Biomaterials in Orthopaedic Fracture Fixation: A Comprehensive Academic Guide
Key Takeaway
Biomaterials in fracture fixation must withstand complex biomechanical forces while maintaining biocompatibility. This guide explores the metallurgical properties of stainless steel, titanium alloys, and porous tantalum, alongside the degradation profiles of bioabsorbable polymers like PGA and PLLA. Understanding material modulus, corrosion resistance, and stress relaxation is critical for orthopaedic surgeons to optimize implant selection, minimize complications, and promote physiological bone healing.
Introduction to Orthopaedic Biomaterials
The evolution of fracture fixation is inextricably linked to the advancement of biomaterials. The primary objective of internal fixation is to provide mechanical stability that facilitates biological bone healing while minimizing iatrogenic soft tissue trauma. To achieve this, orthopaedic implants must possess specific biomechanical properties—such as adequate tensile strength, fatigue resistance, and an appropriate modulus of elasticity—while maintaining strict biocompatibility within the corrosive saline environment of the human body.
Historically, the widespread application of internal fixation was catalyzed by the seminal 1937 report by Venable, Stuck, and Beach. They demonstrated that certain metals, when bathed in the electrolytic environment of interstitial fluids, created an electrical potential leading to galvanic corrosion, local tissue necrosis, and subsequent implant loosening. This discovery mandated the development of metals with the lowest possible electrolytic coefficients, paving the way for modern alloys and, eventually, bioabsorbable polymers.
Biomechanical Principles of Implant Loading
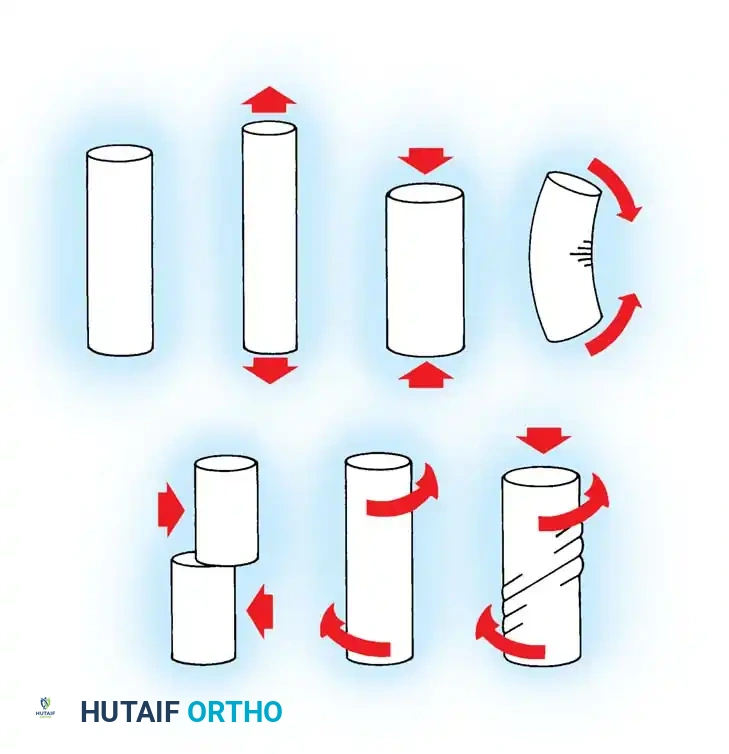
Before selecting a biomaterial, the orthopaedic surgeon must understand the physiological forces the implant will endure. Fracture fixation devices are subjected to repetitive, multi-planar physiological loads.

Unloaded, Tension, Compression, Bending, Shear, Torsion, and Combined loading torsion-compression.
As illustrated in the biomechanical loading diagram above, implants must be engineered to resist several distinct force vectors:
* Tension: Forces pulling the material apart, critical in tension-band constructs.
* Compression: Forces pushing the material together, essential for axial loading in diaphyseal fractures.
* Bending: A combination of tension on the convex side and compression on the concave side, heavily testing the flexural rigidity of plates and intramedullary nails.
* Shear: Unaligned forces pushing parts of the implant in opposite directions, a common cause of screw breakage at the fracture interface.
* Torsion: Twisting forces, particularly relevant in spiral fractures and resisted by interlocking intramedullary devices.
* Combined Loading: The physiological reality where torsion, compression, and bending occur simultaneously during weight-bearing.
Metallic Biomaterials
Metals remain the mainstay of fracture fixation due to their superior ultimate tensile strength, ductility, and fatigue resistance. Currently, the vast majority of orthopaedic implants are manufactured from three primary categories of metals.
316L Stainless Steel
Composed primarily of iron, chromium (17-19%), nickel (13-15%), and molybdenum (2-3%), 316L stainless steel is the traditional workhorse of orthopaedic trauma. The "L" denotes a low carbon content (less than 0.03%), which prevents the precipitation of chromium carbides at grain boundaries, thereby reducing susceptibility to intergranular corrosion.
* Biomechanics: It possesses a high modulus of elasticity (approximately 200 GPa), which is significantly stiffer than cortical bone (15-20 GPa). While this provides rigid fixation, it can lead to stress shielding and subsequent osteopenia if left in situ long-term.
* Corrosion Resistance: The chromium content allows the formation of a thin, self-healing chromium oxide passivation layer that protects the underlying iron from the corrosive physiological environment.
Titanium and Titanium Alloys
Commercially pure titanium (CP Ti, composed of titanium and oxygen) and titanium alloys (most commonly Ti-6Al-4V: titanium, aluminum, and vanadium) have largely superseded stainless steel in many applications.
* Biomechanics: Titanium alloys have a modulus of elasticity closer to that of bone (approximately 110 GPa), which reduces stress shielding and promotes secondary bone healing via callus formation.
* Biocompatibility: Titanium forms a highly stable titanium dioxide (TiO2) passivation layer, offering superior corrosion resistance and excellent osseointegration properties.
* Limitations: Titanium is highly notch-sensitive. A microscopic scratch on a titanium plate can act as a stress riser, significantly reducing its fatigue life and leading to catastrophic failure.
Surgical Warning: Care must be taken not to scratch titanium implants during insertion. Furthermore, titanium and stainless steel implants should never be mixed in the same construct (e.g., a stainless steel screw in a titanium plate), as this creates a galvanic couple, leading to rapid electrolytic corrosion and implant failure.
Tantalum (Trabecular Metal)
A newer addition to the orthopaedic armamentarium, tantalum is utilized as a highly porous trabecular metal. It is manufactured via chemical vapor deposition of elemental tantalum onto a reticulated vitreous carbon substrate.
* Biological Scaffold: The resulting structure is 75% to 80% porous, mimicking the microarchitecture of cancellous bone. This provides an exceptional biological scaffold for osteoconduction and new bone ingrowth.
* Biomechanics: Highly porous tantalum has a modulus of elasticity (approx. 3 GPa) that closely matches cancellous bone, minimizing stress shielding.
* Indications: While historically used for bone markers, its remarkable resistance to corrosion and excellent ingrowth profile make it ideal for reconstructive procedures, avascular necrosis core decompression grafting, and complex revision arthroplasties.
Complications of Metallic Implants
All metals and alloys corrode to some degree in saline environments. This corrosion increases significantly with fretting wear—micromotion between metal components, such as between a screw head and a plate hole.
Concerns regarding metal sensitization, particularly to nickel and chromium, are well-documented. While Type IV delayed hypersensitivity reactions can occur, the actual incidence of metal sensitivity complications that negatively alter fracture healing remains exceedingly low.
Bioabsorbable Materials
The drive to eliminate the need for secondary hardware removal surgeries and to provide a gradual transfer of mechanical load to healing bone led to the development of bioabsorbable polymers.
Evolution and Chemistry
Polyglycolic acid (PGA) was the first totally synthetic bioabsorbable suture, introduced in 1970 as Dexon. This was followed by Vicryl (a copolymer of 92% PGA and 8% polylactic acid [PLA]) in 1975, and polydioxanone (PDS) in 1981. PDS holds the distinction of being the first bioabsorbable material manufactured into orthopaedic screws.
Currently, the primary alpha-polyesters used for bioabsorbable implants include:
* PGA (Polyglycolic Acid): Degrades rapidly.
* PDS (Polydioxanone): Intermediate degradation.
* PLLA (Polylevolactic Acid): Highly crystalline, degrades very slowly.
* PDLLA (Racemic Poly-D,L-lactic Acid): Amorphous, degrades faster than PLLA.
Degradation Pathways
These polymers degrade primarily via hydrolysis—the cleavage of polymer chains by water molecules.
* PGA is hydrolyzed to pyruvic acid, which enters the tricarboxylic acid (Krebs) cycle and is ultimately excreted via respiration as carbon dioxide and water.
* PDLLA follows a similar metabolic pathway to CO2 and H2O.
* PDS is also hydrolyzed but is primarily excreted via the renal system in the urine.
Biomechanical Properties and Manufacturing
Bioabsorbable implants possess a high glass transition temperature (Tg)—the critical temperature at which the amorphous regions of the polymer transition from a hard, glassy state to a soft, rubbery state. Because their Tg is well above body temperature, these implants cannot be contoured intraoperatively without specialized heating equipment, which risks altering their structural integrity.
To enhance their mechanical profile, implants undergo self-reinforcement (SR). This manufacturing process aligns the polymer fibers along the longitudinal axis of the implant, significantly increasing tensile and flexural strength.
However, these polymers are highly viscoelastic and subject to:
* Creep: Progressive deformation under a constant load.
* Stress Relaxation: A decrease in stress over time under a constant strain.
Clinical Pearl: Claes demonstrated that self-reinforced PLA (SR-PLA) and PDLLA-PLLA screws lose 20% of their compressive force within just 20 minutes of insertion. In a physiological saline environment, this loss is even more rapid. Surgeons must account for this rapid loss of compression when utilizing bioabsorbables for fracture fixation.
Strength degradation follows specific timelines based on the polymer. SR-PGA rods retain only 50% of their strength at 2 weeks and 13% at 4 weeks. PLLA exhibits the slowest degradation and longest retention of mechanical strength.
Factors Affecting Biomechanical Properties
The performance of bioabsorbable polymers is multifactorial. Based on the foundational work by Hovis et al., the following variables dictate implant behavior:
- Chemical Composition: Molecular weight, viscosity, molar ratio of copolymers, sequence of chains, and crystallinity.
- Manufacturing Processes: Machining, extrusion, melt molding, compression molding, injection molding, fiber reinforcement, and sterilization methods (gamma irradiation can prematurely degrade polymers; ethylene oxide is preferred).
- Physical Dimensions: Implant diameter and mechanical design (e.g., thread pitch).
- Environmental Factors: Local tissue temperature, pH, regional blood flow, rate of removal of degraded polymer, oxidation, and enzymatic action.
- Time: Viscoelasticity and the inherent rate of degradation.
Clinical Indications for Bioabsorbable Fixation
Because of their rapid loss of mechanical strength, bioabsorbable bone implants must be strictly limited to applications where minimal physiological load is applied until clinical healing is evident. They are ideal for periarticular fractures that are protected by postoperative immobilization.
Primary Indications Include:
* Metatarsal osteotomies (e.g., Hallux Valgus correction)
* Metacarpal and metatarsal fusions
* Malleolar fractures (medial malleolus)
* Osteochondritis dissecans (OCD) lesion fixation
* Fractures of the radial head and olecranon
* Pediatric epiphyseal (physeal) fractures
* Ruptures of the ulnar collateral ligament (UCL) of the thumb
* Arthroscopic fixation of meniscal lesions
* Femoral canal occlusion for cement restriction in arthroplasty
Advantages:
1. Gradual Load Transfer: As the implant degrades, mechanical load is progressively transferred to the healing bone, preventing stress shielding and promoting Wolff's Law of bone remodeling.
2. No Hardware Removal: Eliminates the morbidity and cost of secondary extraction surgeries.
3. Radiolucency: Does not obscure postoperative radiographic evaluation and produces no artifact on MRI or CT scans.
Surgical Handling and Implantation Techniques
The surgical technique for bioabsorbable implants differs significantly from metallic fixation. Orthopaedic surgeons must adhere to strict protocols to prevent intraoperative implant failure.
- Drilling and Tapping: Bioabsorbable screws have low torsional strength. The drill hole must be perfectly aligned, and the near cortex (and often the far cortex) must be meticulously tapped. Forcing a bioabsorbable screw into an untapped hole will result in the screw head shearing off.
- Countersinking: The cortex should be countersunk to prevent the prominent head of the polymer screw from causing local soft tissue irritation before it degrades.
- Insertion Torque: Screws must be inserted with smooth, continuous motion. Avoid excessive final tightening, as the polymer cannot withstand the high torque limits of stainless steel or titanium.
- Postoperative Protocol: Because of stress relaxation and rapid strength degradation, the construct must be protected with rigid external immobilization (cast or strict non-weight-bearing orthosis) until radiographic callus is visible.
Complications of Bioabsorbable Implants
While advantageous, bioabsorbable materials carry unique complication profiles related to their degradation byproducts.
- Aseptic Inflammation and Sinus Tracts: PGA has been heavily implicated in sterile, aseptic inflammatory reactions and sinus tract formation. This occurs when the rapid degradation of PGA overwhelms the local tissue's ability to clear the acidic byproducts (pyruvic acid), leading to a localized drop in pH and a non-specific foreign body reaction.
- Osteolysis: Foreign body reactions to rapidly degrading polymers can manifest as peri-implant osteolysis on radiographs.
- Severe Synovitis: When PGA or PLLA implants are used intra-articularly (e.g., for osteochondral lesions) and the implant head degrades or breaks off into the joint space, severe chemical synovitis can result. Utilizing slower-resorbing, high-strength PLLA may mitigate this risk by releasing debris at a more manageable rate.
- Incomplete Osseous Replacement: Studies by Marumo et al., Barber and Dockery, and Sassmannshausen et al. have definitively shown that while bioabsorbable implants dissolve, the resulting void is not reliably replaced by new bone. Fibrous tissue often fills the tract.
Despite these concerns, large-scale clinical outcomes remain favorable when indications are strictly followed. In a review of over 2,500 fractures fixed with bioabsorbables, Rokkanen et al. reported bacterial wound infections in 3.6%, non-specific foreign body reactions in 2.3%, and fixation failure in 3.7%. Similarly, Sinisaari et al. found that in 3,111 ankle fractures, infection rates were slightly lower with bioabsorbable fixation (3.2%) compared to metallic fixation (4.1%).
Future Horizons in Biomaterials
The future of orthopaedic biomaterials lies in composite technologies and bioactive carriers. Kato et al. have pioneered the use of bioabsorbable implants as delivery vehicles for Bone Morphogenetic Protein-2 (BMP-2) and other osteoinductive chemicals. By impregnating a porous PLLA or PDLLA matrix with BMP-2, the implant not only provides temporary mechanical stability but actively stimulates local osteogenesis as it degrades. Furthermore, composite materials blending PLLA with osteoconductive ceramics like Hydroxyapatite (HA) or Tricalcium Phosphate (TCP) are being developed to solve the issue of incomplete osseous replacement, ensuring that as the polymer dissolves, structural bone takes its place.
Conclusion
The selection of biomaterials in fracture fixation requires a profound understanding of metallurgy, polymer chemistry, and biomechanics. While 316L stainless steel and titanium alloys remain the gold standard for load-bearing diaphyseal fractures due to their high modulus and fatigue resistance, bioabsorbable polymers offer an elegant solution for low-load, periarticular, and pediatric applications. By respecting the biomechanical limitations of these materials—such as stress relaxation in polymers and notch sensitivity in titanium—the orthopaedic surgeon can optimize construct stability, minimize complications, and ensure the highest probability of successful physiological bone healing.
You Might Also Like