INTRODUCTION TO ULNAR TUNNEL SYNDROME
Ulnar tunnel syndrome, clinically synonymous with Guyon’s canal syndrome, results from the mechanical compression or ischemic neuropathy of the ulnar nerve within a tight, triangular fibro-osseous tunnel located at the volar aspect of the carpus. Measuring approximately 1.5 cm in length, this anatomical bottleneck represents the second most common site of ulnar nerve entrapment in the upper extremity, superseded only by cubital tunnel syndrome at the elbow.
Compared with median nerve entrapment in carpal tunnel syndrome, ulnar tunnel syndrome is significantly less common. This discrepancy exists because the anatomical space occupied by the ulnar nerve at the wrist is inherently more yielding and less constrained by rigid osseous boundaries than the carpal tunnel. However, when compression does occur, it can lead to profound functional deficits in the hand, characterized by intrinsic muscle wasting, grip weakness, and debilitating sensory disturbances.
Understanding the precise topographical anatomy of the ulnar tunnel is paramount for the orthopedic surgeon, as the exact level of compression dictates whether the clinical presentation manifests as isolated motor deficits, isolated sensory deficits, or a mixed sensorimotor neuropathy.
SURGICAL ANATOMY AND BIOMECHANICS
The ulnar tunnel (Guyon's canal) is a complex, three-dimensional fibro-osseous space. A thorough mastery of its boundaries and internal contents is critical for safe surgical exploration and effective decompression.
Boundaries of the Ulnar Tunnel
- Roof (Volar): Formed proximally by the volar carpal ligament (a continuation of the deep fascia of the forearm) and distally by the palmaris brevis muscle.
- Floor (Dorsal): Composed of the deep transverse carpal ligament (flexor retinaculum) and the pisohamate ligament.
- Medial Wall (Ulnar): Defined by the pisiform bone, the tendon of the flexor carpi ulnaris (FCU), and the pisometacarpal ligament.
- Lateral Wall (Radial): Formed by the hook of the hamate.
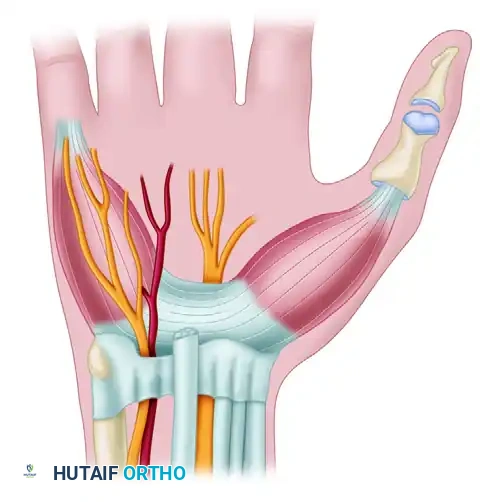
Zones of Compression
The ulnar nerve and artery enter the proximal aspect of the tunnel. Within the canal, the ulnar nerve bifurcates into a superficial sensory branch and a deep motor branch. Based on this bifurcation, the tunnel is anatomically divided into three distinct zones, which correlate directly with clinical presentation:
- Zone I: Encompasses the proximal portion of the canal, prior to the bifurcation of the ulnar nerve. Compression here yields mixed motor and sensory symptoms.
- Zone II: Surrounds the deep motor branch after bifurcation. This branch dives deeply between the abductor digiti minimi and the flexor digiti minimi brevis, passing through the pisohamate hiatus. Compression here yields isolated motor deficits (affecting the interossei, lumbricals to the ring and small fingers, adductor pollicis, and deep head of the flexor pollicis brevis).
- Zone III: Surrounds the superficial sensory branch, which supplies sensation to the volar aspect of the small finger and the ulnar half of the ring finger. Compression here yields isolated sensory deficits without intrinsic muscle weakness.
Clinical Pearl: When both volar and dorsal sensory complaints are present in the ulnar distribution, the site of ulnar nerve involvement is almost certainly proximal to the wrist. The dorsal ulnar cutaneous nerve (DUCN) branches from the parent ulnar nerve at least 8.0 cm proximal to the pisiform. Therefore, intact dorsal sensation with volar sensory loss strongly localizes the lesion to the ulnar tunnel.
ETIOLOGY AND PATHOGENESIS
Unlike carpal tunnel syndrome, which is frequently idiopathic, ulnar tunnel syndrome is almost always secondary to an identifiable structural or space-occupying lesion.
Space-Occupying Lesions
Compression just distal to the tunnel frequently affects the deep motor branch (Zone II). A space-occupying lesion, such as a ganglion cyst (the most common non-traumatic cause) or a benign tumor (e.g., lipoma, giant cell tumor of tendon sheath), can cause insidious compression in this area. Aberrant musculature, such as an accessory abductor digiti minimi or a reversed palmaris longus, has also been documented as a rare cause of dynamic compression.
Traumatic Causes
Fractures of the carpal bones, specifically the hook of the hamate, can precipitate ulnar tunnel syndrome. These fractures often occur in racket sports, golf, or baseball due to the direct impact of the handle against the hypothenar eminence. The resulting hemorrhage, edema, or fracture callus can exert direct pressure on the adjacent deep motor branch of the ulnar nerve.
Vascular Anomalies
The ulnar artery travels alongside the ulnar nerve within Guyon's canal and is highly susceptible to repetitive microtrauma, a condition known as Hypothenar Hammer Syndrome. This is classically seen in manual laborers who use the heel of their hand as a hammer.
Pathologies of the ulnar artery that can compress the ulnar nerve include:
1. Thrombosis of the ulnar artery: Leading to localized inflammation and secondary nerve compression.
2. True Fusiform Aneurysm: A generalized dilation of the arterial wall.
3. Saccular "False" Aneurysm (Pseudoaneurysm): Arising from a traumatic tear in the arterial wall, leading to a contained hematoma that compresses the nerve.
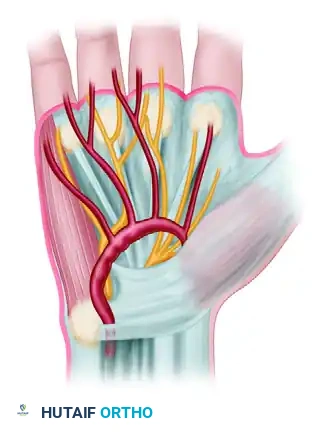
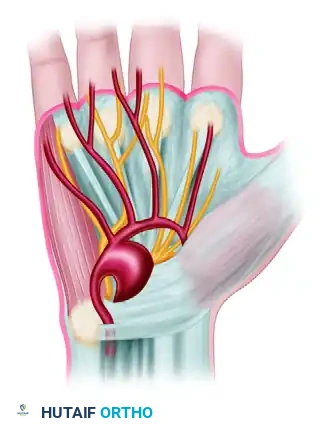
Surgical Warning: Should the ulnar artery be occluded for several millimeters, secondary Raynaud syndrome may be produced in the ulnar three digits. This occurs because the sympathetic nerve fibers supplying these digits travel in the adventitia of the ulnar artery.
Systemic and Inflammatory Conditions
Occasionally, in advanced rheumatoid disease, tenosynovitis can cause simultaneous carpal tunnel and ulnar tunnel syndromes in the same hand.
CLINICAL EVALUATION AND DIAGNOSIS
History and Physical Examination
A meticulous physical examination is required to localize the lesion and rule out proximal entrapment.
* Motor Examination: Assess for intrinsic muscle wasting (interosseous atrophy, hypothenar atrophy). Evaluate for Froment's sign (compensatory interphalangeal joint flexion by the flexor pollicis longus during key pinch due to adductor pollicis weakness) and Wartenberg's sign (abduction posturing of the small finger due to unopposed action of the extensor digiti minimi and weakness of the third palmar interosseous). Severe cases may present with an ulnar claw hand (hyperextension of the MCP joints and flexion of the IP joints of the ring and small fingers).
* Sensory Examination: Map the exact distribution of sensory loss. Crucially, test the dorsum of the ulnar hand. If dorsal sensation is diminished, the lesion is proximal to Guyon's canal (e.g., cubital tunnel syndrome).
* Vascular Examination: Perform an Allen test to assess the patency of the ulnar and radial arteries and the completeness of the superficial palmar arch.
Differential Diagnosis
The differential diagnosis for ulnar-sided hand pain and weakness is broad and must include:
* Cubital tunnel syndrome (ulnar nerve compression at the elbow).
* C8-T1 cervical disc herniation or radiculopathy.
* Thoracic outlet syndrome (lower trunk brachial plexopathy).
* Peripheral neuropathy (e.g., diabetic, alcoholic).
* Syringomyelia or motor neuron disease (e.g., ALS).
Diagnostic Imaging and Electrodiagnostics
- Radiographs: Standard PA, lateral, and carpal tunnel views. A specific carpal tunnel view or a supinated oblique view is essential to evaluate for a hook of hamate fracture.
- Ultrasound/MRI: Highly sensitive for identifying space-occupying lesions (ganglia, lipomas) and vascular anomalies (aneurysms) within the canal.
- Electrodiagnostic Studies (EMG/NCS): Essential for confirming the diagnosis, localizing the exact zone of compression, and ruling out proximal lesions or generalized peripheral neuropathy. Prolonged distal motor latencies to the abductor digiti minimi or first dorsal interosseous, with normal conduction velocities across the elbow, confirm the diagnosis.
INDICATIONS FOR SURGERY
Conservative management (splinting, NSAIDs, activity modification) is generally reserved for mild, idiopathic cases or transient neuropraxias.
Absolute indications for surgical exploration and decompression include:
1. Presence of a space-occupying lesion (ganglion, tumor).
2. Vascular pathology (aneurysm, thrombosis).
3. Displaced or non-union fractures of the hook of the hamate.
4. Progressive motor weakness or intrinsic atrophy.
5. Failure of conservative management after 3 to 6 months.
SURGICAL TECHNIQUE: ULNAR NERVE DECOMPRESSION
Treatment consists of the meticulous exploration of the ulnar nerve at the wrist and the complete removal of any cause of compression.
Preoperative Setup and Positioning
- The patient is placed supine with the operative arm extended on a radiolucent hand table.
- A well-padded pneumatic tourniquet is applied to the proximal arm.
- The limb is exsanguinated, and the tourniquet is inflated (typically to 250 mm Hg).
- Surgical loupe magnification (minimum 2.5x to 3.5x) is highly recommended for the identification of delicate neural and vascular branches.
Incision and Superficial Dissection
- Incision Design: Make a curvilinear or zigzag incision beginning 3 to 4 cm proximal to the distal wrist flexion crease, overlying the flexor carpi ulnaris (FCU) tendon. Extend the incision distally across the wrist crease in an oblique fashion toward the base of the hypothenar eminence, avoiding a perpendicular crossing of the flexion creases to prevent scar contracture.
- Protecting Cutaneous Nerves: Carefully incise the skin and subcutaneous tissue. Identify and protect the palmar cutaneous branches of the ulnar and median nerves.
- Fascial Release: Identify the FCU tendon proximally. The ulnar nerve and artery lie immediately radial and deep to the FCU. Incise the superficial fascia of the forearm and trace the neurovascular bundle distally toward the wrist.
Deep Dissection and Decompression (Zone by Zone)
- Zone I Decompression: Identify the volar carpal ligament (the roof of the proximal tunnel). Divide this ligament longitudinally to unroof the proximal aspect of Guyon's canal. Retract the FCU ulnarward to fully expose the ulnar nerve and artery.
- Identifying the Bifurcation: Trace the parent ulnar nerve distally to its bifurcation into the superficial sensory and deep motor branches.
- Zone III Decompression: Follow the superficial sensory branch distally. Release any fascial bands from the palmaris brevis muscle that may be tethering the nerve.
- Zone II Decompression (The Deep Motor Branch): This is the most critical and technically demanding step. The deep motor branch dives dorsally and radially, passing between the abductor digiti minimi and the flexor digiti minimi brevis.
- Identify the fibrous leading edge of the hypothenar muscles (the pisohamate hiatus).
- Carefully divide the tendinous arch of the hypothenar muscles and the pisohamate ligament to fully decompress the deep branch.
- Follow the deep branch around the hook of the hamate to ensure no distal tethering exists.
Pitfall: Failure to adequately release the deep motor branch as it dives through the pisohamate hiatus is the most common cause of persistent postoperative intrinsic weakness. The surgeon must visually confirm the nerve is free from the pisiform proximally to the hook of the hamate distally.
Management of Specific Pathologies
- Ganglion Cysts: If a ganglion is encountered (typically arising from the triquetrohamate or pisotriquetral joint), it must be meticulously excised along with its stalk to minimize the risk of recurrence.
- Hook of Hamate Fractures: If a non-union of the hook of the hamate is the source of compression, excision of the fracture fragment is generally preferred over internal fixation, followed by decompression of the nerve.
- Vascular Lesions: If an ulnar artery thrombosis or aneurysm is identified, vascular surgical principles apply. Segmental resection of the occluded or aneurysmal section and replacement with a reversed interposition vein graft (often harvested from the distal forearm or dorsal hand) is the preferred procedure when feasible. This restores flow to the superficial palmar arch and mitigates the risk of secondary Raynaud syndrome.
Closure
- Deflate the tourniquet and obtain meticulous hemostasis. Bipolar electrocautery should be used judiciously to avoid thermal injury to the nerve branches.
- Irrigate the wound copiously with sterile saline.
- The ligaments (volar carpal ligament, pisohamate ligament) are left open to prevent recurrent compression.
- Close the skin with interrupted non-absorbable sutures (e.g., 4-0 or 5-0 nylon).
- Apply a bulky, non-adherent compressive dressing and a volar orthosis with the wrist in a neutral position.
POSTOPERATIVE PROTOCOL AND REHABILITATION
Optimal recovery requires a structured postoperative rehabilitation program.
- Days 1-14: The wrist is immobilized in a neutral volar splint to allow for soft tissue healing and to prevent bowstringing of the neurovascular structures. Immediate active range of motion (ROM) of the digits is encouraged to prevent tendon adhesions and reduce edema.
- Weeks 2-4: Sutures are removed at 10 to 14 days. The rigid splint is transitioned to a removable wrist orthosis. Gentle active and active-assisted ROM of the wrist is initiated. Scar massage and desensitization techniques are employed.
- Weeks 4-8: Progressive strengthening of the wrist and hand begins. Particular attention is paid to intrinsic muscle strengthening.
- Months 3-12: Maximum medical improvement for nerve recovery can be prolonged. Usually, sensory symptoms are relieved rapidly, but weakened or atrophic intrinsic muscles may take 3 to 12 months to recover after surgery, depending on the severity and duration of preoperative compression. Wallerian degeneration requires axonal regrowth at a rate of approximately 1 mm per day.
COMPLICATIONS
While generally a safe and highly successful procedure, surgical decompression of the ulnar tunnel carries specific risks:
* Iatrogenic Nerve Injury: The deep motor branch is highly susceptible to injury during its release through the pisohamate hiatus. The palmar cutaneous branch can be injured during the initial skin incision, leading to painful neuromas.
* Incomplete Decompression: Resulting in persistent symptoms, most commonly due to failure to release the distal aspect of the deep motor branch.
* Vascular Compromise: Injury to the ulnar artery during dissection can lead to digital ischemia if the superficial palmar arch is incomplete.
* Pillar Pain: Similar to carpal tunnel release, patients may experience pain over the hypothenar eminence, which usually resolves with time and targeted therapy.
Thorough anatomical knowledge, meticulous surgical technique, and appropriate management of underlying etiologies are the cornerstones of successful outcomes in the treatment of ulnar tunnel syndrome.
📚 Medical References
- ulnar tunnel syndrome, Hand Clin 8:337, 1992.
- Rydevik B, Lundborg G, Bagge U: Effects of graded compression on intraneural blood fl ow, J Hand Surg 6A:3, 1981.
- [Seddon H: Surgical disorders of the peripheral nerves, Baltimore, 1972, Williams & Wilkins. Sunderland S: Nerves and nerve injuries, New York, 1978, Churchill Livingstone.
Diagnostic Aids, Management Plan, and Results Bralliar F: Electromyography: its use and misuse in peripheral nerve injuries, Orthop Clin North Am 12:229, 1981.](https://pubmed.ncbi.nlm.nih.gov/?term=Seddon%20H%3A%20Surgical%20disorders%20of%20the%20peripheral%20nerves%2C%20Baltimore%2C%201972%2C%20Williams%20%26%20Wilkins.%20Sunderland%20S%3A%20Nerves%20and%20nerve%20injuries%2C%20New%20York%2C%201978%2C%20Churchill%20Livingstone.%0A%0ADiagnostic%20Aids%2C%20Management%20Plan%2C%20and%20Results%20Bralliar%20F%3A%20Electromyography%3A%20its%20use%20and%20misuse%20in%20peripheral%20nerve%20injuries%2C%20Orthop%20Clin%20North%20Am%2012%3A229%2C%201981.)
- Cunningham ME, Potter HG, Weiland AJ: Closed partial rupture of a common digital nerve in the palm: a case report, J Hand Surg 30A:100, 2005.
- Dellon AL: Clinical use of vibratory stimuli to evaluate
