Comprehensive Introduction & Overview
The Anterior Lumbar Interbody Fusion (ALIF) procedure represents a cornerstone in modern spinal surgery for addressing a myriad of lumbar spine pathologies. At the heart of a successful ALIF procedure lies the interbody cage – a sophisticated orthopedic implant designed to restore disc height, stabilize the spinal segment, facilitate bone fusion, and indirectly decompress neural elements. This comprehensive guide delves into the intricate world of ALIF interbody cages, providing an exhaustive overview for medical professionals, researchers, and patients seeking a deeper understanding of this critical technology.
An ALIF procedure involves accessing the lumbar spine from the front (anteriorly), typically through an incision in the abdomen, to remove the damaged intervertebral disc. Once the disc is removed, an ALIF interbody cage, packed with bone graft material, is inserted into the disc space. This approach offers several distinct advantages, including direct access to the anterior column, enabling placement of a large interbody graft for robust fusion, and optimizing restoration of lumbar lordosis – the natural inward curve of the lower back. The choice of ALIF cage is paramount, influencing biomechanical stability, fusion rates, and ultimately, patient outcomes. This guide will explore the design, materials, clinical applications, biomechanics, and maintenance considerations that define the efficacy of these advanced implants.
Deep-dive into Technical Specifications / Mechanisms
Design & Materials of ALIF Interbody Cages
The evolution of ALIF interbody cages has been driven by a relentless pursuit of improved biomechanical performance, biocompatibility, and osteointegration. Modern cages incorporate advanced materials and intricate designs to meet these demands.
Materials:
The primary materials utilized in ALIF interbody cages each offer unique characteristics:
- PEEK (Polyetheretherketone):
- Pros: Radiotranslucent (allowing clear visualization of bone graft fusion on X-rays), modulus of elasticity similar to bone (reducing stress shielding), biocompatible, chemically inert.
- Cons: Bioinert (does not promote bone ingrowth directly), lacks inherent osteoconductivity.
- Titanium (Ti):
- Pros: High strength-to-weight ratio, excellent biocompatibility, osteoconductive (promotes bone ingrowth on its surface), radiopaque (visible on X-rays).
- Cons: Stiffer than bone (potential for stress shielding), can create imaging artifacts on MRI.
- Titanium-Coated PEEK:
- Pros: Combines the bone-like modulus of PEEK with the osteoconductive surface properties of titanium, offering a favorable balance for fusion and reduced stress shielding.
- Cons: Manufacturing complexity, potential for coating delamination (though rare with modern designs).
- Trabecular Metal / Porous Titanium (e.g., 3D printed Titanium):
- Pros: Mimics the porous structure of cancellous bone, promoting rapid bone ingrowth and biological fixation. Low modulus of elasticity, excellent osteoconductivity.
- Cons: Higher manufacturing cost, can be radiographically dense.
Design Features:
ALIF cages are engineered with specific features to optimize their function:
- Anatomical Shaping: Often designed with a "kidney bean" shape to conform to the anterior vertebral body anatomy, and lordotic angles (typically 6-15 degrees) to restore the natural lumbar curvature.
- Fenestrations/Windows: Openings within the cage allow for packing with bone graft material (autograft, allograft, or synthetic bone graft substitutes) to facilitate fusion across the disc space.
- Serrations/Teeth/Spikes: Textured surfaces on the superior and inferior aspects of the cage enhance initial stability and resist expulsion or migration after insertion.
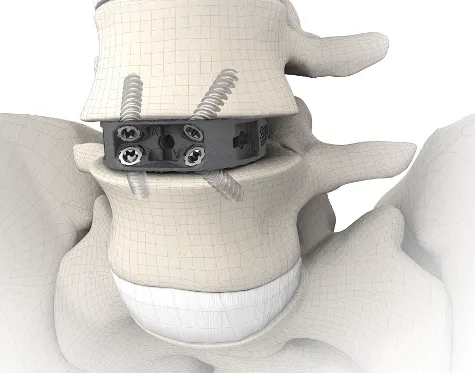
- Integrated Fixation: Some cages incorporate screws or blades that deploy into the vertebral bodies, providing immediate standalone fixation and eliminating the need for supplementary posterior instrumentation in selected cases.
- Porous Structures: Advanced designs, particularly in 3D-printed titanium cages, feature interconnected pores that encourage bone ingrowth and enhance biological fixation.
- Varying Dimensions: Available in a wide range of heights, widths, and lengths to accommodate diverse patient anatomies and disc space dimensions.
Biomechanics of ALIF Cages
The biomechanical role of ALIF cages is critical for achieving successful fusion and restoring spinal function.
- Load Sharing: ALIF cages provide robust anterior column support, bearing a significant portion of the axial compressive loads. This load sharing prevents excessive stress on posterior elements and promotes fusion.
- Stress Shielding: The phenomenon where a stiffer implant bears too much load, leading to reduced stress on the bone and potential bone resorption. PEEK and porous titanium cages, with moduli closer to bone, aim to minimize stress shielding compared to solid titanium.
- Sagittal Balance Restoration: The lordotic angle of the cage is crucial for restoring the natural lumbar lordosis, which is vital for maintaining overall spinal balance, reducing compensatory mechanisms, and improving posture.
- Cage-Endplate Interface: A well-prepared endplate and an optimally designed cage interface are paramount. The cage must engage the strong cortical bone of the endplate to prevent subsidence, where the cage sinks into the softer cancellous bone of the vertebral body. Serrations and porous surfaces enhance this interface.
- Stability: The cage provides immediate mechanical stability to the operated segment, preventing micro-motion that could hinder bone fusion. When combined with integrated or supplemental fixation, this stability is further enhanced.
Extensive Clinical Indications & Usage
Primary Indications for ALIF Interbody Cage Placement
ALIF with interbody cages is indicated for a range of degenerative and deformative conditions of the lumbar spine:
- Degenerative Disc Disease (DDD): Chronic low back pain unresponsive to conservative management, often associated with disc space collapse, instability, and osteophyte formation.
- Spondylolisthesis (Degenerative or Low-Grade Isthmic): Forward slippage of one vertebra over another. ALIF can reduce the slippage, stabilize the segment, and indirectly decompress neural structures.
- Spinal Stenosis with Instability: Narrowing of the spinal canal causing nerve compression, often exacerbated by segmental instability. ALIF can restore disc height and indirectly enlarge the neural foramen.
- Post-Discectomy Syndrome / Failed Back Surgery Syndrome (FBSS): Persistent or recurrent pain after previous lumbar discectomy, often due to recurrent disc herniation, instability, or scar tissue.
- Pseudarthrosis: Failure of a previous spinal fusion to achieve solid bony union.
- Spinal Deformity: Such as adult degenerative scoliosis, where restoration of lumbar lordosis is essential for sagittal balance correction.
Detailed Surgical Applications & Fitting/Usage Instructions
The successful implantation of an ALIF interbody cage requires meticulous pre-operative planning, precise surgical technique, and careful intraoperative assessment.
Pre-operative Planning:
- Patient Selection: Thorough clinical evaluation, including assessment of pain, neurological deficits, and functional limitations.
- Imaging: Comprehensive imaging studies (X-rays, MRI, CT scan) are essential to confirm diagnosis, assess disc height, endplate integrity, degree of instability, and vascular anatomy.
- Cage Sizing & Lordosis Determination: Pre-operative templating using imaging helps determine the appropriate cage height, width, length, and lordotic angle needed to restore anatomical alignment and stability.
- Vascular Assessment: Pre-operative CT angiography may be performed, especially for multi-level ALIF or in cases of significant vascular anomalies or atherosclerosis, to map the great vessels and plan safe access.
Surgical Approach (Anterior Retroperitoneal):
- Patient Positioning: Supine position on a radiolucent table, often with slight Trendelenburg to displace abdominal contents superiorly.
- Incision: Typically a left paramedian or transverse incision in the lower abdomen.
- Muscle Dissection: Careful dissection through abdominal wall muscles.
- Vascular Mobilization: The retroperitoneal space is accessed. The great vessels (aorta, vena cava, iliac arteries, and veins) are identified and carefully mobilized by a vascular surgeon or a spine surgeon with appropriate training, to expose the anterior aspect of the lumbar spine.
- Discectomy and Endplate Preparation: The anterior annulus is incised, and the entire disc material is meticulously removed. Crucially, the cartilaginous endplates are removed down to bleeding subchondral bone to expose a healthy, vascularized bed for fusion, while preserving the strong cortical rim of the endplate to prevent cage subsidence.
Cage Insertion:
- Trial Sizing: Trial implants are used to confirm the appropriate cage height and lordotic angle, ensuring adequate distraction and firm seating.
- Bone Graft Packing: The chosen bone graft material (autograft, allograft, or synthetic) is firmly packed into the cage fenestrations and often around the cage within the disc space.
- Impaction/Insertion: The ALIF cage is carefully inserted into the prepared disc space using specialized instruments (impactors, holders). It should be seated securely within the anterior third of the vertebral bodies, ensuring optimal biomechanical load sharing and preventing posterior neural impingement.
- Adjunctive Fixation: Depending on the cage design and the degree of instability, supplementary fixation (e.g., anterior plate and screws, or posterior pedicle screws and rods) may be applied to enhance stability and fusion rates, particularly for multi-level fusions or high-grade spondylolisthesis.
Intraoperative Checks:
- Fluoroscopy: Real-time X-ray imaging is used throughout the procedure to confirm accurate cage placement, proper spinal alignment, and satisfactory restoration of disc height and lordosis.
- Vascular Protection: Continuous vigilance to protect the mobilized great vessels.
Risks, Side Effects, or Contraindications
While ALIF surgery with interbody cages offers significant benefits, it is a major surgical procedure with inherent risks.
General Surgical Risks:
- Infection: Superficial or deep wound infection.
- Bleeding: Intraoperative or postoperative hemorrhage.
- Anesthesia Risks: Adverse reactions to anesthetic agents.
- Nerve Damage: Although less common with an anterior approach, damage to peripheral nerves or sympathetic plexus is possible.
- Deep Vein Thrombosis (DVT) / Pulmonary Embolism (PE): Blood clot formation.
ALIF-Specific Risks:
- Vascular Injury: The most significant risk due to the proximity of the great vessels (aorta, vena cava, iliac arteries and veins). Injury can lead to life-threatening hemorrhage.
- Retrograde Ejaculation (in males): Damage to the superior hypogastric plexus during vascular dissection can impair sympathetic innervation to the ejaculatory ducts, leading to semen entering the bladder instead of exiting the penis. This complication is often transient but can be permanent.
- Ureteral Injury: Rare, but possible injury to the ureter during exposure.
- Cage Subsidence: The cage sinking into the vertebral body, often due to inadequate endplate preparation, osteoporosis, or inappropriate cage size. Can lead to loss of correction, non-union, or pain.
- Non-union/Pseudarthrosis: Failure of the bone graft to fuse, resulting in persistent instability and pain.
- Cage Migration/Dislodgement: The cage shifting from its intended position, potentially requiring revision surgery.
- Adjacent Segment Disease (ASD): Increased stress and degeneration at the spinal levels immediately above or below the fused segment.
- Ileus: Temporary paralysis of the bowel, leading to nausea, vomiting, and abdominal distension, usually resolving spontaneously.
- Hernia: Incisional hernia at the surgical site.
Contraindications:
- Active Infection: Systemic or local infection, which could lead to implant contamination and severe complications.
- Severe Osteopenia/Osteoporosis: Significantly increases the risk of cage subsidence and non-union.
- Significant Vascular Calcification or Anomalies: Making the anterior approach excessively risky.
- Morbid Obesity: Can make surgical exposure extremely challenging and increase complication rates.
- Allergy to Implant Materials: Although rare, material allergies must be considered.
- Uncontrolled Systemic Diseases: Conditions like uncontrolled diabetes, severe cardiovascular disease, or coagulopathies that increase surgical risk.
Maintenance/Sterilization Protocols
ALIF interbody cages are supplied as single-use, sterile implants. Therefore, maintenance and sterilization protocols specifically apply to the associated surgical instruments used for their insertion and handling, rather than the cage itself.
Handling & Storage of Sterile ALIF Cages:
- Aseptic Technique: Cages must be handled using strict aseptic technique during opening and transfer to the sterile field to prevent contamination.
- Packaging Integrity: Before use, the sterile packaging must be inspected for any signs of damage, tears, or compromise. Damaged packaging renders the implant non-sterile and unusable.
- Storage: Cages should be stored in a clean, dry, controlled environment, away from extreme temperatures or humidity, and protected from physical damage.
- Expiration Dates: Always check the expiration date on the packaging. Expired implants must not be used.
Sterilization of Associated Surgical Instruments:
Reusable surgical instruments (e.g., distractors, trials, impactors, retractors) used during ALIF procedures must undergo rigorous cleaning and sterilization protocols as per hospital guidelines and regulatory standards.
- Point-of-Use Pre-cleaning: Immediately after use, instruments should be wiped clean of gross debris and kept moist to prevent drying of blood and tissue.
- Cleaning:
- Manual Cleaning: Thorough scrubbing with enzymatic detergents and brushes.
- Automated Cleaning: Use of ultrasonic cleaners or washer-disinfectors to remove microscopic debris.
- Inspection: After cleaning, instruments are meticulously inspected for cleanliness, damage, corrosion, or wear. Damaged instruments should be repaired or removed from service.
- Packaging: Instruments are carefully arranged in sterilization trays, wrapped in sterilization wraps, or placed in rigid sterilization containers, ensuring proper steam penetration and maintenance of sterility.
- Sterilization Methods:
- Steam Sterilization (Autoclave): The most common and preferred method for heat- and moisture-stable instruments. Parameters (temperature, pressure, exposure time) must be strictly adhered to.
- Low-Temperature Sterilization: For heat-sensitive instruments, methods like Ethylene Oxide (EtO) gas, Hydrogen Peroxide Plasma, or Vaporized Hydrogen Peroxide (VHP) may be used.
- Storage: Sterilized instruments must be stored in a designated, clean, dry, and secure area to maintain their sterile integrity until needed for surgery.
Patient Outcome Improvements
The primary goal of ALIF surgery with interbody cages is to significantly improve patient outcomes by alleviating pain, restoring function, and enhancing quality of life.
- Pain Reduction: ALIF effectively addresses axial back pain by stabilizing the affected segment and eliminating motion at the painful disc. Radicular pain (leg pain) is often relieved through indirect decompression of neural elements by restoring disc height and widening the neuroforamina.
- Functional Improvement: Patients typically experience improved mobility, increased ability to perform daily activities, and a greater capacity for exercise and work.
- Neurological Recovery: For patients with neurological deficits secondary to nerve compression, ALIF can lead to significant recovery of motor strength and sensory function.
- Spinal Alignment & Sagittal Balance: The ability of ALIF cages to restore physiological lumbar lordosis is crucial. Correcting sagittal balance reduces compensatory mechanisms (e.g., knee flexion, pelvic tilt), leading to improved posture, reduced muscle fatigue, and a more energy-efficient gait.
- High Fusion Rates: With appropriate surgical technique, proper endplate preparation, and adequate bone graft, ALIF procedures boast high fusion rates, providing long-term stability and pain relief.
- Quality of Life Enhancement: Beyond physical improvements, patients often report a significant enhancement in their overall quality of life, including better sleep, reduced reliance on pain medication, and improved psychological well-being.
A Massive FAQ Section
1. What is an ALIF interbody cage?
An ALIF interbody cage is a specialized orthopedic implant used in Anterior Lumbar Interbody Fusion (ALIF) surgery. It's designed to be inserted into the disc space after a damaged intervertebral disc is removed, providing structural support, restoring disc height, and creating a chamber for bone graft to fuse the vertebrae together.
2. How is an ALIF surgery performed?
ALIF surgery involves an incision in the abdomen to access the front of the lumbar spine. A vascular surgeon or trained spine surgeon carefully mobilizes the blood vessels to expose the disc. The damaged disc is removed, and the endplates are prepared. An ALIF cage, packed with bone graft, is then inserted into the disc space. In many cases, additional screws or plates are used for enhanced stability.
3. What are the benefits of ALIF surgery?
Benefits include direct access to the disc for thorough removal and placement of a large fusion device, excellent restoration of disc height and lumbar lordosis (the natural curve of the lower back), indirect decompression of nerves, and high fusion rates. It avoids dissection through posterior back muscles, potentially leading to less muscle damage and pain.
4. What materials are ALIF cages made from?
ALIF cages are commonly made from PEEK (Polyetheretherketone), Titanium, or a combination like Titanium-coated PEEK. Newer designs also utilize porous Titanium or 3D-printed Titanium to promote bone ingrowth. Each material offers unique advantages in terms of strength, flexibility, radiovisibility, and ability to integrate with bone.
5. Is ALIF surgery safe? What are the risks?
ALIF surgery is generally safe when performed by experienced surgeons. However, like any major surgery, it carries risks. Specific risks include injury to major blood vessels (aorta, vena cava, iliacs), retrograde ejaculation in males (temporary or permanent), nerve damage, infection, bleeding, cage subsidence (sinking), non-union (failure to fuse), and cage migration.
6. How long does recovery take after ALIF surgery?
Recovery varies, but most patients can expect to be hospitalized for 1-3 days. Initial recovery involves managing pain and gradually increasing activity. Light activities can usually be resumed within a few weeks. Full fusion can take 6-12 months, and return to strenuous activities typically occurs after 3-6 months, depending on the individual and surgeon's recommendations.
7. Will I need additional surgery after an ALIF?
In some cases, especially for multi-level fusions or certain types of instability (e.g., higher-grade spondylolisthesis), surgeons may recommend supplementary posterior fixation (e.g., pedicle screws and rods) to enhance stability and improve fusion rates. This can be done during the same surgery or as a separate stage.
8. Can I have an MRI with an ALIF cage?
Yes, most modern ALIF cages made from PEEK or biocompatible titanium alloys are MRI-compatible. However, it's crucial to inform your healthcare provider about your implant before any MRI scan. Some metallic implants can cause image artifacts, but they generally do not pose a safety risk in a standard MRI scanner.
9. How do I know if an ALIF is right for me?
An ALIF procedure is typically considered for patients with chronic low back pain, leg pain, or neurological symptoms caused by degenerative disc disease, spondylolisthesis, or spinal stenosis that has not responded to conservative treatments. A thorough evaluation by a spine specialist, including physical examination and imaging studies, is necessary to determine if ALIF is the most appropriate treatment option for your specific condition.
10. What is the success rate of ALIF surgery?
ALIF surgery generally has high success rates, with studies reporting significant pain relief and functional improvement in 80-90% of appropriately selected patients. Fusion rates are also very high, often exceeding 90-95% when proper surgical technique and bone graft materials are used.
11. What is cage subsidence?
Cage subsidence occurs when the interbody cage sinks into the vertebral body bone after implantation. This can happen due to inadequate endplate preparation, a cage that is too small, or poor bone quality (e.g., osteoporosis). Subsidence can lead to loss of disc height correction, nerve compression, non-union, and persistent pain.
12. How does an ALIF cage help restore lordosis?
ALIF cages are often designed with a specific lordotic angle (e.g., 6, 10, or 15 degrees) on their superior and inferior surfaces. When inserted into the disc space, this angled design helps to wedge the anterior portion of the vertebral bodies apart more than the posterior, thereby restoring or enhancing the natural inward curve (lordosis) of the lumbar spine. This is crucial for maintaining overall spinal balance and biomechanics.