Introduction and Epidemiology
Primary tumors of the spine represent a complex intersection of orthopedic oncology and spine surgery. Of all primary benign bone tumors, approximately 8% occur in the spine or sacrum. While the vast majority of spinal column lesions encountered in clinical practice are metastatic or malignant in nature, 20% to 40% of primary spinal tumors are benign.
Understanding the epidemiological distribution is critical for formulating an accurate differential diagnosis. Benign spinal lesions demonstrate a strong predilection for younger demographics, with 60% of these tumors identified in patients during their second and third decades of life. Conversely, in patients older than 21 years, 70% of all spinal tumors are malignant.
Anatomical location within the vertebra also provides significant diagnostic clues. Typically, benign lesions arise within the posterior elements (pedicles, laminae, facets, and spinous processes). In stark contrast, 76% of anteriorly located lesions (within the vertebral body) are malignant. A notable exception to these rules is found in sacral tumors; lesions in the sacrum are more frequently malignant, even within younger age cohorts.
Clinical Pearl: When evaluating a spinal lesion, apply the "Age and Location" rule. A posterior element lesion in a 15-year-old is highly likely to be benign (e.g., osteoid osteoma, aneurysmal bone cyst), whereas a vertebral body lesion in a 55-year-old must be considered metastatic or a myeloma until proven otherwise.
Clinical Presentation and Diagnostic Evaluation
Patients with benign spinal tumors most frequently present with insidious, progressive back pain. Because these lesions are relatively rare, particularly in the thoracolumbar and lumbar spine, they are frequently misdiagnosed as mechanical back pain, leading to diagnostic delays ranging from 1 to 6 years.
Deformity, specifically a rigid and rapidly progressive scoliosis, is a hallmark presentation for certain benign tumors like osteoid osteoma and osteoblastoma. Unlike idiopathic scoliosis, the structural features of vertebral rotation are often absent. The tumor is typically located at the apex of the curve on the concavity, driven by asymmetrical muscle spasm secondary to localized inflammation.
Neurologic compromise is less common with benign lesions compared to malignancies but can occur with aggressive Stage 3 benign tumors (e.g., giant cell tumors or aggressive osteoblastomas) that expand into the epidural space.
Imaging Modalities
- Radiographs: Early plain films may appear deceptively normal. Subtle pedicle sclerosis or asymmetry may be the only initial finding.
- Computed Tomography (CT): The gold standard for defining osseous anatomy, cortical integrity, and identifying the pathognomonic central nidus of an osteoid osteoma.
- Magnetic Resonance Imaging (MRI): Essential for evaluating soft-tissue extension, epidural compression, and neural element involvement. Warning: Benign lesions like osteoblastoma can produce a massive inflammatory "flare" reaction on MRI, showing extensive marrow and soft-tissue edema that mimics a highly malignant extracompartmental sarcoma.
- Radioisotopic Bone Scan: Highly sensitive for localizing active bone-forming tumors, particularly when plain radiographs are equivocal.
Oncologic Principles and Enneking Classification
Surgical management of spinal tumors relies heavily on the Enneking classification system, which categorizes benign tumors based on their biological behavior, radiographic margins, and local aggressiveness.
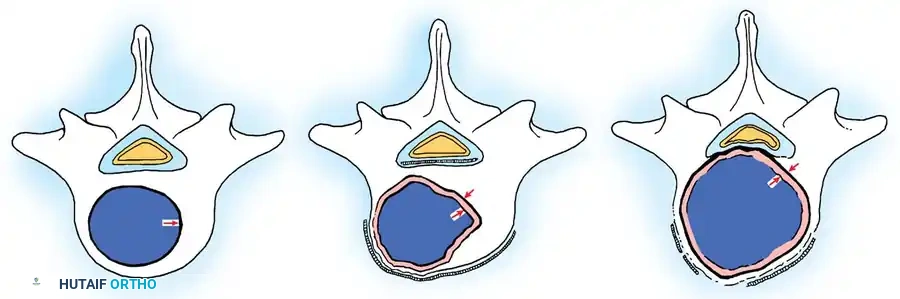
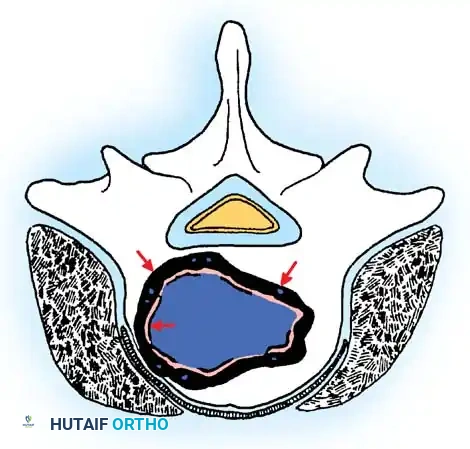
Fig. 41-30: Enneking staging of benign spinal tumors. Capsule of tumor is indicated by 1, and reactive pseudocapsule is indicated by 2. Stage 3 aggressive benign tumors can expand through the posterior vertebral wall and compress the cord. The pseudocapsule is vascularized reactive tissue and can adhere to the dura.
Stage 1: Latent Tumors
These lesions (e.g., asymptomatic osteochondromas, small eosinophilic granulomas) are inactive, well-marginated, and do not progress. They typically require no treatment other than observation. If surgery is mandated for diagnosis or mechanical symptoms, an intralesional excision (curettage) is sufficient.
Stage 2: Active Tumors
Stage 2 tumors (e.g., osteoid osteoma, standard osteoblastoma, aneurysmal bone cysts) grow slowly and cause symptoms, usually pain or deformity. They possess a reactive pseudocapsule. Treatment usually involves en bloc excision where anatomically feasible, though meticulous intralesional excision combined with local adjuvants (liquid nitrogen, phenol, or polymethylmethacrylate [PMMA]) often suffices.
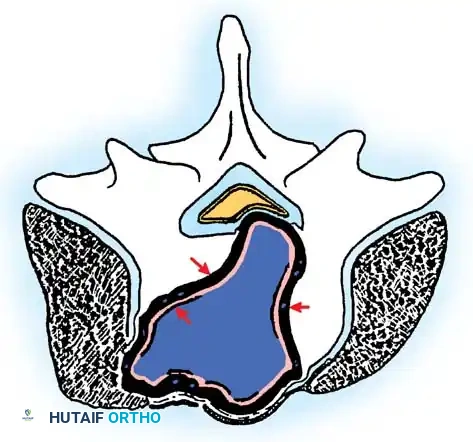
Cross-sectional representation demonstrating the expansion of an active Stage 2 benign lesion within the vertebral body, respecting the outer cortical margins but requiring meticulous intralesional clearance.
Stage 3: Aggressive Tumors
Despite being histologically benign, Stage 3 lesions (e.g., giant cell tumors, aggressive osteoblastomas) are locally destructive. They breach the tumor capsule, invade local compartments, and have a high propensity for recurrence.
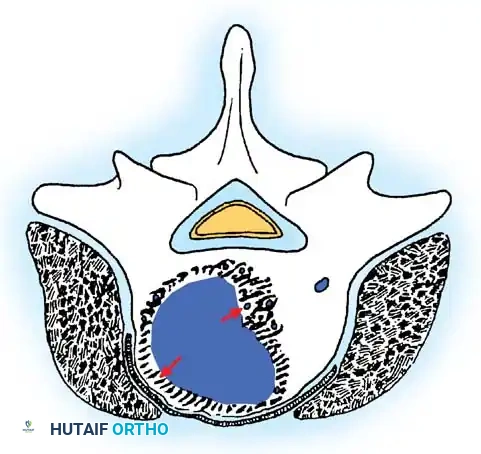
Cross-sectional view of a Stage 3 aggressive benign tumor breaching the posterior vertebral wall, highlighting the necessity for wide excision to prevent local recurrence.
Wide excision—removal of the tumor with a continuous cuff of normal, healthy tissue—is the oncologic treatment of choice. A marginal excision (dissecting through the reactive pseudocapsule) leaves microscopic disease and results in unacceptably high recurrence rates.
Surgical Anatomy and Operative Approaches
During the operative treatment of spinal tumors, the surgeon must balance the oncologic requirement for complete resection against the functional necessity of preserving neural and vascular anatomy.
Neural Preservation and Sacrifice
The spinal cord must be preserved at all costs in cervical and thoracic lesions. However, the management of nerve roots varies by anatomical location:
* Thoracic Roots: Because thoracic nerve roots primarily innervate intercostal muscles, unilateral sacrifice of several thoracic roots does not severely affect function and is often necessary for oncologic clearance. However, sacrificing numerous bilateral roots can impair chest cage mechanics and respiration.
* Sacral Roots: Resection of the sacrum heavily impacts bowel and bladder continence. If both S1 nerve roots and a single S2 nerve root are preserved, approximately 50% of patients will retain continence. If resection extends proximally and only a single S3 root is preserved, severe radiculopathy and incontinence are highly likely.
Vascular Considerations
Paired vascular structures, such as the vertebral arteries in the cervical spine, can occasionally be unilaterally resected if the tumor encases them. However, this mandates preoperative angiography with temporary balloon occlusion to confirm adequate collateral circulation from the contralateral vertebral artery and the Circle of Willis.
Regional Surgical Approaches
Cervical Spine:
Complex cervical resections often require specialized positioning and stabilization. Halo traction is frequently utilized to maintain alignment during destabilizing osteotomies or tumor resections.
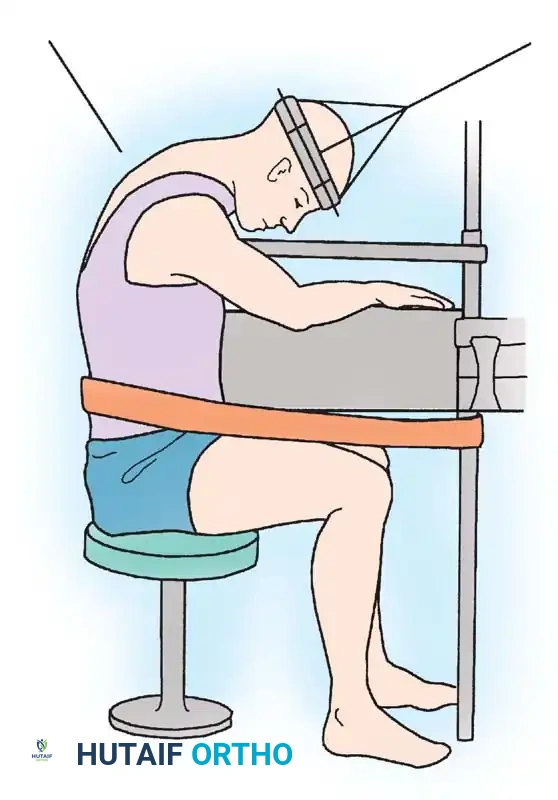
Fig. 41-29: Position of patient for cervical osteotomy—sitting on stool with head suspended by halo and traction. This allows circumferential access and precise alignment control during complex cervical reconstructions.
When performing posterior cervical resections, the osteotomy lines must be meticulously planned to ensure stability if reconstruction is attempted, or to safely widen the canal without plunging into the cord.
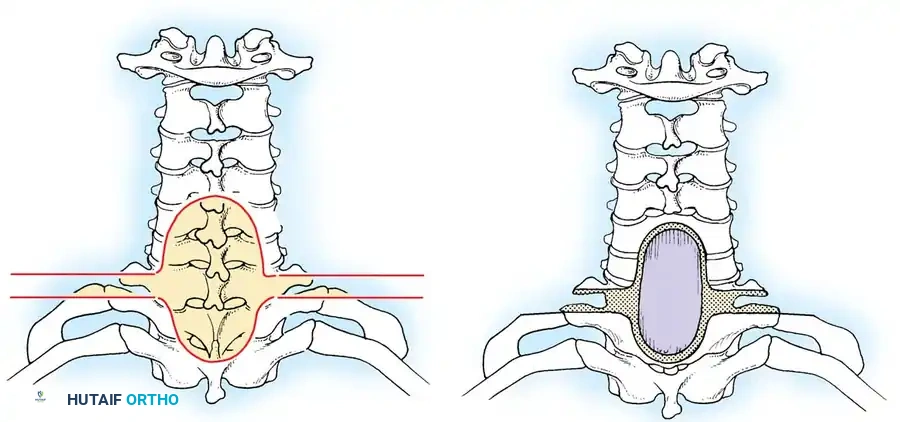
Fig. 41-28: Extent of resection of cervical laminae for safe osteotomy. Lateral resections are beveled toward each other so that opposing surfaces are parallel and in apposition after extension osteotomy.
Thoracic Spine:
A critical surgical axiom is that laminectomy alone does not provide safe access to the anterior column in the thoracic spine. Attempting to resect an anterior thoracic tumor via a standard posterior laminectomy carries an unacceptably high risk of spinal cord paralysis due to the kyphotic nature of the thoracic spine and the tenuous blood supply to the cord.
* Alternative Approach: For patients unable to tolerate a formal thoracotomy, a costotransversectomy or lateral extracavitary approach provides safe, direct access to the anterior column without manipulating the spinal cord.
Biomechanics and Spinal Stability
Tumor resection inherently destabilizes the spine. The criteria for reconstruction differ significantly between pediatric and adult populations, as well as between anterior and posterior resections.
Pediatric vs. Adult Considerations
In the immature pediatric spine, a multi-level laminectomy almost universally leads to progressive post-laminectomy kyphosis due to the loss of the posterior tension band in a growing skeleton. Therefore, arthrodesis is mandatory. The adult spine tolerates laminectomy better, though biomechanical limits still apply.
Anterior Column Stability
Instability created by anterior resection increases proportionally with the volume of the vertebral body removed.
* Rule of Thumb: Structural fusion (using cages, structural allograft, or titanium mesh) must be performed when any significant amount of the vertebral body is resected.
* Exception: If the tumor is managed with simple curettage (leaving the cortical rim intact) and the defect is packed with bone graft or PMMA, formal instrumented fusion is usually unnecessary.
Posterior Column Stability (Bridwell Criteria)
Determining instability after posterior resection is complex. Bridwell established a biomechanical point system, assigning 25% of overall stability to each of four distinct anatomical complexes:
1. The midline osteoligamentous complex (spinous processes, interspinous/supraspinous ligaments).
2. The left facet joint complex.
3. The right facet joint complex.
4. The anterior column (posterior vertebral wall, disc, and annulus).
Surgical Warning: Violation of two of these four complexes, or disruption of 50% of the total stabilizing structures, is an absolute indication for instrumentation and fusion.
Furthermore, impending pathological fractures dictate prophylactic stabilization. Criteria for impending instability include:
* More than 50% collapse of the vertebral body.
* Radiographic translation.
* Segmental kyphosis of >20 degrees above the regional normal.
* Combined involvement of both anterior and posterior columns.
Postoperatively, following anterior or posterior fusion, immobilization with a Thoracolumbosacral Orthosis (TLSO) is generally recommended until radiographic incorporation of the bone graft is identified, typically at 3 to 6 months.
Specific Posterior Element Tumors
Osteoid Osteoma
First described by Jaffe in 1935, osteoid osteoma is a benign, bone-forming lesion. Approximately 42% of all osteoid osteomas occur in the spine, making it the most common benign spinal tumor. It predominantly affects males in their second decade.
Anatomy and Presentation:
The lumbar spine is the most common location, followed by the cervical and thoracic regions. The lesion is almost invariably located in the posterior elements (pars interarticularis, pedicle, or facet). By definition, an osteoid osteoma is less than 2 cm in diameter.
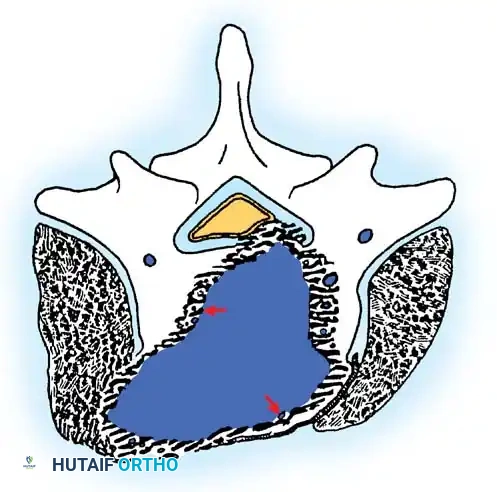
Illustration of a small, well-circumscribed nidus typical of an osteoid osteoma located within the posterior elements, surrounded by dense reactive sclerosis.
Pain is the primary complaint in 83% of patients. It is classically worse at night, awakening the patient in 30% of cases, and is dramatically relieved by NSAIDs (aspirin, ibuprofen) in 27% of patients due to the high concentrations of prostaglandins (PGE2) produced by the tumor nidus.
Scoliosis:
A rigid, painful scoliosis is present in up to 63% of patients. The tumor is typically located on the concavity of the curve. This deformity is secondary to severe, asymmetrical paraspinal muscle spasm rather than structural wedging.
Surgical Management:
Treatment consists of surgical excision of the nidus if medical management fails or if scoliosis is progressive.
* Open Excision: Complete removal of the nidus provides immediate, dramatic pain relief—the best postoperative indicator of success. If the resection destabilizes the facet or pars, a single-level localized fusion is performed simultaneously.
* Deformity Correction: Complete excision usually results in spontaneous resolution of the scoliosis, provided the curve has not been present for more than 15 to 18 months. If the curve persists beyond 18 months post-resection, structural changes may have occurred, necessitating standard scoliosis bracing or fusion.
* Minimally Invasive Techniques: High-frequency radiofrequency ablation (RFA) and percutaneous CT-guided thermocoagulation are highly effective for appendicular osteoid osteomas. However, their use in the spine requires extreme caution due to the proximity of the neural elements to the thermal ablation zone.
Osteoblastoma
Osteoblastoma accounts for 10% of all spinal tumors, and 32% of all osteoblastomas occur in the spine. Like osteoid osteoma, it peaks in the second and third decades with a 2:1 male-to-female predominance.
Pathology and Imaging:
While histologically similar to osteoid osteoma, osteoblastoma is distinguished by its size (greater than 2 cm) and its locally aggressive biological behavior. It almost always involves the posterior elements, though it frequently expands into contiguous vertebral levels and the vertebral body. The cervical spine is the most common location (40%), followed by lumbar (23%) and thoracic (21%).
Radiographic representation of an osteoblastoma: a destructive, expansile lesion with a thin rim of cortical bone, demonstrating significant lytic features and local tissue compromise.
Radiographically, it presents as a destructive, expansile lesion with a thin cortical shell. Lytic features predominate in 50% of cases. Because of its aggressive appearance, it is frequently misdiagnosed as osteosarcoma, Ewing sarcoma, or an aneurysmal bone cyst.
Surgical Management:
Because osteoblastomas are Enneking Stage 2 or 3 lesions, they have a high propensity for local recurrence. Intralesional excision (curettage) carries a recurrence rate of 10% to 20% at 9 years.
* Wide Excision: Whenever anatomically feasible, wide en bloc excision is the treatment of choice to prevent recurrence and the rare but documented risk of malignant transformation.
* Reconstruction: Due to the extensive nature of the resection required to achieve negative margins, complex multi-level instrumented fusion is almost universally required following the excision of a spinal osteoblastoma.
Conclusion
The management of benign tumors of the spine demands a rigorous, multidisciplinary approach. Surgeons must synthesize the patient's age, radiographic findings, and Enneking staging to formulate an appropriate oncologic resection plan. Whether performing a meticulous intralesional curettage for an osteoid osteoma or a complex wide excision and reconstruction for an aggressive osteoblastoma, the dual goals of complete tumor eradication and the preservation of spinal biomechanics and neurologic function remain paramount.
