Anterior Cruciate Ligament Reconstruction: A Comprehensive Surgical Guide
Key Takeaway
Anterior Cruciate Ligament (ACL) reconstruction is a cornerstone procedure in sports orthopaedics, restoring knee stability and function. This guide details contemporary surgical approaches, including autograft and allograft options, tunnel placement, fixation methods, and evidence-based postoperative rehabilitation protocols. Mastering these techniques is crucial for optimal patient outcomes and return to activity.
Anterior Cruciate Ligament Reconstruction: A Comprehensive Surgical Guide
Introduction
The anterior cruciate ligament (ACL) is a critical static stabilizer of the knee, essential for maintaining tibiofemoral kinematics and preventing anterior translation and rotational instability. Rupture of the ACL is a common and debilitating injury, particularly prevalent in athletes participating in pivoting and contact sports. Left untreated, ACL deficiency often leads to recurrent episodes of instability, meniscal tears, chondral damage, and accelerated osteoarthritic changes. Anterior cruciate ligament reconstruction (ACLR) is a well-established surgical procedure aimed at restoring knee stability, protecting secondary intra-articular structures, and facilitating a safe return to pre-injury activity levels. This comprehensive guide provides an in-depth review of the indications, biomechanics, surgical techniques, and postoperative management of ACLR, tailored for advanced orthopedic residents, fellows, and practicing consultant surgeons.
Anatomy and Biomechanics of the ACL
The ACL is an intra-articular, extrasynovial ligament originating from the posteromedomedial aspect of the lateral femoral condyle and inserting into the anteromedial aspect of the tibial plateau, anterior to the intercondylar eminence. It comprises two functional bundles:
* Anteromedial (AM) bundle: Taut in flexion, primarily resists anterior tibial translation.
* Posterolateral (PL) bundle: Taut in extension, primarily resists rotational loads and anterior tibial translation in extension.
Biomechanical studies confirm the ACL's primary role in resisting anterior tibial translation and its secondary role in controlling internal and external rotation of the tibia relative to the femur. Its oblique orientation and bundle arrangement allow it to function throughout the knee's range of motion, providing dynamic stability in conjunction with surrounding musculature. Injury typically occurs via non-contact mechanisms involving sudden deceleration, cutting, or pivoting movements, often with a valgus collapse and internal rotation of the tibia on a fixed foot.
Clinical Pearl: A thorough understanding of the ACL's two functional bundles and their respective tensioning patterns is crucial for achieving anatomical and biomechanically sound graft placement, particularly in double-bundle reconstruction techniques.
Indications for ACL Reconstruction
The decision to proceed with ACLR is individualized, considering patient age, activity level, presence of instability, and associated injuries.
Primary indications include:
* Symptomatic knee instability: Recurrent episodes of "giving way" during activities of daily living or sports.
* Active patients desiring return to pivoting or high-demand sports: Individuals involved in sports requiring cutting, jumping, or rapid changes in direction (e.g., football, basketball, soccer, skiing).
* Associated meniscal tears: Especially reparable meniscal tears, as ACLR provides a stable environment conducive to meniscal healing.
* Associated collateral ligament injuries: Particularly medial collateral ligament (MCL) grade III injuries, which may benefit from concomitant ACLR after initial non-operative management of the MCL.
* Skeletally immature patients: While historically a relative contraindication, physeal-sparing techniques have expanded indications for younger, active patients to prevent recurrent instability and secondary damage.
Contraindications
Absolute contraindications are few, but relative contraindications warrant careful consideration:
* Advanced degenerative joint disease: Significant osteoarthritis may preclude the benefits of ACLR, with arthroplasty potentially being a more appropriate long-term solution.
* Active infection: Must be resolved prior to surgery.
* Non-compliant patient: Inability or unwillingness to adhere to a rigorous postoperative rehabilitation protocol.
* Limited functional demands: Sedentary individuals without symptomatic instability may be managed non-operatively.
* Skeletally immature patients: Traditional transphyseal techniques carry a risk of growth disturbance, necessitating physeal-sparing approaches or delayed surgery.
Preoperative Assessment and Planning
A meticulous preoperative evaluation is paramount for successful ACLR.
* History: Detailed account of injury mechanism, symptoms (pain, swelling, instability), and functional limitations.
* Physical Examination:
* Lachman test: Most sensitive test for ACL rupture, assessing anterior tibial translation at 20-30 degrees of flexion.
* Pivot Shift test: Dynamic test assessing anterolateral rotatory instability, highly specific for ACL deficiency.
* Anterior Drawer test: Performed at 90 degrees of flexion, less sensitive than Lachman.
* Assessment of range of motion, effusion, and concomitant ligamentous or meniscal injuries.
* Imaging:
* Plain radiographs: To rule out fractures, assess skeletal maturity, and evaluate for pre-existing arthritic changes. Weight-bearing views are essential.
* Magnetic Resonance Imaging (MRI): Confirms ACL rupture, identifies associated meniscal tears, chondral lesions, and collateral ligament injuries. Crucial for surgical planning.
* Patient Education and Goal Setting: Discuss realistic expectations, graft options, surgical risks, and the demanding nature of rehabilitation.
* Prehabilitation: Optimizing knee range of motion, strength, and proprioception preoperatively can significantly improve postoperative outcomes.
Graft Choices
The selection of graft material is a critical decision, influencing surgical technique, donor site morbidity, and long-term outcomes.
Autografts
Autografts remain the gold standard due to their biological integration, lack of disease transmission risk, and superior biomechanical properties compared to allografts in younger, active patients.
* Bone-Patellar Tendon-Bone (BPTB) Autograft:
* Harvest: A central 10-12 mm wide strip of patellar tendon, with a 20-25 mm bone block from the patella and a 25-30 mm bone block from the tibial tubercle.
* Advantages: Strong bone-to-bone healing in tunnels, high initial stiffness, excellent long-term results, rapid incorporation.
* Disadvantages: Donor site morbidity (anterior knee pain, patellar fracture, patellar tendon rupture, kneeling pain), potential for patellofemoral dysfunction.
* Hamstring Tendons (Semitendinosus and Gracilis) Autograft:
* Harvest: Semitendinosus and gracilis tendons are harvested through a small incision over the anteromedial tibia. They are typically quadrupled to create a robust graft.
* Advantages: Less donor site morbidity (reduced anterior knee pain), smaller incision, potentially faster early rehabilitation.
* Disadvantages: Slower graft incorporation (tendon-to-bone healing), potential for hamstring weakness, graft elongation, and tunnel widening.
* Quadriceps Tendon (QT) Autograft:
* Harvest: A central strip of quadriceps tendon, typically with a patellar bone block (bone-quadriceps tendon-bone, BQTB) or as a soft tissue graft.
* Advantages: Large, strong graft, lower donor site morbidity than BPTB, versatile for revision cases.
* Disadvantages: Potential for anterior knee pain, patellar fracture, and quadriceps weakness.
Allografts
Allografts (e.g., BPTB, Achilles tendon, tibialis anterior, semitendinosus) are derived from cadaveric donors.
* Advantages: Avoids donor site morbidity, reduced surgical time, larger graft options, useful for multi-ligament injuries or revision surgery.
* Disadvantages: Risk of disease transmission (though extremely low with modern processing), slower incorporation, inferior biomechanical properties, higher re-rupture rates in young, active patients.
Surgical Warning: When using allografts, ensure they are procured from reputable tissue banks and processed using validated sterilization techniques (e.g., gamma irradiation, chemical sterilization) to minimize infection and disease transmission risks. However, these processes can also compromise graft strength.
Patient Positioning and Anesthesia
- Anesthesia: General or regional anesthesia (spinal/epidural) is acceptable. A femoral nerve block can aid postoperative pain control.
- Positioning: Supine on the operating table. A lateral post or leg holder is typically used to allow for knee flexion and extension. A tourniquet is applied to the proximal thigh to ensure a bloodless field, which is crucial for arthroscopic visualization. The contralateral leg is often placed in a well-padded stirrup.
Surgical Technique: General Principles
ACLR is predominantly performed arthroscopically, allowing for precise intra-articular work with minimal soft tissue disruption.
* Arthroscopic Portals: Standard anteromedial (AM) and anterolateral (AL) portals are established. An accessory AM portal may be used for improved visualization or instrument access, particularly for femoral tunnel drilling.
* Diagnostic Arthroscopy: A thorough diagnostic arthroscopy is performed to confirm ACL rupture, assess meniscal integrity, evaluate chondral surfaces, and identify any concomitant pathology.
* Debridement of ACL Remnant: The torn ACL remnant is debrided to improve visualization and prepare the femoral and tibial footprints for graft placement. Care is taken to preserve any viable remnant tissue, as it may contribute to graft revascularization and proprioception.
* Meniscal and Chondral Lesion Management: Any meniscal tears (repairable or non-repairable) or chondral lesions are addressed prior to tunnel drilling.
Surgical Technique: Graft Harvest
BPTB Autograft Harvest
- Incision: A longitudinal incision (approximately 5-7 cm) is made over the patellar tendon, extending from the inferior pole of the patella to the tibial tubercle.
- Exposure: The subcutaneous tissue is incised, and the patellar tendon is exposed. The prepatellar bursa is identified and excised.
- Bone Block Preparation: Using an oscillating saw, a 10-12 mm wide, 20-25 mm long bone block is harvested from the inferior pole of the patella. Similarly, a 10-12 mm wide, 25-30 mm long bone block is harvested from the tibial tubercle.
- Tendon Harvest: The central strip of patellar tendon connecting the two bone blocks is carefully harvested, ensuring adequate length.
- Closure: The patellar tendon defect is typically closed with absorbable sutures, and the skin is closed in layers.
Hamstring Autograft Harvest
- Incision: A 2-3 cm oblique or longitudinal incision is made over the anteromedial aspect of the proximal tibia, approximately 2-3 cm distal to the joint line.
- Exposure: The sartorial fascia is incised, exposing the semitendinosus and gracilis tendons.
- Tendon Identification: The semitendinosus is typically identified first, followed by the gracilis. They are separated from surrounding tissues.
- Harvest: A specialized tendon stripper is used to harvest the tendons proximally, ensuring maximal length. The tendons are then prepared on a graft board, typically quadrupled and secured with sutures, creating a robust graft of appropriate diameter (usually 8-10 mm).
Pitfall: During hamstring harvest, avoid excessive traction on the tendons to prevent avulsion from their musculotendinous junction, which can compromise graft length. Ensure the stripper is advanced smoothly along the tendon.
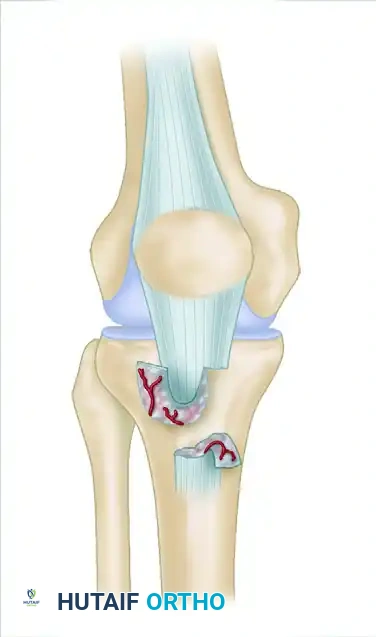
Surgical Technique: Femoral Tunnel Creation
Accurate femoral tunnel placement is paramount for restoring normal knee kinematics and preventing graft impingement.
* Anatomic Considerations: The femoral tunnel should be placed within the native ACL footprint on the lateral femoral condyle, typically between the AM and PL bundles. This ensures isometric graft placement, minimizing tension changes throughout the range of motion.
* Techniques for Femoral Tunnel Drilling:
* Transtibial Technique: Historically common, but often results in a more vertical femoral tunnel, particularly with a steep tibial tunnel, which may not adequately control rotational stability. The tibial tunnel is drilled first, and a guide wire is passed through it into the femur.
* Anteromedial (AM) Portal Technique: The femoral tunnel is drilled independently through the AM portal, allowing for more anatomical and horizontal tunnel placement, better replicating the native ACL's obliquity. This is the preferred method for single-bundle anatomical reconstruction.
* Outside-In Technique: A guide pin is drilled from the lateral femoral cortex into the joint, allowing for precise placement, especially in revision cases or when specific tunnel angles are required.
* Drilling and Reaming: Once the guide wire is accurately positioned, a cannulated reamer is used to create the femoral tunnel to the desired depth and diameter, matching the prepared graft.
Surgical Technique: Tibial Tunnel Creation
The tibial tunnel must also be anatomically placed to avoid impingement and ensure proper graft function.
* Anatomic Considerations: The tibial tunnel should be positioned within the native ACL footprint on the anteromedial aspect of the tibial plateau, posterior to the anterior horn of the lateral meniscus. Avoid placing it too anteriorly (roof impingement in extension) or too posteriorly (PCL impingement, graft laxity).
* Guide Wire Placement: A tibial guide is used to place a guide wire from the anteromedial tibia into the joint, aiming for the center of the native ACL footprint.
* Drilling: A cannulated reamer is used to create the tibial tunnel, matching the graft diameter.
Surgical Warning: Impingement of the graft against the intercondylar notch (roof impingement) or the PCL can lead to graft failure. Ensure adequate notchplasty if necessary, and confirm free graft movement throughout the range of motion before fixation.
Surgical Technique: Graft Passage and Fixation
- Graft Passage: The prepared graft is carefully passed through the tibial tunnel and then into the femoral tunnel, ensuring it is not twisted.
- Fixation Methods: Secure fixation on both the femoral and tibial sides is crucial for initial graft stability and successful healing.
- Femoral Fixation:
- Suspensory Fixation (e.g., Endobutton, TightRope): A cortical button is flipped on the lateral femoral cortex, suspending the graft within the tunnel. Provides strong initial fixation, particularly for soft tissue grafts.
- Interference Screws: Bioabsorbable or metallic screws are inserted alongside the graft within the tunnel, compressing it against the bone. Effective for BPTB grafts (bone-to-bone healing) and can be used for soft tissue grafts.
- Cross-Pin Fixation: Pins are drilled transversely through the femoral condyle, intersecting and securing the graft within the tunnel.
- Tibial Fixation:
- Interference Screws: Similar to femoral side, used for both BPTB and soft tissue grafts.
- Post and Washer: A screw and washer are used to secure the graft to the tibial cortex distally.
- Staples: Used to secure the graft to the tibial cortex.
- Suspensory Fixation: Can also be used on the tibial side, often in conjunction with an interference screw.
- Femoral Fixation:
Clinical Pearl: When using interference screws, ensure the screw diameter is appropriate for the tunnel and graft, and that the screw is inserted parallel to the graft to maximize compression without damaging the graft.
Surgical Technique: Double-Bundle ACL Reconstruction
While single-bundle reconstruction remains the most common technique, double-bundle ACLR aims to more closely replicate the native ACL's anatomy and biomechanics by reconstructing both the AM and PL bundles.
* Rationale: May offer improved rotational stability compared to single-bundle techniques.
* Technique Overview: Involves creating two separate femoral tunnels (for AM and PL bundles) and two separate tibial tunnels, or a single wide tibial tunnel with two femoral tunnels. Each bundle is tensioned independently.
* Considerations: Technically more demanding, increased surgical time, potential for larger bone removal. Evidence for superior clinical outcomes over well-performed single-bundle reconstruction remains debated.
Surgical Technique: Physeal-Sparing ACL Reconstruction
For skeletally immature patients, traditional transphyseal drilling carries a risk of growth arrest or angular deformity. Physeal-sparing techniques aim to reconstruct the ACL while avoiding or minimizing physeal violation.
* Indications: Young, active patients with symptomatic instability and significant growth potential remaining.
* Techniques:
* All-Epiphyseal: Tunnels are drilled entirely within the epiphysis, avoiding the physis.
* Transphyseal with Small Diameter Tunnels: Using small diameter tunnels (e.g., 5-6 mm) and soft tissue grafts, with careful attention to central physeal placement.
* Partial Transphyseal/Over-the-Top: Utilizing a combination of epiphyseal and metaphyseal tunnels, or an "over-the-top" femoral graft placement.
* Graft Choice: Hamstring autografts are typically preferred due to their soft tissue nature and smaller tunnel requirements.
Postoperative Protocol
A structured, progressive rehabilitation program is critical for successful outcomes following ACLR.
* Immediate Postoperative Period (Days 0-14):
* Pain Management: Multimodal analgesia.
* Swelling Control: RICE (Rest, Ice, Compression, Elevation).
* Bracing: Often a hinged knee brace, locked in extension for ambulation, gradually allowing increasing flexion.
* Weight-Bearing: Typically partial weight-bearing with crutches, progressing to full weight-bearing as tolerated.
* Range of Motion (ROM): Early, gentle ROM exercises, aiming for full extension and 90 degrees of flexion by 2 weeks.
* Muscle Activation: Quadriceps and hamstring isometric exercises.
* Phase I: Protection and Early Motion (Weeks 2-6):
* Focus on achieving full knee extension, restoring normal gait, and protecting the healing graft.
* Progressive ROM, gentle strengthening (quadriceps sets, straight leg raises, hamstring curls).
* Proprioceptive exercises (balance board).
* Phase II: Strength and Neuromuscular Control (Weeks 6-12):
* Increase strengthening intensity (closed kinetic chain exercises: squats, lunges, leg press).
* Initiate light cardiovascular activities (stationary bike, elliptical).
* Advanced proprioception and balance training.
* Phase III: Advanced Strengthening and Sport-Specific Training (Months 3-6):
* Progress to open kinetic chain exercises, plyometrics, and agility drills.
* Sport-specific training, gradually increasing intensity.
* Focus on dynamic stability and neuromuscular control.
* Phase IV: Return to Sport (Months 6-12+):
* Criteria-based return to sport, not time-based.
* Requires full ROM, symmetrical strength (quadriceps and hamstrings >90% of contralateral limb), excellent neuromuscular control, and successful completion of sport-specific functional tests (e.g., hop tests).
* Psychological readiness is also crucial.
Clinical Pearl: Early achievement of full knee extension is paramount to prevent arthrofibrosis and patellofemoral pain. Patients should be educated on this from day one.
Complications
While ACLR is generally safe and effective, potential complications include:
* Infection: Rare but serious, requiring aggressive debridement and antibiotics.
* Arthrofibrosis/Stiffness: Can result from inadequate rehabilitation, graft impingement, or an inflammatory response. May require manipulation under anesthesia or arthroscopic lysis of adhesions.
* Graft Failure/Re-rupture: Occurs in 5-10% of cases, higher in young athletes. Often due to premature return to sport, technical error, or subsequent trauma.
* Patellofemoral Pain: More common with BPTB autografts due to donor site morbidity.
* Donor Site Morbidity: Pain, weakness, or numbness at the graft harvest site.
* Neurovascular Injury: Rare, but potential for saphenous nerve injury (hamstring harvest) or popliteal artery/nerve injury during tunnel drilling.
* Residual Laxity: May occur due to graft stretching, tunnel widening, or suboptimal tunnel placement.
* Cyclops Lesion: A localized anterior arthrofibrosis that can cause extension block.
Conclusion
Anterior cruciate ligament reconstruction is a highly successful procedure for restoring knee stability and function in appropriately selected patients. A thorough understanding of ACL anatomy and biomechanics, meticulous preoperative planning, precise surgical technique, and a dedicated postoperative rehabilitation program are all critical components for achieving optimal long-term outcomes. As surgical techniques and rehabilitation protocols continue to evolve, the focus remains on anatomical reconstruction and individualized patient care to facilitate a safe and effective return to desired activity levels.
📚 Medical References
- ACL reconstructions: what happens to the graft? Instr Course Lect 45:229, 1996.
- Arnoczky SP, Tarvin GB, Marshall JL: Anterior cruciate ligament replacement using patellar tendon: an evaluation of graft revascularization in the dog, J Bone Joint Surg 64A:217, 1982.
- Arnoczky SP, Torzilli PA, Warren RF, et al: Biologic fi xation of ligament prostheses and augmentations: an evaluation of bone ingrowth in the dog, Am J Sports Med 16:106, 1988.
- Azar FM: Surgical management of complications in anterior cruciate ligament surgery. Paper presented at the American Academy of Orthopaedic Surgeons Summer Institute, Seattle, Sept 1999.
- Bach BR Jr: Evaluation of ACL injuries and indications for reconstruction. Current treatment and techniques for athletic injuries to the knee. Paper presented at the American Academy of Orthopaedic Surgeons Summer Institute, Seattle, Sept 1999.
- Bach BR Jr, Levy ME, Bojchuk J, et al: Single-incision endoscopic anterior cruciate ligament reconstruction using patellar tendon autograft: minimum two-year follow-up evaluation, Am J Sports Med 26:30, 1998.
- Bach BR Jr, Tradonsky S, Bojchuk J, et al: Arthroscopicallyassisted anterior cruciate ligament reconstruction using patellar tendon autograft: fi veto nine-year follow-up evaluation, Am J Sports Med 26:20, 1998.
- Bach BR Jr, Warren RF, Wickiewicz TL: The pivot shift phenomenon: results and description of a modifi ed clinical test for anterior cruciate ligament insuffi ciency, Am J Sports Med 16:571, 1988.
- Ballock RT, Woo SL, Lyon RM, et al: Use of patellar tendon autograft for anterior cruciate ligament reconstruction in the rabbit: a long-term histologic and biomechanical study, J Orthop Res 7:474, 1989.
- Banovetz JM, Albright JP, Crowley ET: Conservative care of the anterior cruciate ligament–defi cient knee: a review of the literature and a treatment protocol, Sports Med Arthrosc Rev 5:29, 1997.
- Barrack RL, Skinner HB, Buckley SL: Proprioception in the anterior cruciate–defi cient knee, Am J Sports Med 17:1, 1989.
- Barrett DS: Proprioception and function after anterior cruciate reconstruction, J Bone Joint Surg 73B:833, 1991.
- Barrett GR, Line LL, Shelton WR, et al: The Dacron ligament prosthesis in anterior cruciate ligament reconstruction: a fouryear review, Am J Sports Med 21:367, 1993.
- Baugher WH, Warren RF, Marshall JL, et al: Quadriceps atrophy in the anterior cruciate–insuffi cient knee, Am J Sports Med 12:192, 1984.
- Beard DJ, Dodd CAF, Trunde HR, et al: Proprioception enhancement for anterior cruciate ligament defi ciency, J Bone Joint Surg 76B:654, 1994.
- Beard DJ, Kyberd PJ, Fergusson CM, et al: Proprioception after rupture of the anterior cruciate ligament: an objective indication of the need for surgery? J Bone Joint Surg 75B:311, 1993.
- Bechthold JE, Eastlund DT, Butts MK, et al: The effects of freeze-drying and ethylene oxide sterilization on mechanical properties of human patellar tendon, Am J Sports Med 22:562, 1994.
- Berchuck M, Andriacchi TP, Bach BR, et al: Gait adaptations by patients who have a defi cient anterior cruciate ligament, J Bone Joint Surg 72A:871, 1990.
- Beynnon BD, Johnson RJ, Abate JA, et al: Treatment of anterior cruciate ligament injuries, part I, Am J Sports Med 33:1579, 2005.
- Beynnon BD, Johnson RJ, Fleming BC, et al: The measurement of elongation of anterior cruciate ligament grafts in vivo, J Bone Joint Surg 76A:520, 1994.
- Beynnon BD, Pope MH, Wertheimer CM, et al: The effect of functional knee-braces on strain on the anterior cruciate ligament in vivo, J Bone Joint Surg 74A:1298, 1992.
- Blum MF, Garth WP, Lemons JE: The effects of graft rotation on attachment site separation distances in anterior cruciate ligament reconstruction, Am J Sports Med 23:282, 1995.
- Boden BP, Feagin JA Jr: Natural history of the ACL-defi cient knee, Sports Med Arthrosc Rev 65:20, 1997.
- Boeree NR, Ackroyd CE: Magnetic resonance imaging of anterior cruciate ligament rupture, J Bone Joint Surg 74B:614, 1992.
- Boeree NR, Watkinson AF, Ackroyd CE, et al: Magnetic resonance imaging of meniscal and cruciate injuries of the knee, J Bone Joint Surg 73B:452, 1991.
- Bonamo JJ, Krinick RM, Sporn AA: Rupture of the patellar ligament after use of its central third for anterior cruciate ligament reconstruction: a report of two cases, J Bone Joint Surg 66A:1294, 1984.
- Bradley DM, Bergman AG, Dillingham MF: MR imaging of cyclops lesions, AJR Am J Roentgenol 174:719, 2000.
- Bradley J, FitzPatrick D, Daniel D, et al: Orientation of the cruciate ligament in the sagittal plane: a method of predicting its length-change with fl exion, J Bone Joint Surg 70B:94, 1988.
- Bray RC, Dandy DJ: Meniscal lesions and chronic anterior cruciate ligament defi ciency: meniscal tears occurring before and after reconstruction, J Bone Joint Surg 71:128, 1989.
- Brown CH Jr, Carson EW: Revision anterior cruciate ligament surgery, Clin Sports Med 18:109, 1999.
- Burks R, Daniel D, Losse G: The effect of continuous passive motion on anterior cruciate ligament reconstruction stability, Am J Sports Med 12:323, 1984.
- Burks RT, Leland R: Determination of graft tension before fi xation in anterior cruciate ligament reconstruction, Arthroscopy 4:260, 1988.
- Bush-Joseph CA: Evaluation of ACL injuries and indications for reconstruction. Presented at the American Academy of Orthopaedic Surgeons Summer Institute, Toronto, Sept 2000.
- Buss DD, Min R, Skyhar M, et al: Nonoperative treatment of acute anterior cruciate ligament injuries in a selected group of patients, Am J Sports Med 23:160, 1995.
- Buss DD, Warren RF, Wickiewicz TL, et al: Arthroscopically assisted reconstruction of the anterior cruciate ligament with use of autogenous patellar-ligament grafts: results after twentyfour to forty-two months, J Bone Joint Surg 75A:1346, 1993.
- Butler DL: Evaluation of fi xation methods in cruciate ligament replacement, Instr Course Lect 36:173, 1987.
- Bylski-Austrow DI, Grood ES, Hefzy MS, et al: Anterior cruciate ligament replacements: a mechanical study of femoral attachment location, fl exion angle at tensioning, and initial tension, J Orthop Res 8:522, 1990.
- Bynum EB, Barrack RL, Alexander AH: Open versus closed chain kinetic exercises after anterior cruciate ligament recon- struction: a prospective randomized study, Am J Sports Med 23:401, 1995.
- Cabaud HE, Feagin JA, Rodkey WG: Acute anterior cruciate ligament injury and augmented repair: experimental studies, Am J Sports Med 8:395, 1980.
- Cabaud HE, Feagin JA, Rodkey WG: Acute anterior cruciate ligament injury and repair reinforced with a biodegradable intraarticular ligament: experimental studies, Am J Sports Med 10:259, 1982.
- Caborn DNM, Johnson BM: The natural history of the anterior cruciate ligament–defi cient knee, Clin Sports Med 12:625, 1993.
- Cameron M, Buchgraber A, Paller H, et al: The natural history of anterior cruciate ligament–defi cient knee: changes in synovial fl uid cytokine and keratin sulfate concentrations, Am J Sports Med 25:751, 1997.
- Carson EW, Anisko EM, Restrepo C, et al: Revision anterior cruciate ligament reconstruction: etiology of failures and clinical results, J Knee Surg 17:127, 2004.
- Casteleyn PP: Management of anterior cruciate ligament lesions: surgical fashion, personal whim, or scientifi c evidence? Study of mediumand long-term results, Acta Orthop Belg 65:327, 1999.
- Cerabona F, Sherman MF, Bonamo JR, et al: Patterns of meniscal injury with acute anterior cruciate ligament tears, Am J Sports Med 16:603, 1988.
- Cha PS, Brucker PU, West RV, et al: Arthroscopic doublebundle anterior cruciate ligament reconstruction: an anatomic approach, Arthroscopy 21:1275, 2005.
- Chick RP, Collins HR, Rubin BD, et al: The pes anserinus transfer: a long-term follow-up, J Bone Joint Surg 63A:1449, 1981.
- Ciccotti MG, Lombardo SJ, Nonweiler B, et al: Nonoperative treatment of ruptures of anterior cruciate ligament in middleaged patients, J Bone Joint Surg 76A:1315, 1994.
- Clancy WG Jr: Anterior cruciate ligament functional instability: a static intra-articular and dynamic extra-articular procedure, Clin Orthop Relat Res 172:102, 1983.
- Clancy WG Jr: Intra-articular reconstruction of the anterior cruciate ligament, Orthop Clin North Am 16:181, 1985.
- Clancy WG Jr, Narechania RG, Rosenberg TD, et al: Anterior and posterior cruciate ligament reconstruction in Rhesus monkeys: a histological, microangiographic, and biomechanical analysis, J Bone Joint Surg 63A:1270, 1981.
- Clancy WG Jr, Nelson DA, Reider B, et al: Anterior cruciate ligament reconstruction using one-third of the patellar ligament, augmented by extra-articular
You Might Also Like