy cartilage cells harvested from a non-weight-bearing area of the patient's own joint. They are then expanded in a specialized laboratory culture to achieve the necessary cell count for implantation.
- Porcine Collagen Membrane: This is a biocompatible, resorbable, and highly purified collagen type I/III membrane derived from pig tissue. It serves as the scaffold onto which the expanded chondrocytes are uniformly seeded.
Key Material Properties:
- Biocompatibility: The porcine collagen membrane is highly biocompatible, meaning it integrates well with human tissue without eliciting a significant adverse immune response.
- Biodegradability: The membrane gradually degrades over time, typically within weeks to months, as the patient's own cells begin to produce their extracellular matrix, eventually forming new cartilage.
- Porous Structure: The scaffold's porous nature allows for nutrient diffusion and cell migration, crucial for tissue regeneration.
- Mechanical Integrity: While resorbable, the membrane provides sufficient initial mechanical stability to hold the chondrocytes in place within the defect during the critical early healing phase.
2.2. Mechanism of Action
The MACI procedure operates on the principle of biological cartilage regeneration through the following steps:
- Chondrocyte Harvest (Stage 1): A small biopsy (typically 200-300 mg) of healthy articular cartilage is harvested arthroscopically from a non-weight-bearing area of the affected joint.
- Cell Expansion: The harvested cartilage tissue is sent to a specialized laboratory where the chondrocytes are isolated, enzymatically digested, and then cultured and expanded over several weeks (typically 4-6 weeks) to reach a sufficient cell population (millions of cells).
- Matrix Seeding: The expanded autologous chondrocytes are then uniformly seeded onto the porcine collagen membrane. This creates the MACI implant, which is shipped back to the surgeon for implantation.
- Implantation (Stage 2): During the second surgical procedure, the MACI implant is precisely trimmed to match the size and shape of the prepared cartilage defect and secured into place.
- Regeneration: Once implanted, the chondrocytes on the scaffold begin to proliferate and produce a new extracellular matrix, gradually filling the defect with hyaline-like cartilage. The collagen membrane provides a temporary structural framework and a protective environment for these cells.
Cellular Processes:
The implanted chondrocytes maintain their phenotypic characteristics, producing Type II collagen and proteoglycans, which are the hallmarks of healthy articular cartilage. This leads to the formation of a durable, functional repair tissue that closely resembles native hyaline cartilage, contrasting with the fibrocartilage typically formed by simpler repair techniques like microfracture.
3. Extensive Clinical Indications & Usage
MACI is indicated for the repair of symptomatic, full-thickness (International Cartilage Repair Society [ICRS] Grade III or IV) articular cartilage defects of the knee in adults. While primarily used in the knee, its application can extend to other joints in specific, carefully selected cases (e.g., ankle, hip), though these are considered off-label or investigational.
3.1. Patient Selection and Indications
Ideal candidates for MACI typically meet specific criteria:
- Age: Generally between 18 and 55 years old, though individual patient factors are considered.
- Defect Size: Focal, full-thickness cartilage defects typically ranging from 1-10 cm² in area. Smaller defects might be treated with simpler methods, and larger defects might require alternative or adjunctive treatments.
- Defect Location: Commonly involves the femoral condyles, trochlea, or patella.
- Symptomatic: Patients must experience pain, swelling, or mechanical symptoms directly attributable to the cartilage defect.
- No Significant Osteoarthritis: MACI is not a treatment for widespread osteoarthritis. It is designed for isolated, focal defects in otherwise healthy joints.
- Stable Joint: No significant ligamentous instability or malalignment that would compromise the repair. These issues must be addressed concurrently or prior to MACI.
- Compliance: Patients must be willing and able to adhere to a rigorous and extended post-operative rehabilitation protocol.
3.2. Detailed Surgical Application (Fitting/Usage Instructions)
The MACI procedure is a two-stage process:
Stage 1: Cartilage Biopsy
- Anesthesia: General or regional anesthesia.
- Arthroscopy: A minimally invasive arthroscopic procedure is performed to visualize the joint.
- Defect Assessment: The cartilage defect is thoroughly evaluated, confirming its size, depth, and suitability for MACI. Any concomitant pathologies (meniscal tears, ligamentous instability) are noted and may be addressed.
- Biopsy Harvest: A small, cylindrical plug (typically 5-10 mm in diameter, 5-10 mm deep) of healthy articular cartilage is harvested from a low-weight-bearing area, such as the intercondylar notch or superior medial femoral condyle.
- Specimen Handling: The biopsy is immediately placed in a sterile transport medium and shipped to the manufacturing facility for cell processing.
Stage 2: MACI Implantation
- Anesthesia: General or regional anesthesia.
- Surgical Approach: An open arthrotomy or mini-arthrotomy is typically performed to provide direct visualization and access to the cartilage defect. Arthroscopic implantation is technically challenging but evolving.
- Defect Preparation:
- The cartilage defect is thoroughly debrided to remove all unstable or damaged cartilage, leaving a stable, perpendicular shoulder of healthy cartilage surrounding the defect.
- The subchondral bone plate is typically preserved and lightly microfractured or drilled to promote bleeding and release mesenchymal stem cells, which can aid in initial integration.
- The defect bed is meticulously cleaned and dried.
- Implant Preparation:
- The MACI implant (a circular or rectangular membrane with seeded cells) is removed from its sterile packaging.
- Using a sterile template or precise measurements, the implant is carefully trimmed to precisely match the size and shape of the prepared defect. It is crucial to ensure the cell-seeded side faces the subchondral bone.
- Implantation and Fixation:
- The trimmed MACI implant is gently placed into the defect, ensuring full coverage and good apposition to the surrounding healthy cartilage.
- The implant is secured in place using a biocompatible fibrin glue, which helps adhere it to the subchondral bone and surrounding cartilage. Additional sutures (e.g., absorbable PDS sutures) may be used for larger or more challenging defects, particularly on the patella or trochlea, to provide enhanced stability.
- Closure: The joint capsule and overlying tissues are meticulously closed in layers.
3.3. Post-Operative Management and Rehabilitation
A strict and prolonged rehabilitation protocol is critical for successful MACI outcomes.
Typical Phases:
- Phase 1 (Protection & Early Motion, 0-6 weeks):
- Non-weight-bearing or partial weight-bearing (e.g., toe-touch weight-bearing) with crutches.
- Continuous Passive Motion (CPM) machine use for several hours daily to promote cartilage nutrition and prevent adhesions.
- Gentle range of motion exercises (0-90 degrees initially, gradually increasing).
- No impact or twisting movements.
- Phase 2 (Gradual Progression, 6-12 weeks):
- Gradual increase in weight-bearing.
- Progression of range of motion and strengthening exercises.
- Low-impact activities (stationary cycling, swimming).
- Phase 3 (Strength & Endurance, 3-6 months):
- Full weight-bearing as tolerated.
- Advanced strengthening and proprioception exercises.
- Introduction of light sport-specific drills.
- Phase 4 (Return to Activity, 6-12+ months):
- Gradual return to higher-impact activities and sports, under guidance.
- Continued strengthening and maintenance program.
4. Maintenance/Sterilization Protocols
4.1. Product Handling and Sterilization
- MACI Implant: The MACI product itself is a sterile, biological product. It is shipped in a specialized container and must be handled using strict aseptic techniques by the surgical team. It is not designed for re-sterilization or re-use.
- Surgical Instruments: All surgical instruments used during both the biopsy and implantation procedures must adhere to standard hospital sterilization protocols (e.g., steam sterilization/autoclaving) to prevent infection. This includes arthroscopes, cannulas, debridement tools, measuring devices, and fixation instruments.
- Operating Room Environment: The surgical environment must maintain a sterile field throughout the procedure to minimize the risk of contamination.
4.2. Post-Operative Patient Care and Monitoring
- Wound Care: Standard post-operative wound care protocols apply, including keeping the incision site clean and dry.
- Infection Monitoring: Patients are monitored for signs of infection (redness, swelling, warmth, fever).
- Pain Management: Appropriate analgesia is provided to manage post-operative pain.
- Rehabilitation Adherence: Strict adherence to the prescribed rehabilitation protocol is paramount for long-term success. Non-compliance can lead to graft failure or suboptimal outcomes.
- Follow-up Imaging: Regular follow-up appointments with the surgeon are necessary, often including MRI scans at specific intervals (e.g., 6 months, 1 year, 2 years) to assess graft integration, maturation, and overall cartilage regeneration.
5. Biomechanics and Patient Outcome Improvements
5.1. Biomechanics of MACI Repair
The goal of MACI is to restore the biomechanical function of the joint by replacing damaged cartilage with a tissue that structurally and functionally mimics native hyaline cartilage.
- Load Distribution: Healthy articular cartilage provides a low-friction, high-load-bearing surface. A successful MACI repair aims to restore smooth load distribution across the joint, reducing stress concentrations that can lead to further degeneration.
- Friction Reduction: The regenerated hyaline-like cartilage provides a smooth articular surface, reducing friction during joint movement and minimizing wear.
- Shock Absorption: While perhaps not fully replicating the shock-absorbing capacity of native cartilage, the regenerated tissue contributes to cushioning the joint during impact activities.
- Stability: The integration of the MACI graft into the surrounding healthy cartilage and subchondral bone helps to restore the overall structural integrity of the articular surface.
Comparison to Fibrocartilage:
Unlike microfracture, which often results in the formation of mechanically inferior fibrocartilage (rich in Type I collagen), MACI aims for hyaline-like cartilage (rich in Type II collagen). Hyaline-like cartilage is more durable and resilient, offering superior long-term biomechanical performance.
5.2. Patient Outcome Improvements
Clinical studies and long-term follow-up data consistently demonstrate significant improvements in patient outcomes following MACI.
Key Outcome Measures:
- Pain Reduction: Patients typically experience a substantial decrease in joint pain, often allowing them to reduce or eliminate pain medication.
- Functional Improvement: Significant improvements in activities of daily living, range of motion, and ability to participate in recreational and sporting activities.
- Quality of Life: Enhanced overall quality of life due to reduced pain and improved function.
- Graft Durability: Studies show high rates of graft survival and persistence of repair tissue over many years.
- MRI Findings: Post-operative MRI scans often show good to excellent fill of the cartilage defect with tissue resembling native cartilage, with good integration into the surrounding host cartilage and subchondral bone.
Evidence-Based Results:
Numerous clinical trials and registry data have shown MACI to be superior to microfracture for larger defects (typically >2 cm²) and to provide durable results over 5-10 years, with some studies showing positive outcomes extending beyond that. Patients often report satisfaction rates exceeding 80-90%.
6. Risks, Side Effects, or Contraindications
While MACI offers significant benefits, it is a surgical procedure with potential risks and is not suitable for everyone.
6.1. General Surgical Risks
- Infection: Risk associated with any open surgical procedure.
- Bleeding/Hematoma: Accumulation of blood within the joint.
- Nerve or Vascular Damage: Injury to surrounding nerves or blood vessels.
- Deep Vein Thrombosis (DVT) / Pulmonary Embolism (PE): Blood clots, a rare but serious complication.
- Anesthesia Risks: Adverse reactions to anesthetic agents.
6.2. MACI-Specific Risks and Side Effects
- Delamination/Failure of Integration: The implanted membrane may detach or fail to integrate properly with the surrounding cartilage or subchondral bone.
- Hypertrophy: Overgrowth of the repair tissue, which can cause mechanical symptoms (e.g., catching, locking). This is less common with MACI than with earlier ACI techniques but can still occur.
- Persistent Pain/Symptoms: Despite successful graft integration, some patients may continue to experience pain or other symptoms.
- Donor Site Morbidity: Minor pain or discomfort at the site where the cartilage biopsy was taken, though this is typically minimal.
- Allergic Reaction: While rare due to the highly purified nature of the porcine collagen, an allergic reaction to the membrane is theoretically possible.
- Stiffness/Arthrofibrosis: Scar tissue formation within the joint can restrict range of motion.
- Re-operation: Need for subsequent surgery due to complications or failure of the initial procedure.
6.3. Contraindications
- Widespread Osteoarthritis: MACI is not for diffuse arthritic conditions.
- Inflammatory Arthritis: Conditions like rheumatoid arthritis are contraindications.
- Uncorrected Joint Instability or Malalignment: These must be addressed prior to MACI.
- Unresolved Meniscal Pathology: Significant meniscal tears that are not addressed can compromise the MACI repair.
- Active Infection: Any active joint infection.
- Severe Obesity: Can place excessive stress on the graft, leading to failure.
- Unrealistic Patient Expectations: Patients must understand the recovery timeline and potential limitations.
- Pregnancy: Due to the nature of the procedure and post-operative medications.
7. Massive FAQ Section
Q1: What exactly is MACI?
A1: MACI stands for Matrix-Applied Chondrocyte Implantation. It's a two-stage surgical procedure that uses your own cartilage cells (autologous chondrocytes) grown in a lab and then seeded onto a special collagen membrane. This cellularized membrane is then implanted into a cartilage defect in your joint to regenerate new, hyaline-like cartilage.
Q2: How is MACI different from microfracture?
A2: Microfracture involves creating small holes in the bone beneath the cartilage defect to stimulate bleeding and allow bone marrow stem cells to form repair tissue. However, this often results in fibrocartilage, which is less durable and mechanically inferior to native hyaline cartilage. MACI uses your own cultured chondrocytes to grow hyaline-like cartilage, which is structurally and functionally superior.
Q3: Who is a good candidate for MACI?
A3: Ideal candidates are typically adults between 18 and 55 years old with symptomatic, full-thickness articular cartilage defects, usually in the knee, ranging from 1-10 cm² in size. Patients should not have widespread osteoarthritis and must be committed to a rigorous rehabilitation program.
Q4: What is the recovery process like after MACI surgery?
A4: Recovery is a lengthy process, typically lasting 6 to 12 months or more. It involves a strict, phased rehabilitation program starting with non-weight-bearing and continuous passive motion, gradually progressing to full weight-bearing, strengthening, and eventually, return to activity. Adherence to physical therapy is crucial for success.
Q5: How long does the MACI repair last?
A5: Clinical studies have shown durable results for MACI, with many patients experiencing successful outcomes extending beyond 5-10 years. The goal is to create a long-lasting, functional repair tissue that can withstand the demands of daily activity.
Q6: Is MACI covered by insurance?
A6: Coverage for MACI can vary depending on your insurance provider and specific policy. Many insurance companies do cover MACI, especially as it has gained FDA approval and demonstrated clinical effectiveness. It is essential to check with your insurance provider and your surgeon's office for pre-authorization.
Q7: What are the main risks associated with MACI?
A7: General surgical risks include infection, bleeding, and nerve damage. MACI-specific risks include failure of the implant to integrate, delamination (detachment), hypertrophy (overgrowth of tissue), persistent pain, or joint stiffness. Your surgeon will discuss these risks in detail.
Q8: Can MACI treat arthritis?
A8: No, MACI is not a treatment for widespread osteoarthritis. It is specifically designed to repair isolated, focal cartilage defects in otherwise healthy joints. If you have extensive joint degeneration or inflammatory arthritis, MACI is generally not indicated.
Q9: What are the alternatives to MACI for cartilage repair?
A9: Alternatives include microfracture, osteochondral autograft transplantation (OATS), osteochondral allograft transplantation, and other emerging biological repair techniques. The best option depends on the size, location, and nature of the defect, as well as patient factors.
Q10: How successful is MACI typically?
A10: Clinical studies report high success rates for MACI, often exceeding 80-90% in terms of pain reduction, functional improvement, and patient satisfaction over several years. Success is measured by functional outcome scores, reduced pain, and imaging evidence of cartilage regeneration.
Q11: Is the MACI procedure painful?
A11: As with any surgery, there will be post-operative pain, which is managed with medication. The recovery and rehabilitation process can also be challenging and may involve discomfort as you regain strength and motion.
Q12: What role does physical therapy play in MACI recovery?
A12: Physical therapy is absolutely critical. It guides you through the phased rehabilitation protocol, ensuring proper healing, restoring range of motion, strengthening surrounding muscles, and gradually preparing the joint for normal activities. Without dedicated physical therapy, the success of the MACI procedure is significantly compromised.